Essential Fatty Acids
Contents
Summary
- Linoleic acid (LA), an omega-6 fatty acid, and α-linolenic acid (ALA), an omega-3 fatty acid, are considered essential fatty acids because they cannot be synthesized by humans. (More information)
- The long-chain omega-3 fatty acids, eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), can be synthesized from ALA, but due to low conversion efficiency, it is recommended to consume foods rich in EPA and DHA. (More information)
- Both omega-6 and omega-3 fatty acids are important structural components of cell membranes, serve as precursors to bioactive lipid mediators, and provide a source of energy. Long-chain omega-3 polyunsaturated fatty acids (PUFA in particular exert anti-inflammatory effects; it is recommended to increase their presence in the diet. (More information)
- Both dietary intake and endogenous metabolism influence whole body status of essential fatty acids. Genetic polymorphisms in fatty acid synthesizing enzymes can have a significant impact on fatty acid concentrations in the body. (More information)
- DHA supplementation during pregnancy may reduce the risks of early premature birth (birth before 34 weeks' gestation) and very low birth weight (<1.5 kg [<3 pounds 5 ounces]). (More information)
- DHA is important for visual and neurological development. However, supplementation with long-chain during pregnancy or early infancy appears to have no significant effect on children's visual acuity, neurodevelopment, and physical growth. (More information)
- Replacing saturated fat in the diet with omega-6 lowers total blood cholesterol; yet, randomized controlled trials have failed to demonstrate cardiovascular benefits in healthy people and people at risk for or with type 2 diabetes mellitus. Long-chain omega-3 PUFA supplementation may be useful to reduce mortality in patients with prevalent coronary heart disease (CHD) and in those with heart failure without preserved ventricular function. (More information)
- Increasing EPA and DHA intake may benefit individuals with type 2 diabetes mellitus, especially those with elevated serum triglycerides. However, evidence from large-scale randomized trials is insufficient to support the use of omega-3 PUFA supplements for cardiovascular disease prevention in those with type 2 diabetes. (More information)
- Observational studies have found fish intake to be associated with lower risks of cognitive deterioration and Alzheimer’s disease, but it is not yet clear whether supplementation with marine-derived omega-3 PUFA can help prevent cognitive decline. (More information)
- Several omega-3 formulations have been approved by the US Food and Drug Administration for the indication of treating severe hypertriglyceridemia. (More information)
- Although omega-3 PUFA deficiency may not be uncommon in neurodevelopmental and neuropsychiatric disorders, there is little evidence to suggest that supplementation may be a beneficial adjunct in the management of affected individuals. (More information)
- The Food and Nutrition Board of the US Institute of Medicine (now the National Academy of Medicine) established adequate intakes (AI) for omega-6 and omega-3 fatty acids. (More information)
Introduction
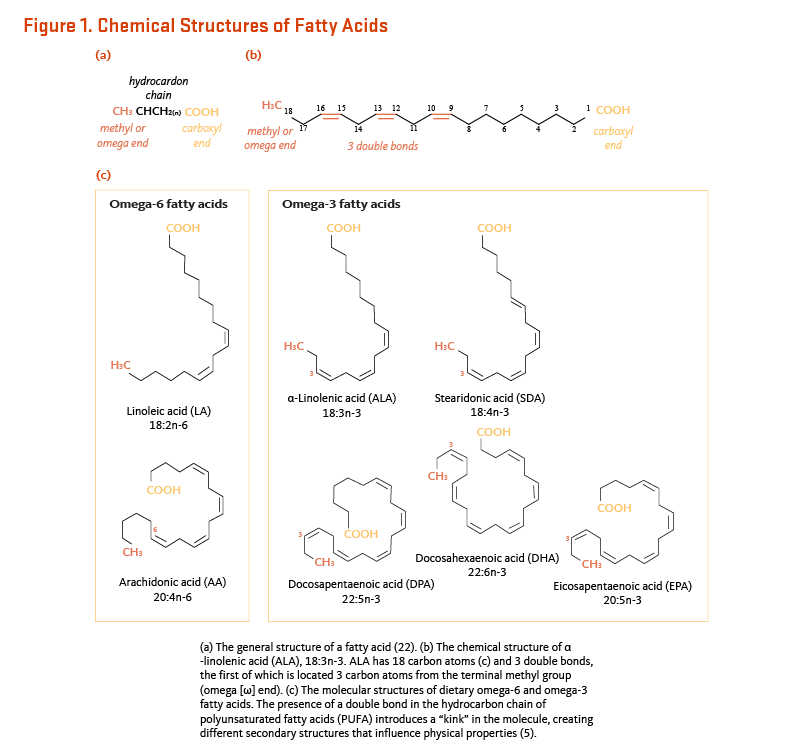
Omega-6 and omega-3 fatty acids are polyunsaturated fatty acids (PUFA), meaning they contain more than one cis double bond (1). In all omega-6 (ω6 or n-6) fatty acids, the first double bond is located between the sixth and seventh carbon atom from the methyl end of the fatty acid. Likewise, all omega-3 fatty acids (ω3 or n-3) have at least one double bond between the third and fourth carbon atom counting from the methyl end of the fatty acid. Scientific abbreviations for fatty acids tell the reader something about their chemical structure. For example, the scientific abbreviation for α-linolenic acid (ALA) is 18:3n-3. The first part (18:3) tells the reader that ALA is an 18-carbon fatty acid with three double bonds, while the second part (n-3) tells the reader that the first double bond is in the n-3 position, which defines this fatty acid as an omega-3 (Figures 1a & b). Double bonds introduce kinks in the hydrocarbon chain that influence the structure and physical properties of the fatty acid molecule (Figure 1c).
Although humans and other mammals can synthesize saturated fatty acids and some monounsaturated fatty acids from carbon groups in carbohydrates and proteins, they lack the delta (Δ) 12 and Δ15 desaturase enzymes necessary to insert a cis double bond at the n-6 or the n-3 position of a fatty acid (1). Consequently, omega-6 and omega-3 fatty acids are essential nutrients. The parent fatty acid of the omega-6 series is linoleic acid (LA; 18:2n-6), and the parent fatty acid of the omega-3 series is ALA (Figure 2 and Table 1). Humans can synthesize long-chain (20 carbons or more) omega-6 fatty acids, such as dihomo-γ-linolenic acid (DGLA; 20:3n-6) and arachidonic acid (AA; 20:4n-6), from LA and long-chain omega-3 fatty acids, such as eicosapentaenoic acid (EPA; 20:5n-3) and docosahexaenoic acid (DHA; 22:6n-3), from ALA (see Metabolism and Bioavailability).
[Figure 1a and 1b - Click to Enlarge] [Figure 1c - Click to Enlarge]
Metabolism and Bioavailability
Prior to absorption in the small intestine, fatty acids must be hydrolyzed from dietary fats (triglycerides and phospholipids) by pancreatic enzymes (2). Bile salts must also be present in the small intestine to allow for the incorporation of fatty acids and other fat digestion products into mixed micelles. Fat absorption from mixed micelles occurs throughout the small intestine and is 85%-95% efficient under normal conditions.
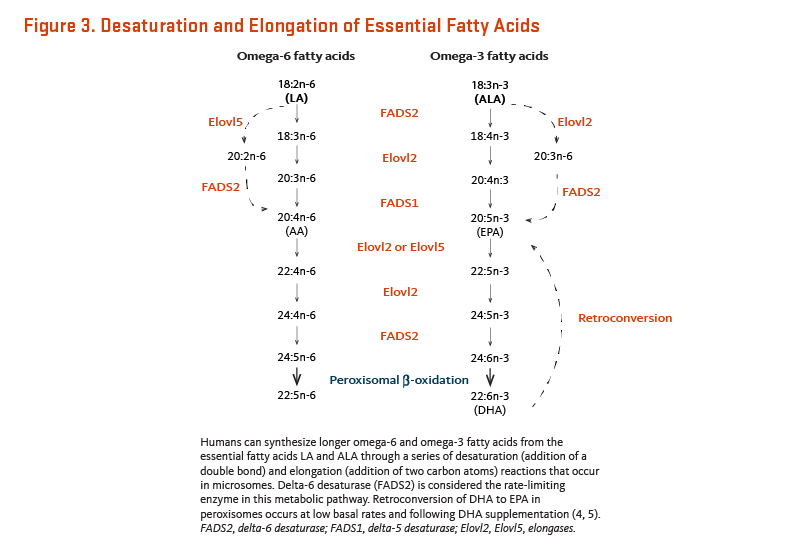
Concentrations of fatty acids in blood (i.e., whole blood, plasma, serum, and red blood cells) reflect both dietary intake and biological processes (3). Humans can synthesize longer omega-6 and omega-3 fatty acids from the essential fatty acids linoleic acid (LA) and α-linolenic acid (ALA), respectively, through a series of desaturation (addition of a double bond between two carbon atoms) and elongation (addition of two carbon atoms) reactions (Figure 3) (4, 5). LA and ALA compete for the same elongase and desaturase enzymes in the synthesis of longer polyunsaturated fatty acids (PUFA), such as arachidonic acid (AA), eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA).
Studies of ALA metabolism in healthy young men indicated that approximately 8% of dietary ALA was converted to EPA and 0%-4% was converted to docosahexaenoic acid (DHA) (6). In healthy young women, approximately 21% of dietary ALA was converted to EPA and 9% was converted to DHA (7). The better capacity to generate long-chain PUFA from ALA in young women compared to men is related to the effects of estrogen (8, 9). Although only the essentiality of ALA is recognized because it cannot be synthesized de novo by humans, the relatively low rate of ALA conversion into EPA and DHA suggests that these long-chain omega-3 PUFA may be considered conditionally essential nutrients.
In addition to gender differences, genetic variability in enzymes involved in fatty acid metabolism influences one’s ability to generate long-chain PUFA. Two key enzymes in fatty acid metabolism are delta 6 desaturase (FADS2) and delta 5 desaturase (FADS1) (Figure 3) (10). Two common haplotypes (a cluster of polymorphisms) in the FADS genes differ dramatically in their ability to generate long-chain PUFA: haplotype D is associated with increased FADS activity (both FADS1 and FADS2) and higher conversion rate of fatty acid precursors (LA and ALA) to long-chain PUFA (EPA, GLA, DHA, and AA) (11). These FADS polymorphisms are relatively common in the population and may explain up to 30% of the variability in blood concentrations of omega-3 and omega-6 fatty acids among individuals (3).
Finally, DHA can be retro-converted to EPA and DPA at a low basal rate and following supplementation (Figure 3) (12). After supplementing omnivores (n=8) and vegetarians (n=12) for six weeks with an EPA-free preparation of DHA (1.62 g/day), EPA, DPA, and DHA concentrations increased in serum and platelet phospholipids (13). Based on the measured changes, the estimated percent retroconversion of DHA to EPA was 7.4%-11.4% (based on serum phospholipid data) and 12.3%-13.8% (based on platelet phospholipid data), with no significant difference between omnivores and vegetarians. Due to this nontrivial retroconversion efficiency, DHA supplementation may represent an alternative to fish oil to increase blood and tissue concentrations of EPA, DPA, and DHA (see Supplements) (5).
Biological Activities
Membrane structure and function
Omega-6 and omega-3 PUFA are important structural components of cell membranes. When incorporated into phospholipids, they affect cell membrane properties, such as fluidity, flexibility, permeability, and the activity of membrane-bound enzymes and cell-signaling pathways (14, 15). In addition to endogenous metabolism, dietary consumption of fatty acids can modify the composition and molecular structure of cellular membranes. Thus, increasing omega-3 fatty acid intake increases the omega-3 content of red blood cells, immune cells (16), atherosclerotic plaques (17), cardiac tissue (18), and other cell types throughout the body.
DHA is selectively incorporated into retinal cell membranes and postsynaptic neuronal cell membranes, suggesting it plays important roles in vision and nervous system function. In fact, DHA represents the predominant PUFA in the retina and neuronal cells (19).
Vision
DHA is found at very high concentrations in the cell membranes of the retina; the retina conserves and recycles DHA even when omega-3 fatty acid intake is low (20). Animal studies indicate that DHA is required for the normal development and function of the retina. Moreover, these studies suggest that there is a critical period during retinal development when inadequate DHA will result in permanent abnormalities in retinal function. Research indicates that DHA plays an important role in the regeneration of the visual pigment rhodopsin, which plays a critical role in the visual transduction system that converts light hitting the retina to visual images in the brain (21).
Nervous system
The phospholipids of the brain's gray matter contain high proportions of long-chain PUFA, suggesting they are important to central nervous system function (22). AA stimulates glucose uptake by cortical astrocytes, meaning that it is important for energy metabolism (23). AA and DHA also increase the release of acetylcholine, which enhances synaptic plasticity and memory, thereby improving learning abilities (24). Although trials of PUFA supplementation during pregnancy and/or early infancy failed to show cognitive improvements in offspring (see Disease Prevention), the availability of omega-3 and omega-6 fatty acids to the fetus and infants is essential for the growth of their brain and development of brain functions. There is compelling evidence to suggest that PUFA are essential to neuronal growth and synapse formation, and for appropriate neurotransmission (reviewed in 25).
Synthesis of lipid mediators
Oxylipins
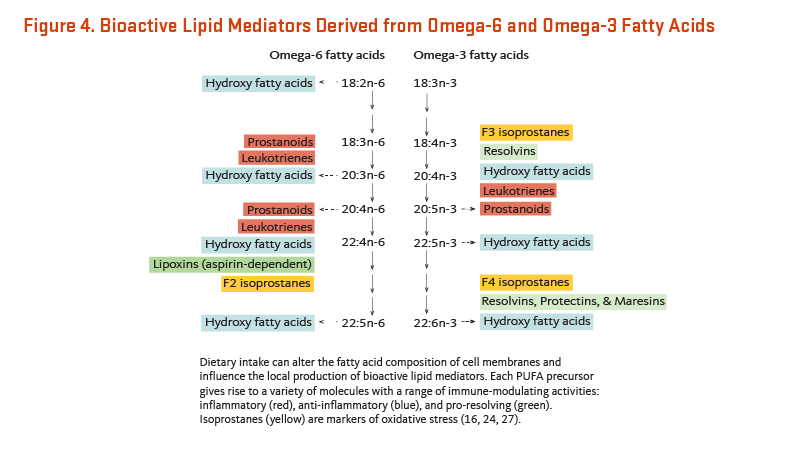
Oxylipins are potent chemical messengers derived from PUFA. They play critical roles in immune and inflammatory responses. The most common oxylipins are eicosanoids that encompass numerous bioactive lipid mediators derived from 20-carbon ("eicosa-") AA. Following stimulation by hormones, cytokines, and other stimuli, PUFA bound to membrane phospholipids are released from cell membranes and become substrates for dodecanoid, eicosanoid, and docosanoid production. Oxylipin synthesis relies primarily on three families of enzymes: cyclooxygenases (COX), lipoxygenases (LOX), and cytochrome p450 mono-oxygenases (P450s) (26). From C18-C22 precursors, COX enzymes produce prostaglandins, prostacyclins, and thromboxanes (collectively known as prostanoids); LOX produces leukotrienes and hydroxy fatty acids; and P450s produce hydroxyeicosatetraenoic acids ("HETEs") and epoxides (Figure 4).
Physiological responses to AA-derived eicosanoids differ from responses to EPA-derived eicosanoids. In general, EPA is a poor substrate for eicosanoid production and EPA-derives eicosanoids are less potent inducers of inflammation, blood vessel constriction, and coagulation than eicosanoids derived from AA (19, 27).
Nonetheless, it is an oversimplification to label all AA-derived eicosanoids as pro-inflammatory. AA-derived prostaglandins induce inflammation but also inhibit pro-inflammatory leukotrienes and cytokines and induce anti-inflammatory lipoxins, thereby modulating the intensity and duration of the inflammatory response via negative feedback (Figure 4) (17).
Pro-resolving mediators
A separate class of PUFA-derived bioactive lipids, specialized pro-resolving mediators (SPMs), has been more recently identified (reviewed in 28). These molecules function as local mediators of the resolution phase of inflammation, actively turning off the inflammatory response. SPMs are derived from both omega-6 and omega-3 PUFA (Figure 4) (29). The S-series of SPMs results from the LOX-mediated oxygenation of EPA and DHA, giving rise to S-resolvins, S-protectins, and S-maresins. A second class of SPMs, the R-series, is generated from the aspirin-dependent acetylation of COX-2 and subsequent generation of aspirin-triggered SPMs from AA, EPA, and DHA. It appears that these mediators may explain many of the anti-inflammatory actions of omega-3 fatty acids that have been described (16, 30).
Isoprostanes
Isoprostanes are prostaglandin-like compounds that are formed by non-enzymatic, free radical-induced oxidation of any PUFA with three or more double bonds (Figure 4) (26). Because they are produced upon exposure to free radicals, isoprostanes are often used as markers for oxidative stress. In contrast to prostanoids, isoprostanes are synthesized from esterified PUFA precursors and remain bound to the membrane phospholipid until cleaved by PLA2 and released into circulation. In addition to being used as markers of oxidative stress, isoprostanes may also function as inflammatory mediators, exerting both pro- and anti-inflammatory effects (26).
Regulation of gene expression
PUFA are pleiotropic regulators of cell function. They can regulate gene expression directly by interacting with transcription factors or indirectly by influencing membrane lipid composition and cell signaling pathways.
The results of cell culture and animal studies indicate that omega-6 and omega-3 fatty acids can modulate the expression of a number of genes, including those involved with fatty acid metabolism and inflammation (31, 32). Omega-6 and omega-3 fatty acids regulate gene expression by interacting with specific transcription factors, such as peroxisome proliferator-activated receptors (PPARs) (33). In many cases, PUFA act like hydrophobic hormones (e.g., steroid hormones) to control gene expression and bind directly to receptors like PPARs. These ligand-activated receptors then bind to the promoters of genes and function to increase/decrease transcription.
In other cases, PUFA regulate the abundance of transcription factors inside the cell's nucleus (14). Two examples include NFκB and SREBP-1. NFκB is a transcription factor involved in regulating the expression of multiple genes involved in inflammation. Omega-3 PUFA suppress NFκB nuclear content, thus inhibiting the production of inflammatory eicosanoids and cytokines. SREBP-1 is a major transcription factor controlling fatty acid synthesis, both de novo lipogenesis and PUFA synthesis. Dietary PUFA can suppress SREBP-1, which decreases the expression of enzymes involved in fatty acid synthesis and PUFA synthesis. In this way, dietary PUFA function as feedback inhibitors of all fatty acid synthesis.
By altering cell membrane fluidity, fatty acids can interfere with the activity of membrane receptor systems and thus indirectly influence signaling pathways and gene expression (34).
Deficiency
Essential fatty acid deficiency
Clinical signs of essential fatty acid deficiency include a dry scaly rash, decreased growth in infants and children, increased susceptibility to infection, and poor wound healing (35). Omega-3, omega-6, and omega-9 fatty acids compete for the same desaturase enzymes. The desaturase enzymes show preference for the different series of fatty acids in the following order: omega-3 > omega-6 > omega-9. Consequently, synthesis of the omega-9 fatty acid eicosatrienoic acid (20:3n-9, mead acid, or 5,8,11-eicosatrienoic acid) increases only when dietary intakes of omega-3 and omega-6 fatty acids are very low; therefore, mead acid is one marker of essential fatty acid deficiency (36). A plasma eicosatrienoic acid:arachidonic acid (triene:tetraene) ratio greater than 0.2 is generally considered indicative of essential fatty acid deficiency (35, 37). In patients who were given total parenteral nutrition containing fat-free, glucose-amino acid mixtures, biochemical signs of essential fatty acid deficiency developed in as little as 7 to 10 days (38). In these cases, the continuous glucose infusion resulted in high circulating insulin concentrations, which inhibited the release of essential fatty acids stored in adipose tissue. When glucose-free amino acid solutions were used, parenteral nutrition up to 14 days did not result in biochemical signs of essential fatty acid deficiency. Essential fatty acid deficiency has also been found to occur in patients with chronic fat malabsorption (39) and in patients with cystic fibrosis (40). It has been proposed that essential fatty acid deficiency may play a role in the pathology of protein-energy malnutrition (36).
Omega-3 fatty acid deficiency
At least one case of isolated omega-3 fatty acid deficiency has been reported. A young girl who received intravenous lipid emulsions with very little ALA developed visual problems and sensory neuropathy; these conditions were resolved when she was administered an emulsion containing more ALA (41). Isolated omega-3 fatty acid deficiency does not result in increased plasma triene:tetraene ratios, and skin atrophy and dermatitis are absent (1). Plasma DHA concentrations decrease when omega-3 fatty acid intake is insufficient, but no accepted plasma omega-3 fatty acid or eicosanoid concentrations indicative of impaired health status have been defined (1). Studies in rodents have revealed significant impairment of n-3 PUFA deficiency on learning and memory (42, 43), prompting research in humans to assess the impact of omega-3 PUFA on cognitive development and cognitive decline (see Cognitive and visual development and Alzheimer's disease).
Omega-3 index
The omega-3 index is defined as the amount of EPA plus DHA in red blood cell membranes expressed as the percent of total red blood cell membrane fatty acids (44). The EPA + DHA content of red blood cell membranes correlates with that of cardiac muscle cells (45, 46), and several observational studies indicate that a lower omega-3 index is associated with an increased risk of coronary heart disease mortality (47). It is therefore proposed that the omega-3 index be used as a biomarker for cardiovascular disease risk, with suggested cutoffs as follows: high risk, <4%; intermediate risk, 4%-8%; and low risk, >8% (48).
Supplementation with EPA + DHA from fish oil capsules for approximately five months dose-dependently increased the omega-3 index in 115 healthy, young adults (ages, 20-45 years), validating the use of the omega-3 index as a biomarker of EPA + DHA intake (49). Before the omega-3 index can be used in routine clinical evaluation, however, clinical reference values in the population must be established (50). Additionally, fatty acid metabolism may be altered in certain disease states, potentially making the omega-3 index less relevant for some cardiovascular conditions (5).
Disease Prevention
Pregnancy and early childhood developmental outcomes
Supplementation during pregnancy
Effect on pregnancy-associated conditions and neonatal outcomes: The results of randomized controlled trials during pregnancy suggest that omega-3 polyunsaturated fatty acid (PUFA) supplementation does not decrease the incidence of gestational diabetes and preeclampsia (51-54) but may result in modest increases in length of gestation, especially in women with low omega-3 fatty acid consumption. A 2006 meta-analysis of six randomized controlled trials in women with low-risk pregnancies found that omega-3 PUFA supplementation during pregnancy resulted in an increased length of pregnancy by 1.6 days (55). A 2007 meta-analysis of randomized controlled trials in women with high-risk pregnancies found that supplementation with long-chain PUFA did not affect pregnancy duration or the overall incidence of premature births (birth before 37 weeks' gestation) but decreased the incidence of early premature births (birth before 34 weeks' gestation; 2 trials, 291 participants) (56). Analyses of the secondary outcomes of the 2010 DHA to Optimize Mother-Infant Outcome (DOMInO) trial in 2,399 participants showed that supplementation with DHA-enriched fish oil capsules (800 mg/day of DHA and 100 mg/day of EPA) during pregnancy (from <21 gestational weeks until birth) reduced the risk of early premature birth but increased the risk of obstetrical interventions like the need for induction or cesarean section, when compared to supplementation with DHA-free vegetable oil capsules (57). A 2016 meta-analysis of trials found evidence to suggest that omega-3 PUFA supplementation during pregnancy reduced the overall risk of prematurity and the risk of early premature births, increased gestational age at delivery and birth weight, and had no effect on the risks of perinatal death and low Apgar scores at 1 minute post birth (58). A dose-response analysis found a continuous reduction of the risks of early premature birth (birth before 34 weeks' gestation) and very low birth weight (birth weight <1,500 g) with daily doses of DHA supplement up to at least 600 mg during pregnancy (59). There is currently limited evidence to support a role for omega-3 supplementation in the prevention of recurrent intrauterine growth restriction (IUGR) (60) or recurrent preterm birth (61).
Effect on children's cognitive and visual development: The effect of maternal omega-3 long-chain PUFA supplementation on early childhood cognitive and visual development was summarized in a 2013 systematic review and meta-analysis (62). Included in this assessment were 11 randomized controlled trials (a total of 5,272 participants) that supplemented maternal diet with omega-3 long-chain PUFA during pregnancy or both pregnancy and lactation. Results regarding visual outcomes (eight trials) could not be pooled together due to variability in assessments; overall, four of six trials had null findings and the remaining two trials had very high rates of attrition. Cognitive outcomes (nine trials) included the Developmental Standard Score (DSS; in infants, toddlers, and preschoolers) or Intelligence Quotient (IQ; in children) and other aspects of neurodevelopment, such as language, behavior, and motor function. No differences were found between DHA and control groups for cognition measured with standardized psychometric scales in infants (<12 months), toddlers (12-24 months), and school-aged children (5-12 years); preschool children (2-5 years) in the DHA treatment group had a substantially higher DSS score compared to controls. The authors noted that many of the trials of long-chain PUFA supplementation in pregnancy had methodological weaknesses (e.g., high rates of attrition, small sample sizes, high risk of bias, multiple comparisons), limiting the confidence and interpretation of the pooled results. Of note, a seven-year follow-up of the DOMInO trial is currently underway to assess the effect of DHA supplementation during pregnancy on child IQ and various measures of cognitive development (e.g., executive functioning, memory, language) (63).
Effect on children’s body composition: The follow-up of 1,531 children whose mothers were randomized to supplemental DHA (800 mg/day) or a control during the second half of pregnancy in the DOMInO trial showed no effect of maternal DHA supplementation on the body mass index (BMI)-for-age z score and percentage of body fat of their children at three and five years of age (64). Measures of insulin resistance in 5-year-old children were unexpectedly higher in children whose mothers were in the DHA group than in those whose mothers were in the control group (64). Further analyses conducted in a subset of children (252) at age seven years again showed no effect of DHA supplementation on BMI z score, percentage of body fat, height, weight, and waist/hip circumference (65). Current evidence from 10 randomized controlled trials primarily conducted in high-income countries (all but one) suggests no influence of maternal supplementation with long-chain PUFA on the body composition and anthropometry of the offspring (66).
Effect on children's risk of allergies and asthma: A 2018 meta-analysis of randomized controlled trials in 2,047 children followed for six months to 16 years found a 19% lower risk of wheezing and/or asthma with maternal supplementation of omega-3 PUFA (primarily EPA and DHA) from as early as the 20th week of gestation until delivery (67). However, there was no effect of prenatal supplementation when the analysis was restricted to the three trials that reported on the incidence of childhood asthma only (67). Another meta-analysis of nine trials in 3,637 children, including three trials in which maternal supplementation with omega-3 PUFA continued after birth, found no effect of prenatal supplements on the risk of any allergy (three trials), the risk of wheeze and/or asthma (seven trials), the risk of eczema (six trials), the development of allergic rhinitis (two trials), and the risk of food allergy (three trials) in children (68). There was, however, some evidence to suggest that prenatal supplementation could lower the incidence of sensitization to specific allergens, namely egg (three trials; -46%) and peanut (two trials; -38%) (68).
Supplementation to breast-feeding mothers
A 2015 systematic review and meta-analysis summarized the results of eight randomized controlled trials that examined the effect of maternal supplementation with long-chain PUFA during either pregnancy and lactation or lactation only on the development and growth of their infants over the first two years of life and beyond (69). All studies were conducted in high-income countries. No differences between long-chain PUFA supplementation and control were observed in terms of language development, intelligence or problem-solving ability, psychomotor development, and anthropometric measurements (weight, length/height, head circumference, BMI, fat mass distribution) (69).
Supplementation in infants
The last trimester of pregnancy and first six months of postnatal life are critical periods for the accumulation of DHA in the brain and retina (70). Human milk contains a mixture of saturated fatty acids (~46%), monounsaturated fatty acids (~41%), omega-6 PUFA (~12%), and omega-3 PUFA (~1.3%) (71). Although human milk contains DHA in addition to ALA and EPA, ALA was the only omega-3 fatty acid present in conventional infant formulas until the year 2001. Although infants can synthesize DHA from ALA, they generally cannot synthesize enough to prevent declines in plasma and cellular DHA concentrations without additional dietary intake. Therefore, it was proposed that infant formulas be supplemented with enough DHA to bring plasma and cellular DHA concentrations of formula-fed infants up to those of breast-fed infants (72).
All infants: Although formulas enriched with DHA raise plasma and red blood cell DHA concentrations in preterm and term infants, the results of randomized controlled trials examining measures of visual acuity and neurological development in infants fed formula with or without added DHA have been mixed. For instance, a 2012 meta-analysis of randomized controlled trials (12 trials, 1,902 infants) comparing long-chain PUFA-supplemented and unsupplemented formula, started within one month of birth, found no effect of long-chain PUFA supplementation on infant cognition assessed at approximately one year of age (73). A lack of effect was observed regardless of the dose of long-chain PUFA or the prematurity status of the infant. With respect to visual acuity, a 2013 meta-analysis of randomized controlled trials (19 trials, 1,949 infants) found a beneficial effect of long-chain PUFA-supplemented formula, started within one month of birth, on infant visual acuity up to 12 months of age (74). Notably, two different types of visual acuity assessment were evaluated in the meta-analysis. Visual acuity assessed by using the Visually Evoked Potential (10 trials, 852 infants) showed a significant positive effect of long-chain PUFA-supplemented formula at 2, 4, and 12 months of age. When assessed by the Behavioral Method (12 trials, 1,095 infants), a significant benefit of long-chain PUFA-supplemented formula on visual acuity was found only at the age of two months. No moderating effects of dose or prematurity status were observed.
Preterm infants: A few trials have been specifically conducted in preterm infants. This is the case of the DHA for the Improvement of Neurodevelopmental Outcome (DINO) trial that initially enrolled 657 very preterm infants (born <33 gestational weeks) in five Australian hospitals (75). The aim of the trial was to examine the effect of enteral feeds with either high DHA (1% of total fatty acids) or standard DHA level (0.3% of total fatty acids) to preterm infants from age 2 to 4 days of life until term's corrected age (mean duration, 9.4 weeks) on their mental and psychomotor development, assessed at 18 months' and 7 years' corrected ages. At the 18-month follow-up, there was no difference in mean Mental Development Index (MDI) and Psychomotor Development Index (PDI) test scores between high-DHA and standard-DHA groups; yet, better MDI scores in girls fed high-DHA versus those fed standard-DHA feeds were reported in subgroup analyses (75). Post-hoc analyses also suggested fewer cases with delayed mental development among girls and infants weighing <1,250 kg at birth in the high- versus standard-DHA group (75). Follow-up at 7 years’ corrected age showed no difference between groups in measures of IQ and cognitive development, including attention, short-term verbal memory and learning ability, executive functioning, visual perception, and academic achievement (76). A 2016 systematic review of 17 trials found little evidence to suggest that supplementing preterm infants with long-chain PUFA (primarily AA and DHA) improved measures of visual acuity, neurodevelopment, and physical growth during infancy (77).
Cardiovascular disease
Omega-6 fatty acids
Linoleic acid (LA) is the most abundant dietary PUFA and accounts for approximately 90% of dietary omega-6 PUFA intake (78).
Observational studies: A pooled analysis of 13 prospective cohort studies, encompassing 310,602 individuals and 12,479 coronary heart disease (CHD) events (of which resulted in 5,882 CHD deaths) over follow-up periods of 5.3 to 30 years, found higher LA intakes to be associated with a 15% lower risk of CHD events and a 21% lower risk of CHD mortality (79). A dose-response analysis found that replacing 5% of energy from saturated fatty acids with LA was associated with a 9% lower risk of coronary events and a 13% lower risk of coronary deaths (79). A 2019 meta-analysis of 30 prospective cohort studies in 68,659 participants found that individuals in the highest versus lowest quintile of LA concentrations in tissues (primarily blood compartments) had a 23% lower risk of cardiovascular mortality (80). No associations were found between LA concentrations in tissues and the risks of CHD, ischemic stroke, or total cardiovascular disease (80).
Randomized controlled trials: Taking into consideration the results from four randomized controlled trials (81-85) that compared the effects of diets either high in saturated fatty acids or PUFA over at least two years, a 2016 systematic review and presidential advisory from the American Heart Association concluded that lowering saturated fat intake and replacing it with vegetable oil rich in PUFA (primarily soybean oil) could reduce the risk of CHD by 29% (86). Of note, these trials were conducted in the 1960s and 1970s, when the use of cholesterol-lowering drug statin was not widespread and the saturated fat content in diets was higher; all but one trial (84, 85) were in men with diagnosed cardiovascular disease (CVD). Among these four trials, the Oslo Diet-Heart Study (83) increased both omega-3 and omega-6 PUFA intake, and the Finnish Mental Hospital Study (84, 85) used a cross-over design — both trials were excluded from a Cochrane systematic review of 19 randomized controlled trials that examined the effect of increasing omega-6 PUFA intake on CVD outcomes (87). Of these 19 trials, seven assessed the effect of supplemental γ-linolenic acid (GLA) and 12 assessed the effect of substituting dietary LA for saturated or monounsaturated fatty acids. The pooled analysis of studies showed no effect of increasing omega-6 intake on the risks of CHD or CVD events, major adverse cardiac and cerebrovascular events, myocardial infarction (MI), stroke, CVD mortality, or all-cause mortality (low-quality evidence) (87). Moreover, many trials that examined the effect of replacing saturated fatty acids with mostly omega-6 PUFA may not have been adequately controlled. For example, in some trials, only the experimental group (the high omega-6 PUFA group) received dietary advice regarding more than just replacing saturated fatty acids by omega-3 PUFA, e.g., to avoid dietary sources of trans fatty acids and processed foods, to consume more whole-plant foods, to lower sugar consumption, to increase consumption of fish and shellfish, which could have biased the results (88). Additionally, a recent meta-analysis of trials with low risk of bias (i.e., free of differences between intervention and control groups other than those under examination) showed no evidence of an effect of substituting omega-6 PUFA for saturated fatty acids on the risks of major CHD events (MI and sudden death), total CHD events, CHD mortality, and all-cause mortality (88).
Yet, replacing dietary saturated fatty acids with omega-6 PUFA was consistently found to lower total blood cholesterol concentrations (87, 89). In fact, LA has been shown to be the most potent fatty acid for lowering total cholesterol when substituted for dietary saturated fatty acids (90). The potential mechanisms by which LA reduces blood cholesterol include (1) the upregulation of LDL receptor and redistribution of LDL-cholesterol from plasma to tissue, (2) the increase in bile acid production and cholesterol catabolism, and (3) the decreased VLDL-to-LDL conversion (91). However, if substituting omega-6 PUFA for saturated fatty acids can reduce blood cholesterol, the most recent systematic reviews and meta-analyses have failed to find evidence of clinical cardiovascular benefits (see above) (87, 88, 92).
Omega-3 fatty acids
Observational studies: A meta-analysis of 17 prospective and two retrospective cohort studies in 45,637 generally healthy participants found that circulating concentrations of α-linolenic acid (ALA) and longer chain omega-3 PUFA (i.e., eicosapentaenoic acid [EPA], docosapentaenoic acid [DPA], docosahexaenoic acid [DHA]) were inversely associated with the risk of fatal coronary heart disease (CHD) (93).
Several observational studies also examined the relationship between dietary ALA intake and the risk of CHD. A 2018 meta-analysis of 14 prospective cohort studies in a total of 345,202 participants free of cardiovascular disease (CVD) evaluated the risk of composite CHD outcomes (combining different CHD events) and fatal CHD in relation to dietary consumption of ALA (94). Overall, the pooled analysis found a 9% lower risk of composite CHD outcomes and a 15% lower risk of fatal CHD with higher ALA exposure (94). Further, a number of prospective cohort studies have examined the consumption of fish, rich in long-chain omega-3 PUFA (mainly EPA and DHA), in relation to various cardiovascular events and mortality. A 2018 review of the evidence and advisory from the American Heart Association concluded that seafood intake was associated with modestly lower risks of CHD, ischemic stroke, and sudden cardiac death, and noted a greater benefit when intake went from zero to one or two seafood meals per week and when seafood was substituted for less healthy options like processed meat (95). In contrast, recently published meta-analyses of prospective cohort studies found little evidence of inverse associations between fish consumption and either CHD or stroke (96, 97). Higher fish consumption was found to be associated with lower risks of myocardial infarction (MI) (98) and congestive heart failure (96). In addition, one meta-analysis of 12 prospective cohort studies found a 6% lower risk of all-cause mortality with the highest versus lowest level of fish consumption (99). Yet, another meta-analysis found no association between fish intake and all-cause mortality but a 4% lower risk of CVD mortality for each 20-g/day increment in fish intake (100).
The potential cardiovascular benefit of seafood consumption appears to be tightly linked to the type of seafood (e.g., fatty or lean fish), the way it is prepared (e.g., baked, broiled, or fried), the presence of toxic metals and environmental contaminants, and the habitual level of consumption (high versus low) — these factors may be confounding the results reported in observational studies and pooled analyses (95). Although seafood is a good source of long-chain omega-3 PUFA, health benefits associated with fish consumption could be attributed to the presence of other nutritional factors (e.g., micronutrients and high-quality protein) and that seafood consumption is usually a marker of higher socioeconomic status, as well as healthy lifestyles (101, 102).
Randomized controlled trials: A 2018 Cochrane systematic review assessed the evidence for a cardioprotective effect of ALA and long-chain omega-3 PUFA in individuals either at low or high risk of CVD (103). Moderate-to-high quality evidence from randomized controlled trials (of at least 12 months) suggested no effect of omega-3 PUFA (either supplemented, enriched in meals, or advised to be consumed) on the risk of CHD events, CVD events, arrhythmia, stroke, CHD mortality, CVD mortality, or all-cause mortality. There was also no evidence of an effect on secondary outcomes, including major adverse cerebrovascular or cardiovascular events, MI, sudden cardiac death, angina pectoris, heart failure, revascularization, peripheral arterial disease, and acute coronary syndrome (103). A 2017 review and advisory from the American Heart Association found no evidence to suggest a benefit of long-chain omega-3 PUFA supplementation for the prevention of cardiovascular mortality in patients with or at risk of type 2 diabetes mellitus, the prevention of CHD in patients with atherosclerotic disease (e.g., with prior stroke, peripheral vascular disease, diabetes, hypercholesterolemia), the prevention of stroke in patients with or without a history of stroke, and the prevention of atrial fibrillation in patients with prior atrial fibrillation or in those undergoing cardiac surgery (104). There was some evidence to suggest that supplementation with long-chain omega-3 PUFA in patients with prior clinical CHD might reduce the risk of CHD death, possibly because of a reduction in the risk of ischemia-induced sudden cardiac death (104).
Hypertriglyceridemia (borderline high: serum triglycerides 150-199 mg/dL; high: serum triglycerides >200 mg/dL) is an independent risk factor for cardiovascular disease (105). Numerous controlled clinical trials have demonstrated that increasing intakes of EPA and DHA significantly lower serum triglyceride concentrations (103). The triglyceride-lowering effects of EPA and DHA increase with dose (106), but clinically meaningful reductions in serum triglyceride concentrations have been demonstrated at doses of 2 g/day of EPA + DHA (107). Although long-chain omega-3 PUFA can reduce triglyceride concentrations, they have no effect on total cholesterol, LDL-cholesterol, or HDL-cholesterol in blood (103). Of note, the mechanisms by which long-chain omega-3 PUFA supplements may reduce CHD death are unlikely to involve a lowering of triglycerides as doses used in the studies (~1 g/day) were generally too low (104). Some studies in cell culture indicated that long-chain omega-3 PUFA may decrease the excitability of cardiac muscle cells (myocytes) by modulating ion channel conductance, which would be consistent with anti-arrhythmic effects observed in animal models (see also Hypertriglyceridemia) (108, 109).
Summary
Replacing dietary saturated fatty acids with omega-6 PUFA lowers total blood cholesterol, yet there is no convincing evidence of an effect of omega-6 PUFA on the risk of major CVD events. Although evidence supports the adoption of a heart-healthy dietary pattern that includes two servings of seafood per week (95), supplementation with long-chain omega-3 fatty acids is unlikely to result in cardiovascular benefits in generally healthy people with a low CVD risk or in individuals at risk of or with type 2 diabetes mellitus (104). In its recommendations regarding omega-3 fatty acids and cardiovascular disease (see Intake Recommendations), the American Heart Association indicates that long-chain omega-3 PUFA supplementation may be useful to reduce mortality in patients with prevalent CHD (e.g., who suffered a recent MI) and in those with heart failure without preserved ventricular function (104).
Cardiometabolic risk factors in individuals with diabetes mellitus
Type 2 diabetes mellitus: Cardiovascular disease is the leading cause of death in individuals with diabetes mellitus. The dyslipidemia typically associated with diabetes is characterized by a combination of hypertriglyceridemia (serum triglycerides >200 mg/dL), low HDL-cholesterol, and abnormal LDL-cholesterol (110). Lipid-lowering therapy to normalize diabetic dyslipidemia and reduce cardiovascular risk includes lifestyle modification and medications — particularly the use of cholesterol-lowering statins (111, 112). Additionally, achieving glucose control in people with type 2 diabetes has been shown to decrease the occurrence of major microvascular and macrovascular events (113).
A 2014 meta-analysis of 19 randomized controlled trials, including 24,788 individuals with either impaired glucose metabolism or type 2 diabetes mellitus, found that long-chain omega-3 PUFA supplementation (doses, 360-10,000 mg/day; for 6 weeks to 6 years) lowered serum triglyceride concentrations by 0.25 mmol/L but had no substantial effect on total cholesterol, LDL-cholesterol, or HDL-cholesterol (114). There was also no significant effect on HbA1c, fasting glucose, blood pressure, heart rate, or a measure of endothelial function. Four trials that lasted over a year reported on cardiovascular outcomes, including mortality. The pooled analysis of these trials found no effect of supplementation with omega-3 PUFA on the risk of major cardiovascular events, cardiovascular mortality, all-cause mortality, or a composite endpoint of all-cause mortality and hospitalization for a cardiovascular cause. It is worth noting that two of these trials — the Alpha Omega Trial (115) and the ORIGIN trial (116) — included a high proportion of participants who took cardiovascular medications (i.e., cholesterol-lowering statins) (114). Another meta-analysis of 45 randomized controlled trials in 2,674 participants with type 2 diabetes found that supplementation with omega-3 (400-1,800 mg/day for 2 weeks to 2 years) led to small decreases in blood concentrations of triglycerides, VLDL-triglycerides, LDL-cholesterol, and vLDL-cholesterol (117). There was no evidence of an effect on total cholesterol, HDL-cholesterol, non-esterified fatty acids, apolipoprotein-A1, and apolipoprotein-B. There was a reduction in circulating concentrations of pro-inflammatory cytokines, TNF-α and IL-6, in response to omega-3 supplementation, yet not of C-reactive protein (CRP) — a marker of low-grade inflammation. Omega-3 PUFA supplementation had no effect on systolic or diastolic blood pressure. Finally, a small decrease in HbA1c was reported in response to supplemental omega-3 fatty acids, yet there was no effect on other indicators of glycemic control, especially fasting glucose, fasting insulin, connecting (C-) peptide, and a measure of insulin resistance (117).
Lifestyle changes involving dietary modifications, such as the substitution of healthy fats (mono- and poly-unsaturated fatty acids) for saturated and trans fats, are recommended to reduce the risk of cardiovascular disease in people with type 2 diabetes mellitus (118). In their most recent updated recommendations on the prevention of cardiovascular disease in adults with type 2 diabetes, the American Diabetes Association and American Heart Association found insufficient evidence from large-scale randomized trials in individuals with type 2 diabetes to support the use of omega-3 fatty acid supplements (combined with a heart-healthy diet) in the prevention of cardiovascular events (118).
Gestational diabetes: Poor glycemic control during pregnancy, whether due to type 1 diabetes, type 2 diabetes, or gestational diabetes, increases the risk of fetal anomalies, preeclampsia, spontaneous abortion, stillbirth, macrosomia, neonatal hypoglycemia, and neonatal hyperbilirubinemia (119). Diabetes during pregnancy is also associated with a higher risk of metabolic disorders in offspring later in life (119). A team of investigators in Iran examined the effect of omega-3 PUFA supplementation during pregnancy, beginning at 24 to 28 weeks' gestation for six weeks, in women with gestational diabetes. Overall, there was evidence of beneficial effects of 1,000 mg/day of omega-3 alone (120) or together with vitamin E (121) or vitamin D (122) on markers of glucose homeostasis and, to a lesser extent, on markers of oxidative stress and inflammation and blood lipid profile. In one randomized, placebo-controlled trial in 60 women with gestational diabetes, supplementation with omega-3 fatty acids and vitamin E reduced the risk of neonatal hyperbilirubinemia yet had no effect on the rate of cesarean section, need for insulin therapy, maternal hospitalization, newborns' hospitalization, gestational age, birth size, and Apgar score (122).
Current recommendations by the American Diabetes Association for the management of gestational diabetes encourage the development of an individualized nutrition plan between a woman and a registered dietitian, highlighting the importance of the amount and type of carbohydrates in the diet (119). The use of omega-3 supplements in the management of gestational diabetes is not currently under consideration.
Type 2 diabetes mellitus
A meta-analysis of 13 randomized, controlled feeding trials that substituted plant-derived PUFA (primarily linoleic acid [LA]) for saturated fatty acids or carbohydrates for 3 to 16 weeks in generally healthy adults showed a decrease in fasting insulin concentration and insulin resistance but no effect on fasting glucose concentration (123). Most studies used a mixture of omega-3 and omega-6 PUFA in the form of plant-derived oils such that potential differences in effect between them could not be examined.
A meta-analysis of 20 prospective cohort studies conducted in 10 countries, in a total of 39,740 participants free from diabetes at baseline, examined biomarkers of omega-6 intake in relation to the risk of developing type 2 diabetes mellitus (124). LA ranged from 8.3% of total fatty acids in erythrocyte phospholipids to 54.5% in plasma cholesterol esters. The lowest percentage of arachidonic acid (AA) was found in adipose tissue (0.3%) and the highest in erythrocyte phospholipids (17.0%). The highest versus lowest concentration of LA markers in each compartment (phospholipids, plasma or serum, cholesterol esters) except adipose tissue was associated with a 35% lower risk of type 2 diabetes. In contrast, only AA in plasma or serum was inversely associated with the risk of type 2 diabetes (124). If LA concentration in blood and adipose tissue can provide an objective assessment of dietary LA intake (125), these results suggest that dietary LA may be important for glycemic control and diabetes prevention.
Metabolic syndrome
A 2019 meta-analysis of 13 observational (9 cross-sectional, 2 case-control, 1 nested case-control, and 1 prospective cohort; 36,542 participants) studies showed higher concentrations of omega-3 in blood and adipose tissue and higher level of omega-3 intake to be associated with a lower risk of metabolic syndrome (126). No association was found between tissue omega-6 concentration or dietary omega-6 intake level and the risk of metabolic syndrome (126).
Cognitive decline and Alzheimer's disease
Alzheimer’s disease is the most common cause of dementia in older adults (127). Alzheimer's disease is characterized by the formation of amyloid plaque in the brain and nerve cell degeneration. Disease symptoms, including memory loss and confusion, worsen over time (128).
Observational studies: Several observational studies have examined dietary fish and PUFA consumption in relation to risks of cognitive decline, dementia, and Alzheimer's disease. The pooled analysis of five large prospective cohort studies (Three-City Study, Nurses' Health Study, Women's Health Study, Chicago Health and Aging Project, and Rush Memory and Aging Project) that followed a total of 23,688 older (ages, ≥65 years) participants (88% women) for 3.9 to 9.1 years found slower rates of decline in episodic memory and global cognition with increasing fish intakes (129). Previous studies have suggested that the effect of fish or PUFA consumption on cognition may be dependent on apolipoprotein E (APOE) genotype (130, 131). Of three common APOE alleles (epsilon 2 [ε2], ε3, and ε4), the presence of the APOE ε4 (E4) allele has been associated with increased risk and earlier onset of Alzheimer's disease (132). It was found that long-chain omega-3 PUFA supplementation did not increase plasma omega-3 concentrations to the same extent in E4 carriers than in non-carriers (133) and that DHA metabolism differs in E4 carriers compared to non-carriers, with greater oxidation and lower plasma concentrations in E4 carriers (134). However, neither APOE genotype nor polymorphisms in 11 other genes associated with Alzheimer's disease were found to modify the inverse relationship between fish intake and risk of cognitive decline in the pooled analysis of the five cohorts (129).
In a recent meta-analysis of observational studies, each one-serving increase of fish intake per week was found to be associated with a 5% lower risk of dementia and a 7% lower risk of Alzheimer's disease (135). Dietary intake level of marine-derived DHA — but not blood DHA concentration — was also inversely associated with the risks of dementia and Alzheimer's disease; for instance, a 100 mg/day increment in dietary DHA intake was associated with lower risks of dementia (-14%) and Alzheimer's disease (-37%) (135). Results from two large cohort studies published after this dose-response meta-analysis showed blood DHA concentration to be positively associated with cognitive performance in adults (136, 137). Findings from preclinical studies suggest that long-chain omega-3 fatty acids may have neuroprotective effects, potentially through mitigating neuroinflammation, improving cerebral blood flow, and/or reducing amyloid aggregation (138).
Randomized controlled trials: A 2012 systematic review identified three randomized controlled trials that examined the effect of omega-3 supplementation on the risk of cognitive decline in cognitively healthy older or elderly adults (139). There was no evidence showing an effect of omega-3 on measures of cognitive functions in these clinical trials. In a more recent systematic review that identified seven trials conducted in cognitively healthy participants, the authors reported positive effects of long-chain omega-3 supplementation on measures of cognitive outcomes in all studies but the second longest and the two largest trials (140). Another seven trials examined the effect of long-chain omega-3 supplementation in individuals with mild cognitive impairment; all but three trials showed a significant benefit on measures of cognitive function or specific memory tasks (140). Yet, two trials that found no improvement in cognitive performance included omega-3 supplements in both intervention and control arms (141, 142).
Overall, the data favor a role for diets rich in long-chain omega-3 fatty acids in slowing cognitive decline, but larger trials with longer intervention periods may be necessary to see a consistent beneficial effect of omega-3 supplementation in older individuals with normal or declining cognitive functions.
Disease Treatment
Hypertriglyceridemia
About one-third of US adults have serum triglycerides >150 mg/dL, and 16% of US adults have serum triglycerides >200 mg/dL (143). The 2011 American Heart Association guidelines on triglyceride management recommended the use of marine-derived omega-3 fatty acid supplements (2-4 g/day of EPA plus DHA) under medical supervision to reduce triglyceride concentrations below 100 mg/dL (143). Hypertriglyceridemia can have various causes, such as inherited and acquired disorders of triglyceride metabolism, poor diet, and/or use of certain medications (143).
Several omega-3 fatty acid preparations have been approved by the US Food and Drug Administration for the treatment of hypertriglyceridemia (104). Out of the five currently available preparations, four contain ethyl esters of EPA and/or DHA and one contains long-chain omega-3 PUFA as free fatty acids (104). The Epanova for lowering very high triglycerides (EVOLVE) randomized controlled trial demonstrated that the omega-3 free fatty acid formulation (2-4 g/day for 12 weeks) effectively reduced triglycerides and other atherogenic factors, including vLDL-cholesterol and remnant-like cholesterol particles, when compared to olive oil (4 g/day) in patients with severe hypertriglyceridemia (serum triglycerides >500 mg/dL) (reviewed in 144). Omega-3 supplementation also decreased inflammation (as shown by a reduction in lipoprotein-associated phospholipase A2) and platelet activation (as shown by a reduction in circulating concentrations of arachidonic acid) (144, 145). This omega-3 formulation also proved to be effective in reducing persistent hypertriglyceridemia (serum triglycerides, 200-499 mg/dL) in patients treated with statins (cholesterol-lowering drugs) (146). Statin use has been found to effectively reduce triglyceride concentrations by about 5%-20% (147). However, a residual elevation in triglycerides and triglyceride-rich lipoprotein cholesterol may remain in a substantial fraction of patients treated with statins. Compared to 4 g/day of olive oil, omega-3 supplementation with 2 or 4 g/day for six weeks reduced triglycerides by 14.6% and 20.6% and non-HDL-cholesterol by 3.9% and 6.9%, respectively (146). The magnitude of these reductions in triglyceride and non-HDL-cholesterol concentrations was similar to what has been observed in other trials that examined the use of ethyl ester omega-3 supplements as add-ons to statin therapy (146, 148-150). A study is underway to assess the benefit of combining omega-3 fatty acids and statins on the risk of major cardiovascular events over a three- to five-year period in patients with hypertriglyceridemia (144, 151).
Nonalcoholic fatty liver disease
Often associated with metabolic disorders, nonalcoholic fatty liver disease (NAFLD) is a condition characterized by an excessive lipid accumulation in the liver (i.e., hepatosteatosis). NAFLD can progress to nonalcoholic steatohepatitis (NASH) in about one-third of the patients with NAFLD, thereby increasing the risk of cirrhosis and hepatocellular carcinoma (152, 153). An emerging feature of NAFLD is the decline in hepatic omega-3 and omega-6 PUFA with disease progression (154). Considering that C20-22 omega-3 PUFA can reduce fatty acid synthesis and inflammation, a possible therapeutic strategy would be to increase dietary intake of long-chain omega-3 PUFA. A 2018 meta-analysis of 18 randomized controlled trials in 1,424 participants with NAFLD found that omega-3 supplementation showed beneficial effects on liver fat, specific liver enzymatic activities, serum triglycerides, fasting glucose, and insulin resistance (155). However, there was no evidence of an effect on total cholesterol, LDL-cholesterol, HDL-cholesterol, fasting insulin, blood pressure, BMI, and waist circumference (155). Other recent meta-analyses have also reported that supplementation with long-chain omega-3 fatty acids from fish/seal oil (0.25-6.8 g/day for 3-25 months) improved hepatosteatosis and other metabolic disorders in both children and adults with NAFLD (reviewed in 153). Additional studies are needed to examine their efficacy in more severe cases of NASH.
Inflammatory diseases
Rheumatoid arthritis
A 2017 meta-analysis of 20 randomized controlled trials in 1,252 participants with rheumatoid arthritis assessed the efficacy of long-chain omega-3 PUFA supplementation on a series of clinical outcomes (156). Omega-3 supplementation (0.3-9.6 g/day) for 3 to 18 months reduced the number of tender joints (14 trials), as well as early morning stiffness (15 trials) and pain level (16 trials) compared to placebo. Blood concentrations of triglycerides (3 trials) and pro-inflammatory leukotriene B4 (5 trials) were also decreased with supplemental omega-3 PUFA (156). Another 2017 meta-analysis of 42 randomized controlled trials examined the effect of omega-3 supplementation (mainly as fish oil) on arthritic pain in patients diagnosed with different types of arthritis (157). Daily administration of marine-derived EPA (0.01-4.1 g) and DHA (0.01-2.7 g) for up to 18 months resulted in a reduction in patients’ reported pain (using a visual analog scale [VAS] for pain) in those suffering from rheumatoid arthritis (22 trials) and those with other types of arthritis (i.e., juvenile arthritis, psoriatic arthritis) or mixed diagnoses (3 trials), yet not in those with osteoarthritis (5 trials). The evidence of an effect of omega-3 supplements in patients with rheumatoid arthritis was deemed of moderate quality (157). In a 2017 systematic review of 18 trials, including 1,143 subjects with rheumatoid arthritis, only 4 of 18 placebo-controlled trials showed a benefit of omega-3 PUFA supplementation (2.2-3.6 g/day for 12-36 weeks) on pain level — reported by patients and/or assessed by physicians (158). In most trials, the use of medications (nonsteroidal anti-inflammatory drugs [NSAIDs] and/or disease-modifying anti-rheumatic drugs [DMARDs]) was continued throughout the intervention period. Results of a few trials suggested that omega-3 PUFA could spare the need for anti-inflammatory medications in some patients yet failed to show superiority of PUFA in pain management (159, 160).
The limited body of evidence that suggests potential benefits of omega-3 supplementation in rheumatoid arthritis treatment needs strengthening with data from larger studies conducted for longer intervention periods (157, 158).
Inflammatory bowel disease
Crohn's disease: A 2013 systematic review evaluated the efficacy of omega-3 supplementation in patients with Crohn's disease, considering the evidence base from both short-term (9 to 24 weeks) and long-term (1 year) trials (161). Among five trials that evaluated the efficacy of omega-3 supplementation on relapse rates, conflicting outcomes were reported. Most trials were limited by small sample sizes and short duration — up to three years may be necessary to see an effect on relapse rates given the natural relapsing-remitting course of the disease. The two largest and most recent trials (EPIC-1 and EPIC-2) showed no significant effect of omega-3 supplementation on indicators of Crohn's disease remission compared to placebo (162). Other systematic reviews of the literature reached similar conclusions (163-165). Three short-term trials showed positive effects of omega-3 supplementation on plasma biochemical parameters (e.g., reduced inflammatory cytokine expression, increased plasma EPA and DHA concentrations) compared to controls (161). In spite of its impact on biochemical changes in the short-term, however, the ability of omega-3 supplementation to maintain remission or effect clinically meaningful changes in Crohn's disease is not supported by the current evidence (164).
Ulcerative colitis: Seven randomized controlled trials of fish oil supplementation in patients with active ulcerative colitis reported significant improvement in at least one outcome measure, such as decreased corticosteroid use, improved disease activity scores, or improved histology scores (163). In patients with inactive ulcerative colitis, omega-3 supplementation had no effect on relapse rates compared to placebo in four separate trials (163, 165).
While no serious side effects were reported in any trials of fish oil supplementation for the maintenance or remission of inflammatory bowel disease, diarrhea and upper gastrointestinal symptoms occurred more frequently with omega-3 treatment (163-165).
Asthma
Inflammatory eicosanoids (leukotrienes) derived from arachidonic acid (AA; 20:4n-6) are thought to play an important role in the pathology of asthma (32). Because increasing omega-3 fatty acid intake has been found to decrease the formation of AA-derived leukotrienes, a number of clinical trials have examined the effects of long-chain omega-3 fatty acid supplementation on asthma. Although there is some evidence that omega-3 fatty acid supplementation can decrease the production of inflammatory mediators in asthmatic patients (166, 167), evidence that omega-3 fatty acid supplementation decreases the clinical severity of asthma in controlled trials has been inconsistent (168). Three systematic reviews of randomized controlled trials of long-chain omega-3 fatty acid supplementation in asthmatic adults and children found no consistent effects on clinical outcome measures, including pulmonary function tests, asthmatic symptoms, medication use, or bronchial hyperreactivity (169-171).
Immunoglobulin A nephropathy
Immunoglobulin A (IgA) nephropathy is a kidney disorder that results from the deposition of IgA in the glomeruli of the kidneys. The cause of IgA nephropathy is not clear, but progressive renal failure may eventually develop in 15%-40% of patients (172). Since glomerular IgA deposition results in increased production of inflammatory mediators, omega-3 fatty acid supplementation could potentially modulate the inflammatory response and preserve renal function.
A 2012 meta-analysis assessed the efficacy of omega-3 fatty acid supplementation on adult IgA nephropathy (173). Five randomized controlled trials were included in an analysis involving 239 patients (mean age, 37-41 years) who received placebo or supplemental EPA + DHA at doses of 1.4 to 5.1 g/day for 6 to 24 months. Compared with control groups, omega-3 supplementation had no significant effect on urine protein excretion or glomerular filtration rate. Only two trials measured changes in serum creatinine (a marker of renal function) and end-stage renal disease — omega-3 treatment had a beneficial effect on these two parameters in both trials. No adverse events associated with omega-3 supplementation were reported in any of the trials. A more recent review of the literature identified six trials showing evidence of omega-3 supplementation slowing IgA nephropathy disease progression and three trials reporting no effect (174). Additionally, preliminary data suggested that the potential synergistic actions of aspirin and long-chain omega-3 PUFAs might constitute a promising treatment option (168).
Neuropsychiatric disorders
Autism spectrum disorders
Autism spectrum disorders (ASD) refer to three neurodevelopmental disorders of variable severity, namely autism, Asperger syndrome, and pervasive development disorder. ASD are characterized by abnormal information processing in the brain due to alterations in the way nerve cells and their synapses connect and organize. ASD are thought to have a strong genetic basis, yet environmental factors including diet may play an important role. Given that omega-3 and omega-6 PUFA are necessary for neuronal growth and synapse formation (see Biological Activities), they may be of significant benefit in the prevention and/or management of ASD. This is supported by observations of PUFA abnormalities in blood of children with ASD, when compared to their peers with no neurodevelopmental disorders (175). A meta-analysis of case-control studies reported lower blood concentrations of DHA and EPA in children with ASD compared to typically developing children; yet, the ratio of total omega-6 to omega-3 fatty acids was similar between children with and without ASD symptoms (176). A systematic review by the same authors identified six randomized controlled trials that examined the effect of primarily long-chain omega-3 PUFA on ASD symptoms (176). All the studies included children; one study also included adults ≤28 years (177). Four trials used EPA (0.70-0.84 g/day) plus DHA (0.46-0.70 g/day) (178-181), one trial used DHA (0.24 g/day) plus AA (0.24 g/day) (177), and one trial only used only DHA (0.20 g/day) (182). A pooled analysis of four (177-180) of these trials, including a total of 107 participants, showed a small improvement in measures of social interaction and repetitive and restrictive interests and behaviors with long-chain PUFA supplementation for 6 to 16 weeks; however, there was no effect on measures of communication and ASD co-existing conditions, such as hyperactivity, irritability, sensory issues, and gastrointestinal symptoms (176). Two additional systematic reviews and meta-analyses, also published in 2017, identified the same set of trials. One meta-analysis suggested a benefit of long-chain PUFA on measures of lethargy and stereotypy but found no overall clinical improvement compared to placebo (183). The other meta-analysis suggested an improvement regarding lethargy yet a worsening of externalizing behavior and social skills in children supplemented with omega-3 PUFA (184).
The available evidence is based on few trials of small sample sizes and is thus too limited to draw firm conclusions regarding the potential benefit of long-chain PUFA supplementation in ASD management.
Major depression and bipolar disorder
Data from ecologic studies across different countries suggested an inverse association between seafood consumption and national rates of major depression (185) and bipolar disorder (186).
Several small studies have found omega-3 fatty acid concentrations to be lower in plasma (187-189) and adipose tissue (190) of individuals suffering from depression compared to controls. Although it is not known how omega-3 fatty acid intake affects the incidence of depression, modulation of neuronal signaling pathways and eicosanoid production have been proposed as possible mechanisms (191). There may be some benefit of omega-3 PUFA supplementation on depressive disorders, but it is difficult to compare studies and draw conclusions due to great heterogeneity among the trials (192, 193). Small sample sizes, lack of standardization of therapeutic doses, type of omega-3 PUFA administered, co-treatment with pharmacological agents, and diagnostic criteria vary among the trials. A 2012 systematic review of all published randomized controlled trials investigated the effect of omega-3 PUFA supplementation on the prevention and treatment of several types of depression and other neuropsychiatric disorders (192). With respect to major depression, most studies reported a positive effect of omega-3 supplements on depressive symptoms, though efficacy is still considered inconclusive given the great variability among trials. A few themes emerged from this review: more trials reported positive effect for omega-3 PUFA supplements as an adjunct to pharmacological treatment; in monotherapy trials, EPA alone was more effective than DHA alone; and in combination trials, positive effects were more likely if an EPA:DHA ratio of >1.5–2.0 was administered.
A 2014 meta-analysis grouped trials by type of diagnosis of depression (194). A positive effect of omega-3 supplementation was found in 11 trials in participants with a diagnosis of major depressive disorder (according to the Diagnostic and Statistical Manual of Mental Disorders [DSM] criteria). Omega-3 supplementation also appeared to be effective in the pooled analysis of eight trials in participants not formally diagnosed with major depressive disorder, i.e., adults with depressive symptoms despite ongoing treatment, untreated patients with mild-to-severe depressed mood, patients with a history of at least one major depressive episode, women with borderline personality disorder, patients with recurrent self-harm, and postmenopausal women with psychological distress and depressive symptoms. There was no mood improvement with omega-3 supplements in generally healthy adults experiencing depressive symptoms, as suggested by the pooled analysis of six trials (194).
Finally, a 2017 Cochrane systematic review and meta-analysis of 20 randomized controlled trials reported a small benefit of omega-3 supplementation on depressive symptoms when compared to placebo, yet the evidence was deemed of very low quality and the positive effect was judged likely to be biased and not clinically significant (195).
Unipolar depression and bipolar disorder are considered distinct psychiatric conditions, although major depression occurs in both. A 2016 meta-analysis of eight case-control studies that compared the PUFA composition of red blood cell membranes between patients with bipolar disorder and healthy subjects showed abnormally low red blood cell DHA concentrations with bipolar disorder (196). As with major depression, reviews of trials indicated that omega-3 supplementation may have a positive effect as an adjunct to therapy in patients with bipolar disorder (192, 194). Additionally, a 2016 randomized, placebo-controlled trial in 100 participants with bipolar disorder reported a reduction in the severity of manic episodes with daily supplementation of 1,000 mg omega-3 PUFA for three months (197).
While there is some promising evidence for the use of omega-3 fatty acids for major depression and bipolar disorder, additional trials that account for dietary omega-3 intake, changes in red blood cell PUFA concentrations, the ratio of EPA:DHA provided, and co-treatment with medications are necessary.
Schizophrenia
A 2013 meta-analysis of 18 studies compared the PUFA composition of red blood cell membranes in patients with schizophrenia to individuals without the disorder (198). The majority of studies investigated medicated patients, though the authors separated the analysis into three groups of patients at time of measurement in order to account for possible confounding from pharmacologic agents: antipsychotic-medicated, antipsychotic-naïve, and antipsychotic-free. Overall, decreased concentrations of DPA, DHA, and AA in red blood cell membranes were associated with the schizophrenic state. Several mechanisms may account for PUFA abnormalities in schizophrenia, such as altered lipid metabolism, increased oxidative stress, or changes in diet consequent to disease-related behavior.
The use of long-chain omega-3 fatty acid supplements to alleviate symptoms of schizophrenia or to mitigate adverse effects of antipsychotic medications has been investigated in a number of clinical trials (194, 199). In a recent randomized, placebo-controlled trial in 50 subjects with recent onset of schizophrenia who were medicated, daily supplementation with EPA (740 mg) and DHA (400 mg) reduced psychotic symptoms (assessed with the Brief Psychiatric Rating Scale) only in those who were not taking the anxiolytic, lorazepam (Ativan) (200). Overall, however, there was no effect of long-chain PUFA supplements on schizophrenia symptoms. Yet, given the high safety profile of fish oil supplements and some evidence of a positive effect of EPA supplementation in a subset of trials, some clinicians may consider EPA a useful adjunct to antipsychotic therapy in patients with schizophrenia.
Alzheimer's disease and dementia
Several mechanisms suggest that omega-3 PUFA supplementation may improve the cognitive performance of individuals with Alzheimer's disease and other types of dementia. In particular, the antioxidative and anti-inflammatory properties of these PUFA may help protect neurons, promote synaptic plasticity, and limit cellular death. The PUFA composition of the diet appears to influence blood cholesterol, which may play a role in the pathology of Alzheimer's disease. However, the current evidence from clinical trials is not supportive of omega-3 supplementation in the treatment of Alzheimer’s disease in humans. A 2016 Cochrane review identified three randomized, placebo-controlled trials in patients with Alzheimer's disease of mild-to-moderate severity (201). These trials compared daily supplementation with DHA (between 675 mg and 1,700 mg) and EPA (between 600 mg and 975 mg) to a placebo for 12 months (202, 203) or 18 months (204). Of note, the study by Quinn et al. (204) also included 4 mg/day of vitamin E (used as preservative — see also Nutrient interactions) in the intervention arm, and the study by Freund-Levi et al. (202) included DHA (900-1,100 mg/day) but no EPA. The pooled analysis of these trials showed no beneficial effect of omega-3 supplementation on measures of global and specific cognitive functions, measures of functional outcomes, and measures of dementia severity (201). There was no difference between intervention and placebo arms regarding the occurrence of adverse effects (201).
Sources
Food sources
Humans can synthesize arachidonic acid (AA) from linoleic acid (LA) and eicosapentaenoic acid (EPA) and docosapentaenoic acid (DHA) from α-linolenic acid (ALA) through a series of desaturation and elongation reactions. EPA and docosapentaenoic acid (DPA) are also obtained from the retroconversion of DHA (see Metabolism and Bioavailability). Due to low conversion efficiency, it is advised to obtain EPA and DHA from additional sources.
Omega-6 fatty acids
Linoleic acid (LA): Food sources of LA include vegetable oils, such as soybean, safflower, and corn oil; nuts; seeds; and some vegetables. Dietary surveys in the US indicate that the average adult intake of LA ranges from 17 to 20 g/day for men and 12 to 13 g/day for women (78). Some foods that are rich in LA are listed in Table 2.
| Food | Serving | Linoleic Acid (g) |
|---|---|---|
| Safflower oil | 1 tablespoon |
10.1
|
| Sunflower seeds, oil roasted | 1 ounce |
9.7
|
| Pine nuts | 1 ounce |
9.4
|
| Sunflower oil | 1 tablespoon |
8.9
|
| Corn oil | 1 tablespoon |
7.3
|
| Soybean oil | 1 tablespoon |
6.9
|
| Pecans, oil roasted | 1 ounce |
6.4
|
| Brazil nuts | 1 ounce |
5.8
|
| Sesame oil | 1 tablespoon |
5.6
|
Arachidonic acid: Animals, but not plants, can convert LA to AA. Therefore, AA is absent in vegetable oils and fats and present in small amounts in meat, poultry, and eggs.
Omega-3 fatty acids
α-Linolenic acid (ALA): Flaxseeds, walnuts, and their oils are among the richest dietary sources of ALA. Canola oil is also an excellent source of ALA. Dietary surveys in the US indicate that average adult intakes for ALA range from 1.8 to 2.0 g/day for men and from 1.4 to 1.5 g/day for women (78). Some foods that are rich in ALA are listed in Table 3.
| Food | Serving | α-Linolenic acid (g) |
|---|---|---|
| Flaxseed oil | 1 tablespoon |
7.3
|
| Chia seeds, dried | 1 ounce |
5.1
|
| Walnuts, English | 1 ounce |
2.6
|
| Flaxseeds, ground | 1 tablespoon |
1.6
|
| Walnut oil | 1 tablespoon |
1.4
|
| Canola oil | 1 tablespoon |
1.3
|
| Soybean oil | 1 tablespoon |
0.9
|
| Mustard oil | 1 tablespoon |
0.8
|
| Walnuts, black | 1 ounce |
0.6
|
| Tofu, firm | ½ cup |
0.2
|
Eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA): Dietary surveys in the US indicate that average adult intakes of EPA range from 0.03 to 0.06 g/day, and average adult intakes of DHA range from 0.05 to 0.10 g/day (78). Oily fish are the major dietary source of EPA and DHA; omega-3 fatty acid-enriched eggs are also available in the US. Some foods that are rich in EPA and DHA are listed in Table 4.
| Food | Serving | EPA (g) | DHA (g) |
Amount Providing 1 g of EPA + DHA |
|---|---|---|---|---|
| Herring, Pacific | 3 ounces* |
1.06
|
0.75
|
1.5 ounces
|
| Salmon, chinook | 3 ounces |
0.86
|
0.62
|
2 ounces
|
| Sardines, Pacific | 3 ounces |
0.45
|
0.74
|
2.5 ounces
|
| Salmon, Atlantic | 3 ounces |
0.28
|
0.95
|
2.5 ounces
|
| Oysters, Pacific | 3 ounces |
0.75
|
0.43
|
2.5 ounces
|
| Salmon, sockeye | 3 ounces |
0.45
|
0.60
|
3 ounces
|
| Trout, rainbow | 3 ounces |
0.40
|
0.44
|
3.5 ounces
|
| Tuna, canned, white | 3 ounces |
0.20
|
0.54
|
4 ounces
|
| Crab, Dungeness | 3 ounces |
0.24
|
0.10
|
9 ounces
|
| Tuna, canned, light | 3 ounces |
0.04
|
0.19
|
12 ounces
|
| *A three-ounce serving of fish is about the size of a deck of cards. | ||||
Supplements
Omega-6 fatty acids
Borage seed oil, evening primrose oil, and black currant seed oil are rich in γ-linolenic acid (GLA; 18:3n-6) and are often marketed as GLA or essential fatty acid (EFA) supplements (206).
Omega-3 fatty acids
Flaxseed oil (also known as flax oil or linseed oil) is available as an ALA supplement. A number of fish oils are marketed as omega-3 fatty acid supplements. The omega-3 fatty acids from natural fish oil are in the triglyceride form, often with only one of three attached fatty acids an omega-3; thus, up to 70% of fatty acids provided may be other types (3). Ethyl esters of EPA and DHA (ethyl-EPA and ethyl-DHA) are concentrated sources of long-chain omega-3 fatty acids that provide more EPA and DHA per gram of oil. Krill oil contains both EPA and DHA and is considered comparable to fish oil as a source of these long-chain PUFA (207). Cod liver oil is also a rich source of EPA and DHA, but some cod liver oil preparations may contain excessive amounts of preformed vitamin A (retinol) and vitamin D (206). DHA supplements derived from algal and fungal sources are also available. Because dietary DHA can be retroconverted to EPA and DPA in humans, DHA supplementation represents yet another alternative to fish oil supplements (see Metabolism and Bioavailability).
The content of EPA and DHA varies in each of these preparations, making it necessary to read product labels in order to determine the EPA and DHA levels provided by a particular supplement. All omega-3 fatty acid supplements are absorbed more efficiently with meals. Dividing one's daily dose into two or three smaller doses throughout the day will decrease the risk of gastrointestinal side effects (see Safety).
Infant formula
In 2001, the FDA began permitting the addition of DHA and AA to infant formula in the United States (208). Presently, manufacturers are not required to list the amounts of DHA and AA added to infant formula on the label. However, most infant formula manufacturers provide this information. The amounts added to formulas in the US range from 8 to 17 mg DHA/100 calories (5 fl oz) and from 16 to 34 mg AA/100 calories. For example, an infant drinking 20 fl oz of DHA-enriched formula daily would receive 32 to 68 mg/day of DHA and 64 to 136 mg/day of AA.
Safety
Adverse effects
γ-Linolenic acid (18:3n-6)
Supplemental γ-linolenic acid is generally well tolerated, and serious adverse side effects have not been observed at doses up to 2.8 g/day for 12 months (209). High doses of borage seed oil, evening primrose oil, or black currant seed oil may cause gastrointestinal upset, loose stools, or diarrhea (206). Because of case reports that supplementation with evening primrose oil induced seizure activity in people with undiagnosed temporal lobe epilepsy (210), people with a history of seizures or a seizure disorder are generally advised to avoid evening primrose oil and other γ-linolenic acid-rich oils (206).
α-Linolenic acid (18:3n-3)
Although flaxseed oil is generally well tolerated, high doses may cause loose stools or diarrhea (211). Allergic and anaphylactic reactions have been reported with flaxseed and flaxseed oil ingestion (212).
Eicosapentaenoic acid (20:5n-3) and docosahexaenoic acid (22:6n-3)
Serious adverse reactions have not been reported in those using fish oil or other EPA and DHA supplements. The most common adverse effect of fish oil or EPA and DHA supplements is a fishy aftertaste. Belching and heartburn have also been reported. Additionally, high doses may cause nausea and loose stools.
Potential for excessive bleeding: The potential for high omega-3 fatty acid intakes, especially EPA and DHA, to prolong bleeding times has been well studied and may play a role in the cardioprotective effects of omega-3 fatty acids. Although excessively long bleeding times and increased incidence of hemorrhagic stroke have been observed in Greenland Eskimos with very high intakes of EPA + DHA (6.5 g/day), it is not known whether high intakes of EPA and DHA are the only factor responsible for these observations (1). The US FDA has ruled that intakes up to 3 g/day of long-chain omega-3 fatty acids (EPA and DHA) are Generally Recognized As Safe (GRAS) for inclusion in the diet, and available evidence suggests that intakes less than 3 g/day are unlikely to result in clinically significant bleeding (107). Although the US Institute of Medicine did not establish a tolerable upper intake level (UL) for omega-3 fatty acids, caution was advised with the use of supplemental EPA and DHA, especially in those who are at increased risk of excessive bleeding (see Drug interactions and Nutrient interactions) (1, 206).
Potential for immune system suppression: Although the suppression of inflammatory responses resulting from increased omega-3 fatty acid intakes may benefit individuals with inflammatory or autoimmune diseases, anti-inflammatory doses of omega-3 fatty acids could decrease the potential of the immune system to destroy pathogens (213). Studies comparing measures of immune cell function outside the body (ex vivo) at baseline and after supplementing people with omega-3 fatty acids, mainly EPA and DHA, have demonstrated immunosuppressive effects at doses as low as 0.9 g/day for EPA and 0.6 g/day for DHA (1). Although it is not clear if these findings translate to impaired immune responses in vivo, caution should be observed when considering omega-3 fatty acid supplementation in individuals with compromised immune systems.
Potential other effects: Although fish oil supplements are unlikely to affect glucose homeostasis, people with diabetes mellitus who are considering fish oil supplements should inform their physician and be monitored if they choose to take them (206).
Infant formula
In early studies of DHA-enriched infant formula, EPA- and DHA-rich fish oil was used as a source of DHA. However, some preterm infants receiving fish oil-enriched formula had decreased plasma AA concentrations, which were associated with decreased weight (but not length and head circumference) (214, 215). This effect was attributed to the potential for high concentrations of EPA to interfere with the synthesis of AA, which is essential for normal growth. Consequently, EPA was removed and AA was added to DHA-enriched formula. Currently available infant formulas in the US contain only AA and DHA derived from algal or fungal sources, rather than fish oil. Randomized controlled trials have not found any adverse effects on growth in infants fed formulas enriched with AA and DHA for up to one year (216).
Pregnancy and lactation
The safety of supplemental omega-3 and omega-6 fatty acids, including borage seed oil, evening primrose oil, black currant seed oil, and flaxseed oil, has not been established in pregnant or lactating (breast-feeding) women (217). Studies of fish oil supplementation during pregnancy and lactation have not reported any serious adverse effects, but use of omega-6/omega-3 PUFA-containing supplements and fish oil supplements in pregnant or nursing women should be monitored by a physician (see Contaminants in fish and Contaminants in supplements) (206).
Contaminants in fish
Some species of fish may contain significant levels of methylmercury, polychlorinated biphenyls (PCBs), or other environmental contaminants (218). In general, larger predatory fish, such as swordfish, tend to contain the highest levels of these contaminants. Removing the skin, fat, and internal organs of the fish prior to cooking and allowing the fat to drain from the fish while it cooks will decrease exposure to a number of fat-soluble pollutants, such as PCBs (219). However, methylmercury is found throughout the muscle of fish, so these cooking precautions will not reduce exposure to methylmercury. Organic mercury compounds are toxic and excessive exposure can cause brain and kidney damage. The developing fetus, infants, and young children are especially vulnerable to the toxic effects of mercury on the brain. In order to limit their exposure to methylmercury, the US Food and Drug Administration (FDA) and Environmental Protection Agency have formulated joint recommendations for women who may become pregnant, pregnant women, breast-feeding women, and parents. These recommendations are presented in Table 5.
For more information about the FDA/Environmental Protection Agency advisory for pregnant women and parents of young children on eating fish, see their online brochure. More information about mercury levels in commercial fish and shellfish is available from the FDA.
Of note, the 2015-2020 Dietary Guidelines for Americans recommend the consumption of salmon, anchovies, herring, shad, sardines, Pacific oysters, trout, and Atlantic and Pacific mackerel (not king mackerel), which are higher in EPA and DHA and lower in methylmercury (220).
Contaminants in supplements
Although concerns have been raised regarding the potential for omega-3 fatty acid supplements derived from fish oil to contain methylmercury, PCBs, and dioxins, several independent laboratory analyses in the US have found commercially available omega-3 fatty acid supplements to be free of methylmercury, PCBs, and dioxins (221). The absence of methylmercury in omega-3 fatty acid supplements can be explained by the fact that mercury accumulates in the muscle, rather than the fat of fish (107). In general, fish body oils contain lower concentrations of PCBs and other fat-soluble contaminants than fish liver oils. Additionally, fish oils that have been more highly refined and deodorized contain lower concentrations of PCBs (222). Pyrrolizidine alkaloids, potentially hepatotoxic and carcinogenic compounds, are found in various parts of the borage plant. People who take borage oil supplements should use products that are certified free of unsaturated pyrrolizidine alkaloids (206).
| 1. Eat 8-12 ounces of a variety of fish a week |
|
| 2. Choose fish lower in mercury. |
|
| 3. Avoid 4 types of fish: tilefish from the Gulf of Mexico, shark, swordfish, and king mackerel. |
|
| 4. When eating fish you or others have caught from streams, rivers, and lakes, pay attention to fish advisories on those waterbodies. |
|
| 5. When adding more fish to your diet, be sure to stay within your calorie needs. |
Drug interactions
γ-Linolenic acid supplements, such as evening primrose oil or borage seed oil, may increase the risk of seizures in people on phenothiazines (neuroleptic agents), such as chlorpromazine (210). High doses of black currant seed oil, borage seed oil, evening primrose oil, flaxseed oil, and fish oil may inhibit platelet aggregation; therefore, these supplements should be used with caution in people on anticoagulant medications (206). In particular, people taking fish oil or long-chain omega-3 fatty acid (EPA and DHA) supplements in combination with anticoagulant drugs, including aspirin, clopidogrel (Plavix), dalteparin (Fragmin), dipyridamole (Persantine), enoxaparin (Lovenox), heparin, ticlopidine (Ticlid), and warfarin (Coumadin), should have their coagulation status monitored using a standardized prothrombin time assay (international normalized ratio [INR]). One small study found that 3 g/day or 6 g/day of fish oil did not affect INR values in 10 patients on warfarin over a four-week period (223). However, a case report described an individual who required a reduction of her warfarin dose when she doubled her fish oil dose from 1 g/day to 2 g/day (224).
Nutrient interactions
Vitamin E
Outside the body, PUFA become rancid (oxidized) more easily than saturated fatty acids. Fat-soluble antioxidants, such as vitamin E (α-tocopherol), play an important role in preventing the oxidation of PUFA. Inside the body, results of animal studies and limited data in humans suggest that the amount of vitamin E required to prevent lipid peroxidation increases with the amount of PUFA consumed (225). One widely used recommendation for vitamin E intake is 0.6 mg of α-tocopherol per gram of dietary PUFA. This recommendation was based on a small study in men and the ratio of α-tocopherol to LA in the US diet and has not been verified in more comprehensive studies. Although EPA and DHA are easily oxidized outside the body, it is presently unclear whether they are more susceptible to oxidative damage within the body (226). High vitamin E intakes have not been found to decrease biomarkers of oxidative damage when EPA and DHA intakes are increased (227, 228), but some experts believe that an increase in PUFA intake, particularly omega-3 PUFA intake, should be accompanied by an increase in vitamin E intake (1).
Intake Recommendations
US Institute of Medicine
The Food and Nutrition Board of the US Institute of Medicine (now the National Academy of Medicine) has established adequate intake (AI) for omega-6 and omega-3 fatty acids (Tables 6 and 7) (1).
| Life Stage | Age | Source | Males (g/day) | Females (g/day) |
|---|---|---|---|---|
| Infants | 0-6 months | Omega-6 PUFA* | 4.4 | 4.4 |
| Infants | 7-12 months | Omega-6 PUFA* | 4.6 | 4.6 |
| Children | 1-3 years | LA# | 7 | 7 |
| Children | 4-8 years | LA | 10 | 10 |
| Children | 9-13 years | LA | 12 | 10 |
| Adolescents | 14-18 years | LA | 16 | 11 |
| Adults | 19-50 years | LA | 17 | 12 |
| Adults | 51 years and older | LA | 14 | 11 |
| Pregnancy | all ages | LA | - | 13 |
| Breast-feeding | all ages | LA | - | 13 |
| *The various omega-6 polyunsaturated fatty acids (PUFA) present in human milk can contribute to the AI for infants. # LA, linoleic acid | ||||
| Life Stage | Age | Source | Males (g/day) | Females (g/day) |
|---|---|---|---|---|
| Infants | 0-6 months | ALA, EPA, DHA* |
0.5
|
0.5
|
| Infants | 7-12 months | ALA, EPA, DHA |
0.5
|
0.5
|
| Children | 1-3 years | ALA |
0.7
|
0.7
|
| Children | 4-8 years | ALA |
0.9
|
0.9
|
| Children | 9-13 years | ALA |
1.2
|
1.0
|
| Adolescents | 14-18 years | ALA |
1.6
|
1.1
|
| Adults | 19 years and older | ALA |
1.6
|
1.1
|
| Pregnancy | all ages | ALA |
-
|
1.4
|
| Breast-feeding | all ages | ALA |
-
|
1.3
|
| *All omega-3 polyunsaturated fatty acids present in human milk can contribute to the AI for infants. ALA, α-linolenic acid; EPA, eicosapentaenoic acid; DHA, docosahexaenoic acid. | ||||
Given the established health benefits of consuming at least two servings of oily fish per week, providing approximately 400 to 500 mg EPA + DHA, some researchers have proposed that the US Institute of Medicine (now the National Academy of Medicine) establish dietary reference intakes (DRIs) for EPA + DHA (27). For now, there are no DRIs for EPA and DHA specifically.
Because maternal dietary intake of long-chain PUFA determines the DHA status of the newborn, several expert panels in the US recommend that pregnant and lactating women consume at least 200 mg DHA per day, close to the amount recommended for adults in general (250 mg/day) (70, 229). The potential benefits associated with obtaining long-chain omega-3 fatty acids through moderate consumption of fish (e.g., 1-2 servings weekly) during pregnancy and lactation outweigh any risks of contaminant exposure, though fish with high concentrations of methylmercury should be avoided (218). For information about contaminants in fish and guidelines for fish consumption by women of childbearing age, see Contaminants in fish.
2015-2020 Dietary Guidelines for Americans
The 2015-2020 Dietary Guidelines provide recommendations for nutritional goals for linoleic acid and α-linolenic acid based on the DRIs (see Tables 6 and 7). Seafood, nuts, seeds, and oils, which are all part of healthy dietary patterns, provide essential fatty acids. The 2015-2020 Dietary Guidelines provide dietary recommendations regarding the amounts of these foods for those who choose to follow a healthy US-style eating pattern, a healthy Mediterranean-style eating pattern, or a healthy vegetarian eating pattern (Table 8).
| Food | Healthy Eating Patterns | ||
|---|---|---|---|
| US-style | Mediterranean-style | Vegetarian | |
| Seafood (oz-eq/week) | 8 | 15 | – |
|
Nuts, seeds, soy products (oz-eq/week)
|
5 | 5 | 7 |
| Oils (g/week) | 27 | 27 | 27 |
|
*Recommendations for total daily energy needs of 2,000 calories per day. Estimates of daily calorie needs according to age, gender, and physical activity can be found in the Appendix 2 of the ‘2015-2020 Dietary Guidelines for Americans’ report (220). Oz-eq, ounce-equivalent |
|||
American Heart Association recommendation
The American Heart Association recommends that people without documented coronary heart disease (CHD) eat a variety of fish (preferably oily) at least twice weekly (230). Two servings of oily fish provide approximately 500 mg of EPA plus DHA. Pregnant women and children should avoid fish that typically have higher levels of methylmercury (see Contaminants in fish). People with documented CHD and those with heart failure without preserved left ventricular function are advised to consume approximately 1 g/day of EPA + DHA preferably from oily fish, or to consider EPA + DHA supplements in consultation with a physician (104, 107). Patients who need to lower serum triglycerides may take 2 to 4 g/day of EPA + DHA supplements under a physician's care (see Hypertriglyceridemia).
International recommendations
Upon request of the European Commission, the European Food Safety Authority (EFSA) proposed adequate intakes (AI) for the essential fatty acids LA and ALA, as well as the long-chain omega-3 fatty acids EPA and DHA (231). EFSA recommends an LA intake of 4% of total energy and an ALA intake of 0.5% of total energy; an AI of 250 mg/day is recommended for EPA plus DHA (232). The European Food and Safety Authority (EFSA) recommends that pregnant and lactating women consume an additional 100 to 200 mg of preformed DHA on top of the 250 mg/day EPA plus DHA recommended for healthy adults (231).
For adults, the World Health Organization recommends an acceptable macronutrient distribution range (AMDR) for omega-6 fatty acid intake of 2.5%-9% of energy and for omega-3 fatty acid intake of 0.5%-2% of energy (233). Their AMDR for EPA plus DHA is 0.25 to 2 g/day (the upper level applying to secondary prevention of coronary heart disease).
The International Society for the Study of Fatty Acids and Lipids (ISSFAL) recommends healthy adults have an LA intake of 2% energy, an ALA intake of 0.7% energy, and a minimum of 500 mg/day of EPA plus DHA for cardiovascular health (234).
Linus Pauling Institute recommendation
The Linus Pauling Institute supports the AI for the essential fatty acids (see Tables 6 and 7) and recommends that generally healthy adults increase their intake of long-chain omega-3 fatty acids by eating fish twice weekly and consuming foods rich in ALA, such as walnuts, flaxseeds, and flaxseed or canola oil. If you don't regularly consume fish, consider taking a two-gram fish oil supplement several times a week. If you are prone to bleeding or take anticoagulant drugs, consult your physician.
Authors and Reviewers
Originally written in 2003 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in December 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in April 2009 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in April 2014 by:
Giana Angelo, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in May 2019 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in June 2019 by:
Donald B. Jump, Ph.D.
Professor, School of Biological and Population Health Sciences
Principal Investigator, Linus Pauling Institute
Oregon State University
Copyright 2003-2024 Linus Pauling Institute
References
1. Food and Nutrition Board, Institute of Medicine. Dietary Fats: Total Fat and Fatty Acids. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids. Washington, D.C.: National Academies Press; 2002:422-541. (The National Academies Press)
2. Lichtenstein A, Jones PJ. Lipids: Absorption and Transport. In: Erdman JWJ, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition. 10th ed: ILSI Wiley-Blackwell; 2012:118-131.
3. Davidson MH. Omega-3 fatty acids: new insights into the pharmacology and biology of docosahexaenoic acid, docosapentaenoic acid, and eicosapentaenoic acid. Curr Opin Lipidol. 2013;24(6):467-474. (PubMed)
4. Nakamura MT, Nara TY. Structure, function, and dietary regulation of delta6, delta5, and delta9 desaturases. Annu Rev Nutr. 2004;24:345-376. (PubMed)
5. Jump DB, Depner CM, Tripathy S. Omega-3 fatty acid supplementation and cardiovascular disease. J Lipid Res. 2012;53(12):2525-2545. (PubMed)
6. Burdge GC, Jones AE, Wootton SA. Eicosapentaenoic and docosapentaenoic acids are the principal products of α-linolenic acid metabolism in young men. Br J Nutr. 2002;88(4):355-364. (PubMed)
7. Burdge GC, Wootton SA. Conversion of α-linolenic acid to eicosapentaenoic, docosapentaenoic and docosahexaenoic acids in young women. Br J Nutr. 2002;88(4):411-420. (PubMed)
8. Burdge G. α-Linolenic acid metabolism in men and women: nutritional and biological implications. Curr Opin Clin Nutr Metab Care. 2004;7(2):137-144. (PubMed)
9. Giltay EJ, Gooren LJ, Toorians AW, Katan MB, Zock PL. Docosahexaenoic acid concentrations are higher in women than in men because of estrogenic effects. Am J Clin Nutr. 2004;80(5):1167-1174. (PubMed)
10. Tosi F, Sartori F, Guarini P, Olivieri O, Martinelli N. Delta-5 and delta-6 desaturases: crucial enzymes in polyunsaturated fatty acid-related pathways with pleiotropic influences in health and disease. Adv Exp Med Biol. 2014;824:61-81. (PubMed)
11. Ameur A, Enroth S, Johansson A, et al. Genetic adaptation of fatty-acid metabolism: a human-specific haplotype increasing the biosynthesis of long-chain omega-3 and omega-6 fatty acids. Am J Hum Genet. 2012;90(5):809-820. (PubMed)
12. Brossard N, Croset M, Pachiaudi C, Riou JP, Tayot JL, Lagarde M. Retroconversion and metabolism of [13C]22:6n-3 in humans and rats after intake of a single dose of [13C]22:6n-3-triacylglycerols. Am J Clin Nutr. 1996;64(4):577-586. (PubMed)
13. Conquer JA, Holub BJ. Dietary docosahexaenoic acid as a source of eicosapentaenoic acid in vegetarians and omnivores. Lipids. 1997;32(3):341-345. (PubMed)
14. Jump DB, Tripathy S, Depner CM. Fatty acid-regulated transcription factors in the liver. Ann Rev Nutr. 2013;33:249-269. (PubMed)
15. Stillwell W, Wassall SR. Docosahexaenoic acid: membrane properties of a unique fatty acid. Chem Phys Lipids. 2003;126(1):1-27. (PubMed)
16. Calder PC. n-3 fatty acids, inflammation and immunity: new mechanisms to explain old actions. Proc Nutr Soc. 2013;72(3):326-336. (PubMed)
17. Calder PC. Polyunsaturated fatty acids and inflammatory processes: New twists in an old tale. Biochimie. 2009;91(6):791-795. (PubMed)
18. Harris WS, Sands SA, Windsor SL, et al. Omega-3 fatty acids in cardiac biopsies from heart transplantation patients: correlation with erythrocytes and response to supplementation. Circulation. 2004;110(12):1645-1649. (PubMed)
19. Jump DB. The biochemistry of n-3 polyunsaturated fatty acids. J Biol Chem. 2002;277(11):8755-8758. (PubMed)
20. Jeffrey BG, Weisingerb HS, Neuringer M, Mitcheli DC. The role of docosahexaenoic acid in retinal function. Lipids. 2001;36(9):859-871. (PubMed)
21. SanGiovanni JP, Chew EY. The role of omega-3 long-chain polyunsaturated fatty acids in health and disease of the retina. Prog Retin Eye Res. 2005;24(1):87-138. (PubMed)
22. Innis SM. Dietary omega 3 fatty acids and the developing brain. Brain Res. 2008;1237:35-43. (PubMed)
23. Yu N, Martin JL, Stella N, Magistretti PJ. Arachidonic acid stimulates glucose uptake in cerebral cortical astrocytes. Proc Natl Acad Sci U S A. 1993;90(9):4042-4046. (PubMed)
24. Das UN, Fams. Long-chain polyunsaturated fatty acids in the growth and development of the brain and memory. Nutrition. 2003;19(1):62-65. (PubMed)
25. Das UN. Autism as a disorder of deficiency of brain-derived neurotrophic factor and altered metabolism of polyunsaturated fatty acids. Nutrition. 2013;29(10):1175-1185. (PubMed)
26. American Oil Chemists' Society. The AOCS Lipid Library. August 15, 2012. Available at: http://lipidlibrary.aocs.org/. Accessed 2/25/14.
27. Flock MR, Harris WS, Kris-Etherton PM. Long-chain omega-3 fatty acids: time to establish a dietary reference intake. Nutr Rev. 2013;71(10):692-707. (PubMed)
28. Serhan CN, Chiang N. Resolution phase lipid mediators of inflammation: agonists of resolution. Curr Opin Pharmacol. 2013;13(4):632-640. (PubMed)
29. Bannenberg G, Serhan CN. Specialized pro-resolving lipid mediators in the inflammatory response: An update. Biochim Biophys Acta. 2010;1801(12):1260-1273. (PubMed)
30. Nicolaou A, Mauro C, Urquhart P, Marelli-Berg F. Polyunsaturated Fatty Acid-Derived Lipid Mediators and T Cell Function. Front Immunol. 2014;5:75. (PubMed)
31. Price PT, Nelson CM, Clarke SD. Omega-3 polyunsaturated fatty acid regulation of gene expression. Curr Opin Lipidol. 2000;11(1):3-7. (PubMed)
32. Calder PC. Dietary modification of inflammation with lipids. Proc Nutr Soc. 2002;61(3):345-358. (PubMed)
33. Sampath H, Ntambi JM. Polyunsaturated fatty acid regulation of gene expression. Nutr Rev. 2004;62(9):333-339. (PubMed)
34. Shaikh SR. Biophysical and biochemical mechanisms by which dietary N-3 polyunsaturated fatty acids from fish oil disrupt membrane lipid rafts. J Nutr Biochem. 2012;23(2):101-105. (PubMed)
35. Jeppesen PB, Hoy CE, Mortensen PB. Essential fatty acid deficiency in patients receiving home parenteral nutrition. Am J Clin Nutr. 1998;68(1):126-133. (PubMed)
36. Smit EN, Muskiet FA, Boersma ER. The possible role of essential fatty acids in the pathophysiology of malnutrition: a review. Prostaglandins Leukot Essent Fatty Acids. 2004;71(4):241-250. (PubMed)
37. Mascioli EA, Lopes SM, Champagne C, Driscoll DF. Essential fatty acid deficiency and home total parenteral nutrition patients. Nutrition. 1996;12(4):245-249. (PubMed)
38. Stegink LD, Freeman JB, Wispe J, Connor WE. Absence of the biochemical symptoms of essential fatty acid deficiency in surgical patients undergoing protein sparing therapy. Am J Clin Nutr. 1977;30(3):388-393. (PubMed)
39. Jeppesen PB, Hoy CE, Mortensen PB. Deficiencies of essential fatty acids, vitamin A and E and changes in plasma lipoproteins in patients with reduced fat absorption or intestinal failure. Eur J Clin Nutr. 2000;54(8):632-642. (PubMed)
40. Lepage G, Levy E, Ronco N, Smith L, Galeano N, Roy CC. Direct transesterification of plasma fatty acids for the diagnosis of essential fatty acid deficiency in cystic fibrosis. J Lipid Res. 1989;30(10):1483-1490. (PubMed)
41. Holman RT, Johnson SB, Hatch TF. A case of human linolenic acid deficiency involving neurological abnormalities. Am J Clin Nutr. 1982;35(3):617-623. (PubMed)
42. Fedorova I, Hussein N, Baumann MH, Di Martino C, Salem N, Jr. An n-3 fatty acid deficiency impairs rat spatial learning in the Barnes maze. Behav Neurosci. 2009;123(1):196-205. (PubMed)
43. Fedorova I, Salem N, Jr. Omega-3 fatty acids and rodent behavior. Prostaglandins Leukot Essent Fatty Acids. 2006;75(4-5):271-289. (PubMed)
44. Harris WS, Von Schacky C. The omega-3 index: a new risk factor for death from coronary heart disease? Prev Med. 2004;39(1):212-220. (PubMed)
45. Metcalf RG, James MJ, Gibson RA, et al. Effects of fish-oil supplementation on myocardial fatty acids in humans. Am J Clin Nutr. 2007;85(5):1222-1228. (PubMed)
46. Owen AJ, Peter-Przyborowska BA, Hoy AJ, McLennan PL. Dietary fish oil dose- and time-response effects on cardiac phospholipid fatty acid composition. Lipids. 2004;39(10):955-961. (PubMed)
47. von Schacky C. Omega-3 index and cardiovascular health. Nutrients. 2014;6(2):799-814. (PubMed)
48. Harris WS. The omega-3 index as a risk factor for coronary heart disease. Am J Clin Nutr. 2008;87(6):1997S-2002S. (PubMed)
49. Flock MR, Skulas-Ray AC, Harris WS, Etherton TD, Fleming JA, Kris-Etherton PM. Determinants of erythrocyte omega-3 fatty acid content in response to fish oil supplementation: a dose-response randomized controlled trial. J Am Heart Assoc. 2013;2(6):e000513. (PubMed)
50. Harris WS, Pottala JV, Varvel SA, Borowski JJ, Ward JN, McConnell JP. Erythrocyte omega-3 fatty acids increase and linoleic acid decreases with age: observations from 160,000 patients. Prostaglandins Leukot Essent Fatty Acids. 2013;88(4):257-263. (PubMed)
51. Olsen SF, Sorensen JD, Secher NJ, et al. Randomised controlled trial of effect of fish-oil supplementation on pregnancy duration. Lancet. 1992;339(8800):1003-1007. (PubMed)
52. Onwude JL, Lilford RJ, Hjartardottir H, Staines A, Tuffnell D. A randomised double blind placebo controlled trial of fish oil in high risk pregnancy. Br J Obstet Gynaecol. 1995;102(2):95-100. (PubMed)
53. Smuts CM, Huang M, Mundy D, Plasse T, Major S, Carlson SE. A randomized trial of docosahexaenoic acid supplementation during the third trimester of pregnancy. Obstet Gynecol. 2003;101(3):469-479. (PubMed)
54. Zhou SJ, Yelland L, McPhee AJ, Quinlivan J, Gibson RA, Makrides M. Fish-oil supplementation in pregnancy does not reduce the risk of gestational diabetes or preeclampsia. Am J Clin Nutr. 2012;95(6):1378-1384. (PubMed)
55. Szajewska H, Horvath A, Koletzko B. Effect of n-3 long-chain polyunsaturated fatty acid supplementation of women with low-risk pregnancies on pregnancy outcomes and growth measures at birth: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2006;83(6):1337-1344. (PubMed)
56. Horvath A, Koletzko B, Szajewska H. Effect of supplementation of women in high-risk pregnancies with long-chain polyunsaturated fatty acids on pregnancy outcomes and growth measures at birth: a meta-analysis of randomized controlled trials. Br J Nutr. 2007;98(2):253-259. (PubMed)
57. Makrides M, Gibson RA, McPhee AJ, Yelland L, Quinlivan J, Ryan P. Effect of DHA supplementation during pregnancy on maternal depression and neurodevelopment of young children: a randomized controlled trial. JAMA. 2010;304(15):1675-1683. (PubMed)
58. Kar S, Wong M, Rogozinska E, Thangaratinam S. Effects of omega-3 fatty acids in prevention of early preterm delivery: a systematic review and meta-analysis of randomized studies. Eur J Obstet Gynecol Reprod Biol. 2016;198:40-46. (PubMed)
59. Carlson SE, Gajewski BJ, Alhayek S, Colombo J, Kerling EH, Gustafson KM. Dose-response relationship between docosahexaenoic acid (DHA) intake and lower rates of early preterm birth, low birth weight and very low birth weight. Prostaglandins Leukot Essent Fatty Acids. 2018;138:1-5. (PubMed)
60. Saccone G, Berghella V, Maruotti GM, Sarno L, Martinelli P. Omega-3 supplementation during pregnancy to prevent recurrent intrauterine growth restriction: systematic review and meta-analysis of randomized controlled trials. Ultrasound Obstet Gynecol. 2015;46(6):659-664. (PubMed)
61. Saccone G, Berghella V. Omega-3 supplementation to prevent recurrent preterm birth: a systematic review and metaanalysis of randomized controlled trials. Am J Obstet Gynecol. 2015;213(2):135-140. (PubMed)
62. Gould JF, Smithers LG, Makrides M. The effect of maternal omega-3 (n-3) LCPUFA supplementation during pregnancy on early childhood cognitive and visual development: a systematic review and meta-analysis of randomized controlled trials. Am J Clin Nutr. 2013;97(3):531-544. (PubMed)
63. Gould JF, Treyvaud K, Yelland LN, et al. Does n-3 LCPUFA supplementation during pregnancy increase the IQ of children at school age? Follow-up of a randomised controlled trial. BMJ Open. 2016;6(5):e011465. (PubMed)
64. Muhlhausler BS, Yelland LN, McDermott R, et al. DHA supplementation during pregnancy does not reduce BMI or body fat mass in children: follow-up of the DHA to Optimize Mother Infant Outcome randomized controlled trial. Am J Clin Nutr. 2016;103(6):1489-1496. (PubMed)
65. Wood K, Mantzioris E, Lingwood B, et al. The effect of maternal DHA supplementation on body fat mass in children at 7 years: follow-up of the DOMInO randomized controlled trial. Prostaglandins Leukot Essent Fatty Acids. 2018;139:49-54. (PubMed)
66. Vahdaninia M, Mackenzie H, Dean T, Helps S. The effectiveness of omega-3 polyunsaturated fatty acid interventions during pregnancy on obesity measures in the offspring: an up-to-date systematic review and meta-analysis. Eur J Nutr. 2018; doi: 10.1007/s00394-018-1824-9. [Epub ahead of print]. (PubMed)
67. Lin J, Zhang Y, Zhu X, Wang D, Dai J. Effects of supplementation with omega-3 fatty acids during pregnancy on asthma or wheeze of children: a systematic review and meta-analysis. J Matern Fetal Neonatal Med. 2018:1-10. (PubMed)
68. Vahdaninia M, Mackenzie H, Dean T, Helps S. Omega-3 LCPUFA supplementation during pregnancy and risk of allergic outcomes or sensitization in offspring: A systematic review and meta-analysis. Ann Allergy Asthma Immunol. 2019;122(3):302-313.e302. (PubMed)
69. Delgado-Noguera MF, Calvache JA, Bonfill Cosp X, Kotanidou EP, Galli-Tsinopoulou A. Supplementation with long chain polyunsaturated fatty acids (LCPUFA) to breastfeeding mothers for improving child growth and development. Cochrane Database Syst Rev. 2015(7):Cd007901. (PubMed)
70. Guesnet P, Alessandri JM. Docosahexaenoic acid (DHA) and the developing central nervous system (CNS) - Implications for dietary recommendations. Biochimie. 2011;93(1):7-12. (PubMed)
71. Gibson RA, Kneebone GM. Fatty acid composition of human colostrum and mature breast milk. Am J Clin Nutr. 1981;34(2):252-257. (PubMed)
72. Larque E, Demmelmair H, Koletzko B. Perinatal supply and metabolism of long-chain polyunsaturated fatty acids: importance for the early development of the nervous system. Ann N Y Acad Sci. 2002;967:299-310. (PubMed)
73. Qawasmi A, Landeros-Weisenberger A, Leckman JF, Bloch MH. Meta-analysis of long-chain polyunsaturated fatty acid supplementation of formula and infant cognition. Pediatrics. 2012;129(6):1141-1149. (PubMed)
74. Qawasmi A, Landeros-Weisenberger A, Bloch MH. Meta-analysis of LCPUFA supplementation of infant formula and visual acuity. Pediatrics. 2013;131(1):e262-272. (PubMed)
75. Makrides M, Gibson RA, McPhee AJ, et al. Neurodevelopmental outcomes of preterm infants fed high-dose docosahexaenoic acid: a randomized controlled trial. JAMA. 2009;301(2):175-182. (PubMed)
76. Collins CT, Gibson RA, Anderson PJ, et al. Neurodevelopmental outcomes at 7 years' corrected age in preterm infants who were fed high-dose docosahexaenoic acid to term equivalent: a follow-up of a randomised controlled trial. BMJ Open. 2015;5(3):e007314. (PubMed)
77. Moon K, Rao SC, Schulzke SM, Patole SK, Simmer K. Longchain polyunsaturated fatty acid supplementation in preterm infants. Cochrane Database Syst Rev. 2016;12:Cd000375. (PubMed)
78. US Department of Agriculture. Agricultural Research Service. Nutrient intakes from food: mean amounts consumed per individual, by gender and age. Available at: www.ars.usda.gov/ba/bhnrc/fsrg. Accessed 4/25/14.
79. Farvid MS, Ding M, Pan A, et al. Dietary linoleic acid and risk of coronary heart disease: a systematic review and meta-analysis of prospective cohort studies. Circulation. 2014;130(18):1568-1578. (PubMed)
80. Marklund M, Wu JHY, Imamura F, et al. Biomarkers of dietary omega-6 fatty acids and incident cardiovascular disease and mortality: an individual-level pooled analysis of 30 cohort studies. Circulation. 2019; 139(21):2422-2436. (PubMed)
81. UK Medical Research Council. Controlled trial of soya-bean oil in myocardial infarction. Lancet. 1968;2(7570):693-699. (PubMed)
82. Dayton S, Pearce ML, Hashimoto S, Dixon WJ, Tomiyasu U. A controlled clinical trial of a diet high in unsaturated fat in preventing complications of atherosclerosis. Circulation. 1969;40(1s2):II-1-II-63.
83. Leren P. The Oslo diet-heart study. Eleven-year report. Circulation. 1970;42(5):935-942. (PubMed)
84. Miettinen M, Turpeinen O, Karvonen MJ, Pekkarinen M, Paavilainen E, Elosuo R. Dietary prevention of coronary heart disease in women: the Finnish mental hospital study. Int J Epidemiol. 1983;12(1):17-25. (PubMed)
85. Turpeinen O, Karvonen MJ, Pekkarinen M, Miettinen M, Elosuo R, Paavilainen E. Dietary prevention of coronary heart disease: the Finnish Mental Hospital Study. Int J Epidemiol. 1979;8(2):99-118. (PubMed)
86. Sacks FM, Lichtenstein AH, Wu JHY, et al. Dietary fats and cardiovascular disease: a presidential advisory from the American Heart Association. Circulation. 2017;136(3):e1-e23. (PubMed)
87. Hooper L, Al-Khudairy L, Abdelhamid AS, et al. Omega-6 fats for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst Rev. 2018;11:Cd011094. (PubMed)
88. Hamley S. The effect of replacing saturated fat with mostly n-6 polyunsaturated fat on coronary heart disease: a meta-analysis of randomised controlled trials. Nutr J. 2017;16(1):30. (PubMed)
89. Mensink RP, World Health Organization. Effects of saturated fatty acids on serum lipids and lipoproteins: a systematic review and regression analysis. 2016.
90. Mensink RP, Katan MB. Effect of dietary fatty acids on serum lipids and lipoproteins. A meta-analysis of 27 trials. Arterioscler Thromb. 1992;12(8):911-919. (PubMed)
91. Fernandez ML, West KL. Mechanisms by which dietary fatty acids modulate plasma lipids. J Nutr. 2005;135(9):2075-2078. (PubMed)
92. Ramsden CE, Zamora D, Leelarthaepin B, et al. Use of dietary linoleic acid for secondary prevention of coronary heart disease and death: evaluation of recovered data from the Sydney Diet Heart Study and updated meta-analysis. BMJ. 2013;346:e8707. (PubMed)
93. Del Gobbo LC, Imamura F, Aslibekyan S, et al. Omega-3 polyunsaturated fatty Acid biomarkers and coronary heart disease: pooling project of 19 cohort studies. JAMA Intern Med. 2016;176(8):1155-1166. (PubMed)
94. Wei J, Hou R, Xi Y, et al. The association and dose-response relationship between dietary intake of alpha-linolenic acid and risk of CHD: a systematic review and meta-analysis of cohort studies. Br J Nutr. 2018;119(1):83-89. (PubMed)
95. Rimm EB, Appel LJ, Chiuve SE, et al. Seafood long-chain n-3 polyunsaturated fatty acids and cardiovascular disease: a science advisory from the American Heart Association. Circulation. 2018;138(1):e35-e47. (PubMed)
96. Bechthold A, Boeing H, Schwedhelm C, et al. Food groups and risk of coronary heart disease, stroke and heart failure: A systematic review and dose-response meta-analysis of prospective studies. Crit Rev Food Sci Nutr. 2017:1-20. (PubMed)
97. Zhao W, Tang H, Yang X, et al. Fish consumption and stroke risk: a meta-analysis of prospective cohort studies. J Stroke Cerebrovasc Dis. 2019;28(3):604-611. (PubMed)
98. Jayedi A, Zargar MS, Shab-Bidar S. Fish consumption and risk of myocardial infarction: a systematic review and dose-response meta-analysis suggests a regional difference. Nutr Res. 2019;62:1-12. (PubMed)
99. Zhao LG, Sun JW, Yang Y, Ma X, Wang YY, Xiang YB. Fish consumption and all-cause mortality: a meta-analysis of cohort studies. Eur J Clin Nutr. 2016;70(2):155-161. (PubMed)
100. Jayedi A, Shab-Bidar S, Eimeri S, Djafarian K. Fish consumption and risk of all-cause and cardiovascular mortality: a dose-response meta-analysis of prospective observational studies. Public Health Nutr. 2018;21(7):1297-1306. (PubMed)
101. Chowdhury R, Stevens S, Gorman D, et al. Association between fish consumption, long chain omega 3 fatty acids, and risk of cerebrovascular disease: systematic review and meta-analysis. BMJ. 2012;345:e6698. (PubMed)
102. Mozaffarian D, Appel LJ, Van Horn L. Components of a cardioprotective diet: new insights. Circulation. 2011;123(24):2870-2891. (PubMed)
103. Abdelhamid AS, Brown TJ, Brainard JS, et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst Rev. 2018;11:Cd003177. (PubMed)
104. Siscovick DS, Barringer TA, Fretts AM, et al. Omega-3 polyunsaturated fatty acid (fish oil) supplementation and the prevention of clinical cardiovascular disease: a science advisory from the American Heart Association. Circulation. 2017;135(15):e867-e884. (PubMed)
105. Di Angelantonio E, Sarwar N, Perry P, et al. Major lipids, apolipoproteins, and risk of vascular disease. JAMA. 2009;302(18):1993-2000. (PubMed)
106. Balk EM, Lichtenstein AH, Chung M, Kupelnick B, Chew P, Lau J. Effects of omega-3 fatty acids on serum markers of cardiovascular disease risk: a systematic review. Atherosclerosis. 2006;189(1):19-30. (PubMed)
107. Kris-Etherton PM, Harris WS, Appel LJ. Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Circulation. 2002;106(21):2747-2757. (PubMed)
108. Leaf A, Xiao YF, Kang JX, Billman GE. Prevention of sudden cardiac death by n-3 polyunsaturated fatty acids. Pharmacol Ther. 2003;98(3):355-377. (PubMed)
109. Mozaffarian D, Wu JH. Omega-3 fatty acids and cardiovascular disease: effects on risk factors, molecular pathways, and clinical events. J Am Col Cardiol. 2011;58(20):2047-2067. (PubMed)
110. Howard BV. Lipoprotein metabolism in diabetes mellitus. J Lipid Res. 1987;28(6):613-628. (PubMed)
111. Khavandi M, Duarte F, Ginsberg HN, Reyes-Soffer G. Treatment of dyslipidemias to prevent cardiovascular disease in patients with type 2 diabetes. Curr Cardiol Rep. 2017;19(1):7. (PubMed)
112. Scicali R, Di Pino A, Ferrara V, et al. New treatment options for lipid-lowering therapy in subjects with type 2 diabetes. Acta Diabetol. 2018;55(3):209-218. (PubMed)
113. Patel A, MacMahon S, Chalmers J, et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med. 2008;358(24):2560-2572. (PubMed)
114. Zheng T, Zhao J, Wang Y, et al. The limited effect of omega-3 polyunsaturated fatty acids on cardiovascular risk in patients with impaired glucose metabolism: a meta-analysis. Clin Biochem. 2014;47(6):369-377. (PubMed)
115. Kromhout D, Giltay EJ, Geleijnse JM. n-3 fatty acids and cardiovascular events after myocardial infarction. New Engl J Med. 2010;363(21):2015-2026. (PubMed)
116. Bosch J, Gerstein HC, Dagenais GR, et al. n-3 fatty acids and cardiovascular outcomes in patients with dysglycemia. New Engl J Med. 2012;367(4):309-318. (PubMed)
117. O'Mahoney LL, Matu J, Price OJ, et al. Omega-3 polyunsaturated fatty acids favourably modulate cardiometabolic biomarkers in type 2 diabetes: a meta-analysis and meta-regression of randomized controlled trials. Cardiovasc Diabetol. 2018;17(1):98. (PubMed)
118. Fox CS, Golden SH, Anderson C, et al. Update on prevention of cardiovascular disease in adults with type 2 diabetes mellitus in light of recent evidence: a scientific statement from the American Heart Association and the American Diabetes Association. Diabetes Care. 2015;38(9):1777-1803. (PubMed)
119. American Diabetes Association. Management of diabetes in pregnancy: standards of medical care in diabetes-2019. Diabetes Care. 2019;42(Suppl 1):S165-s172. (PubMed)
120. Samimi M, Jamilian M, Asemi Z, Esmaillzadeh A. Effects of omega-3 fatty acid supplementation on insulin metabolism and lipid profiles in gestational diabetes: Randomized, double-blind, placebo-controlled trial. Clin Nutr. 2015;34(3):388-393. (PubMed)
121. Taghizadeh M, Jamilian M, Mazloomi M, Sanami M, Asemi Z. A randomized-controlled clinical trial investigating the effect of omega-3 fatty acids and vitamin E co-supplementation on markers of insulin metabolism and lipid profiles in gestational diabetes. J Clin Lipidol. 2016;10(2):386-393. (PubMed)
122. Jamilian M, Samimi M, Ebrahimi FA, et al. The effects of vitamin D and omega-3 fatty acid co-supplementation on glycemic control and lipid concentrations in patients with gestational diabetes. J Clin Lipidol. 2017;11(2):459-468. (PubMed)
123. Wanders AJ, Blom WAM, Zock PL, Geleijnse JM, Brouwer IA, Alssema M. Plant-derived polyunsaturated fatty acids and markers of glucose metabolism and insulin resistance: a meta-analysis of randomized controlled feeding trials. BMJ Open Diabetes Res Care. 2019;7(1):e000585. (PubMed)
124. Wu JHY, Marklund M, Imamura F, et al. Omega-6 fatty acid biomarkers and incident type 2 diabetes: pooled analysis of individual-level data for 39 740 adults from 20 prospective cohort studies. Lancet Diabetes Endocrinol. 2017;5(12):965-974. (PubMed)
125. Hodson L, Skeaff CM, Fielding BA. Fatty acid composition of adipose tissue and blood in humans and its use as a biomarker of dietary intake. Prog Lipid Res. 2008;47(5):348-380. (PubMed)
126. Jang H, Park K. Omega-3 and omega-6 polyunsaturated fatty acids and metabolic syndrome: A systematic review and meta-analysis. Clin Nutr. 2019; doi: 10.1016/j.clnu.2019.03.032. [Epub ahead of print]. (PubMed)
127. US Centers for Disease Control and Prevention (CDC). Alzheimer's Disease. October 1, 2018. Available at: https://www.cdc.gov/aging/aginginfo/alzheimers.htm. Accessed 4/30/19.
128. Maccioni RB, Munoz JP, Barbeito L. The molecular bases of Alzheimer's disease and other neurodegenerative disorders. Arch Med Res. 2001;32(5):367-381. (PubMed)
129. Samieri C, Morris MC, Bennett DA, et al. Fish intake, genetic predisposition to Alzheimer disease, and decline in global cognition and memory in 5 cohorts of older persons. Am J Epidemiol. 2018;187(5):933-940. (PubMed)
130. Huang TL, Zandi PP, Tucker KL, et al. Benefits of fatty fish on dementia risk are stronger for those without APOE epsilon4. Neurology. 2005;65(9):1409-1414. (PubMed)
131. Whalley LJ, Deary IJ, Starr JM, et al. n-3 Fatty acid erythrocyte membrane content, APOE varepsilon4, and cognitive variation: an observational follow-up study in late adulthood. Am J Clin Nutr. 2008;87(2):449-454. (PubMed)
132. Corder EH, Saunders AM, Strittmatter WJ, et al. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer's disease in late onset families. Science. 1993;261(5123):921-923. (PubMed)
133. Plourde M, Vohl MC, Vandal M, Couture P, Lemieux S, Cunnane SC. Plasma n-3 fatty acid response to an n-3 fatty acid supplement is modulated by apoE epsilon4 but not by the common PPAR-alpha L162V polymorphism in men. Br J Nutr. 2009;102(8):1121-1124. (PubMed)
134. Chouinard-Watkins R, Rioux-Perreault C, Fortier M, et al. Disturbance in uniformly 13C-labelled DHA metabolism in elderly human subjects carrying the apoE epsilon4 allele. Br J Nutr. 2013;110(10):1751-1759. (PubMed)
135. Zhang Y, Chen J, Qiu J, Li Y, Wang J, Jiao J. Intakes of fish and polyunsaturated fatty acids and mild-to-severe cognitive impairment risks: a dose-response meta-analysis of 21 cohort studies. Am J Clin Nutr. 2016;103(2):330-340. (PubMed)
136. D'Ascoli TA, Mursu J, Voutilainen S, Kauhanen J, Tuomainen TP, Virtanen JK. Association between serum long-chain omega-3 polyunsaturated fatty acids and cognitive performance in elderly men and women: The Kuopio Ischaemic Heart Disease Risk Factor Study. Eur J Clin Nutr. 2016;70(8):970-975. (PubMed)
137. van der Lee SJ, Teunissen CE, Pool R, et al. Circulating metabolites and general cognitive ability and dementia: Evidence from 11 cohort studies. Alzheimers Dement. 2018;14(6):707-722. (PubMed)
138. Fotuhi M, Mohassel P, Yaffe K. Fish consumption, long-chain omega-3 fatty acids and risk of cognitive decline or Alzheimer disease: a complex association. Nat Clin Pract Neurol. 2009;5(3):140-152. (PubMed)
139. Sydenham E, Dangour AD, Lim WS. Omega 3 fatty acid for the prevention of cognitive decline and dementia. Cochrane Database Syst Rev. 2012(6):Cd005379. (PubMed)
140. Rangel-Huerta OD, Gil A. Effect of omega-3 fatty acids on cognition: an updated systematic review of randomized clinical trials. Nutr Rev. 2018;76(1):1-20. (PubMed)
141. Jackson PA, Forster JS, Bell JG, Dick JR, Younger I, Kennedy DO. DHA supplementation alone or in combination with other nutrients does not modulate cerebral hemodynamics or cognitive function in healthy older adults. Nutrients. 2016;8(2):86. (PubMed)
142. Kobe T, Witte AV, Schnelle A, et al. Combined omega-3 fatty acids, aerobic exercise and cognitive stimulation prevents decline in gray matter volume of the frontal, parietal and cingulate cortex in patients with mild cognitive impairment. Neuroimage. 2016;131:226-238. (PubMed)
143. Miller M, Stone NJ, Ballantyne C, et al. Triglycerides and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2011;123(20):2292-2333. (PubMed)
144. Benes LB, Bassi NS, Davidson MH. Omega-3 carboxylic acids monotherapy and combination with statins in the management of dyslipidemia. Vasc Health Risk Manag. 2016;12:481-490. (PubMed)
145. Kastelein JJ, Maki KC, Susekov A, et al. Omega-3 free fatty acids for the treatment of severe hypertriglyceridemia: the EpanoVa fOr Lowering Very high triglyceridEs (EVOLVE) trial. J Clin Lipidol. 2014;8(1):94-106. (PubMed)
146. Maki KC, Orloff DG, Nicholls SJ, et al. A highly bioavailable omega-3 free fatty acid formulation improves the cardiovascular risk profile in high-risk, statin-treated patients with residual hypertriglyceridemia (the ESPRIT trial). Clin Ther. 2013;35(9):1400-1411.e1401-1403. (PubMed)
147. Nicholls SJ, Brandrup-Wognsen G, Palmer M, Barter PJ. Meta-analysis of comparative efficacy of increasing dose of atorvastatin versus rosuvastatin versus simvastatin on lowering levels of atherogenic lipids (from VOYAGER). Am J Cardiol. 2010;105(1):69-76. (PubMed)
148. Ballantyne CM, Bays HE, Kastelein JJ, et al. Efficacy and safety of eicosapentaenoic acid ethyl ester (AMR101) therapy in statin-treated patients with persistent high triglycerides (from the ANCHOR study). Am J Cardiol. 2012;110(7):984-992. (PubMed)
149. Bays HE, McKenney J, Maki KC, Doyle RT, Carter RN, Stein E. Effects of prescription omega-3-acid ethyl esters on non--high-density lipoprotein cholesterol when coadministered with escalating doses of atorvastatin. Mayo Clin Proc. 2010;85(2):122-128. (PubMed)
150. Davidson MH, Stein EA, Bays HE, et al. Efficacy and tolerability of adding prescription omega-3 fatty acids 4 g/d to simvastatin 40 mg/d in hypertriglyceridemic patients: an 8-week, randomized, double-blind, placebo-controlled study. Clin Ther. 2007;29(7):1354-1367. (PubMed)
151. ClinicalTrials.gov. Outcomes study to assess statin residual risk reduction with Epanova in high CV risk patients with hypertriglyceridemia (STRENGTH). Available at: https://clinicaltrials.gov/ct2/show/NCT02104817. Accessed 5/24/19.
152. Michelotti GA, Machado MV, Diehl AM. NAFLD, NASH and liver cancer. Nat Rev Gastroenterol Hepatol. 2013;10(11):656-665. (PubMed)
153. Spooner MH, Jump DB. Omega-3 fatty acids and nonalcoholic fatty liver disease in adults and children: where do we stand? Curr Opin Clin Nutr Metab Care. 2019;22(2):103-110. (PubMed)
154. Arendt BM, Comelli EM, Ma DW, et al. Altered hepatic gene expression in nonalcoholic fatty liver disease is associated with lower hepatic n-3 and n-6 polyunsaturated fatty acids. Hepatology. 2015;61(5):1565-1578. (PubMed)
155. Yan JH, Guan BJ, Gao HY, Peng XE. Omega-3 polyunsaturated fatty acid supplementation and non-alcoholic fatty liver disease: A meta-analysis of randomized controlled trials. Medicine (Baltimore). 2018;97(37):e12271. (PubMed)
156. Gioxari A, Kaliora AC, Marantidou F, Panagiotakos DP. Intake of omega-3 polyunsaturated fatty acids in patients with rheumatoid arthritis: A systematic review and meta-analysis. Nutrition. 2018;45:114-124.e114. (PubMed)
157. Senftleber NK, Nielsen SM, Andersen JR, et al. Marine oil supplements for arthritis pain: a systematic review and meta-analysis of randomized trials. Nutrients. 2017;9(1). (PubMed)
158. Abdulrazaq M, Innes JK, Calder PC. Effect of omega-3 polyunsaturated fatty acids on arthritic pain: A systematic review. Nutrition. 2017;39-40:57-66. (PubMed)
159. Lau CS, Morley KD, Belch JJ. Effects of fish oil supplementation on non-steroidal anti-inflammatory drug requirement in patients with mild rheumatoid arthritis--a double-blind placebo controlled study. Br J Rheumatol. 1993;32(11):982-989. (PubMed)
160. Skoldstam L, Borjesson O, Kjallman A, Seiving B, Akesson B. Effect of six months of fish oil supplementation in stable rheumatoid arthritis. A double-blind, controlled study. Scand J Rheumatol. 992;21(4):178-185. (PubMed)
161. Swan K, Allen PJ. Omega-3 fatty acid for the treatment and remission of Crohn's disease. J Complement Integr Med. 2013;10. (PubMed)
162. Feagan BG, Sandborn WJ, Mittmann U, et al. Omega-3 free fatty acids for the maintenance of remission in Crohn disease: the EPIC Randomized Controlled Trials. JAMA. 2008;299(14):1690-1697. (PubMed)
163. Cabre E, Manosa M, Gassull MA. Omega-3 fatty acids and inflammatory bowel diseases - a systematic review. Br J Nutr. 2012;107 Suppl 2:S240-252. (PubMed)
164. Lev-Tzion R, Griffiths AM, Leder O, Turner D. Omega 3 fatty acids (fish oil) for maintenance of remission in Crohn's disease. Cochrane Database Syst Rev. 2014(2):Cd006320. (PubMed)
165. Turner D, Shah PS, Steinhart AH, Zlotkin S, Griffiths AM. Maintenance of remission in inflammatory bowel disease using omega-3 fatty acids (fish oil): a systematic review and meta-analyses. Inflamm Bowel Dis. 2011;17(1):336-345. (PubMed)
166. Hodge L, Salome CM, Hughes JM, et al. Effect of dietary intake of omega-3 and omega-6 fatty acids on severity of asthma in children. Eur Respir J. 1998;11(2):361-365. (PubMed)
167. Okamoto M, Mitsunobu F, Ashida K, et al. Effects of dietary supplementation with n-3 fatty acids compared with n-6 fatty acids on bronchial asthma. Intern Med. 2000;39(2):107-111. (PubMed)
168. Wong KW. Clinical efficacy of n-3 fatty acid supplementation in patients with asthma. J Am Diet Assoc. 2005;105(1):98-105. (PubMed)
169. Schachter HM, Reisman J, Tran K, et al. Health effects of omega-3 fatty acids on asthma. Evid Rep Technol Assess (Summ). 2004(91):1-7. (PubMed)
170. Woods RK, Thien FC, Abramson MJ. Dietary marine fatty acids (fish oil) for asthma in adults and children. Cochrane Database Syst Rev. 2002(3):CD001283. (PubMed)
171. Reisman J, Schachter HM, Dales RE, et al. Treating asthma with omega-3 fatty acids: where is the evidence? A systematic review. BMC Complement Altern Med. 2006;6:26. (PubMed)
172. Donadio JV, Grande JP. IgA nephropathy. N Engl J Med. 2002;347(10):738-748. (PubMed)
173. Liu LL, Wang LN. Omega-3 fatty acids therapy for IgA nephropathy: a meta-analysis of randomized controlled trials. Clin Nephrol. 2012;77(2):119-125. (PubMed)
174. Hirahashi J. Omega-3 polyunsaturated fatty acids for the treatment of IgA nephropathy. J Clin Med. 2017;6(7). (PubMed)
175. Brigandi SA, Shao H, Qian SY, Shen Y, Wu BL, Kang JX. Autistic children exhibit decreased levels of essential fatty acids in red blood cells. Int J Mol Sci. 2015;16(5):10061-10076. (PubMed)
176. Mazahery H, Stonehouse W, Delshad M, et al. Relationship between long chain n-3 polyunsaturated fatty acids and autism spectrum disorder: systematic review and meta-analysis of case-control and randomised controlled trials. Nutrients. 2017;9(2). (PubMed)
177. Yui K, Koshiba M, Nakamura S, Kobayashim Y, Ohnishi M. Efficacy of adding large doses of arachidonic acid to docosahexaenoic acid against restricted repetitive behaviors in individuals with autism spectrum disorders: a placebo-controlled trial. J Addict Res Ther. 2011;S4(006).
178. Amminger GP, Berger GE, Schafer MR, Klier C, Friedrich MH, Feucht M. Omega-3 fatty acids supplementation in children with autism: a double-blind randomized, placebo-controlled pilot study. Biol Psychiatry. 2007;61(4):551-553. (PubMed)
179. Bent S, Bertoglio K, Ashwood P, Bostrom A, Hendren RL. A pilot randomized controlled trial of omega-3 fatty acids for autism spectrum disorder. J Autism Dev Disord. 2011;41(5):545-554. (PubMed)
180. Bent S, Hendren RL, Zandi T, et al. Internet-based, randomized, controlled trial of omega-3 fatty acids for hyperactivity in autism. J Am Acad Child Adolesc Psychiatry. 2014;53(6):658-666. (PubMed)
181. Mankad D, Dupuis A, Smile S, et al. A randomized, placebo controlled trial of omega-3 fatty acids in the treatment of young children with autism. Mol Autism. 2015;6:18. (PubMed)
182. Voigt RG, Mellon MW, Katusic SK, et al. Dietary docosahexaenoic acid supplementation in children with autism. J Pediatr Gastroenterol Nutr. 2014;58(6):715-722. (PubMed)
183. Cheng YS, Tseng PT, Chen YW, et al. Supplementation of omega 3 fatty acids may improve hyperactivity, lethargy, and stereotypy in children with autism spectrum disorders: a meta-analysis of randomized controlled trials. Neuropsychiatr Dis Treat. 2017;13:2531-2543. (PubMed)
184. Horvath A, Lukasik J, Szajewska H. Omega-3 fatty acid supplementation does not affect autism spectrum disorder in children: a systematic review and meta-analysis. J Nutr. 2017;147(3):367-376. (PubMed)
185. Hibbeln JR. Fish consumption and major depression. Lancet. 1998;351(9110):1213. (PubMed)
186. Noaghiul S, Hibbeln JR. Cross-national comparisons of seafood consumption and rates of bipolar disorders. Am J Psychiatry. 2003;160(12):2222-2227. (PubMed)
187. Maes M, Christophe A, Delanghe J, Altamura C, Neels H, Meltzer HY. Lowered omega3 polyunsaturated fatty acids in serum phospholipids and cholesteryl esters of depressed patients. Psychiatry Res. 1999;85(3):275-291. (PubMed)
188. Peet M, Murphy B, Shay J, Horrobin D. Depletion of omega-3 fatty acid levels in red blood cell membranes of depressive patients. Biol Psychiatry. 1998;43(5):315-319. (PubMed)
189. Tiemeier H, van Tuijl HR, Hofman A, Kiliaan AJ, Breteler MM. Plasma fatty acid composition and depression are associated in the elderly: the Rotterdam Study. Am J Clin Nutr. 2003;78(1):40-46. (PubMed)
190. Mamalakis G, Tornaritis M, Kafatos A. Depression and adipose essential polyunsaturated fatty acids. Prostaglandins Leukot Essent Fatty Acids. 2002;67(5):311-318. (PubMed)
191. Locke CA, Stoll AL. Omega-3 fatty acids in major depression. World Rev Nutr Diet. 2001;89:173-185. (PubMed)
192. Ortega RM, Rodriguez-Rodriguez E, Lopez-Sobaler AM. Effects of omega 3 fatty acids supplementation in behavior and non-neurodegenerative neuropsychiatric disorders. Br J Nutr. 2012;107 Suppl 2:S261-270. (PubMed)
193. Prior PL, Galduroz JC. (N-3) Fatty acids: molecular role and clinical uses in psychiatric disorders. Adv Nutr. 2012;3(3):257-265. (PubMed)
194. Grosso G, Pajak A, Marventano S, et al. Role of omega-3 fatty acids in the treatment of depressive disorders: a comprehensive meta-analysis of randomized clinical trials. PLoS One. 2014;9(5):e96905. (PubMed)
195. Appleton KM, Sallis HM, Perry R, Ness AR, Churchill R. Omega-3 fatty acids for major depressive disorder in adults: an abridged Cochrane review. BMJ Open. 2016;6(3):e010172. (PubMed)
196. McNamara RK, Welge JA. Meta-analysis of erythrocyte polyunsaturated fatty acid biostatus in bipolar disorder. Bipolar Disord. 2016;18(3):300-306. (PubMed)
197. Shakeri J, Khanegi M, Golshani S, et al. Effects of omega-3 supplement in the treatment of patients with bipolar I disorder. Int J Prev Med. 2016;7:77. (PubMed)
198. Hoen WP, Lijmer JG, Duran M, Wanders RJ, van Beveren NJ, de Haan L. Red blood cell polyunsaturated fatty acids measured in red blood cells and schizophrenia: a meta-analysis. Psychiatry Res. 2013;207(1-2):1-12. (PubMed)
199. Akter K, Gallo DA, Martin SA, et al. A review of the possible role of the essential fatty acids and fish oils in the aetiology, prevention or pharmacotherapy of schizophrenia. J Clin Pharm Ther. 2012;37(2):132-139. (PubMed)
200. Robinson DG, Gallego JA, John M, et al. A potential role for adjunctive omega-3 polyunsaturated fatty acids for depression and anxiety symptoms in recent onset psychosis: Results from a 16week randomized placebo-controlled trial for participants concurrently treated with risperidone. Schizophr Res. 2019;204:295-303. (PubMed)
201. Burckhardt M, Herke M, Wustmann T, Watzke S, Langer G, Fink A. Omega-3 fatty acids for the treatment of dementia. Cochrane Database Syst Rev. 2016;4:Cd009002. (PubMed)
202. Freund-Levi Y, Eriksdotter-Jonhagen M, Cederholm T, et al. Omega-3 fatty acid treatment in 174 patients with mild to moderate Alzheimer disease: OmegAD study: a randomized double-blind trial. Arch Neurol. 2006;63(10):1402-1408. (PubMed)
203. Shinto L, Quinn J, Montine T, et al. A randomized placebo-controlled pilot trial of omega-3 fatty acids and alpha lipoic acid in Alzheimer's disease. J Alzheimers Dis. 2014;38(1):111-120. (PubMed)
204. Quinn JF, Raman R, Thomas RG, et al. Docosahexaenoic acid supplementation and cognitive decline in Alzheimer disease: a randomized trial. JAMA. 2010;304(17):1903-1911. (PubMed)
205. US Department of Agriculture ARS. USDA National Nutrient Database for Standard Reference, Release 26. Available at: http://www.ars.usda.gov/ba/bhnrc/ndl. Accessed 4/25/14.
206. Hendler SS, Rorvik DM. PDR for nutritional supplements: Thomson Reuters New Jersey; 2008.
207. Ulven SM, Kirkhus B, Lamglait A, et al. Metabolic effects of krill oil are essentially similar to those of fish oil but at lower dose of EPA and DHA, in healthy volunteers. Lipids. 2011;46(1):37-46. (PubMed)
208. US Food and Drug Administration. CfFSaAN. Agency Response Letter: GRAS Notice No. GRN 000080. 2001. Available at: http://www.cfsan.fda.gov/~rdb/opa-g080.html. Accessed 11/7/08.
209. Zurier RB, Rossetti RG, Jacobson EW, et al. gamma-Linolenic acid treatment of rheumatoid arthritis. A randomized, placebo-controlled trial. Arthritis Rheum. 1996;39(11):1808-1817. (PubMed)
210. Vaddadi KS. The use of gamma-linolenic acid and linoleic acid to differentiate between temporal lobe epilepsy and schizophrenia. Prostaglandins Med. 1981;6(4):375-379. (PubMed)
211. Nordstrom DC, Honkanen VE, Nasu Y, Antila E, Friman C, Konttinen YT. Alpha-linolenic acid in the treatment of rheumatoid arthritis. A double-blind, placebo-controlled and randomized study: flaxseed vs. safflower seed. Rheumatol Int. 1995;14(6):231-234. (PubMed)
212. Alonso L, Marcos ML, Blanco JG, et al. Anaphylaxis caused by linseed (flaxseed) intake. J Allergy Clin Immunol. 1996;98(2):469-470. (PubMed)
213. Harbige LS. Fatty acids, the immune response, and autoimmunity: a question of n-6 essentiality and the balance between n-6 and n-3. Lipids. 2003;38(4):323-341. (PubMed)
214. Carlson SE, Cooke RJ, Werkman SH, Tolley EA. First year growth of preterm infants fed standard compared to marine oil n-3 supplemented formula. Lipids. 1992;27(11):901-907. (PubMed)
215. Carlson SE, Werkman SH, Tolley EA. Effect of long-chain n-3 fatty acid supplementation on visual acuity and growth of preterm infants with and without bronchopulmonary dysplasia. Am J Clin Nutr. 1996;63(5):687-697. (PubMed)
216. Schulzke SM, Patole SK, Simmer K. Long-chain polyunsaturated fatty acid supplementation in preterm infants. Cochrane Database Syst Rev. 2011(2):CD000375. (PubMed)
217. Hendler SS, Rorvik DR, eds. PDR for Nutritional Supplements. Montvale: Medical Economics Company, Inc; 2001.
218. Mozaffarian D, Rimm EB. Fish intake, contaminants, and human health: evaluating the risks and the benefits. JAMA. 2006;296(15):1885-1899. (PubMed)
219. Environmental Protection Agency. Fish Advisories [Web site]. April 14, 2003. Available at: http://www.epa.gov/waterscience/fish/. Accessed 4/28/03.
220. 2015-2020 Dietary Guidelines for Americans. Available at: https://health.gov/dietaryguidelines/2015/guidelines/. Accessed 5/24/19.
221. Consumer Lab. Product Review: FIsh oil and omega-3 fatty acid supplements review (including krill, algae, calamari, green-lipped mussel oil). Available at: http://www.consumerlab.com/results/omega3.asp. Accessed 4/25/14.
222. Hilbert G, Lillemark L, Balchen S, Hojskov CS. Reduction of organochlorine contaminants from fish oil during refining. Chemosphere. 1998;37(7):1241-1252. (PubMed)
223. Bender NK, Kraynak MA, Chiquette E, Linn WD, Clark GM, Bussey HI. Effects of marine fish oils on the anticoagulation status of patients receiving chronic warfarin therapy. J Thromb Thrombolysis. 1998;5(3):257-261. (PubMed)
224. Buckley MS, Goff AD, Knapp WE. Fish oil interaction with warfarin. Ann Pharmacother. 2004;38(1):50-52. (PubMed)
225. Valk EE, Hornstra G. Relationship between vitamin E requirement and polyunsaturated fatty acid intake in man: a review. Int J Vitam Nutr Res. 2000;70(2):31-42. (PubMed)
226. Higdon JV, Liu J, Du SH, Morrow JD, Ames BN, Wander RC. Supplementation of postmenopausal women with fish oil rich in eicosapentaenoic acid and docosahexaenoic acid is not associated with greater in vivo lipid peroxidation compared with oils rich in oleate and linoleate as assessed by plasma malondialdehyde and F(2)-isoprostanes. Am J Clin Nutr. 2000;72(3):714-722. (PubMed)
227. Wander RC, Du SH, Ketchum SO, Rowe KE. alpha-Tocopherol influences in vivo indices of lipid peroxidation in postmenopausal women given fish oil. J Nutr. 1996;126(3):643-652. (PubMed)
228. Wander RC, Du SH. Oxidation of plasma proteins is not increased after supplementation with eicosapentaenoic and docosahexaenoic acids. Am J Clin Nutr. 2000;72(3):731-737. (PubMed)
229. Larque E, Gil-Sanchez A, Prieto-Sanchez MT, Koletzko B. Omega 3 fatty acids, gestation and pregnancy outcomes. Br J Nutr. 2012;107 Suppl 2:S77-84. (PubMed)
230. American Heart Association. Frequently Asked Questions About Fish. Available at: http://www.heart.org/HEARTORG/General/Frequently-Asked-Questions-About-Fish_UCM_306451_Article.jsp. Accessed 4/25/14.
231. EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). Scientific opinion on dietary reference values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids, and cholesterol. EFSA Journal. 2010;8(3):107.
232. European Food Safety Authority. The DRV Finder. Available at: https://www.efsa.europa.eu/en/interactive-pages/drvs. Accessed 5/24/19.
233. FAO/WHO. Interim Summary of Conclusions and Dietary Recommendations on Total Fat & Fatty Acids. Joint FAO/WHO Expert Consultation on Fats and Fatty Acids in Human Nutrition. Geneva: WHO; 2008:1-14.
234. International Society for the Study of Fatty Acids and Lipids (ISSFAL). Recommendations for intake of polyunsaturated fatty acids in healthy adults. Available at: https://www.issfal.org/statement-3. Accessed 4/24/19.