Food and Beverages
Plant-based foods, including fruit, vegetables, legumes, whole grains, and nuts, are prominent features of healthy dietary patterns. In addition to providing energy and essential micronutrients, plant-based foods contribute thousands of biologically active phytochemicals (plant chemicals that may affect health) to the human diet. While there is ample evidence to support the health benefits of diets rich in fruit, vegetables, legumes, whole grains, and nuts, evidence that these effects are due to specific nutrients or phytochemicals is limited. Although scientists are very interested in the potential for specific phytochemicals to prevent or treat disease, current scientific evidence suggests that plant-based foods are the healthiest phytochemical delivery system.
Fruit and Vegetables
Contents
Summary
- Dietary patterns characterized by high intakes of fruit and vegetables are consistently associated with lower risks of cardiovascular disease and type 2 diabetes mellitus. (More information)
- Although prospective cohort studies provide weak support for an association between total fruit and vegetable consumption and cancer risk, they provide some evidence that high intakes of certain classes of fruit or vegetables are associated with reduced risk of individual cancers. (More information)
- Most observational studies suggest that diets rich in fruit and vegetables may have beneficial effects on bone health during aging. Yet, large randomized controlled trials are needed to confirm such benefits. (More information)
- Adherence to diets rich in fruit and vegetables has been associated with a lower risk of age-related macular degeneration (AMD), a leading cause of blindness in older people. Randomized controlled trials found supplementation with carotenoids — compounds found in fruit and vegetables — may delay the progression to advanced AMD in subjects with the disease. (More information)
- Oxidative stress and inflammation play key roles in the etiology of chronic obstructive lung disease (COPD). Current evidence suggests that fruit and vegetables high in antioxidants like vitamin C, β-carotene, or flavonoids could play a protective role against COPD. (More information)
- Regular consumption of fruit and vegetables may reduce risk of some chronic diseases, thereby improving overall health and longevity. (More information)
- The 2015-2020 Dietary Guidelines for Americans recommend consuming a healthy diet that includes, among other things, a variety of vegetables from all of the subgroups and fruit. (More information)
Introduction
Despite disagreements regarding the optimal components of a healthy diet, the importance of fruit and vegetables is rather undisputed. The results of numerous epidemiological studies provide consistent evidence suggesting associations between diets rich in fruit and vegetables and lower risks of chronic disease. On the other hand, evidence that very high doses of individual micronutrients or phytochemicals found in fruit and vegetables can provide similar health benefits is inconsistent and relatively weak. In addition to essential micronutrients, fruit and vegetables contain thousands of biologically active phytochemicals that are likely to interact in a number of ways to prevent disease and promote health (1). Fruit and vegetables are rich in antioxidants, which help protect the body from oxidative damage induced by pro-oxidants. The best way to take advantage of these complex interactions is to eat a variety of fruit and vegetables.
Disease Prevention
Cardiovascular disease
Prospective cohort studies have consistently reported inverse associations between high intakes of fruit and vegetables and risk of cardiovascular disease (CVD), including coronary heart disease (CHD) and stroke (2-4). A 2017 meta-analysis of prospective cohort studies found a 16% lower risk of CVD with high versus low fruit and vegetable intake and an 8% lower risk with every 200 grams (g)/day increment in daily fruit and vegetable intake in a dose-response analysis (2). Similar risk reductions were reported for daily 200-g increments in fruit and/or vegetable intake and risks of CHD and stroke (2). However, regarding primary CVD prevention in high-risk subjects, evidence that increasing intakes of fruit and vegetable can improve cardiovascular risk factors is scarce because of a lack of long-term intervention studies (5). In a small 12-week intervention study in 109 overweight adults at high risk for CVD, no differences in blood pressure and serum concentrations of low-density lipoprotein (LDL)-cholesterol and C-reactive protein (CRP; a marker of inflammation) were found between participants consuming two (~160 g), four (~320 g), or seven (~560 g) daily servings of fruit and vegetables (6).
Nonetheless, adherence to the Dietary Approaches to Stop Hypertension (DASH) diet, which emphasizes fruit, vegetables, whole grains, poultry, fish, nuts, and low-fat dairy, was found to substantially lower systolic/diastolic blood pressures by 11.4/5.5 mm Hg in hypertensive and 3.5/2.1 mm Hg in normotensive people compared to a typical US diet (for additional information on the DASH diet, see the National Institutes of Health website) (7). Blood pressure lowering effectively reduces CVD risk (8). Adherence to a Mediterranean-style dietary pattern, also characterized by high fruit and vegetable intake, might further contribute to preventing cardiovascular events in healthy and high-risk subjects through normalizing total and LDL-cholesterol concentrations in the circulation (9).
A number of compounds may play a part in the cardioprotective effects of fruit and vegetables, including vitamin C, folate, potassium, fiber, and various phytochemicals (10, 11). However, supplementation with individual micronutrients or phytochemicals has not generally resulted in significantly decreased incidence of cardiovascular events in randomized controlled trials. Thus, in the case of fruit and vegetables, the benefit of the whole may be greater than the sum of its parts. Of note, the World Health Organization (WHO) recommends the daily consumption of >400 g of fruit and vegetables as part of a healthy diet low in fat, sugar, and salt (sodium chloride), in order to minimize the risk of certain non-communicable diseases like CVD and type 2 diabetes mellitus (12).
Type 2 diabetes mellitus
Nearly 10% of the US population is affected by type 2 diabetes mellitus and another 34% has impaired glucose control (prediabetes) that places them at high risk of developing type 2 diabetes (13). Adherence to a Mediterranean-style or Dietary Approaches to Stop Hypertension (DASH) diet — both rich in vegetables, legumes, fruit, and nuts — has been associated with a lower risk of developing type 2 diabetes (14). Yet, a recent meta-analysis of prospective cohort studies failed to find any association between intakes of vegetables, legumes, fruit, or nuts and the risk of type 2 diabetes (15). In contrast, high intakes of whole-grain and dairy foods were linked to a lower risk of type 2 diabetes, while consumption of sugar-sweetened beverages and red and processed meat were associated with an increased risk of diabetes (15). Whether evidence for a potential protective effect of fruit and vegetables regarding type 2 diabetes is more easily detectable when they are combined with other food groups within a diet rather than when their effect is singled out is unclear. A recent meta-analysis of 12 cross-sectional and two prospective cohort studies found a 27% lower risk of type 2 diabetes with the consumption of a vegetarian diet (16). Yet, it is unclear whether the potential benefit of such a diet is linked to the fact that it does not include foods that are associated with a higher diabetes risk (i.e., red and processed meat) and/or because of the inclusion of foods like fruit and vegetables that may be protective. Nevertheless, in the European Prospective Investigation into Cancer and nutrition (EPIC)-Norfolk, a prospective study that followed 3,704 participants for nearly 11 years, a lower risk of type 2 diabetes was linked to higher intakes of vegetables (but not fruit), as well as with a greater diversity of consumed fruit and vegetables (17). A dose-response analysis found an 8% lower risk of diabetes with every increase of two items added to the variety of fruit or vegetables consumed each week (17). A follow-up study also showed that a composite score reflecting fruit and vegetable consumption, derived from circulating concentrations of vitamin C, β-carotene, and lutein, was also inversely associated with the risk of type 2 diabetes in this cohort (18).
Without changes in lifestyle behavior, especially regarding dietary habits and physical activity, individuals with prediabetes will eventually progress to develop overt type 2 diabetes. Strategies promoting healthier eating habits to improve glucose control usually encourage the consumption of more fruit and vegetables and the concomitant reduction of sugar and fat intake. The American Diabetes Association does not specifically emphasize an increase in fruit and vegetable intake for diabetes prevention yet recommends dietary strategies that include reducing caloric and fat intake, as well as increasing intake of whole-grain foods and dietary fiber that can be sourced in fruit and vegetables (19).
Cancer
A plethora of observational studies has investigated the relationship between intakes of fruit and vegetables and risk of developing site-specific cancers. A 2014 report summarized the findings of 27 studies that examined fruit and vegetable consumption in relation to incidental cancer in participants of the ongoing, multicenter European Prospective Investigation into Cancer and nutrition (EPIC) study (20). This report found the highest versus lowest quintile of fruit intake (≥356 g/day vs. ≤89 g/day) to be associated with a 40% lower risk of oropharyngeal, laryngeal, and esophageal cancers and a 20% lower risk of lung cancer. No associations were observed between fruit intake and cancers of the lymphatic system, stomach, pancreas, breast, cervix, prostate, or bladder. There were also no inverse associations between intakes of vegetables and risk of cancer. More recently published EPIC data analyses also failed to find significant inverse associations between fruit and/or vegetable intakes and risk of hepatocellular carcinoma (21), colorectal cancer (22), hormone-receptor-positive breast cancer (23), or differentiated thyroid carcinoma (24). Further, mixed results from recent meta-analyses of observational studies are reported in Table 1, with significant associations noted in bold. Some of the discrepancies might be attributed to study design. Compared to prospective cohort studies, which collect dietary information from participants before they are diagnosed with cancer, retrospective case-control studies are more susceptible to bias in the selection of participants (cases and controls) and with dietary recall (25). For example, this might explain why fruit intake is associated with the risk of esophageal adenocarcinoma in the meta-analysis of six case-control studies but not in that of three cohort studies in the meta-analysis by Li et al. (26). Inaccurate measurements (introducing measurement bias) and changes in the diet during follow-up in prospective cohort studies may also contribute to reporting associations that are proven spurious or to missing true associations. A 2012 meta-analysis of prospective studies suggested that, when compared to direct measurements of plasma carotenoids, the use of a food-frequency questionnaire to assess carotenoid intake introduced measurements errors that led to underestimating the strength of the association between carotenoid intake and reduced breast cancer risk (27).
| Type of Cancer | References | Highest versus Lowest Quantile of Intake | Type of Observational Studies | Risk Ratio [RR] or Odds Ratio [OR] (95% Confidence Interval)* |
|---|---|---|---|---|
| All cancer types |
Aune et al. (2017) (2) |
Total fruit & vegetables | 14 prospective cohort studies | RR: 0.97 (0.95-0.99) |
| Vegetables | 19 prospective cohort studies | RR: 0.96 (0.93-0.99) | ||
| Cruciferous vegetables | 5 prospective cohort studies | RR: 0.84 (0.72-0.97) | ||
| Green yellow vegetables | 5 prospective cohort studies | RR: 0.88 (0.77-1.00) | ||
| Fruit | 25 prospective cohort studies | RR: 0.96 (0.94-0.99) | ||
| Citrus fruit | 5 prospective cohort studies | RR: 0.97 (0.90-1.04) | ||
| Bladder cancer |
Vieira et al. (2015) (28) |
Total fruit & vegetables | 9 prospective cohort studies | RR: 0.89 (0.75-1.05) |
| Vegetables | 10 prospective cohort studies | RR: 0.92 (0.84-1.01) | ||
| Cruciferous vegetables | 7 prospective cohort studies | RR: 0.85 (0.69-1.06) | ||
| Leafy vegetables | 6 prospective cohort studies | RR: 0.90 (0.78-1.04) | ||
| Fruit | 12 prospective cohort studies | RR: 0.91 (082-1.00) | ||
| Citrus fruit | 8 prospective cohort studies | RR: 0.87 (0.76-0.99) | ||
| Liu et al. (2015) (29) | Vegetables | 14 case-control studies | RR: 0.76 (0.58-1.00) | |
| 10 prospective cohort studies | RR: 0.92 (0.84-1.02) | |||
| Fruit | 15 case-control studies | RR: 0.76 (0.66-0.88) | ||
| 12 prospective cohort studies | RR: 0.85 (0.73-0.98) | |||
| Breast cancer | Aune et al. (2012) (30) | Total fruit & vegetables | 6 prospective cohort studies | RR: 0.89 (0.80-0.99) |
| Vegetables | 9 prospective cohort studies | RR: 0.99 (0.92-1.06) | ||
| Fruit | 10 prospective cohort studies | RR: 0.92 (0.86-0.98) | ||
| Colorectal adenoma | Ben et al. (2015) (31) | Total fruit & vegetables | 5 case-control and 3 prospective cohort studies | RR: 0.82 (0.75-0.91) |
| Vegetables | 12 case-control and 5 prospective cohort studies | RR: 0.91 (0.80-1.02) | ||
| Fruit | 15 case-control and 5 prospective cohort studies | RR: 0.94 (0.92-0.97) | ||
| Colorectal cancer | Aune et al. (2011) (32) | Total fruit & vegetables | 11 prospective cohort studies | OR: 0.92 (0.86-0.99) |
| Vegetables | 16 prospective cohort studies | OR: 0.91 (0.86-0.96) | ||
| Fruit | 14 prospective cohort studies | OR: 0.90 (0.83-0.98) | ||
| Kashino et al. (2015) (33) | Vegetables | 9 case-control studies | OR: 0.75 (0.59-0.96) | |
| 6 prospective cohort studies | RR: 1.00 (0.92-1.10) | |||
| Esophageal adenocarcinoma |
Li et al. (2014) (26) |
Total fruit & vegetables | 4 case-control studies | RR: 0.61 (0.44-0.84) |
| Vegetables | 6 case-control studies | RR: 0.75 (0.53-1.06) | ||
| 3 prospective cohort studies | RR: 0.76 (0.54-1.05) | |||
| Fruit | 6 case-control studies | RR: 0.59 (0.38-0.90) | ||
| 3 prospective cohort studies | RR: 0.99 (0.72-1.36) | |||
| Esophageal cancer | Vingeliene et al. (2016) (34) | Citrus fruit | 5 studies with a prospective design# | RR: 0.85 (0.73-0.99) |
| Gastric cancer | Wang et al. (2015) (35) | Vegetables | 19 prospective cohort studies | RR: 0.96 (0.88-1.06) |
| Fruit | 22 prospective cohort studies | RR: 0.90 (0.83-0.98) | ||
| Bae et al. (2016) (36) | Citrus fruit | 5 prospective cohort studies | RR: 0.87 (0.76-0.99) | |
| Vingeliene et al. (2016) (34) | Citrus fruit | 4 studies with a prospective design# | RR: 0.93 (0.82-1.07) | |
| Hepatocellular carcinoma | Yang et al. (2014) (37) | Vegetables | 9 case-control studies | RR: 0.76 (0.48-1.20) |
| 8 prospective cohort studies | RR: 0.66 (0.51-0.86) | |||
| Fruit | 19 case-control studies | RR: 0.77 (0.67-0.88) | ||
| 19 prospective cohort studies | RR: 0.84 (0.75-0.94) | |||
| Lung cancer | Wang et al. (2015) (38) | Vegetables | 19 case-control studies | RR: 0.62 (0.54-0.70) |
| 18 prospective cohort studies | RR: 0.88 (0.81-0.97) | |||
| Fruit | 19 case-control studies | RR: 0.77 (0.67-0.88) | ||
| 19 prospective cohort studies | RR: 0.84 (0.75-0.94) | |||
| Vieira et al. (2016) (39) | Total fruit and vegetables | 18 studies with a prospective design# | RR: 0.86 (0.78-0.94) | |
| Vegetables | 25 studies with a prospective design | RR: 0.92 (0.87-0.97) | ||
| Cruciferous vegetables | 11 studies with a prospective design | RR: 0.87 (0.79-0.97) | ||
| Green leafy vegetables | 9 studies with a prospective design | RR: 0.85 (0.75-0.96) | ||
| Fruit | 29 studies with a prospective design | RR: 0.82 (0.76-0.89) | ||
| Citrus fruit | 15 studies with a prospective design | RR: 0.85 (0.78-0.93) | ||
|
*Statistically significant associations noted in bold. #Cohort, nested case-control, and case-cohort studies |
||||
Additional evidence from observational studies, discussed in MIC articles focusing specifically on cruciferous vegetables, garlic, carotenoid-rich vegetables, and legumes suggests that high intakes of certain classes of vegetables are associated with reduced risk of individual cancers. The 2007 report from the World Cancer Research Fund International (WCRF)/American Institute for Cancer Research (AICR) concluded that fruit and non-starchy vegetables were probably protective against some cancers, recommending a daily consumption of five portions (400 g; based on an average portion weighing 80 g) of a variety of vegetables and fruit (40). AICR continuously collates evidence from cohort and randomized controlled studies through its Continuous Update Project (CUP), and updated CUP-derived Cancer Prevention Recommendations are expected to be published in late 2017 (41).
Osteoporosis
Bone turnover
Fruit and vegetables are rich in precursors of bicarbonate ions (HCO3–), which serve to buffer acids in the body. When the quantity of bicarbonate ions is insufficient to maintain normal pH, the body is capable of mobilizing alkaline calcium salts from bone in order to neutralize acids consumed in the diet and generated by metabolism (42). It has been hypothesized that higher consumption of fruit and vegetables could help reduce the net acid content of the diet and preserve calcium in bones, which might otherwise be mobilized to maintain normal pH to the detriment of bone health. Results from the ancillary DASH-Sodium study, which emphasizes the intake of fruit, vegetables, whole-grain foods, and low-fat dairy, supported a beneficial link between bone health and fruit and vegetable intake. Compared to a control diet, one month-administration of the DASH diet to (pre)hypertensive middle-aged adults significantly lowered the rate of bone turnover, as shown by reduced serum concentrations of osteocalcin (OC), a marker of bone formation released by osteoblasts into circulation during the mineralization of newly synthesized collagen. C-terminal telopeptide of type 1 collagen (CTX) — a marker of bone resorption released from bone collagen into circulation following bone degradation by osteoclasts — was also decreased (43). However, more recent randomized controlled interventions found no effect of increasing fruit and vegetable intake on markers of bone turnover (44-46). The results of a placebo-controlled trial in 276 healthy postmenopausal women suggested that supplementing the diet with alkali, either through supplemental potassium citrate or an additional 300 g/day of fruit and vegetables, did not reduce bone turnover, increase bone mineral density (BMD), or blunt the age-associated bone loss over a two-year period (45). The effect of five portions of fruit and vegetables on measures of dietary acid/base balance and bone turnover markers was also reported as a secondary outcome in the Ageing and Dietary Intervention Trial that included 83 healthy older participants (ages, 65-85 years) (46). Compared to habitual fruit and vegetable intakes of ≤2 portions/day, the consumption of five portions per day for 16 weeks increased the alkalinity of the diet but failed to reduce markers of bone turnover (46). There were also no significant reductions in circulating bone turnover markers in postmenopausal women with osteopenia supplemented with six extra servings of fruit and vegetables per day (~400 g/day) for 12 weeks (44).
Bone mineral density
Several observational studies have examined intakes of fruit and vegetables in relation to bone mineral density (BMD) in men and women, providing mixed results (reviewed in 47). In an early cross-sectional analysis of the longitudinal Framingham Osteoporosis Study in elderly adults (mean age, 75 years), baseline intakes of fruit and vegetables were positively associated with BMD at various sites, including trochanter, femoral neck, and radius (48). Baseline fruit and vegetable intakes were also positively associated with longitudinal changes of trochanter BMD over four years in elderly men but not elderly women (48, 49). Two recent cross-sectional studies in Chinese cohorts reported positive relationships between BMD and intakes of fruit rather than vegetables (50, 51). In the cross-sectional analysis of two Hong-Kong-based cohorts of 3,995 older participants (≥65 years), higher daily intakes of fruit — but not vegetables — were associated with significantly higher whole-body and femoral neck BMD in men and women (50). The cross-sectional study of 3,089 Chinese adults (ages, 40-75 years) reported positive associations between whole-body, total hip, and femoral neck BMD and higher intakes of apples, pears, peaches, pineapples, plums, and to a lesser extent, citrus fruit (51). However, the two-year supplementation of 300 g/day of fruit and vegetables failed to reduce BMD loss in healthy postmenopausal women (45).
Risk of fracture
A pooled analysis of data from several prospective cohort studies, including the EPIC-Elderly Greece (9,534 participants), EPIC-Elderly Sweden (3,276 participants), Cohort of Swedish Men (COSM; 20,150 men), Swedish Mammography Cohort (SMC; 17,506 women), and Nurses' Health Study (NHS; 91,552 women), found a 39% higher risk of hip fracture with intakes of fruit and vegetables ≤1 serving/day compared to 4-5 daily servings (52). Further subgroup analysis linked higher hip fracture risk specifically with low intakes of vegetables rather than fruit. Finally, compared to 4-5 daily servings of fruit and vegetables, there was no reduction in hip fracture risk with intakes greater than 5 servings/day (52). The consumption of fruit and vegetables has been recently examined in relation to 415 fracture-related hospitalizations identified during a 14.5-year follow-up of 1,468 elderly participants (≥70 years) of the Perth Longitudinal Study of Aging in Women (PLSAW) (53). Whereas no association was found between fruit intakes and fracture risk, high versus low intake of total vegetables (≥3 servings/day versus <2 servings/day) was associated with a 27% reduced risk of all fractures and a 39% reduced risk of hip fractures. Further analyses suggested an inverse association between consumption of allium vegetables (onion, leek, and garlic) and risk of fracture (53).
Although observational studies suggest a positive relationship between diets rich in fruit and vegetables and bone health during aging, randomized controlled studies are needed to examine the nature of this association.
Eye diseases
Cataracts
Cataracts are thought to be caused by oxidative damage of proteins in the eye's lens induced by long-term exposure to ultraviolet (UV) light (54). The resulting cloudiness and discoloration of the lens leads to vision loss that becomes more severe with age. A 2015 meta-analysis of nine observational studies found a 28% lower risk of age-related cataracts with the highest versus lowest intakes of vegetables (55). In a large Swedish prospective study that followed 30,607 middle-aged and elderly women for a mean 7.7 years, the risk of age-related cataracts has been examined in relation to the estimated total antioxidant capacity of the diet (56). Higher versus lower estimates of total dietary antioxidant capacity were associated with a 13% lower risk of cataracts. Subgroup analyses showed that this inverse association was statistically significant in women younger than 65 years and in corticosteroid users (56). Pooled analyses of observational studies that investigated the relationships between individual nutrients with antioxidant properties and the risk of cataracts have also suggested a lower risk of cataracts with higher intakes or higher blood concentrations of vitamin C, vitamin A, or β-carotene, although the results vary according to study design (i.e., case-control versus cross-sectional versus longitudinal) (57, 58). However, a 2012 review of nine randomized clinical trials found no substantial effect of β-carotene, vitamin C, and vitamin E, administered individually or in combination over 2.1 to 12 years, on the risk of cataracts or cataract surgery (59). In addition, a large randomized controlled trial in 5,802 subjects at high risk for cardiovascular disease recently reported no difference in cataract surgery incidence over a seven-year follow-up period between participants assigned to a Mediterranean diet that included the whole range of antioxidant nutrients and those assigned to a control diet (60). A secondary analysis reported a 29% lower cataract surgery risk in participants in the highest versus lowest tertile of vitamin K1 intake (61).
Age-related macular degeneration
In industrialized countries, degeneration of the macula, located in the center of the retina, is the leading cause of blindness in older adults (62). Long-term light exposure and oxidative damage in the outer segments of photoreceptors may lead to drusen and/or pigment abnormalities in the macula, increasing the risk of age-related macular degeneration (AMD) and central blindness.
Several recent observational studies have examined AMD prevalence, incidence, progression, or severity in relation to dietary patterns. Most of them used constructed scoring systems reflecting the level of adherence to specific dietary patterns by individuals. The European Eye (EUREYE) study examined associations between the prevalence of AMD and dietary patterns among 4,753 individuals from seven European countries. Adherence to the Mediterranean diet was assessed using a Mediterranean Diet Score system that captured high intakes of key food items, such as olive oil, wine, fruit, vegetables or salad, fish, and legumes, and low intakes of meat. High adherence to the Mediterranean Diet was associated with a reduced risk of developing large drusen, but there was no association with the risk of early or advanced AMD (63). In the Carotenoids in the Age-Related Eye Disease Study (CAREDS) that included 1,313 US women (ages, 50-79 years), high adherence to a Mediterranean-like dietary pattern characterized by high intakes of fruit, vegetables, whole grains, legumes, nuts, and fish, and moderate intakes of red meat and alcohol, were found to be associated with a 66% lower risk of early AMD (64). This Mediterranean-like dietary pattern, which is closer to dietary patterns occurring in the US, was also associated with a 26% lower risk of progression to advanced AMD in 2,525 subjects followed for a mean 8.7 years in the Age-Related Eye Disease Study (AREDS) (65). Of note, this association was no longer valid when the analysis was restricted to individuals with a genetically determined susceptibility to AMD, i.e., those homozygous for the risk allele of the complement factor H [CFH] gene (rs1410996) (65). A cross-sectional study that analyzed baseline data from 4,088 AREDS participants, among whom 2,739 had no AMD, 4,599 had early AMD, and 765 had advanced AMD, identified two main dietary patterns: so-called "Oriental" and "Western" patterns (66). High adherence to the "Oriental" dietary pattern characterized by consumption of vegetables, legumes, fruit, fish, whole grains, poultry, and low-fat dairy products was associated with lower risks of early and advanced AMD. In contrast, higher risks of early and advanced AMD were found in individuals with high adherence to a "Western" diet that included red and processed meat, potatoes, French fries, butter, high-fat dairy products, eggs, refined grains, and sweets and desserts (66).
Among observational studies that focused on individual food groups or nutrients, some have suggested that high intakes of fruit, vegetables, or antioxidant nutrients, such as vitamin C, vitamin E, and carotenoids, might be protective against AMD. In two early case-control studies, high intakes of dark-green leafy vegetables especially rich in lutein and zeaxanthin, two carotenoids present in the retina, were associated with a significantly lower risk of developing AMD (67, 68). In a prospective cohort study of more than 118,000 men and women, those who consumed ≥3 servings/day of fruit had a 36% lower risk of developing AMD over the next 12 to 18 years than those who consumed <1.5 servings/day (69). Interestingly, vegetable intake was not associated with the risk of AMD in this cohort. Another study combining lutein and zeaxanthin intake was not associated with the prevalence of intermediate AMD in a cohort of women aged 50-79 years (70). However, further analysis of the data revealed that women younger than 75 years with stable intakes of lutein and zeaxanthin had a 43% lower risk of developing intermediate AMD (70). In the AREDS trial, oral supplementation with β-carotene (15 mg/day), vitamin C (500 mg/day), vitamin E (400 IU/day), zinc (80 mg/day as zinc oxide), and copper (2 mg/day as cupric oxide) for five years was shown to reduce the risk of developing advanced AMD by 25% (71). In a follow-up study — the AREDS2 trial — supplemental lutein (10 mg/day) and zeaxanthin (2 mg/day) in combination with the 'AREDS formulation' only reduced the risk of progression to late AMD in the subset of participants with the lowest dietary intakes of lutein and zeaxanthin (72). A more detailed account of the epidemiological evidence regarding the relationship between dietary and supplemental carotenoids and AMD risk can be found in the article on Carotenoids.
Chronic obstructive pulmonary disease
Chronic obstructive pulmonary disease (COPD) is a condition that combines emphysema and chronic bronchitis, two chronic lung conditions that are characterized by airway obstruction. Smoking and indoor/outdoor pollution are considered to be primary contributors to COPD development, but dietary patterns low in fruit and vegetables and providing inadequate vitamin intakes may also affect lung function and risk for COPD (reviewed in 73, 74). Early observational studies in Europe indicated that higher fruit intakes, especially apple intakes, were associated with higher spirometric values (including forced expiratory volume in 1 second [FEV1]), indicative of better lung function (75-77). In a cross-sectional study of 2,500 middle-aged Welsh men, slower declines in lung function were associated with the consumption of at least five apples weekly compared to no consumption (76). Another study of 2,917 European men followed over 20 years found that each 100 g (3.5 oz) increase in daily fruit consumption was associated with a 24% lower risk of COPD-related mortality (78). Additionally, when compared to a Western dietary pattern of refined grains, cured and red meats, French fries, and desserts, a prudent diet emphasizing fruit, vegetables, fish, and whole grains was associated with a 25%-50% lower risk of COPD in two large cohorts of men (79) and women (80). In a large prospective cohort study that followed 44,335 Swedish men (mean age, 60.2 years) for a mean 13.2 years, the highest versus lowest quintile (≥5.3 servings/day versus <2 servings/day) of fruit and vegetable intakes was associated with a 35% lower risk of developing COPD (81). Subgroup analyses showed that fruit and vegetable intakes were inversely associated with COPD risk in current and former smokers, but not in men who never smoked. Because oxidative stress and inflammation play key roles in the etiology of chronic obstructive lung disease, it has been suggested that fruit and vegetables high in antioxidants, such as vitamin C, β-carotene, or flavonoids, could play a protective role against COPD. A three-year randomized controlled study in 120 patients with COPD (mean age, 68.1 years) assigned to a diet rich in antioxidants, such as fresh fruit, fruit juice, and vegetables, or a control diet has provided support for the antioxidant hypothesis (82). Shifting to a higher consumption of fruit and vegetables prevented the decline of lung function observed in subjects who consumed the control diet (82).
Asthma
Environment and lifestyle changes, including shifts toward unhealthy diets, are thought to contribute to the increasing prevalence of asthma and allergic diseases in industrialized countries. Observational studies that examined asthma and allergic symptoms in relation to fruit and vegetable intakes have provided mixed results (83). A cross-sectional analysis of a population-based study in 32,644 Portuguese adults identified five dietary patterns, of which one included fish, fruit, and vegetables — three essential components of the Mediterranean diet. This dietary pattern was found to be inversely associated with self-reported asthma symptoms and self-reported use of asthma drugs — yet not with self-reported medical diagnosis of asthma (84). In contrast, the most recent cross-sectional study of 3,202 participants in the European Global Allergy and Asthma Network of Excellence (GA2LEN) showed no association between fruit and vegetable intake and risk of asthma and chronic rhino-sinusitis (85). A 2017 systematic review identified 58 studies (in addition to the two above-cited studies) reporting fruit and vegetable intake in relation to lung function, wheeze, or asthma, of which 30 were cross-sectional studies and 41 conducted in children and/or adolescents (83). A majority of studies (8 in adults and 22 in children) reported inverse associations between diets high in fruit and vegetables and risk of asthma and/or wheeze: 20 studies (8 in adults and 12 in children) showed that intakes of either fruit or vegetables were inversely associated with asthma and/or wheeze, and eight studies (one in adults and seven in children) found no associations. Pooled data analyses showed an inverse association between vegetable intake and risk of asthma, as well as fruit intake and asthma severity and risk of wheeze (83). A previous meta-analysis of observational studies reported lower risks of wheeze and asthma with higher intakes of fruit and vegetables in adults and children in cross-sectional studies, but not all prospective cohort studies have supported these findings (86). Subgroup analyses also suggested inverse associations between intakes of apples, citrus fruit, and tomatoes and risks of wheeze and asthma. Finally, pooled analyses of prospective cohort studies have revealed no association between fruit or vegetable intake during pregnancy and risk of wheeze or asthma in the offspring (86).
Cognitive decline and neurodegenerative disease
Observational studies
Results from most cross-sectional and longitudinal studies suggest that diets rich in fruit and vegetables might help prevent age-related cognitive deterioration and reduce the risk of neurodegenerative diseases like Alzheimer's disease (AD) (87). A 2014 systematic review identified 11 prospective cohort studies (88), of which four examined fruit and vegetable intakes in relation to incidence of neurodegenerative diseases. All four prospective studies reported inverse associations between consumption of fruit and vegetables and risk of developing mild cognitive impairments or dementia, including AD (89-92). Among the seven prospective cohort studies positively linking fruit and vegetable intakes to better cognitive performance (reviewed in 88), a two-year follow-up of 13,388 women (mean age, 74 years) in the Nurses’ Health Study (NHS) found less cognitive decline in those in the highest versus lowest intakes of green leafy vegetables, cruciferous vegetables, and legumes (93). Fruit consumption was not associated with changes in cognitive performance in this study (93). However, a more recently published NHS study in 16,010 women analyzed intakes of major flavonoid-containing foods in relation to cognitive test scores and reported less cognitive decline with higher long-term intakes of strawberries and blueberries (94). Finally, a meta-analysis of 13 prospective cohort studies showed better global cognition in healthy older adults consuming the Mediterranean diet compared to control diets. In contrast, there were no differences in measures of episodic, semantic, and working memory between diets (95).
Dietary interventions
To date, only a few interventions have examined the overall effect of fruit- and vegetable-rich diets on cognition in cognitively healthy older adults. One trial assessed cognitive changes in 334 older adults (mean age, 66.9 years) at high risk for cardiovascular disease (CVD) randomly assigned to either a Mediterranean diet supplemented with extra-virgin olive oil, a Mediterranean diet supplemented with nuts, or a control diet (96). Following a median of 4.1 years, both Mediterranean diets prevented the deterioration of cognitive function that was observed in those ascribed the control diet. Compared to participants in the control diet group, those who followed the Mediterranean diet plus nuts had improved composite cognitive test scores for memory, while those in the Mediterranean diet plus olive oil group had better composite scores for frontal function and global cognition (96). However, in a six-month trial in 137 CVD-free Australian adults (mean age, 72 years), consumption of a Mediterranean diet rich in fruit, legumes, dairy, nuts, olive oil, and seafood did not result in improved cognitive function — measured by a battery of 13 neuropsychological tests — compared to the habitual diet (97, 98).
Mortality
Because regular consumption of fruit and vegetables may reduce the risk of some chronic diseases, it may also improve overall health and longevity. A 2017 meta-analysis of 95 prospective cohort studies found that daily consumption of fruit and vegetables was inversely associated with cause-specific and all-cause mortality (2). The risk of all-cause mortality was found to be 24% lower with five daily serving of fruit and vegetables combined (~400 mg/day) compared to little or no daily intake of fruit and vegetables. Five daily servings of fruit and vegetables were also associated with lower risks of cardiovascular-related (-19%) and cancer-related (-12%) mortality. In addition, the risk of all-cause mortality was lower with higher intakes of specific types of fruit and vegetables. There was evidence of lower risk of all-cause mortality with the highest versus lowest intake level of apples (-20%), berries (-8%), citrus fruit (-10%), cruciferous vegetables (-12%), and green leafy vegetables (-8%). This meta-analysis further reported a reduced risk of all-cause mortality with both cooked (-13%) and raw vegetable (-12%) intakes (2).
Although the Dietary Guidelines for Americans recommend fresh, frozen, and canned fruit equally, the consumption of the latter has been associated with increased risks of all-cause and cardiovascular related mortality in a pooled analysis of three UK-based prospective studies found with consumption of tinned fruit (99). While added sugar content in tinned fruit or exposure to bisphenol A (a chemical component of tin cans) may explain these results, further studies are needed to clarify whether fresh fruit and canned fruit can provide similar health benefits when included as part of a healthy diet.
Finally, although increasing daily intakes of fruit and vegetables would very likely reduce the number of premature deaths caused by cardiovascular disease and cancer in the US population, it is estimated that about 8 in 10 Americans do not meet the current intake recommendations (100).
Intake Recommendations
The 2015-2020 Dietary Guidelines for Americans — issued jointly by the US Department of Health and Human Services and the US Department of Agriculture — recommend to consume a healthy diet which includes, among other things, a variety of vegetables from all of the subgroups and fruit, especially whole fruit (101). In the 2015-2020 Dietary Guidelines for Americans, the unit of measure of a fruit or vegetable serving size is the cup-equivalent (c-eq). In general, one cup-equivalent of fruit corresponds to (1) one cup of cut-up, raw, or canned fruit; (2) one cup (eight fluid ounces) of 100% fruit juice; or (3) one-half cup of dried fruit. Table 2 provides examples of the size of specific fruit counting as one cup-equivalent.
| Fruit | Examples of One Cup-equivalent of Fruit |
|---|---|
| Apple | 1 small |
| Banana | 1 large |
| Grapefruit | 1 medium |
| Grapes | 32 seedless grapes |
| Peach | 1 large |
| Pear | 1 medium |
| Plum | 2 large or 3 medium |
| Strawberries | 8 large berries |
| 100% fruit juice | 8 fluid ounces (1 cup) |
| Dried fruit (e.g., raisins, apricots) | ½ cup |
One cup-equivalent of vegetables generally corresponds to one cup of raw or cooked vegetables or vegetable juice. One exception is leafy greens (e.g., spinach, romaine, watercress, dark-green leafy lettuce, endive, escarole) for which one cup-equivalent corresponds to one cup of cooked or two cups of raw vegetables. Table 3 provides specific examples of what counts as one cup-equivalent of vegetables.
| Vegetables | Examples of One Cup-equivalent of Vegetables |
|---|---|
| Dark-green vegetables | |
| Greens (e.g., collards, kale) | 1 cup, cooked |
| Raw leafy greens (e.g., watercress, endive, romaine) | 2 cups, raw |
| Spinach |
1 cup, cooked 2 cups, raw |
| Red and orange vegetables | |
| Carrots |
1 cup, chopped, raw or cooked 2 medium 1 cup of baby carrots |
| Red peppers | 1 large pepper |
| Tomatoes |
1 large, raw 1 cup, chopped, raw, canned, or cooked |
| Sweet potatoes | 1 large, baked |
| Legumes | |
| Dry beans and peas | 1 cup, whole or mashed, cooked |
| Starchy vegetables | |
| Corn, yellow or white | 1 large ear |
| White potatoes | 1 medium, boiled or baked |
| Other vegetables | |
| Celery |
1 cup, diced or sliced, raw or cooked 2 large stalks |
| Green peppers | 1 large pepper |
| Lettuce | 2 cups, raw, shredded or chopped |
| Onions | 1 cup, chopped, raw or cooked |
The 2015-2020 Dietary Guidelines for Americans provides dietary recommendations, including amounts of fruit and vegetables, designed to meet nutrient needs and Dietary Guidelines standards, for those who choose to follow either a healthy US-style eating pattern, a healthy Mediterranean-style eating pattern, or a healthy vegetarian eating pattern (101). The recommendations are based on estimated energy needs that vary with age, gender, and level of physical activity. Recommended daily intakes of fruit and vegetables at all calorie requirement levels can be found in the '2015-2020 Dietary Guidelines for Americans' report (see Appendices 3-5) (101). Table 4 provides the amounts of fruit and vegetables (expressed in cup-equivalents) that are recommended at the 2,000-calorie per day level. Regardless of the chosen eating pattern, consumption of a variety of different vegetables and fruit is recommended, including all fresh, frozen, and canned dark-green, red, and orange vegetables, starchy vegetables, legumes (peas and beans), and all fresh, frozen, canned, and dried fruit and 100% fruit juice.
| Food | Healthy Eating Patterns | ||
|---|---|---|---|
| US-style | Mediterranean-style | Vegetarian | |
| Vegetables (c-eq/day) | 2½ | 2½ | 2½ |
| Dark-green vegetables (c-eq/week) | 1½ | 1½ | 1½ |
| Red and orange vegetables (c-eq/week) | 5½ | 5½ | 5½ |
| Legumes (c-eq/week) | 1½ | 1½ | 1½ |
| Starchy vegetables (c-eq/week) | 5 | 5 | 5 |
| Other vegetables (c-eq/week) | 4 | 4 | 4 |
| Fruit (c-eq/day) | 2 | 2½ | 2 |
|
*Recommendations for fruit and vegetable intakes at the 2,000-calorie per day level. Estimates of daily calorie needs according to age, gender, and physical activity can be found in the Appendix 2 of the ‘2015-2020 Dietary Guidelines for Americans’ report (101). c-eq, cup-equivalents |
|||
The nonprofit organization, Produce for Better Health Foundation (PBH), has partnered with the US Centers for Disease Control and Prevention (CDC) to develop the Fruits & Veggies — More Matters®health initiative, which aims to help Americans increase their consumption of fruit and vegetables for better health (104). Other initiatives like the US Department of Agriculture (USDA)'s ChooseMyPlate.gov have been developed to help everyone make healthier dietary choices, particularly by adding more fruit and vegetables to daily meals.
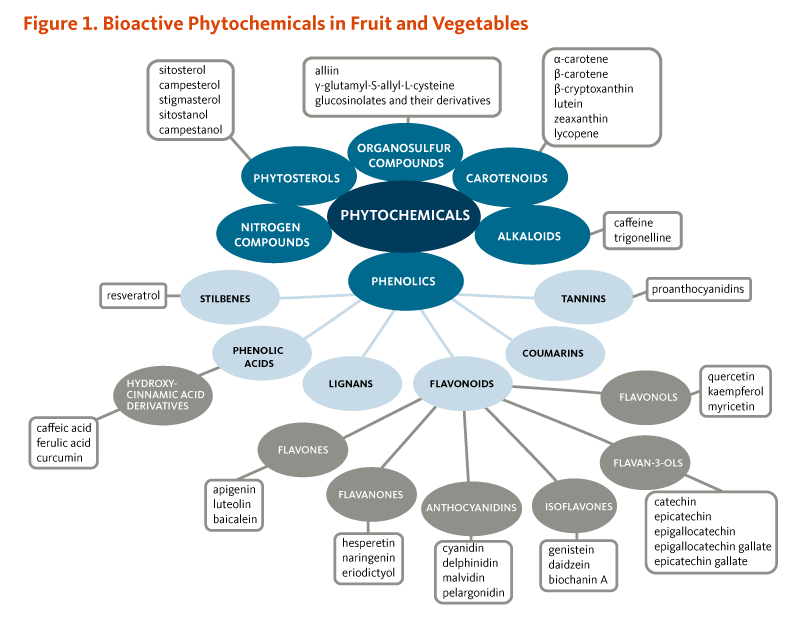
Finally, vegetables and fruit not only are a great source of micronutrients, dietary fiber, and unsaturated fat, they also supply a wide range of biologically active phytochemicals (Figure 1) that contribute to the health benefits of plant foods. Information regarding the functions and health benefits of specific micronutrients and phytochemicals can be found in articles on vitamins, minerals, and dietary phytochemicals.
Authors and Reviewers
Originally written in 2003 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in December 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in May 2009 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in October 2017 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in January 2018 by:
Dagfinn Aune, Ph.D.
Department of Epidemiology and Biostatistics
School of Public Health
Imperial College London, St. Mary's Campus
London, United Kingdom
Copyright 2003-2024 Linus Pauling Institute
References
1. Liu RH. Health-promoting components of fruits and vegetables in the diet. Adv Nutr. 2013;4(3):384s-392s. (PubMed)
2. Aune D, Giovannucci E, Boffetta P, et al. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose-response meta-analysis of prospective studies. Int J Epidemiol. 2017;46(3):1029-1056. (PubMed)
3. Gan Y, Tong X, Li L, et al. Consumption of fruit and vegetable and risk of coronary heart disease: a meta-analysis of prospective cohort studies. Int J Cardiol. 2015;183:129-137. (PubMed)
4. He FJ, Nowson CA, Lucas M, MacGregor GA. Increased consumption of fruit and vegetables is related to a reduced risk of coronary heart disease: meta-analysis of cohort studies. J Hum Hypertens. 2007;21(9):717-728. (PubMed)
5. Hartley L, Igbinedion E, Holmes J, et al. Increased consumption of fruit and vegetables for the primary prevention of cardiovascular diseases. Cochrane Database Syst Rev. 2013(6):Cd009874. (PubMed)
6. McEvoy CT, Wallace IR, Hamill LL, et al. Increasing fruit and vegetable intake has no dose-response effect on conventional cardiovascular risk factors in overweight adults at high risk of developing cardiovascular disease. J Nutr. 2015;145(7):1464-1471. (PubMed)
7. Appel LJ, Moore TJ, Obarzanek E, et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N Engl J Med. 1997;336(16):1117-1124. (PubMed)
8. Law MR, Morris JK, Wald NJ. Use of blood pressure lowering drugs in the prevention of cardiovascular disease: meta-analysis of 147 randomised trials in the context of expectations from prospective epidemiological studies. BMJ. 2009;338:b1665. (PubMed)
9. Rees K, Hartley L, Flowers N, et al. 'Mediterranean' dietary pattern for the primary prevention of cardiovascular disease. Cochrane Database Syst Rev. 2013(8):Cd009825. (PubMed)
10. Alissa EM, Ferns GA. Dietary fruits and vegetables and cardiovascular diseases risk. Crit Rev Food Sci Nutr. 2017;57(9):1950-1962. (PubMed)
11. Tang GY, Meng X, Li Y, Zhao CN, Liu Q, Li HB. Effects of Vegetables on Cardiovascular Diseases and Related Mechanisms. Nutrients. 2017;9(8). (PubMed)
12. World Health Organization. Increasing fruit and vegetable consumption to reduce the risk of noncommunicable diseases. 17 June 2017. Available at: http://www.who.int/elena/titles/fruit_vegetables_ncds/en/. Accessed 10/5/17.
13. Centers for Disease Control and Prevention. US National Diabetes Statistics Report. 17 July 2017. Available at: https://www.cdc.gov/diabetes/data/statistics/statistics-report.html. Accessed 10/4/17.
14. Jannasch F, Kroger J, Schulze MB. Dietary patterns and type 2 diabetes: a systematic literature review and meta-analysis of prospective studies. J Nutr. 2017;147(6):1174-1182. (PubMed)
15. Schwingshackl L, Hoffmann G, Lampousi AM, et al. Food groups and risk of type 2 diabetes mellitus: a systematic review and meta-analysis of prospective studies. Eur J Epidemiol. 2017;32(5):363-375. (PubMed)
16. Lee Y, Park K. Adherence to a vegetarian diet and diabetes risk: a systematic review and meta-analysis of observational studies. Nutrients. 2017;9(6). (PubMed)
17. Cooper AJ, Sharp SJ, Lentjes MA, et al. A prospective study of the association between quantity and variety of fruit and vegetable intake and incident type 2 diabetes. Diabetes Care. 2012;35(6):1293-1300. (PubMed)
18. Cooper AJ, Sharp SJ, Luben RN, Khaw KT, Wareham NJ, Forouhi NG. The association between a biomarker score for fruit and vegetable intake and incident type 2 diabetes: the EPIC-Norfolk study. Eur J Clin Nutr. 2015;69(4):449-454. (PubMed)
19. Bantle JP, Wylie-Rosett J, Albright AL, et al. Nutrition recommendations and interventions for diabetes: a position statement of the American Diabetes Association. Diabetes Care. 2008;31 Suppl 1:S61-78. (PubMed)
20. Bradbury KE, Appleby PN, Key TJ. Fruit, vegetable, and fiber intake in relation to cancer risk: findings from the European Prospective Investigation into Cancer and Nutrition (EPIC). Am J Clin Nutr. 2014;100 Suppl 1:394s-398s. (PubMed)
21. Bamia C, Lagiou P, Jenab M, et al. Fruit and vegetable consumption in relation to hepatocellular carcinoma in a multi-centre, European cohort study. Br J Cancer. 2015;112(7):1273-1282. (PubMed)
22. Leenders M, Siersema PD, Overvad K, et al. Subtypes of fruit and vegetables, variety in consumption and risk of colon and rectal cancer in the European Prospective Investigation into Cancer and Nutrition. Int J Cancer. 2015;137(11):2705-2714. (PubMed)
23. Emaus MJ, Peeters PH, Bakker MF, et al. Vegetable and fruit consumption and the risk of hormone receptor-defined breast cancer in the EPIC cohort. Am J Clin Nutr. 2016;103(1):168-177. (PubMed)
24. Zamora-Ros R, Beraud V, Franceschi S, et al. Consumption of fruits, vegetables and fruit juices and differentiated thyroid carcinoma risk in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Int J Cancer. 2017;142(3):449-459. (PubMed)
25. Willett W. Nutritional Epidemiology. 2nd ed. New York: Oxford University Press; 1998.
26. Li B, Jiang G, Zhang G, et al. Intake of vegetables and fruit and risk of esophageal adenocarcinoma: a meta-analysis of observational studies. Eur J Nutr. 2014;53(7):1511-1521. (PubMed)
27. Aune D, Chan DS, Vieira AR, et al. Dietary compared with blood concentrations of carotenoids and breast cancer risk: a systematic review and meta-analysis of prospective studies. Am J Clin Nutr. 2012;96(2):356-373. (PubMed)
28. Vieira AR, Vingeliene S, Chan DS, et al. Fruits, vegetables, and bladder cancer risk: a systematic review and meta-analysis. Cancer Med. 2015;4(1):136-146. (PubMed)
29. Liu H, Wang XC, Hu GH, et al. Fruit and vegetable consumption and risk of bladder cancer: an updated meta-analysis of observational studies. Eur J Cancer Prev. 2015;24(6):508-516. (PubMed)
30. Aune D, Chan DS, Vieira AR, et al. Fruits, vegetables and breast cancer risk: a systematic review and meta-analysis of prospective studies. Breast Cancer Res Treat. 2012;134(2):479-493. (PubMed)
31. Ben Q, Zhong J, Liu J, et al. Association between consumption of fruits and vegetables and risk of colorectal adenoma: a PRISMA-compliant meta-analysis of observational studies. Medicine (Baltimore). 2015;94(42):e1599. (PubMed)
32. Aune D, Lau R, Chan DS, et al. Nonlinear reduction in risk for colorectal cancer by fruit and vegetable intake based on meta-analysis of prospective studies. Gastroenterology. 2011;141(1):106-118. (PubMed)
33. Kashino I, Mizoue T, Tanaka K, et al. Vegetable consumption and colorectal cancer risk: an evaluation based on a systematic review and meta-analysis among the Japanese population. Jpn J Clin Oncol. 2015;45(10):973-979. (PubMed)
34. Vingeliene S, Chan DS, Aune D, et al. An update of the WCRF/AICR systematic literature review on esophageal and gastric cancers and citrus fruits intake. Cancer Causes Control. 2016;27(7):837-851. (PubMed)
35. Wang Q, Chen Y, Wang X, Gong G, Li G, Li C. Consumption of fruit, but not vegetables, may reduce risk of gastric cancer: results from a meta-analysis of cohort studies. Eur J Cancer. 2014;50(8):1498-1509. (PubMed)
36. Bae JM, Kim EH. Dietary intakes of citrus fruit and risk of gastric cancer incidence: an adaptive meta-analysis of cohort studies. Epidemiol Health. 2016;38:e2016034. (PubMed)
37. Yang Y, Zhang D, Feng N, et al. Increased intake of vegetables, but not fruit, reduces risk for hepatocellular carcinoma: a meta-analysis. Gastroenterology. 2014;147(5):1031-1042. (PubMed)
38. Wang M, Qin S, Zhang T, Song X, Zhang S. The effect of fruit and vegetable intake on the development of lung cancer: a meta-analysis of 32 publications and 20,414 cases. Eur J Clin Nutr. 2015;69(11):1184-1192. (PubMed)
39. Vieira AR, Abar L, Vingeliene S, et al. Fruits, vegetables and lung cancer risk: a systematic review and meta-analysis. Ann Oncol. 2016;27(1):81-96. (PubMed)
40. World Cancer Research Fund International/American Institute for Cancer Research. Second Expert Report: Food, Nutrition, Physical activity and the Prevention of Cancer: a Global Perspective. Washington DC: AICR; 2007.
41. World Cancer Research Fund International. Continuous Update Project (CUP). Available at: http://www.wcrf.org/int/research-we-fund/continuous-update-project-cup. Accessed 10/2/17.
42. New SA. Nutrition Society Medal lecture. The role of the skeleton in acid-base homeostasis. Proc Nutr Soc. 2002;61(2):151-164. (PubMed)
43. Lin PH, Ginty F, Appel LJ, et al. The DASH diet and sodium reduction improve markers of bone turnover and calcium metabolism in adults. J Nutr. 2003;133(10):3130-3136. (PubMed)
44. Ebrahimof S, Hoshiarrad A, Hossein-Nezhad A, Larijani B, Kimiagar SM. Effects of increasing fruit and vegetable intake on bone turnover in postmenopausal osteopenic women. Daru. 2010;0(1):30-37.
45. Macdonald HM, Black AJ, Aucott L, et al. Effect of potassium citrate supplementation or increased fruit and vegetable intake on bone metabolism in healthy postmenopausal women: a randomized controlled trial. Am J Clin Nutr. 2008;88(2):465-474. (PubMed)
46. Neville CE, Young IS, Gilchrist SE, et al. Effect of increased fruit and vegetable consumption on bone turnover in older adults: a randomised controlled trial. Osteoporos Int. 2014;25(1):223-233. (PubMed)
47. Hamidi M, Boucher BA, Cheung AM, Beyene J, Shah PS. Fruit and vegetable intake and bone health in women aged 45 years and over: a systematic review. Osteoporos Int. 2011;22(6):1681-1693. (PubMed)
48. Tucker KL, Hannan MT, Chen H, Cupples LA, Wilson PW, Kiel DP. Potassium, magnesium, and fruit and vegetable intakes are associated with greater bone mineral density in elderly men and women. Am J Clin Nutr. 1999;69(4):727-736. (PubMed)
49. Tucker KL, Chen H, Hannan MT, et al. Bone mineral density and dietary patterns in older adults: the Framingham Osteoporosis Study. Am J Clin Nutr. 2002;76(1):245-252. (PubMed)
50. Liu ZM, Leung J, Wong SY, Wong CK, Chan R, Woo J. Greater fruit intake was associated with better bone mineral status among Chinese elderly men and women: results of Hong Kong Mr. Os and Ms. Os studies. J Am Med Dir Assoc. 2015;16(4):309-315. (PubMed)
51. Qiu R, Cao WT, Tian HY, He J, Chen GD, Chen YM. Greater Intake of Fruit and Vegetables Is Associated with Greater Bone Mineral Density and Lower Osteoporosis Risk in Middle-Aged and Elderly Adults. PLoS One. 2017;12(1):e0168906. (PubMed)
52. Benetou V, Orfanos P, Feskanich D, et al. Fruit and vegetable intake and hip fracture incidence in older men and women: The CHANCES Project. J Bone Miner Res. 2016;31(9):1743-1752. (PubMed)
53. Blekkenhorst LC, Hodgson JM, Lewis JR, et al. Vegetable and Fruit Intake and Fracture-Related Hospitalisations: A Prospective Study of Older Women. Nutrients. 2017;9(5). (PubMed)
54. Vinson JA. Oxidative stress in cataracts. Pathophysiology. 2006;13(3):151-162. (PubMed)
55. Huang G, Wu L, Qiu L, Lai J, Huang Z, Liao L. Association between vegetables consumption and the risk of age-related cataract: a meta-analysis. Int J Clin Exp Med. 2015;8(10):18455-18461. (PubMed)
56. Rautiainen S, Lindblad BE, Morgenstern R, Wolk A. Total antioxidant capacity of the diet and risk of age-related cataract: a population-based prospective cohort of women. JAMA Ophthalmol. 2014;132(3):247-252. (PubMed)
57. Wang A, Han J, Jiang Y, Zhang D. Association of vitamin A and beta-carotene with risk for age-related cataract: a meta-analysis. Nutrition. 2014;30(10):1113-1121. (PubMed)
58. Wei L, Liang G, Cai C, Lv J. Association of vitamin C with the risk of age-related cataract: a meta-analysis. Acta Ophthalmol. 2016;94(3):e170-176. (PubMed)
59. Mathew MC, Ervin AM, Tao J, Davis RM. Antioxidant vitamin supplementation for preventing and slowing the progression of age-related cataract. Cochrane Database Syst Rev. 2012(6):Cd004567. (PubMed)
60. Garcia-Layana A, Ciufo G, Toledo E, et al. The effect of a Mediterranean diet on the incidence of cataract surgery. Nutrients. 2017;9(5). (PubMed)
61. Camacho-Barcia ML, Bullo M, Garcia-Gavilan JF, et al. Association of dietary vitamin K1 intake with the incidence of cataract surgery in an adult Mediterranean population: a secondary analysis of a randomized clinical trial. JAMA Ophthalmol. 2017;135(6):657-661. (PubMed)
62. US National Eye Institute. Facts About Age-Related Macular Degeneration. September 2015. Available at: https://nei.nih.gov/health/maculardegen/armd_facts. Accessed 10/6/17.
63. Hogg RE, Woodside JV, McGrath A, et al. Mediterranean diet score and its association with age-related macular degeneration: The European Eye Study. Ophthalmology. 2017;124(1):82-89. (PubMed)
64. Mares JA, Voland RP, Sondel SA, et al. Healthy lifestyles related to subsequent prevalence of age-related macular degeneration. Arch Ophthalmol. 2011;129(4):470-480. (PubMed)
65. Merle BM, Silver RE, Rosner B, Seddon JM. Adherence to a Mediterranean diet, genetic susceptibility, and progression to advanced macular degeneration: a prospective cohort study. Am J Clin Nutr. 2015;102(5):1196-1206. (PubMed)
66. Chiu CJ, Chang ML, Zhang FF, et al. The relationship of major American dietary patterns to age-related macular degeneration. Am J Ophthalmol. 2014;158(1):118-127.e111. (PubMed)
67. Seddon JM, Ajani UA, Sperduto RD, et al. Dietary carotenoids, vitamins A, C, and E, and advanced age-related macular degeneration. Eye Disease Case-Control Study Group. JAMA. 1994;272(18):1413-1420. (PubMed)
68. Snellen EL, Verbeek AL, Van Den Hoogen GW, Cruysberg JR, Hoyng CB. Neovascular age-related macular degeneration and its relationship to antioxidant intake. Acta Ophthalmol Scand. 2002;80(4):368-371. (PubMed)
69. Cho E, Seddon JM, Rosner B, Willett WC, Hankinson SE. Prospective study of intake of fruits, vegetables, vitamins, and carotenoids and risk of age-related maculopathy. Arch Ophthalmol. 2004;122(6):883-892. (PubMed)
70. Moeller SM, Parekh N, Tinker L, et al. Associations between intermediate age-related macular degeneration and lutein and zeaxanthin in the Carotenoids in Age-related Eye Disease Study (CAREDS): ancillary study of the Women's Health Initiative. Arch Ophthalmol. 2006;124(8):1151-1162. (PubMed)
71. Age-Related Eye Disease Study 2 Research Group. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: the Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA. 2013;309(19):2005-2015. (PubMed)
72. Chew EY, Clemons TE, Sangiovanni JP, et al. Secondary analyses of the effects of lutein/zeaxanthin on age-related macular degeneration progression: AREDS2 report No. 3. JAMA Ophthalmol. 2014;132(2):142-149. (PubMed)
73. Hanson C, Rutten EP, Wouters EF, Rennard S. Diet and vitamin D as risk factors for lung impairment and COPD. Transl Res. 2013;162(4):219-236. (PubMed)
74. Tsiligianni IG, van der Molen T. A systematic review of the role of vitamin insufficiencies and supplementation in COPD. Respir Res. 2010;11:171. (PubMed)
75. Tabak C, Smit HA, Rasanen L, et al. Dietary factors and pulmonary function: a cross sectional study in middle aged men from three European countries. Thorax. 1999;54(11):1021-1026. (PubMed)
76. Butland BK, Fehily AM, Elwood PC. Diet, lung function, and lung function decline in a cohort of 2512 middle aged men. Thorax. 2000;55(2):102-108. (PubMed)
77. Tabak C, Arts IC, Smit HA, Heederik D, Kromhout D. Chronic obstructive pulmonary disease and intake of catechins, flavonols, and flavones: the MORGEN Study. Am J Respir Crit Care Med. 2001;164(1):61-64. (PubMed)
78. Walda IC, Tabak C, Smit HA, et al. Diet and 20-year chronic obstructive pulmonary disease mortality in middle-aged men from three European countries. Eur J Clin Nutr. 2002;56(7):638-643. (PubMed)
79. Varraso R, Fung TT, Hu FB, Willett W, Camargo CA. Prospective study of dietary patterns and chronic obstructive pulmonary disease among US men. Thorax. 2007;62(9):786-791. (PubMed)
80. Varraso R, Fung TT, Barr RG, Hu FB, Willett W, Camargo CA, Jr. Prospective study of dietary patterns and chronic obstructive pulmonary disease among US women. Am J Clin Nutr. 2007;86(2):488-495. (PubMed)
81. Kaluza J, Larsson SC, Orsini N, Linden A, Wolk A. Fruit and vegetable consumption and risk of COPD: a prospective cohort study of men. Thorax. 2017;72(6):500-509. (PubMed)
82. Keranis E, Makris D, Rodopoulou P, et al. Impact of dietary shift to higher-antioxidant foods in COPD: a randomised trial. Eur Respir J. 2010;36(4):774-780. (PubMed)
83. Hosseini B, Berthon BS, Wark P, Wood LG. Effects of fruit and vegetable consumption on risk of asthma, wheezing and immune responses: a systematic review and meta-analysis. Nutrients. 2017;9(4). (PubMed)
84. Barros R, Moreira A, Padrao P, et al. Dietary patterns and asthma prevalence, incidence and control. Clin Exp Allergy. 2015;45(11):1673-1680. (PubMed)
85. Garcia-Larsen V, Arthur R, Potts JF, et al. Is fruit and vegetable intake associated with asthma or chronic rhino-sinusitis in European adults? Results from the Global Allergy and Asthma Network of Excellence (GA2LEN) Survey. Clin Transl Allergy. 2017;7:3. (PubMed)
86. Seyedrezazadeh E, Moghaddam MP, Ansarin K, Vafa MR, Sharma S, Kolahdooz F. Fruit and vegetable intake and risk of wheezing and asthma: a systematic review and meta-analysis. Nutr Rev. 2014;72(7):411-428. (PubMed)
87. Miller MG, Thangthaeng N, Poulose SM, Shukitt-Hale B. Role of fruits, nuts, and vegetables in maintaining cognitive health. Exp Gerontol. 2017;94:24-28. (PubMed)
88. Lamport DJ, Saunders C, Butler LT, Spencer JP. Fruits, vegetables, 100% juices, and cognitive function. Nutr Rev. 2014;72(12):774-789. (PubMed)
89. Barberger-Gateau P, Raffaitin C, Letenneur L, et al. Dietary patterns and risk of dementia: the Three-City cohort study. Neurology. 2007;69(20):1921-1930. (PubMed)
90. Dai Q, Borenstein AR, Wu Y, Jackson JC, Larson EB. Fruit and vegetable juices and Alzheimer's disease: the Kame Project. Am J Med. 2006;119(9):751-759. (PubMed)
91. Hughes TF, Andel R, Small BJ, et al. Midlife fruit and vegetable consumption and risk of dementia in later life in Swedish twins. Am J Geriatr Psychiatry. 2010;18(5):413-420. (PubMed)
92. Ritchie K, Carriere I, Ritchie CW, Berr C, Artero S, Ancelin ML. Designing prevention programmes to reduce incidence of dementia: prospective cohort study of modifiable risk factors. BMJ. 2010;341:c3885. (PubMed)
93. Kang JH, Ascherio A, Grodstein F. Fruit and vegetable consumption and cognitive decline in aging women. Ann Neurol. 2005;57(5):713-720. (PubMed)
94. Devore EE, Kang JH, Breteler MM, Grodstein F. Dietary intakes of berries and flavonoids in relation to cognitive decline. Ann Neurol. 2012;72(1):135-143. (PubMed)
95. Loughrey DG, Lavecchia S, Brennan S, Lawlor BA, Kelly ME. The impact of the Mediterranean diet on the cognitive functioning of healthy older adults: a systematic review and meta-analysis. Adv Nutr. 2017;8(4):571-586. (PubMed)
96. Valls-Pedret C, Sala-Vila A, Serra-Mir M, et al. Mediterranean diet and age-related cognitive decline: a randomized clinical trial. JAMA Intern Med. 2015;175(7):1094-1103. (PubMed)
97. Knight A, Bryan J, Wilson C, Hodgson J, Murphy K. A randomised controlled intervention trial evaluating the efficacy of a Mediterranean dietary pattern on cognitive function and psychological wellbeing in healthy older adults: the MedLey study. BMC Geriatr. 2015;15:55. (PubMed)
98. Knight A, Bryan J, Wilson C, Hodgson JM, Davis CR, Murphy KJ. The Mediterranean diet and cognitive function among healthy older adults in a 6-month randomised controlled trial: The MedLey Study. Nutrients. 2016;8(9). (PubMed)
99. Aasheim ET, Sharp SJ, Appleby PN, et al. Tinned fruit consumption and mortality in three prospective cohorts. PLoS One. 2015;10(2):e0117796. (PubMed)
100. National Cancer Institute. Usual dietary intakes: food intakes, US population, 2007-10. https://epi.grants.cancer.gov/diet/usualintakes/pop/2007-10/. Accessed 1/13/18.
101. US Department of Health & Human Services and the US Department of Agriculture. 2015-2020 Dietary Guidelines for Americans. Available at: https://health.gov/dietaryguidelines/2015/guidelines/. Accessed 10/2/17.
102. US Department of Agriculture. All about the fruit group. 5 April 2017. Available at: https://www.choosemyplate.gov/eathealthy/fruits. Accessed 10/2/17.
103. US Department of Agriculture. All about the vegetable group. 05 April 2017. Available at: https://www.choosemyplate.gov/eathealthy/vegetables. Accessed 10/2/17.
104. Produce for Better Health Foundation. Fruits & Veggies — More Matters®. Available at: https://wicworks.fns.usda.gov/resources/fruits-and-veggies-more-mattersr. Accessed 10/22/19.
Cruciferous Vegetables
Contents
Summary
- Cruciferous vegetables are unique in that they are rich sources of sulfur-containing compounds known as glucosinolates. (More information)
- Chopping or chewing raw cruciferous vegetables results in the formation of bioactive glucosinolate hydrolysis products, such as isothiocyanates and indole-3-carbinol. These metabolites are also generated by colonic bacteria, following the ingestion of cooked cruciferous vegetables. (More information)
- The biological activities of glucosinolate-derived isothiocyanates and indole-3-carbinol likely contribute to the potential health-promoting effects of cruciferous vegetables. (More information)
- Variations in the sequence of genes coding for Phase II detoxification enzymes, glutathione S-transferases (GSTs), may influence the potential health benefits of consuming cruciferous vegetables. (More information)
- A few observational studies have examined the link between cruciferous vegetable consumption and cardiovascular health, but the results have been largely inconsistent. (More information)
- High intakes of cruciferous vegetables have been associated with lower risk of bladder, breast, colorectal, endometrial, gastric, lung, ovarian, pancreatic, prostate, and renal cancer. However, evidence of associations remains largely limited to case-control studies. (More information)
- The Dietary Guidelines for Americans recommend for adults to consume 1½-2½ cup-equivalents of dark-green vegetables per week, including cruciferous vegetables, as part of healthy meals. (More information)
Introduction
Cruciferous or Brassica vegetables come from plants in the family known to botanists and biologists as Cruciferae or alternately, Brassicaceae. The Brassicaceae family, which includes the model plant Arabidopsis thaliana, comprises approximately 375 genera and over 3,000 species (1). Many, but not all, commonly consumed cruciferous vegetables come from the Brassica genus; examples include broccoli, Brussels sprouts, cabbage, cauliflower, collard greens, kale, kohlrabi, mustard, rutabaga, turnips, bok choy, and Chinese cabbage (2). Examples of other edible crucifers include radish (Raphanus sativus), horseradish (Armoracia rusticana), watercress (Nasturtium officinale), and wasabi (Wasabia japonica) (2).
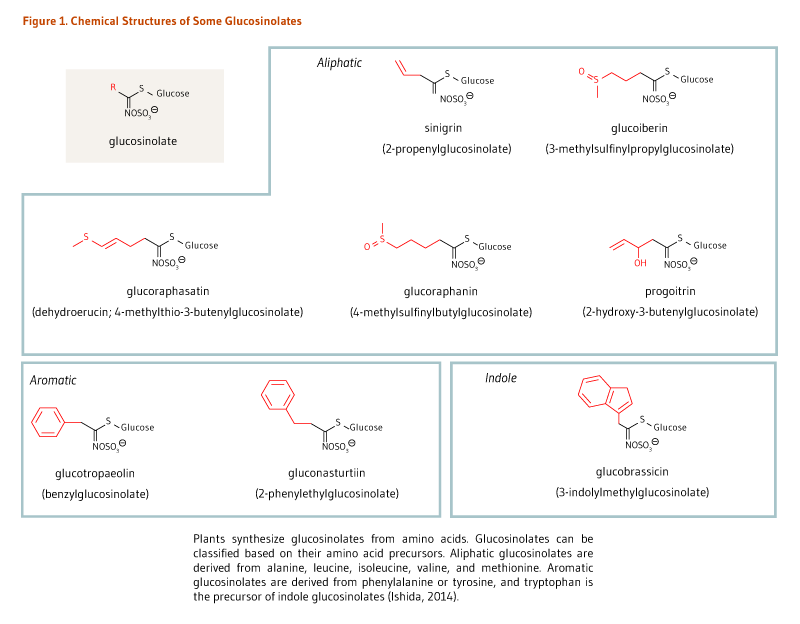
Cruciferous vegetables are unique in that they are a rich source of sulfur-containing compounds called glucosinolates (β-thioglucoside N-hydroxysulfates) that impart a pungent aroma and spicy (some say bitter) taste (Figure 1). Glucosinolates can be classified into three categories based on the chemical structure of their amino acid precursors: aliphatic glucosinolates (e.g., glucoraphanin), indole glucosinolates (e.g., glucobrassicin), and aromatic glucosinolates (e.g., gluconasturtiin) (Figure 1) (1). Around 130 glucosinolate structures have been described to date (3), but only a subset can be found in the human diet. In a cohort of 2,121 German participants in the European Prospective Investigation into Cancer and Nutrition (EPIC study), glucobrassicin, sinigrin, glucoraphasatin (dehydroerucin), glucoraphanin, and glucoiberin were found to contribute most to total glucosinolate intake (4).
Glucosinolates and their breakdown derivatives (metabolites), especially isothiocyanates and indole-3-carbinol, exert a variety of biological activities that may be relevant to health promotion and disease prevention in humans (see the MIC articles on Indole-3-Carbinol and Isothiocyanates).
Metabolism and Bioavailability of Glucosinolates
Metabolism
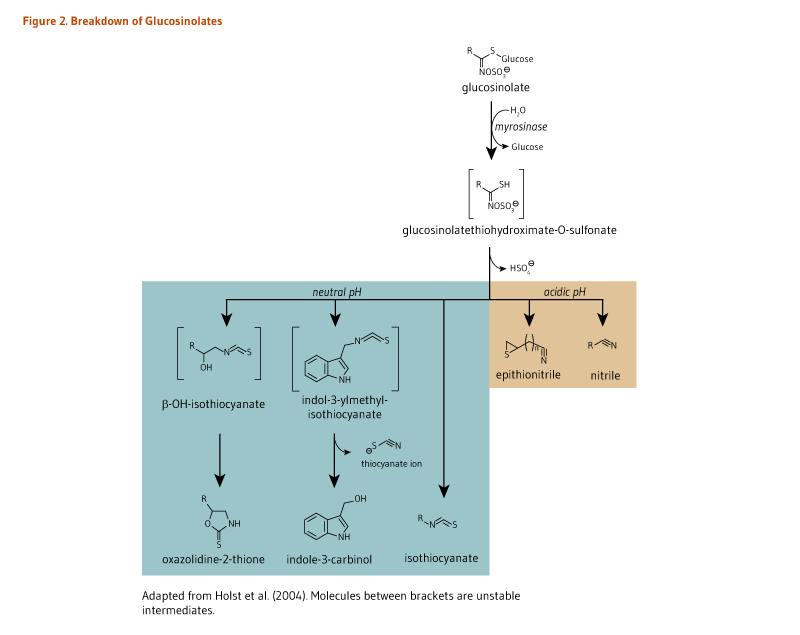
The hydrolysis of glucosinolates, which is catalyzed by a class of enzymes called myrosinases (β-thioglucosidases), leads to the formation of breakdown compounds, such as thiocyanates, isothiocyanates, indoles, oxazolidine-2-thiones (e.g., goitrin), epithionitrile, and nitrile (Figure 2). In intact plant cells, myrosinase is physically separated from glucosinolates. Yet, when plant cells are damaged, myrosinase is released and comes in contact with glucosinolates, catalyzing their conversion into highly reactive metabolites. In plants, thiocyanates, isothiocyanates, epithionitrile, and nitrile are defensive compounds against pathogens, insects, and herbivores (1). When raw cruciferous vegetables are chopped during the cooking process, glucosinolates are rapidly hydrolyzed by myrosinase, generating metabolites that are then absorbed in the proximal intestine. In contrast, boiling cruciferous vegetables before consumption inactivates myrosinase, thus preventing the breakdown of glucosinolates. A small fraction of intact glucosinolates may be absorbed in the small intestine, but a large proportion reaches the colon (5). Of note, boiling cruciferous vegetables has also been found to reduce their glucosinolate content to a much greater extent than steam cooking, microwaving, and stir-frying do (5). Nonetheless, when cruciferous vegetables are cooked, bacterial myrosinase-like activity in the colon is mainly responsible for glucosinolate degradation, generating a wide range of metabolites (5, 6).
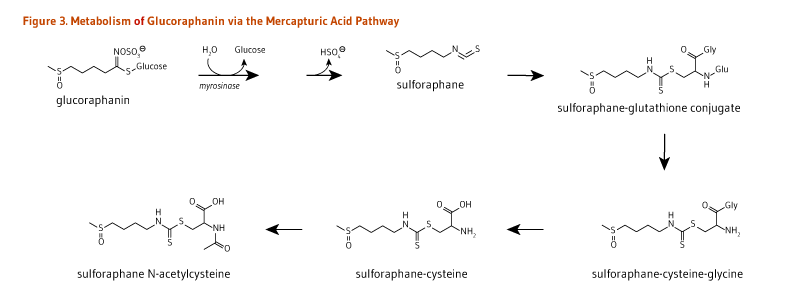
A neutral pH may favor the formation of isothiocyanates from glucosinolates (Figure 2). Once absorbed, isothiocyanates, such as glucoraphanin-derived sulforaphane, are conjugated to glutathione in the liver, and then sequentially metabolized in the mercapturic acid pathway (Figure 3). Sulforaphane metabolites — sulforaphane-glutathione, sulforaphane-cysteine-glycine, sulforaphane-cysteine, and sulforaphane N-acetylcysteine (Figure 3) — collectively known as dithiocarbamates, are ultimately excreted in the urine (5).
Bioavailability
The composition and content of glucosinolates in cruciferous vegetables are relatively stable, yet depend on the genus and species and can vary with plant growing and post-harvest storage conditions and culinary processing (7, 8). Since most cruciferous vegetables are cooked prior to eating, bacterial myrosinase-like activity in the gut rather than plant myrosinase is responsible for the initial step in glucosinolate degradation (Figure 2). In a feeding study involving 45 healthy subjects, the mean conversion rate of glucosinolates (of which 85% was glucoraphanin) to dithiocarbamates over a 24-hour period was estimated to be around 12% with wide variations among participants (range, 1.1 to 40.7%) (7). In contrast, 70%-75% of ingested isothiocyanates were found to be metabolized to dithiocarbamates. Therefore, following the ingestion of cooked cruciferous vegetables, the conversion of glucosinolates into isothiocyanates by gut bacteria appears to be a limiting step in the generation of dithiocarbamates (7). However, differences in individuals’ capacity to metabolize glucosinolates have not been linked to differences in gut microbiota composition (9).
Disease Prevention
Like most other vegetables, cruciferous vegetables are good sources of a variety of nutrients and phytochemicals that synergistically contribute to health promotion (see Bioactive compounds in cruciferous vegetables) (10). One challenge in studying the relationships between cruciferous vegetable intake and disease risk in humans is dissociating the benefits of whole diets that are generally rich in vegetables from those that are specifically rich in cruciferous vegetables (11). One characteristic that sets cruciferous vegetables apart from other vegetables is their high glucosinolate content (see Introduction). Glucosinolate hydrolysis products may play important roles in disease prevention by triggering antioxidant and anti-inflammatory response and contributing to the maintenance of cell homeostasis (see the MIC articles on Isothiocyanates and Indole-3-Carbinol).
Genetic influences
Once absorbed, glucosinolate-derived isothiocyanates (like sulforaphane) are promptly conjugated to glutathione by a class of phase II detoxification enzymes known as glutathione S-transferases (GSTs) (Figure 3). This mechanism is meant to increase the solubility of isothiocyanates, thereby promoting a rapid excretion in the urine. Isothiocyanates are thought to play a prominent role in the potential anticancer and cardiovascular benefits associated with cruciferous vegetable consumption (12, 13). Genetic variations in the sequence of genes coding for GSTs may affect the activity of these enzymes. Such variations have been identified in humans. Specifically, null variants of the GSTM1 and GSTT1 alleles contain large deletions, and individuals who inherit two copies of the GSTM1-null or GSTT1-null alleles cannot produce the corresponding GST enzymes (14). It has been proposed that a reduced GST activity in these individuals would slow the rate of excretion of isothiocyanates, thereby increasing tissue exposure to isothiocyanates after cruciferous vegetable consumption (15). However, human interventional studies with watercress report there is no difference in the isothiocyanate excretion rate between positive (+/+) and null (-/-) genotypes (16). Similar studies with broccoli have shown that GSTM1-/- individuals excreted a greater proportion of ingested sulforaphane via mercapturic acid metabolism than GSTM1+/+ individuals (17, 18). In addition, GSTs are involved in "detoxifying" potentially harmful substances like carcinogens, suggesting that individuals with reduced GST activity might also be more susceptible to cancer (19-21). Finally, induction of the expression and activity of GSTs and other phase II detoxification/antioxidant enzymes by isothiocyanates is an important defense mechanism against oxidative stress and damage associated with the development of diseases like cancer and cardiovascular disease (22). The ability of sulforaphane (glucoraphanin-derived isothiocyanate) to reduce oxidative stress in different settings is linked to activation of the nuclear factor E2-related factor 2 (Nrf2)-dependent pathway. Yet, whether potential protection conferred by isothiocyanates via the Nrf2-dependent pathway is diminished in individuals carrying GST-/- variants is currently unknown.
Some, but not all, observational studies have found that GST genotypes could influence the associations between isothiocyanate intake from cruciferous vegetables and risk of disease (23).
Cardiovascular disease
High intakes of fruit and vegetables have been consistently associated with a reduced risk of cardiovascular disease (CVD) (24, 25). Yet, few observational studies have specifically examined the potential benefits of cruciferous vegetable consumption. In the Shanghai Women’s Health Study (mean follow-up, 10.2 years) and the Shanghai Men’s Health Study (mean follow-up, 4.6 years), which included a total of 134,796 Chinese adults, participants in the highest versus lowest quintile of cruciferous vegetable intakes had a 22% reduced risk of all cause-mortality and a 31% reduced risk of CVD-related mortality (26). In contrast, a pooled analysis of two large US prospective cohort studies, the Nurses’ Health Study (70,870 women) and the Health Professionals’ Follow-Up Study (38,918 men), found no significant association between cruciferous vegetable intake and combined risk of myocardial infarction (MI) and ischemic stroke (27). A case-control study conducted in 2,042 subjects (ages, <75 years) who survived a first acute myocardial infarction (MI), and matched healthy controls with no CVD history found that the individuals in the highest versus lowest tertile of cruciferous vegetable intakes (6 times/week versus <1 time/week) had 27% lower odds of MI (28). However, further analyses showed that the association between cruciferous vegetable intake and MI events was significant in individuals with two functional GSTT1 alleles but not in carriers of two alleles of the GSTT1 null variant (-/-) (28).
Analysis of data from two 12-week randomized controlled trials in 130 participants with mild or moderate CVD risk found that the consumption of 400 g/week of high-glucosinolate broccoli (containing 3 to 6 times more glucoraphanin and glucoiberin than standard broccoli) resulted in a significant reduction in low-density lipoprotein (LDL)-cholesterol concentration in plasma compared with standard broccoli (29). Whether the effect of glucosinolates on cholesterol metabolism might be beneficial in the prevention of CVD needs further investigation.
Cancer
A recent intervention study demonstrated that cruciferous vegetables could increase the detoxification of carcinogens and other xenobiotics in humans. In this 12-week randomized controlled trial in 391 healthy Chinese adults exposed to high levels of air pollution, daily consumption of a broccoli sprout-rich beverage (providing 600 µmol/day of glucoraphanin and 40 µmol/day of sulforaphane) significantly increased the urinary excretion of a known carcinogen, benzene, and a toxicant, acrolein, compared to placebo (20). The biological activities of glucosinolate derivatives, isothiocyanates and indole-3-carbinol, which include modulation of xenobiotic metabolism, but also antioxidant and anti-inflammatory properties, induction of cell cycle arrest and apoptosis, and inhibition of angiogenesis, likely contribute to the potential benefits of cruciferous vegetables in the prevention of cancer (see the MIC articles on Isothiocyanates and Indole-3-Carbinol) (23).
Evidence from observational studies
Numerous observational studies have examined the relationship between cruciferous vegetable intake and cancer risk. Results from recent published meta-analyses of observational studies are reported in Table 1 (adapted from 23).
| Type of Cancer | Type of Observational Studies | Relative Risk [RR] or Odds Ratio [OR] (95% Confidence Interval) | Relative Risk [RR] in Subgroup Analyses (e.g., by food group or study type) | References |
|---|---|---|---|---|
| Bladder cancer | 5 prospective cohort and 5 case-control studies | RR: 0.80 (0.69-0.92) |
RR: 0.78 (0.67-0.89) with case-control studies only RR: 0.86 (0.61-1.11) with cohort studies only |
Liu et al. (2013) (30) |
| 12 prospective cohort and case-control studies | RR: 0.84 (0.77-0.91) | Yao et al. (2014) (31) | ||
| 7 prospective cohort and case-control studies | RR: 0.85 (0.69-1.06) | Vieira et al. (2015) (32) | ||
| 8 prospective cohort studies | RR: 0.97 (0.93-1.01) | Xu et al. (2015) (33) | ||
| Breast cancer | 11 case-control studies | RR: 0.85 (0.77-0.94) | Liu et al. (2013) (34) | |
| Colorectal cancer | 24 case-control and 11 prospective cohort studies | RR: 0.82 (0.75-0.90) |
RR: 0.76 (0.60-0.97) for studies reporting on cabbage intake RR: 0.82 (0.65-1.02) for studies reporting on broccoli intake |
Wu et al. (2013) (35) |
| 11 prospective cohort and 18 case-control studies | OR: 0.92 (0.83-1.01) |
OR: 0.84 (0.72-0.98) for colon cancer OR: 0.99 (0.67-1.46) for rectal cancer OR: 1.09 (0.90-1.33) for colonic adenoma OR: 0.80 (0.65-0.99) for studies reporting on broccoli intake OR: 0.95 (0.80-1.14) for studies reporting on cabbage intake OR: 1.0 (0.75-1.34) for studies reporting on Brussels sprouts |
Tse et al. (2014) (36) | |
| Endometrial cancer | 1 prospective cohort study and 16 case-control studies | OR: 0.79 (0.69-0.90) per 100 g/day | Bandera et al. (2007) (37) | |
| Gastric cancer | 6 prospective cohort and 16 case-control studies | RR: 0.81 (0.75-0.88) |
RR: 0.78 (0.71-0.86) for case-control studies RR: 0.89 (0.77-1.02) for cohort studies RR: 0.68 (0.58-0.80) for studies reporting on cabbage intake |
Wu et al. (2013) (38) |
| Lung cancer | 6 prospective cohort and 13 case-control studies |
RR: 0.77 (0.68-0.88) for case-control studies RR: 0.83 (0.62-1.08) for cohort studies |
Lam et al. (2009) (39) | |
| 5 prospective cohort and 6 case-control studies | RR: 0.75 (0.63-0.89) | Wu et al. (2013) (40) | ||
| Ovarian cancer | 5 prospective cohort and 6 case-control studies | RR: 0.90 (0.82-0.98) |
RR: 0.84 (0.75-0.94) for case-control studies RR: 1.0 (0.85-1.11) for cohort studies |
Han et al. (2014) (41) |
| 4 prospective cohort and 4 case-control studies | RR: 0.89 (0.81-0.99) | Hu et al. (2015) (42) | ||
| Pancreatic cancer | 4 prospective cohort and 5 case-control studies | OR: 0.79 (0.64-0.91) |
OR: 0.72 (0.55-0.89) for case-control studies OR: 0.87 (0.67-1.06) for cohort studies OR: 0.78 (0.55-1.01) for high-quality studies OR: 0.80 (0.66-0.94) for low-quality studies |
Li et al. (2015) (43) |
| Prostate cancer | 7 prospective cohort and 6 case-control studies | RR: 0.90 (0.85-0.96) |
RR: 0.79 (0.69-0.89) for case-control studies RR: 0.95 (0.89-1.02) for cohort studies |
Liu et al. (2012) (44) |
| Renal cell carcinoma | 6 prospective cohort and 6 case-control studies | RR: 0.81 (0.72-0.91) |
RR: 0.89 (0.82-0.98) for high-quality studies RR: 0.72 (0.64-0.81) for case-control studies RR: 0.92 (0.84-1.00) for cohort studies |
Zhao et al. (2013) (45) |
| 3 prospective cohort and 7 case-control studies | RR: 0.73 (0.63-0.83) |
RR: 0.69 (0.60-0.78) for case-control studies RR: 0.96 (0.71-1.21) for cohort studies |
Liu et al. (2013) (46) |
Most meta-analyses found inverse associations between cruciferous vegetable intake and risk of bladder, breast, colorectal, endometrial, gastric, lung, ovarian, pancreatic, prostate, and renal cancer. Subgroup analyses showed that inverse associations remained significant in pooled analyses of case-control studies but not in pooled analyses of prospective cohort studies (see Table 1). Retrospective case-control studies are susceptible to bias in the selection of participants (cases and controls) and prone to dietary recall bias compared to prospective cohort studies, which collect dietary information from participants before they are diagnosed with cancer (47). The method of cooking cruciferous vegetables, which strongly affects the bioavailability and potential anticancer benefits of isothiocyanates (see Metabolism and Bioavailability of Glucosinolates) may be a source of bias and explain variation in the results of the studies (heterogeneity among studies). The lack of information regarding cooking methods prevented data adjustment to reduce bias.
In the past decades, some observational studies have examined the effect of individuals’ genetic variations on the relationship between cruciferous vegetable intake and the risk of different cancer types. For example, a pooled analysis of two prospective cohort studies and six case-control studies found an inverse association between cruciferous vegetable consumption and risk of colorectal neoplasm in carriers of the GSTT1 null variant but not in individuals with the GSTM1 null variant or those with both the GSTT1and GSTM1 null variants (-/-) (36). The results of a pooled analysis of five case-control studies also suggested a stronger association between cruciferous vegetable intake and lung cancer in carriers of both the GSTT1-/- and GSTM1-/- variants compared to carriers of wild-type alleles (+/+); however, it was not reported whether results from these two groups of individuals were significantly different (39). There is also a significant body of evidence suggesting that GSTM1+/+ individuals gain greater cancer protection from consumption total cruciferous vegetables or broccoli compared to GSTM1-/- variant carriers (25, 48, 49). Current evidence is scarce, and adequately powered, well-designed studies are required to assess and explain potential interactions between cruciferous vegetable intake and GST genotypes.
A few observational studies have looked at whether cruciferous vegetable intake could be associated with reduced risks of disease progression and mortality. The highest versus lowest intake of cruciferous vegetables (assessed before diagnosis) was associated with a better survival rate over 72 months after diagnosis in 547 women with lung cancer (50). A prospective study in 29,361 men who underwent a prostate-specific antigen (PSA) test found that intake of cruciferous vegetables was inversely associated with risk of metastatic prostate cancer — cancer that has spread beyond the prostate (i.e., late-stage prostate cancer) — during a mean follow-up of 4.2 years (51). Another prospective study in 1,560 men diagnosed with non-metastatic prostate cancer reported that higher post-diagnosis intake of cruciferous vegetables was associated with a 59% lower risk of prostate cancer progression during a two-year period after completion of the dietary assessment (52). In contrast, cruciferous vegetable consumption in a cohort of 11,390 women with stage I-III invasive breast cancer (from four US and Chinese prospective studies), assessed about two years after diagnosis, was not found to be associated with risk of cancer recurrence or total mortality (53).
Nutrient Interactions
Iodine and thyroid function
Very high intakes of cruciferous vegetables, such as cabbage and turnips, have been found to cause hypothyroidism (insufficient production of thyroid hormones) in animals (54). Two mechanisms can potentially explain this effect. The hydrolysis of progoitrin, found in cruciferous vegetables (see Figure 1), may yield a compound known as goitrin, which may interfere with thyroid hormone synthesis. The hydrolysis of another class of glucosinolates, known as indole glucosinolates, results in the release of thiocyanate ions (see Figure 2) that can compete with iodine for uptake by the thyroid gland (55). However, increased exposure to thiocyanate ions from cruciferous vegetable consumption or, more commonly, from cigarette smoking, does not appear to increase the risk of hypothyroidism unless accompanied by iodine deficiency. One study in humans found that the consumption of 150 g/day (5 oz/day) of cooked Brussels sprouts for four weeks had no adverse effects on thyroid function (56). Similarly, consumption of high amounts of cruciferous vegetables has been associated with increased thyroid cancer risk only in iodine-deficient areas (57).
Intake Recommendations
The 2015-2020 Dietary Guidelines for Americans recommend eating a variety of vegetables daily (2½ cup-equivalents/day for a 2,000 calorie diet) from all of the five vegetable subgroups (dark green, red and orange, legumes, starchy, and other; see 58). No separate recommendations have been established for cruciferous vegetables, yet the 2015-2020 Dietary Guidelines for Americans recommend that adults consume 1½-2½ cup-equivalents of dark-green vegetables (which include cruciferous vegetables) per week (58).
Bioactive compounds in cruciferous vegetables
Cruciferous vegetables are important sources of some vitamins and minerals, fiber, and various phytochemicals other than glucosinolates (Table 2). Many of these compounds likely contribute to the potential health-promoting benefits of cruciferous vegetables.
| Vitamins | Minerals | Phytochemicals |
|---|---|---|
| Folate | Potassium | Carotenoids |
| Vitamin C | Selenium | Chlorophyll |
| Vitamin K | Calcium | Fiber |
| Flavonoids | ||
| Indole-3-Carbinol | ||
| Isothiocyanates | ||
| Lignans | ||
| Phytosterols | ||
| Sulfur bioactives (other than glucosinolates) (59) |
Authors and Reviewers
Originally written in 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in December 2008 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in December 2016 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in April 2017 by:
Maria Traka, Ph.D.
Senior Research Scientist
Chair of the Athena SWAN SAT
Food and Health Programme
Institute of Food Research
Norwich, United Kingdom
Copyright 2005-2024 Linus Pauling Institute
References
2. International Agency for Research on Cancer. Cruciferous vegetables. Cruciferous vegetables, isothiocyanates and indoles. Lyon, France: IARC; 2004:1-12.
3. Agerbirk N, Olsen CE. Glucosinolate structures in evolution. Phytochemistry. 2012;77:16-45. (PubMed)
4. Steinbrecher A, Linseisen J. Dietary intake of individual glucosinolates in participants of the EPIC-Heidelberg cohort study. Ann Nutr Metab. 2009;54(2):87-96. (PubMed)
5. Barba FJ, Nikmaram N, Roohinejad S, Khelfa A, Zhu Z, Koubaa M. Bioavailability of glucosinolates and their breakdown products: impact of processing. Front Nutr. 2016;3:24. (PubMed)
6. Luang-In V, Albaser AA, Nueno-Palop C, Bennett MH, Narbad A, Rossiter JT. Glucosinolate and desulfo-glucosinolate metabolism by a selection of human gut bacteria. Curr Microbiol. 2016;73(3):442-451. (PubMed)
7. Fahey JW, Wehage SL, Holtzclaw WD, et al. Protection of humans by plant glucosinolates: efficiency of conversion of glucosinolates to isothiocyanates by the gastrointestinal microflora. Cancer Prev Res (Phila). 2012;5(4):603-611. (PubMed)
8. Verkerk R, Schreiner M, Krumbein A, et al. Glucosinolates in Brassica vegetables: the influence of the food supply chain on intake, bioavailability and human health. Mol Nutr Food Res. 2009;53 Suppl 2:S219. (PubMed)
9. Li F, Hullar MA, Beresford SA, Lampe JW. Variation of glucoraphanin metabolism in vivo and ex vivo by human gut bacteria. Br J Nutr. 2011;106(3):408-416. (PubMed)
10. Liu RH. Potential synergy of phytochemicals in cancer prevention: mechanism of action. J Nutr. 2004;134(12 Suppl):3479S-3485S. (PubMed)
11. McNaughton SA, Marks GC. Development of a food composition database for the estimation of dietary intakes of glucosinolates, the biologically active constituents of cruciferous vegetables. Br J Nutr. 2003;90(3):687-697. (PubMed)
12. Bai Y, Wang X, Zhao S, Ma C, Cui J, Zheng Y. Sulforaphane protects against cardiovascular disease via Nrf2 activation. Oxid Med Cell Longev. 2015;2015:407580. (PubMed)
13. Higdon JV, Delage B, Williams DE, Dashwood RH. Cruciferous vegetables and human cancer risk: epidemiologic evidence and mechanistic basis. Pharmacol Res. 2007;55(3):224-236. (PubMed)
14. Coles BF, Kadlubar FF. Detoxification of electrophilic compounds by glutathione S-transferase catalysis: determinants of individual response to chemical carcinogens and chemotherapeutic drugs? Biofactors. 2003;17(1-4):115-130. (PubMed)
15. Seow A, Shi CY, Chung FL, et al. Urinary total isothiocyanate (ITC) in a population-based sample of middle-aged and older Chinese in Singapore: relationship with dietary total ITC and glutathione S-transferase M1/T1/P1 genotypes. Cancer Epidemiol Biomarkers Prev. 1998;7(9):775-781. (PubMed)
16. Dyba M, Wang A, Noone AM, et al. Metabolism of isothiocyanates in individuals with positive and null GSTT1 and M1 genotypes after drinking watercress juice. Clin Nutr. 2010;29(6):813-818. (PubMed)
17. Gasper AV, Al-Janobi A, Smith JA, et al. Glutathione S-transferase M1 polymorphism and metabolism of sulforaphane from standard and high-glucosinolate broccoli. Am J Clin Nutr. 2005;82(6):1283-1291. (PubMed)
18. Steck SE, Gammon MD, Hebert JR, Wall DE, Zeisel SH. GSTM1, GSTT1, GSTP1, and GSTA1 polymorphisms and urinary isothiocyanate metabolites following broccoli consumption in humans. J Nutr. 2007;137(4):904-909. (PubMed)
19. Economopoulos KP, Sergentanis TN. GSTM1, GSTT1, GSTP1, GSTA1 and colorectal cancer risk: a comprehensive meta-analysis. Eur J Cancer. 2010;46(9):1617-1631. (PubMed)
20. Egner PA, Chen JG, Zarth AT, et al. Rapid and sustainable detoxication of airborne pollutants by broccoli sprout beverage: results of a randomized clinical trial in China. Cancer Prev Res (Phila). 2014;7(8):813-823. (PubMed)
21. Sergentanis TN, Economopoulos KP. GSTT1 and GSTP1 polymorphisms and breast cancer risk: a meta-analysis. Breast Cancer Res Treat. 2010;121(1):195-202. (PubMed)
22. Bryan HK, Olayanju A, Goldring CE, Park BK. The Nrf2 cell defence pathway: Keap1-dependent and -independent mechanisms of regulation. Biochem Pharmacol. 2013;85(6):705-717. (PubMed)
23. Traka MH. Chapter nine - Health benefits of glucosinolates. Advances in Botanical Research. 2016;80:247-279.
24. Nothlings U, Schulze MB, Weikert C, et al. Intake of vegetables, legumes, and fruit, and risk for all-cause, cardiovascular, and cancer mortality in a European diabetic population. J Nutr. 2008;138(4):775-781. (PubMed)
25. Wang X, Ouyang Y, Liu J, et al. Fruit and vegetable consumption and mortality from all causes, cardiovascular disease, and cancer: systematic review and dose-response meta-analysis of prospective cohort studies. BMJ. 2014;349:g4490. (PubMed)
26. Zhang X, Shu XO, Xiang YB, et al. Cruciferous vegetable consumption is associated with a reduced risk of total and cardiovascular disease mortality. Am J Clin Nutr. 2011;94(1):240-246. (PubMed)
27. Joshipura KJ, Hung HC, Li TY, et al. Intakes of fruits, vegetables and carbohydrate and the risk of CVD. Public Health Nutr. 2009;12(1):115-121. (PubMed)
28. Cornelis MC, El-Sohemy A, Campos H. GSTT1 genotype modifies the association between cruciferous vegetable intake and the risk of myocardial infarction. Am J Clin Nutr. 2007;86(3):752-758. (PubMed)
29. Armah CN, Derdemezis C, Traka MH, et al. Diet rich in high glucoraphanin broccoli reduces plasma LDL cholesterol: Evidence from randomised controlled trials. Mol Nutr Food Res. 2015;59(5):918-926. (PubMed)
30. Liu B, Mao Q, Lin Y, Zhou F, Xie L. The association of cruciferous vegetables intake and risk of bladder cancer: a meta-analysis. World J Urol. 2013;31(1):127-133. (PubMed)
31. Yao B, Yan Y, Ye X, et al. Intake of fruit and vegetables and risk of bladder cancer: a dose-response meta-analysis of observational studies. Cancer Causes Control. 2014;25(12):1645-1658. (PubMed)
32. Vieira AR, Vingeliene S, Chan DS, et al. Fruits, vegetables, and bladder cancer risk: a systematic review and meta-analysis. Cancer Med. 2015;4(1):136-146. (PubMed)
33. Xu C, Zeng XT, Liu TZ, et al. Fruits and vegetables intake and risk of bladder cancer: a PRISMA-compliant systematic review and dose-response meta-analysis of prospective cohort studies. Medicine (Baltimore). 2015;94(17):e759. (PubMed)
34. Liu X, Lv K. Cruciferous vegetables intake is inversely associated with risk of breast cancer: a meta-analysis. Breast. 2013;22(3):309-313. (PubMed)
35. Wu QJ, Yang Y, Vogtmann E, et al. Cruciferous vegetables intake and the risk of colorectal cancer: a meta-analysis of observational studies. Ann Oncol. 2013;24(4):1079-1087. (PubMed)
36. Tse G, Eslick GD. Cruciferous vegetables and risk of colorectal neoplasms: a systematic review and meta-analysis. Nutr Cancer. 2014;66(1):128-139. (PubMed)
37. Bandera EV, Kushi LH, Moore DF, Gifkins DM, McCullough ML. Fruits and vegetables and endometrial cancer risk: a systematic literature review and meta-analysis. Nutr Cancer. 2007;58(1):6-21. (PubMed)
38. Wu QJ, Yang Y, Wang J, Han LH, Xiang YB. Cruciferous vegetable consumption and gastric cancer risk: a meta-analysis of epidemiological studies. Cancer Sci. 2013;104(8):1067-1073. (PubMed)
39. Lam TK, Gallicchio L, Lindsley K, et al. Cruciferous vegetable consumption and lung cancer risk: a systematic review. Cancer Epidemiol Biomarkers Prev. 2009;18(1):184-195. (PubMed)
40. Wu QJ, Xie L, Zheng W, et al. Cruciferous vegetables consumption and the risk of female lung cancer: a prospective study and a meta-analysis. Ann Oncol. 2013;24(7):1918-1924. (PubMed)
41. Han B, Li X, Yu T. Cruciferous vegetables consumption and the risk of ovarian cancer: a meta-analysis of observational studies. Diagn Pathol. 2014;9:7. (PubMed)
42. Hu J, Hu Y, Hu Y, Zheng S. Intake of cruciferous vegetables is associated with reduced risk of ovarian cancer: a meta-analysis. Asia Pac J Clin Nutr. 2015;24(1):101-109. (PubMed)
43. Li LY, Luo Y, Lu MD, Xu XW, Lin HD, Zheng ZQ. Cruciferous vegetable consumption and the risk of pancreatic cancer: a meta-analysis. World J Surg Oncol. 2015;13:44. (PubMed)
44. Liu B, Mao Q, Cao M, Xie L. Cruciferous vegetables intake and risk of prostate cancer: a meta-analysis. Int J Urol. 2012;19(2):134-141. (PubMed)
45. Zhao J, Zhao L. Cruciferous vegetables intake is associated with lower risk of renal cell carcinoma: evidence from a meta-analysis of observational studies. PLoS One. 2013;8(10):e75732. (PubMed)
46. Liu B, Mao Q, Wang X, et al. Cruciferous vegetables consumption and risk of renal cell carcinoma: a meta-analysis. Nutr Cancer. 2013;65(5):668-676. (PubMed)
47. Song JW, Chung KC. Observational studies: cohort and case-control studies. Plast Reconstr Surg. 2010;126(6):2234-2242. (PubMed)
48. Joseph MA, Moysich KB, Freudenheim JL, et al. Cruciferous vegetables, genetic polymorphisms in glutathione S-transferases M1 and T1, and prostate cancer risk. Nutr Cancer. 2004;50(2):206-213. (PubMed)
49. Spitz MR, Duphorne CM, Detry MA, et al. Dietary intake of isothiocyanates: evidence of a joint effect with glutathione S-transferase polymorphisms in lung cancer risk. Cancer Epidemiol Biomarkers Prev. 2000;9(10):1017-1020. (PubMed)
50. Wu QJ, Yang G, Zheng W, et al. Pre-diagnostic cruciferous vegetables intake and lung cancer survival among Chinese women. Sci Rep. 2015;5:10306. (PubMed)
51. Kirsh VA, Peters U, Mayne ST, et al. Prospective study of fruit and vegetable intake and risk of prostate cancer. J Natl Cancer Inst. 2007;99(15):1200-1209. (PubMed)
52. Richman EL, Carroll PR, Chan JM. Vegetable and fruit intake after diagnosis and risk of prostate cancer progression. Int J Cancer. 2012;131(1):201-210. (PubMed)
53. Nechuta S, Caan BJ, Chen WY, et al. Postdiagnosis cruciferous vegetable consumption and breast cancer outcomes: a report from the After Breast Cancer Pooling Project. Cancer Epidemiol Biomarkers Prev. 2013;22(8):1451-1456. (PubMed)
54. Fenwick GR, Heaney RK, Mullin WJ. Glucosinolates and their breakdown products in food and food plants. Crit Rev Food Sci Nutr. 1983;18(2):123-201. (PubMed)
55. Felker P, Bunch R, Leung AM. Concentrations of thiocyanate and goitrin in human plasma, their precursor concentrations in brassica vegetables, and associated potential risk for hypothyroidism. Nutr Rev. 2016;74(4):248-258. (PubMed)
56. McMillan M, Spinks EA, Fenwick GR. Preliminary observations on the effect of dietary brussels sprouts on thyroid function. Hum Toxicol. 1986;5(1):15-19. (PubMed)
57. Cho YA, Kim J. Dietary factors affecting thyroid cancer risk: a meta-analysis. Nutr Cancer. 2015;67(5):811-817. (PubMed)
58. US Department of Health and Human Services and US Department of Agriculture. 2015-2020 Dietary Guidelines for Americans. 8th ed.; 2015.
59. Traka MH, Saha S, Huseby S, et al. Genetic regulation of glucoraphanin accumulation in Beneforte broccoli. New Phytol. 2013;198(4):1085-1095. (PubMed)
Garlic
Contents
Garlic and Organosulfur Compounds
Summary
- Garlic (Allium sativum L.) is a particularly rich source of organosulfur compounds, which are currently under investigation for their potential to prevent and treat disease. (More information)
- The two main classes of organosulfur compounds found in whole garlic cloves are L-cysteine sulfoxides and γ-glutamyl-L-cysteine peptides. (More information)
- Crushing or chopping garlic releases an enzyme called alliinase that catalyzes the formation of allicin from S-allyl-L-cysteine sulfoxide (Allin). Allicin rapidly breaks down to form a variety of organosulfur compounds. (More information)
- In vivo studies indicate that allicin-derived organosulfur compounds may be poorly bioavailable, whereas water-soluble derivatives of γ-glutamyl-L-cysteine peptides have been detected in plasma, liver, and kidney following oral consumption. (More information)
- Several different types of garlic supplements are available commercially, and each type provides a different profile of organosulfur compounds depending on how it was processed. (More information)
- Numerous preclinical studies reported that organosulfur compounds from garlic could exert antioxidant, anti-inflammatory, antimicrobial, anticancer, and cardioprotective activities in various experimental settings. (More information)
- The results of randomized controlled trials suggested that garlic supplementation modestly improves serum lipid profiles in individuals with elevated serum cholesterol and reduces blood pressure in hypertensive subjects, at least in the short term. It is not known whether garlic supplementation can help prevent cardiovascular disease. (More information)
- Current evidence from observational studies does not support an association between high intakes of garlic and prevention of cancer, including gastric and colorectal cancer. It is not known whether garlic-derived organosulfur compounds may be effective in preventing or treating human cancers. (More information)
Introduction
Garlic (Allium sativum L.) has been used for culinary and medicinal purposes in many cultures for centuries (1). Garlic is a particularly rich source of organosulfur compounds, which are thought to be responsible for its flavor and aroma, as well as its potential health benefits (2). Consumer interest in the health benefits of garlic is strong enough to place it among the best-selling herbal supplements in the United States (3). Scientists are interested in the potential for organosulfur compounds derived from garlic to prevent and treat chronic diseases, such as cancer and cardiovascular disease (4).
Organosulfur compounds from garlic
Two classes of organosulfur compounds are found in whole garlic cloves: L-cysteine sulfoxides and γ-glutamyl-L-cysteine peptides.
L-Cysteine sulfoxides
S-allyl-L-cysteine sulfoxide (alliin) accounts for approximately 80% of cysteine sulfoxides in garlic (Figure 1) (5). When raw garlic cloves are crushed, chopped, or chewed, an enzyme known as alliinase is released. Alliinase catalyzes the formation of sulfenic acids from L-cysteine sulfoxides (Figure 2). Sulfenic acids spontaneously react with each other to form unstable compounds called thiosulfinates. In the case of alliin, the resulting sulfenic acids react with each other to form a thiosulfinate known as allicin (half-life in crushed garlic at 23°C is 2.5 days). The formation of thiosulfinates is very rapid and has been found to be complete within 10 to 60 seconds of crushing garlic. Allicin breaks down in vitro to form a variety of fat-soluble organosulfur compounds (Figure 2), including diallyl trisulfide (DATS), diallyl disulfide (DADS), and diallyl sulfide (DAS), or in the presence of oil or organic solvents, ajoene and vinyldithiins (6). In vivo, allicin can react with glutathione and L-cysteine to produce S-allylmercaptoglutathione (SAMG) and S-allylmercaptocysteine (SAMC), respectively (Figure 2) (4).
γ-Glutamyl-L-cysteine peptides
Crushing garlic does not change its γ-glutamyl-L-cysteine peptide content. γ-Glutamyl-L-cysteine peptides include an array of water-soluble dipeptides, including γ-glutamyl-S-allyl-L-cysteine, γ-glutamylmethylcysteine, and γ-glutamylpropylcysteine (see Figure 1). Water-soluble organosulfur compounds, such as S-allylcysteine and SAMC (Figure 3), are formed from γ-glutamyl-S-allyl-L-cysteine during long-term incubation of crushed garlic in aqueous solutions, as in the manufacture of aged garlic extracts (see Sources).
Non-sulfur garlic phytochemicals
Although little is known about their bioavailability and biological activities, non-sulfur garlic phytochemicals, including flavonoids, steroid saponins, organoselenium compounds, and allixin, likely work in synergy with organosulfur compounds (6).
Metabolism and Bioavailability
S-Allyl-L-cysteine sulfoxide (Alliin)
In studies conducted in rodents, orally administrated alliin was found to be absorbed intact and to reach plasma and liver without being converted to allicin. There are no thiosulfinates (like allicin) in intact garlic cloves, and none can be generated in the stomach because alliinase would be irreversibly inhibited under acidic conditions (6).
Allicin and derivatives
The absorption and metabolism of allicin and allicin-derived compounds (see Figure 2) are only partially understood (7). In humans, no allicin has been detected in the serum or urine up to 24 hours after the ingestion of 25 g of raw garlic containing a significant amount of allicin (8). Before ingestion in garlic preparations and after ingestion in the stomach, allicin likely breaks down to release a number of volatile compounds, including DAS and DADS. These organosulfur compounds are metabolized to allyl mercaptan, allyl methyl sulfide, and allyl methyl disulfide, which have been detected in human breath after garlic consumption (9-11). Although a number of biological activities have been attributed to various allicin-derived compounds, it is not yet clear which of these compounds or metabolites actually reaches target tissues (5). Allyl methyl sulfide — but not allyl mercaptan — has been detected in the urine within four hours of garlic ingestion, suggesting that this compound is absorbed into the circulation and rapidly excreted (11). Other allicin-derived compounds, including diallyl sulfides, ajoenes, and vinyldithiins, have not been detected in human blood, urine, or stool, even after the consumption of up to 25 g of fresh garlic or 60 mg of pure allicin (5). These findings suggest that, if they are absorbed, allicin and allicin-derived compounds are rapidly metabolized.
γ-Glutamyl-S-allyl-L-cysteine and derivatives
γ-Glutamyl-S-allyl-L-cysteine is thought to be absorbed intact and hydrolyzed to S-allyl-L-cysteine (SAC) and trans-S-1-propenyl-L-cysteine (see Figure 3), since metabolites of these compounds have been measured in human urine after garlic consumption (12, 13). The consumption of aged garlic extract, a commercial garlic preparation that contains SAC, has been found to increase plasma SAC concentrations in humans (14-16). SAC has been detected in plasma, liver, and kidney of SAC-fed animals (17). Water-soluble organosulfur compounds like SAC and its metabolite, N-acetyl-S-allyl-L-cysteine, may be used as reliable markers of compliance in clinical trials involving garlic intake (6, 18).
Biological Activities
Antioxidant activity
Glutathione
Low cellular concentrations of glutathione, a major intracellular antioxidant, and/or overproduction of reactive oxygen species (ROS) can lead to oxidative stress-induced damage to biological macromolecules and contribute to the development and progression of pathological conditions. In endothelial cells (that line the inner wall of blood vessels), garlic-derived allicin lowered ROS production and increased the concentration of glutathione (19). Oral administration of allicin to mice lowered ROS production and prevented ROS-induced cardiac hypertrophy by inhibiting pro-inflammatory pathways like mitogen-activated protein kinase (MAPK) and phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt)/glycogen synthase kinase 3β (GSK3β) signaling pathways (20). It is thought that, upon crossing cell membranes, allicin interacts with glutathione and forms SAMG (see Figure 2), which could prolong the antioxidant activity of allicin (19).
Nrf2-dependent antioxidant pathway
Allicin was also found to upregulate the expression of glutamate-cysteine ligase (GCL), the rate-limiting enzyme in glutathione synthesis, and other Phase II detoxifying/antioxidant enzymes, likely via the activation of the nuclear factor E2-related factor 2 (Nrf2)-dependent pathway (19). Briefly, Nrf2 is a transcription factor that is bound to the protein Kelch-like ECH-associated protein 1 (Keap1) in the cytosol. Keap1 responds to oxidative stress signals by freeing Nrf2. Upon release, Nrf2 translocates to the nucleus and binds to the antioxidant response element (ARE) located in the promoter of genes coding for antioxidant/detoxifying enzymes and scavengers. Nrf2/ARE-dependent genes code for numerous mediators of the antioxidant response, including GCL, glutathione S-transferases (GSTs), thioredoxin, NAD(P)H quinone oxidoreductase 1 (NQO-1), and heme oxygenase 1 (HO-1) (21). Like allicin, oil-soluble organosulfides, DADS and DATS (see Figure 2), have been shown to stimulate Nrf2-dependent antioxidant pathway (4). For example, antioxidant and cytoprotective effects of DADS against acute ethanol-induced liver damage in mice were associated with the ability to trigger Nrf2-dependent HO-1 activation (22). DATS protected cardiac cells in vitro and in experimental diabetic rats from high glucose-induced oxidative stress and apoptosis by inducing PI3K/Akt-dependent Nrf2 antioxidant signaling (23).
Aged garlic extract have also been shown to increase expression of antioxidant enzymes via the Nrf2/ARE pathway (24). SAC, a major organosulfur compound in aged garlic extract, prevented renal damage caused by ROS in cisplatin-treated rats, by limiting cisplatin-induced reduction of glutathione level, Nrf2 expression, and activity of several antioxidant enzymes (catalase, glutathione reductase, glutathione peroxidase) (25). SAC also protected neurons from oxidative damage and apoptosis in wild-type mice but not in mice without a functional Nrf2 signaling pathway (26).
Nitric oxide (NO) signaling cascade
The generation of nitric oxide (NO) catalyzed by endothelial nitric oxide synthase (eNOS) plays a critical role in protecting the vascular endothelium from oxidative and inflammatory insults (27). ROS-induced NO inactivation can impair vascular endothelial function, contributing to various pathologies like atherosclerosis, hypertension, cardiovascular disease, and central nervous system disorders (27, 28). Interestingly, ingestion of 2 g of fresh garlic was found to increase NO plasma concentrations within two to four hours in healthy volunteers (29). DADS and DATS protected eNOS activity and NO bioavailability in cultured endothelial cells challenged with oxidized low-density lipoprotein (LDL) (30). In a model of traumatic brain injury in rats, allicin attenuated brain edema, neurological deficits, and apoptotic neuronal death, and exhibited antioxidant and anti-inflammatory effects, partly by increasing Akt-mediated eNOS activation (31). Aged garlic extract and SAC were also found to stimulate NO production in different experimental settings (32). In a model of erectile dysfunction in diabetic rats, SAC restored electrically-induced penile erection by stimulating eNOS activity and inhibiting the expression of NADPH oxidase (Nox) responsible for ROS overproduction (33).
Anti-inflammatory activity
Garlic-derived organosulfur compounds have been found to inhibit mediators of the inflammatory response, including cytokines, chemokines, adhesion molecules, and enzymes like cyclooxygenase (COX), lipoxygenase (LOX), and inducible nitric oxide synthase (iNOS) (34-36). Nuclear factor-kappa B (NF-κB) is a transcription factor that binds DNA and induces the transcription of the COX-2 gene, other pro-inflammatory genes, as well as genes involved in cell proliferation, adhesion, survival, and differentiation. The anti-inflammatory effects of organosulfur compounds result from their ability to counteract the activation of pro-inflammatory pathways — like NF-κB-, MAPK-, and PI3K/Akt-dependent signaling pathways — by pro-inflammatory stimuli (4). DATS inhibited bacterial lipopolysaccharide (LPS)-induced macrophage activation by limiting LPS binding to toll-like receptor 4 (TLR4) and blocking the upregulation of TLR4 and TLR4-associated molecule MyoD88 expression (37). DATS also inhibited LPS-induced NF-κB-dependent expression of COX-2, iNOS, tumor necrosis factor-α (TNF-α), and interleukin-1β (IL-1β) (37). In a mouse model of inflammation, the decrease of LPS-induced paw edema by DATS was associated with reduced serum concentrations of the pro-inflammatory cytokines, TNF-α, IL-6, and monocyte chemotactic protein-1 (MCP-1) (36).
Protection of the cardiovascular system
Inhibition of cholesterol synthesis
Garlic and garlic-derived organosulfur compounds have been found to decrease the synthesis of cholesterol by hepatocytes (38). Several garlic-derived organosulfur compounds, including S-allylcysteine and ajoene, have been found to inhibit 3-hydroxy-3-methyl-glutaryl-coenzyme A reductase (HMG-CoA reductase), a critical enzyme in the cholesterol biosynthesis pathway (39, 40). Garlic-derived compounds may also inhibit other enzymes in this pathway, including sterol 4α-methyl oxidase (41).
Inhibition of platelet aggregation
An increase in the ability of platelets to aggregate has been linked to the narrowing of blood vessels and the occurrence of acute thrombotic events. A variety of garlic-derived organosulfur compounds have been found to inhibit platelet aggregation in the test tube (42-44). Aged garlic extract was found to inhibit chemically stimulated platelet aggregation by downregulating the fibrinogen binding activity of glycoprotein IIb/IIIa fibrinogen receptor found on platelets (45, 46) and/or by preventing intraplatelet calcium mobilization (42).
Inhibition of vascular smooth muscle cell (VSMC) proliferation
The proliferation and migration of normally quiescent VSMCs are central features of vascular diseases, including atherosclerosis and coronary restenosis (when treated arteries become blocked again) (47). Although the significance of these findings for human cardiovascular disease is not yet clear, limited cell culture research suggested that organosulfur compounds from garlic could inhibit the proliferation and migration of VSMCs (39, 48, 49).
Inhibition of vascular cell adhesion molecules
An elevation of oxidized low-density lipoprotein (LDL) concentration in plasma has been involved in the pathogenesis of atherosclerosis. Oxidized LDL may stimulate the recruitment of inflammatory white blood cells from the blood to the arterial wall by inducing the expression of vascular cell adhesion molecules. DADS and DATS inhibited the expression of adhesion molecules, E-selectin and vascular cell adhesion molecule-1 (VCAM-1), on endothelial cell surface by reversing oxidized LDL-induced inhibition of PI3K/Akt and cAMP responsive element binding protein (CREB) signaling pathways (50).
Hydrogen sulfide-mediated vasodilatory activity
The preservation of normal arterial function plays an important role in cardiovascular disease prevention. Hydrogen sulfide (H2S), a gaseous signaling molecule produced by some cells within the body, acts as a vasodilator (relaxes blood vessels) and thus may have cardioprotective properties (51, 52). H2S production may be involved in vascular smooth muscle cell relaxation through regulating the opening/closing of potassium channels and/or enhancing NO-dependent signaling pathway (reviewed in 53). A study found that garlic-derived compounds are converted to hydrogen sulfide by red blood cells in vitro (54). However, human consumption of a high dose of raw garlic does not increase breath hydrogen sulfide levels, suggesting that significant metabolism of garlic compounds to hydrogen sulfide does not occur in vivo (11).
Note that the potential benefits of garlic consumption/supplementation on cardiovascular health may also be attributed to antioxidant and anti-inflammatory activities described above.
Anticancer activity
Effects on carcinogen metabolism
Inhibition of metabolic activation of carcinogens: Some chemical carcinogens do not become active carcinogens until they have been metabolized by Phase I biotransformation enzymes, such as those belonging to the cytochrome P450 (CYP) family. Inhibition of specific CYP enzymes involved in carcinogen activation inhibits the development of cancer in some animal models (55). In particular, DAS and its metabolites have been found to inhibit CYP2E1 activity in vitro (56, 57) and when administered orally at high doses to animals (58, 59). Oral administration of garlic oil and DAS to humans has also resulted in evidence of decreased CYP2E1 activity (60-62).
Induction of Phase II detoxifying enzymes: Reactions catalyzed by phase II detoxifying enzymes generally promote the elimination of drugs, toxins, and carcinogens from the body. Consequently, increasing the activity of phase II enzymes, such as glutathione S-transferases (GSTs) and NQO-1, may help prevent cancer by enhancing the elimination of potential carcinogens (see the Nrf2-dependent antioxidant pathway) (63). In animal studies, oral administration of garlic preparations and organosulfur compounds was found to increase the expression and activity of phase II enzymes in a variety of tissues (64-66). For example, DADS protected rodent liver against carbon tetrachloride (CCl4; an environmental pollutant)-induced lipid peroxidation and cell necrosis by blocking CYP2E1-mediated CCL4 metabolic activation and by upregulating Nrf2 downstream genes for NQO-1, HO-1, GCL, GST, and superoxide dismutase (SOD1) (67, 68).
Induction of cell cycle arrest
In normal cells, the cell cycle is tightly regulated to ensure faithful DNA replication and chromosomal segregation prior to cell division. When defects occur during DNA replication or chromosomal segregation and in case of DNA damage, the cell cycle can be transiently arrested at check points to allow for repair. Apoptosis is triggered when repair fails. Defective check points and evasion of apoptosis allow the unregulated division of cancer cells (69). Organosulfur compounds, including allicin, DAS, DADS, DATS, ajoene, and SAMC, have been found to induce cell cycle arrest when added to cancer cells in cell culture experiments (reviewed in 4, 70). DATS reduced the incidence of poorly differentiated prostate tumors and limited the number of metastatic lesions in the lungs of mice genetically modified to develop prostate adenocarcinomas (71). DATS was shown to inhibit cancer cell proliferation, as well as neuroendocrine differentiation — a hallmark of prostate cancer malignancy — but had no effect on apoptosis and markers of invasion (71). In a rat model of chemically induced colon cancer, inhibition of cell proliferation by aged garlic extract was associated with a reduction in the incidence of precancerous lesions and dysplastic adenomas, but not of adenocarcinomas (72).
Induction of apoptosis
Apoptosis is a physiological process of programmed death of cells that are genetically damaged or no longer necessary. Precancerous and cancerous cells are resistant to signals that induce apoptosis (73). Garlic-derived organosulfur compounds, including allicin, ajoene, DAS, DADS, DATS, and SAMC, have been found to induce apoptosis when added to various cancer cell lines grown in culture (reviewed in 4, 70). Oral administration of aqueous garlic extract and S-allylcysteine has been reported to enhance apoptosis in an animal model of oral cancer (74, 75). Garlic oil reduced the incidence of N-nitrosodiethylamine-induced liver nodules by preventing oxidative damage to lipids and DNA and by promoting apoptosis (76). Garlic oil upregulated the activity of various antioxidant enzymes and expression of pro-apoptotic effectors like Bax and Caspase-3 and downregulated the expression of the anti-apoptotic genes β-arrestin-2, Bcl-2, and Bcl-X (76).
Inhibition of angiogenesis
To fuel their rapid growth, invasive tumors must develop new blood vessels by a process known as angiogenesis. Anti-angiogenic properties of several organosulfur compounds, including alliin, DATS, and ajoene, have been observed in in vitro or ex vivo experiments (70). In human breast cancer cells, DADS inhibited TNF-α-induced release of MCP-1, a chemokine that promotes tissue remodeling, angiogenesis, and metastasis (77). Aged garlic extract was also found to suppress in vitro angiogenesis by inhibiting endothelial cell proliferation, loss of adhesion, motility, and tube formation (78).
Antimicrobial activity
Garlic extracts have been found to have antibacterial and antifungal properties (79, 80). Thiosulfinates, particularly allicin, are thought to play an important role in the antimicrobial activity of garlic (80-82). Allicin-derived compounds, including DATS and ajoene, also have some antimicrobial activity in vitro (5). To date, randomized controlled trials using oral garlic preparations have not provided strong evidence for such activity in humans (83-85). A small randomized controlled trial found that application of 1% ajoene cream to the skin twice daily was as effective in treating tinea pedis (fungal skin infection known as athlete’s foot) as 1% terbinafine (Lamisil) cream (86). In another preliminary randomized controlled trial, circulating immune innate cells (γδT-lymphocytes and natural killer (NK) cells), isolated from healthy adults supplemented with aged garlic extract, proliferated better in ex vivo culture than those from volunteers who consumed a placebo, suggesting a greater pathogen-fighting ability. The number of self-reported illnesses was similar between groups after 90 days of aged garlic extract or placebo supplementation, but aged garlic extract significantly reduced the severity of self-reported cold or flu symptoms (87).
Disease Prevention
Cardiovascular disease
Interest in garlic and its potential to prevent cardiovascular disease began with observations that people living near the Mediterranean basin had lower mortality from cardiovascular disease (88). Garlic is a common ingredient in Mediterranean cuisine, but a number of characteristics of the "Mediterranean diet" have been proposed to explain its cardioprotective effects (89). Although few observational studies have examined associations between garlic consumption and cardiovascular disease risk, numerous intervention trials have explored the effects of garlic supplementation on cardiovascular disease risk factors.
Platelet aggregation
Platelet aggregation is one of the first steps in the formation of blood clots that can occlude coronary or cerebral arteries, leading to myocardial infarction or ischemic stroke, respectively. Evidence that garlic inhibits platelet aggregation is based mainly on in vitro experiments and a small number of ex vivo assays. Of 10 randomized controlled trials that tested the antithrombotic effect of garlic preparation, four reported a modest but significant decrease in ex vivo platelet aggregation with garlic supplementation compared to placebo (reviewed in 90). Because garlic oil extract in particular may have antithrombotic activity, a small randomized controlled trial in 12 healthy adults was conducted to test the acute effect of one large dose of garlic oil (extracted from 9.9 g of fresh garlic) on ex vivo platelet aggregation (91). The garlic oil extract had a mild effect on adrenaline-induced platelet aggregation (12% reduction) but had no effect on adenosine diphosphate (ADP)- or collagen-induced aggregation measured four hours post-consumption. Another study in 14 healthy volunteers showed that aged garlic extract dose-dependently inhibited ADP-stimulated platelet aggregation by downregulating the fibrinogen binding activity of glycoprotein IIb/IIIa fibrinogen receptor found on platelets (46).
Serum lipid profiles
A recent systematic review of randomized controlled trials examining the effect of supplementation with various garlic preparations on serum lipid profiles in individuals with elevated and normal serum cholesterol levels reported mixed results (92). The most recent and comprehensive meta-analysis compared the results from 39 randomized controlled trials published between 1955 and 2011 that tested the effect of garlic preparations on serum lipid concentrations (93). These 39 trials studied 2,298 adult participants (mean age, 49.5 years), administered garlic-only preparations, used a true placebo, and lasted for at least two weeks. The majority of included trials recruited subjects with elevated total cholesterol at baseline (>200 mg/dL [>5.2 mmol/L], 29 trials) and lasted more than eight weeks (30 trials). The authors found that garlic preparations significantly lowered total cholesterol and low-density lipoprotein (LDL)-cholesterol compared to placebo. High-density lipoprotein (HDL)-cholesterol concentrations were mildly increased and triglyceride concentrations were not affected by garlic supplementation. All administered garlic preparations (garlic powder, aged garlic extract, garlic oil, and fresh garlic) were well tolerated and associated with only minor side effects (garlic odor and mild gastrointestinal discomfort) (93).
Although garlic supplementation for a minimum of two months may lower total- and LDL-cholesterol concentrations in individuals with elevated total cholesterol, the benefits may not last beyond the short term (90, 92). Whether garlic possesses long-lasting lipid-lowering effects remain questionable and future investigations may focus on ways to maximize potential benefits of garlic preparations on serum lipids.
Atherosclerosis
Very few studies have attempted to assess the effect of garlic supplementation on the progression of atherosclerosis in humans. One early study in Germany used ultrasound imaging to assess the effect of 900 mg/day of dehydrated garlic on the progression of atherosclerotic plaque in the carotid and femoral arteries (94). After four years, the increase in plaque volume was significantly greater in women taking the placebo (+53.1%) than in women taking the garlic supplement (-4.6%), while no significant difference in plaque volume was found between garlic (+1.1%) and placebo (+5.5%) in men (94, 95). In a smaller pilot study, investigators measured coronary artery calcium using electron-beam computed tomography to assess the effect of supplementation with aged garlic extract on the progression of atherosclerosis in 19 adults already taking HMG-CoA reductase inhibitors (lipid-lowering drugs also known as statins) (18). After one year, increases in coronary artery calcium score were significantly lower in those taking aged garlic extract than in those taking a placebo. Nevertheless, although coronary calcium scores may have a predictive value regarding future cardiac events in asymptomatic subjects, it may not be a reliable marker of plaque burden in symptomatic patients (96, 97). In a recent double-blind, controlled study, the extent of coronary atherosclerosis was assessed with cardiac computed tomography angiography in 72 individuals (55 at study completion) at high risk of coronary heart disease randomized to receive either 2,400 mg of aged garlic extract or placebo for 52 weeks (98). The result suggested a significant decrease in the extent of coronary plaques with low-attenuation area (a type of vulnerable plaques prone to rupture) (99, 100) with aged garlic extract compared to placebo, but no differences in total plaque volume and proportions of non-calcified plaques and dense calcium were found between treatment and placebo groups (98).
Hypertension
Most systematic reviews and/or meta-analyses of randomized controlled trials to date have provided mixed results regarding the potential blood pressure-lowering effect of garlic, possibly because most of these trials enrolled both normotensive and hypertensive subjects (90, 101-105).
A systematic review and meta-analysis by Xiong et al. (106) included seven randomized, placebo-controlled trials that exclusively enrolled individuals with high blood pressure, i.e., with systolic blood pressure (SBP) ≥140 mm Hg and/or diastolic blood pressure (DBP) ≥90 mm Hg. Five out of seven trials identified in this systematic review reported statistically significant reductions in SBP and DBP with several garlic preparations (dried garlic homogenate, garlic powder, and aged garlic extract) (106). Another recent meta-analysis included nine randomized controlled trials in 482 hypertensive individuals who were given garlic powder (six studies), garlic homogenate (one study), aged garlic extract (two studies), or placebo for 8 to 26 weeks (107). Garlic preparations were found to significantly reduce SBP by a mean of 9.1 mm Hg and DBP by a mean of 3.8 mm Hg compared to placebo. The most recent meta-analysis found that garlic preparations reduced SBP by a mean 8.7 mm Hg (10 trials, 440 subjects) and DBP by 6.1 mm Hg (8 trials, 257 subjects) (102). Such reductions in blood pressure seem comparable to those reported with currently used classes of blood pressure-lowering medications (average reduction, -9.1 mm Hg for SBP and -5.5 mm Hg for DBP) (108). The effect of blood pressure reduction from such medications at standard dose has been estimated to lower the risk of coronary heart disease events by about one-quarter and the risk of stroke by about one-third (108). Nonetheless, evidence showing that garlic supplements may reduce the risk of cardiovascular morbidity and mortality is still lacking (109).
In a recent 12-week, randomized, placebo-controlled trial in untreated hypertensive subjects, daily intake of aged garlic extract (1.2 g of which contained 1.2 mg of S-allyl-L-cysteine [SAC]) was shown to significantly lower SBP by 11 mm Hg and DBP by 6 mm Hg on average in 50%-60% of participants, but reductions in blood pressure were not reported in 40%-50% of participants compared to placebo (110). Whether interindividual differences in nutritional status and genetic polymorphisms can explain differences in blood pressure response to garlic treatment need to be explored in future studies (53, 110).
Overall, short-term garlic supplementation appears to effectively reduce blood pressure with minimal side effects in hypertensive patients.
Summary
The results of randomized controlled trials have suggested that garlic supplementation modestly improves serum lipid profiles in individuals with elevated serum cholesterol and reduces blood pressure in hypertensive subjects. It is not yet clear whether garlic supplementation can reduce atherosclerosis or prevent cardiovascular events, such as myocardial infarction or stroke.
Cancer
Gastric cancer
A recent meta-analysis of 17 studies (mostly case-control studies) reported an inverse association between high versus low garlic consumption and the risk of gastric cancer (111). Nevertheless, this conclusion is hindered by a number of limitations, especially related to the retrospective design of most studies included in the analysis, as well as great variations in the amount and duration of garlic intakes. In a 2009 review of the literature, Kim et al. (112) identified 20 human studies that examined garlic intake in relation to gastric cancer risk: three intervention studies, one case-cohort study, 13 case-control studies, and three cross-sectional/ecologic studies. Using the Food and Drug Administration (FDA)’s evidence-based criteria for the scientific evaluation of health claims (113), the authors excluded 16 studies for methodological flaws; only four studies (two case-control (114, 115), one case-cohort (116), and one intervention (85)) received moderate-to-high quality ratings (112). Among these four studies, garlic intake during adolescence or 20 years prior to the interview was not found to be associated with the risk of gastric cancer in one of the case-control studies in Sweden (338 gastric cancer patients and 669 control subjects) (114). Another case-control study in Korea failed to show an association between past garlic consumption and gastric cancer in 136 people diagnosed with gastric cancer and 136 cancer-free subjects (115). In addition, a prospective case-cohort study in the Netherlands found no association between the use of garlic supplements (unknown composition) and gastric cancer risk (116). Finally, a randomized, double-blind, placebo-controlled intervention study in 3,365 subjects from the Shandong province of China found that supplementation with aged garlic extract and steam-distilled garlic oil for 7.3 years did not reduce the prevalence of precancerous gastric lesions or the incidence of gastric cancer (85). An updated analysis of the data collected 7.3 years after garlic supplementation ended provided further confirmation for a lack of significant reduction in gastric cancer incidence or mortality with supplemental garlic (117).
Helicobacter pylori (H. pylori) infection and gastric cancer: Infection with some strains of H. pylori bacteria markedly increases the risk of gastric cancer. Although garlic preparations and organosulfur compounds could inhibit the growth of H. pylori in the laboratory (118, 119), there is little evidence to suggest that high garlic intakes or garlic supplementation may help prevent or eradicate H. pylori infection in humans (120). Higher intakes of garlic were not associated with a significantly lower prevalence of H. pylori infection in China or Turkey (121, 122). Moreover, clinical trials using garlic cloves (123), aged garlic extract (84), steam-distilled garlic oil (84, 124), garlic oil macerate (125), or garlic powder (126) have not found garlic supplementation to be effective in eradicating H. pylori infection in humans.
Colorectal cancer
A 2014 meta-analysis of prospective cohort studies in 335,923 subjects (including 4,610 colorectal cancer [CRC] cases) found no association of consuming raw or cooked garlic (three studies, four cohorts) or supplemental garlic (four studies, five cohorts) with CRC (127). Another recent systematic review and meta-analysis that combined data from seven cohort and seven case-control studies also failed to find a statistically significant reduction in CRC risk with garlic intake (128). Yet, these results are in contrast with previous pooled analyses of data from case-control studies (129) or from both case-control and prospective studies (130) that reported an approximate 30% lower CRC risk in individuals with the highest garlic intakes compared to those with the lowest intakes. Inclusion of case-control studies, which are more susceptible to bias, may explain these discrepancies among meta-analyses (128). For information regarding different types of epidemiological studies, see the Spring/Summer 2016 LPI Research Newsletter.
A small preliminary intervention trial in 37 patients with colorectal adenomas examined whether supplementation with aged garlic extract for 12 months affected adenoma size and recurrence. Both the number and size of adenomas were significantly reduced in patients given a high dose of aged garlic extract compared to those given a much lower dose (0.16 mL/day) (131, 132). Larger randomized controlled trials are needed to determine whether garlic or garlic extracts can substantially reduce adenoma progression to advanced cancer and recurrence.
Other types of cancer
In a small, placebo-controlled intervention study in 50 patients with cancer (42 with liver cancer, seven with pancreatic cancer, and one with colon cancer), supplementation with 500 mg/day of aged garlic extract for six months failed to prevent quality of life deterioration caused by disease progression and chemotherapy-associated adverse effects (133). Yet, the active treatment limited the decline in natural killer cell count and activity that accompanies digestive cancer progression and reduces patient survival (133).
At present, evidence from trials is limited and results from observational studies do not suggest a role of high intakes of garlic in the prevention of cancer in humans (112).
Sources
Food sources
Allium vegetables, including garlic and onions, are the richest sources of organosulfur compounds in the human diet (134). To date, the majority of scientific research relating to the health effects of organosulfur compounds has focused on those derived from garlic. Fresh garlic cloves contain about 2 to 6 mg/g of γ-glutamyl-S-allyl-L-cysteine (0.2%-0.6% fresh weight) and 6 to 14 mg/g of alliin (0.6%-1.4% fresh weight). Garlic cloves yield about 2.5 to 4.5 mg of allicin per gram of fresh weight when crushed. One fresh garlic clove weighs 2 to 4 g (5).
Effects of cooking
The enzyme alliinase can be inactivated by heat. In one study, microwave cooking of unpeeled, uncrushed garlic totally destroyed alliinase enzyme activity (135). An in vitro study found that prolonged oven heating or boiling (i.e., six minutes or longer) suppressed the inhibitory effect of uncrushed and crushed garlic on platelet aggregation, but crushed garlic retained more anti-aggregatory activity compared to uncrushed garlic (136). Administering raw garlic to rats significantly decreased the amount of DNA damage caused by a chemical carcinogen, but heating uncrushed garlic cloves for 60 seconds in a microwave oven or 45 minutes in a convection oven prior to administration blocked the protective effect of garlic (137). The protective effect of garlic against DNA damage can be partially conserved by crushing garlic and allowing it to stand for 10 minutes prior to microwave heating for 60 seconds or by cutting the tops off garlic cloves and allowing them to stand for 10 minutes before heating in a convection oven. Because organosulfur compounds derived from alliinase-catalyzed reactions may play a role in some of the biological effects of garlic, some scientists recommend that crushed or chopped garlic be allowed to "stand" for at least 10 minutes prior to cooking (135).
Supplements
Several different types of garlic preparations are available commercially, and each type provides a different profile of organosulfur compounds depending on how it was processed (see Table 1). Not all garlic preparations are standardized, and even standardized brands may vary with respect to the amount and the bioavailability of the organosulfur compounds they provide (5).
Powdered (dehydrated) garlic
Powdered or dehydrated garlic is made from garlic cloves that are usually sliced and dried at a low temperature to prevent alliinase inactivation (138). The dried garlic is pulverized and often made into tablets. To meet United States Pharmacopeial Convention (USP) standards, powdered garlic supplements must contain no less than 0.1% γ-glutamyl-S-allyl-L-cysteine and no less than 0.3% alliin (dry weight) (139). Although powdered garlic supplements do not actually contain allicin, the manufacturer may provide a value for the "allicin potential" or "allicin yield" of a supplement on the label. These values represent the maximum achievable allicin yield of a supplement (140). It is determined by dissolving powdered garlic in water at room temperature and measuring the allicin content after 30 minutes (139). Because alliinase is inactivated by the acidic pH of the stomach, most powdered garlic tablets are enteric-coated to keep them from dissolving before they reach the neutral pH of the small intestine. It has been argued that it is more appropriate to measure "allicin release" using a USP method for assessing drug release from enteric-coated tablets under conditions that mimic those of the stomach and intestine (139). Allicin release by this method has been shown to parallel true bioavailability (140). Most tablet brands have been found to produce little allicin under these conditions, due mainly to low alliinase activity and prolonged disintegration times (140, 141). Many manufacturers provide information on the "allicin potential" of their powdered garlic supplements, but few provide information on the "allicin release." A number of controlled clinical trials have examined the effect of powdered or dehydrated garlic supplements on cardiovascular risk factors (see Cardiovascular disease). The most commonly used doses ranged from of 600 to 900 mg/day and provided 3.6 to 5.4 mg/day of potential allicin (90).
Garlic fluid extracts (aged garlic extract™)
When garlic cloves are incubated in a solution of ethanol and water for up to 20 months, allicin is mainly converted to allyl sulfides, which are lost by evaporation or converted to other compounds (138). The resulting extract contains primarily water-soluble organosulfur compounds, such as SAC and SAMC (see Figure 3) (142). Garlic fluid extracts, including aged garlic extracts, are standardized to their S-allyl-L-cysteine content. In controlled clinical trials, daily intakes of aged garlic extract at doses between 1.2 g-2.4 g (containing 1.2 to 2.4 mg of S-allyl-L-cysteine) consistently resulted in reductions in SBP by 9 mm Hg-10 mm Hg and reductions in DBP by 4 mm Hg-8 mm Hg in a majority of patients with uncontrolled hypertension (110, 143). Additionally, aged garlic extract at doses of 2.4 to 7.2 g/day resulted in short-term reductions in ex vivo platelet aggregation (144) and reductions in serum cholesterol concentrations up to 12 weeks (16).
Steam-distilled garlic oil
Steam distillation of crushed garlic cloves results in a product that contains mainly allyl sulfides, including DATS, DADS, and DAS (see Figure 2) (138). These fat-soluble steam distillation products are usually dissolved in vegetable oil.
Garlic oil macerates
Incubation of crushed garlic cloves in oil at room temperature results in the formation of vinyldithiins and ajoene from allicin, in addition to allyl sulfides, such as DADS and DATS (see Figure 2) (5). Ether extracts are similar in composition to garlic oil macerates but more concentrated (145).
| Product | Principal Organosulfur Compounds | Delivers Allicin-derived Compounds? |
|---|---|---|
| Fresh garlic cloves |
Cysteine sulfoxides (Alliin) |
Yes, when chopped, crushed, or chewed raw. Minimal, when garlic cloves are cooked before crushing or chopping. |
| Garlic powder (tablets) |
Cysteine sulfoxides (Alliin) γ-Glutamyl-L-cysteine peptides |
Varies greatly among commercial products. Enteric-coated tablets that pass the USP allicin release test are likely to provide the most. |
| Steam-distilled garlic oil (capsules) |
Diallyl disulfide (DADS) Diallyl trisulfide (DATS) Allyl methyl trisulfide |
Yes, but there is only 1% of oil-soluble sulfur compounds in 99% of vegetable oil. |
| Garlic oil macerate (capsules) |
Vinyldithiins (E/Z)-ajoene Diallyl trisulfide |
Yes |
|
Aged garlic extract™ (tablets or capsules) |
S-Allyl-L-cysteine (SAC) S-Allylmercaptocysteine (SAMC) trans-S-1-Propenyl-L-cysteine |
Minimal |
Safety
Adverse effects
The most commonly reported adverse effects of oral ingestion of garlic and garlic supplements are breath and body odor (90, 146). Gastrointestinal symptoms have also been reported, including heartburn, abdominal pain, belching, nausea, vomiting, flatulence, constipation, and diarrhea (106). The most serious adverse effects associated with oral garlic supplementation are related to uncontrolled bleeding. Several cases of serious postoperative or spontaneous bleeding associated with garlic supplementation have been reported in the medical literature (147-150). Garlic may also trigger allergic responses in some individuals, including asthma in people with occupational exposure to garlic powder or dust (151). Exposure of the skin to garlic has been reported to cause contact dermatitis in some individuals (146, 152). More serious skin lesions, including blisters and burns, have also been reported with topical exposure to garlic for six or more hours.
The safety characteristics of the various garlic preparations likely depend on their specific chemical composition (see Table 1). Aged garlic extract — the only water-based garlic supplement — showed a safe profile in toxicity studies and exhibited no undesirable side effects when combined with anticoagulants (warfarin), antiplatelets (aspirin), cholesterol-lowering (statins) drugs, or anticancer drugs (doxorubicin, 5-fluorouracil, methotrexate) in clinical settings (reviewed in 6). Safety and toxicity data are lacking for lipophilic (hydrophobic) garlic preparations, but some of their constituents have been shown to interfere with drug-metabolizing enzymes and transporters (see Drug interactions).
Pregnancy and lactation
No adverse effects on pregnancy outcomes have been reported when garlic is consumed in the diet. Although no adverse pregnancy outcomes were reported in a study of Iranian women who took dehydrated garlic tablets (800 mg/day) for two months during the third trimester of pregnancy (153), the safety of garlic supplements in pregnancy has not been established. There is some evidence that garlic consumption alters the odor and possibly the flavor of breast milk. In a controlled cross-over trial, oral consumption of 1.5 g of garlic extract by lactating women increased the perceived intensity of breast milk odor (154). Infants spent more time breast-feeding after their mothers consumed the garlic extract compared to a placebo, but the amount of milk consumed and number of feedings were not significantly different. Additionally, it is not known if topical use of garlic is safe during pregnancy or lactation.
Drug interactions
Anticoagulant medications
Garlic may enhance the anticoagulant effects of warfarin (Coumadin). There have been two case reports in which prothrombin time (INR) increased in patients who started taking garlic tablets or garlic oil without changing their warfarin dose or other habits (155). However, a more recent study in closely monitored patients on warfarin therapy found that garlic fluid extracts (aged garlic extract) did not increase hemorrhagic risk (156). Since garlic supplements have been found to inhibit platelet aggregation (90), there is a potential for additive effects when garlic supplements are taken together with other medications or supplements that inhibit platelet aggregation, such as high-dose fish oil or vitamin E (157). More research is needed to determine whether garlic supplements are safe for people on anticoagulatory therapy.
HIV protease inhibitors
Supplementation of healthy volunteers with garlic caplets twice daily (allicin yield, 7.2 mg/day) for three weeks resulted in a 50% decrease in the bioavailability of the protease inhibitor, saquinavir (Fortovase) (158). Although saquinavir undergoes significant metabolism by CYP3A4, supplementation with garlic extract for two weeks did not significantly alter a measure of CYP3A4 activity in healthy volunteers (159). Garlic extract supplementation (10 mg/day) for four days did not significantly alter single-dose pharmacokinetics of the protease inhibitor, ritonavir (Norvir), but further research is needed to determine steady-state interactions between well-characterized garlic supplements and ritonavir (160). In vitro hepatic models suggested that flavonoids and sulfur-containing compounds in garlic supplements might interfere with the activity of efflux drug transporters of the ATP-binding cassette (ABC) family, including P-glycoprotein, multidrug resistance protein (MRP), and breast cancer-resistance protein (BCRP), which function as ATP-dependent efflux pumps that actively regulate the excretion of a number of drugs limiting their systemic bioavailability. They may also affect the activity of phase I biotransformation enzymes like cytochrome P450 (CYP) 3A4 (CYP3A4) (161, 162). Modifications of efflux transporter and CYP3A4 activities may explain how supplementation with garlic phytochemicals might hinder the therapeutic efficacy of medications like antiretroviral drugs (162).
Authors and Reviewers
Originally written in 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in July 2008 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in September 2016 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in December 2016 by:
Karin Ried, Ph.D., MSc.
Research Director
National Institute of Integrative Medicine
Copyright 2005-2024 Linus Pauling Institute
References
1. Guercio V, Galeone C, Turati F, La Vecchia C. Gastric cancer and allium vegetable intake: a critical review of the experimental and epidemiologic evidence. Nutr Cancer. 2014;66(5):757-773. (PubMed)
2. Block E. The chemistry of garlic and onions. Sci Am. 1985;252(3):114-119.
3. Blumenthal M. Herb Sales Down 7.4 Percent in Mainstream Market. HerbalGram: American Botanical Council; 2005:63.
4. Trio PZ, You S, He X, He J, Sakao K, Hou DX. Chemopreventive functions and molecular mechanisms of garlic organosulfur compounds. Food Funct. 2014;5(5):833-844. (PubMed)
5. Lawson LD. Garlic: a review of its medicinal effects and indicated active compounds. In: Lawson LD, Bauer R, eds. Phytomedicines of Europe: Chemistry and Biological Activity. Washington, D. C.: American Chemical Society; 1998:177-209.
6. Amagase H. Clarifying the real bioactive constituents of garlic. J Nutr. 2006;136(3 Suppl):716S-725S. (PubMed)
7. Lawson LD, Wang ZJ. Allicin and allicin-derived garlic compounds increase breath acetone through allyl methyl sulfide: use in measuring allicin bioavailability. J Agric Food Chem. 2005;53(6):1974-1983. (PubMed)
8. Lawson LD, Hughes BG. Characterization of the formation of allicin and other thiosulfinates from garlic. Planta Med. 1992;58(4):345-350. (PubMed)
9. Minami T, Boku T, Inada K, Morita M, Okasaki Y. Odor components of human breath after the ingestion of grated raw garlic. J Food Sci. 1989;54:763-765.
10. Rosen RT, Hiserodt RD, Fukuda EK, et al. Determination of allicin, S-allylcysteine and volatile metabolites of garlic in breath, plasma or simulated gastric fluids. J Nutr. 2001;131(3s):968S-971S. (PubMed)
11. Suarez F, Springfield J, Furne J, Levitt M. Differentiation of mouth versus gut as site of origin of odoriferous breath gases after garlic ingestion. Am J Physiol. 1999;276(2 Pt 1):G425-430. (PubMed)
12. de Rooij BM, Boogaard PJ, Rijksen DA, Commandeur JN, Vermeulen NP. Urinary excretion of N-acetyl-S-allyl-L-cysteine upon garlic consumption by human volunteers. Arch Toxicol. 1996;70(10):635-639. (PubMed)
13. Jandke J, Spiteller G. Unusual conjugates in biological profiles originating from consumption of onions and garlic. J Chromatogr. 1987;421(1):1-8. (PubMed)
14. Kodera Y, Suzuki A, Imada O, et al. Physical, chemical, and biological properties of s-allylcysteine, an amino acid derived from garlic. J Agric Food Chem. 2002;50(3):622-632. (PubMed)
15. Percival SS. Aged Garlic Extract Modifies Human Immunity. J Nutr. 2016;146(2):433S-436S. (PubMed)
16. Steiner M, Khan AH, Holbert D, Lin RI. A double-blind crossover study in moderately hypercholesterolemic men that compared the effect of aged garlic extract and placebo administration on blood lipids. Am J Clin Nutr. 1996;64(6):866-870. (PubMed)
17. Nagae S, Ushijima M, Hatono S, et al. Pharmacokinetics of the garlic compound S-allylcysteine. Planta Med. 1994;60(3):214-217. (PubMed)
18. Budoff MJ, Takasu J, Flores FR, et al. Inhibiting progression of coronary calcification using Aged Garlic Extract in patients receiving statin therapy: a preliminary study. Prev Med. 2004;39(5):985-991. (PubMed)
19. Horev-Azaria L, Eliav S, Izigov N, et al. Allicin up-regulates cellular glutathione level in vascular endothelial cells. Eur J Nutr. 2009;48(2):67-74. (PubMed)
20. Liu C, Cao F, Tang QZ, et al. Allicin protects against cardiac hypertrophy and fibrosis via attenuating reactive oxygen species-dependent signaling pathways. J Nutr Biochem. 2010;21(12):1238-1250. (PubMed)
21. Chen C, Kong AN. Dietary chemopreventive compounds and ARE/EpRE signaling. Free Radic Biol Med. 2004;36(12):1505-1516. (PubMed)
22. Zeng T, Zhang CL, Song FY, et al. The activation of HO-1/Nrf-2 contributes to the protective effects of diallyl disulfide (DADS) against ethanol-induced oxidative stress. Biochim Biophys Acta. 2013;1830(10):4848-4859. (PubMed)
23. Tsai CY, Wang CC, Lai TY, et al. Antioxidant effects of diallyl trisulfide on high glucose-induced apoptosis are mediated by the PI3K/Akt-dependent activation of Nrf2 in cardiomyocytes. Int J Cardiol. 2013;168(2):1286-1297. (PubMed)
24. Hiramatsu K, Tsuneyoshi T, Ogawa T, Morihara N. Aged garlic extract enhances heme oxygenase-1 and glutamate-cysteine ligase modifier subunit expression via the nuclear factor erythroid 2-related factor 2-antioxidant response element signaling pathway in human endothelial cells. Nutr Res. 2016;36(2):143-149. (PubMed)
25. Gomez-Sierra T, Molina-Jijon E, Tapia E, et al. S-allylcysteine prevents cisplatin-induced nephrotoxicity and oxidative stress. J Pharm Pharmacol. 2014;66(9):1271-1281. (PubMed)
26. Shi H, Jing X, Wei X, et al. S-allyl cysteine activates the Nrf2-dependent antioxidant response and protects neurons against ischemic injury in vitro and in vivo. J Neurochem. 2015;133(2):298-308. (PubMed)
27. Higashi Y, Noma K, Yoshizumi M, Kihara Y. Endothelial function and oxidative stress in cardiovascular diseases. Circ J. 2009;73(3):411-418. (PubMed)
28. Lundblad C, Grande PO, Bentzer P. Hemodynamic and histological effects of traumatic brain injury in eNOS-deficient mice. J Neurotrauma. 2009;26(11):1953-1962. (PubMed)
29. Bhattacharyya M, Girish GV, Karmohapatra SK, Samad SA, Sinha AK. Systemic production of IFN-α by garlic (Allium sativum) in humans. J Interferon Cytokine Res. 2007;27(5):377-382. (PubMed)
30. Lei YP, Liu CT, Sheen LY, Chen HW, Lii CK. Diallyl disulfide and diallyl trisulfide protect endothelial nitric oxide synthase against damage by oxidized low-density lipoprotein. Mol Nutr Food Res. 2010;54 Suppl 1:S42-52. (PubMed)
31. Chen W, Qi J, Feng F, et al. Neuroprotective effect of allicin against traumatic brain injury via Akt/endothelial nitric oxide synthase pathway-mediated anti-inflammatory and anti-oxidative activities. Neurochem Int. 2014;68:28-37. (PubMed)
32. Shouk R, Abdou A, Shetty K, Sarkar D, Eid AH. Mechanisms underlying the antihypertensive effects of garlic bioactives. Nutr Res. 2014;34(2):106-115. (PubMed)
33. Yang J, Wang T, Yang J, et al. S-allyl cysteine restores erectile function through inhibition of reactive oxygen species generation in diabetic rats. Andrology. 2013;1(3):487-494. (PubMed)
34. Ho SC, Su MS. Evaluating the anti-neuroinflammatory capacity of raw and steamed garlic as well as five organosulfur compounds. Molecules. 2014;19(11):17697-17714. (PubMed)
35. Liu KL, Chen HW, Wang RY, Lei YP, Sheen LY, Lii CK. DATS reduces LPS-induced iNOS expression, NO production, oxidative stress, and NF-κB activation in RAW 264.7 macrophages. J Agric Food Chem. 2006;54(9):3472-3478. (PubMed)
36. You S, Nakanishi E, Kuwata H, et al. Inhibitory effects and molecular mechanisms of garlic organosulfur compounds on the production of inflammatory mediators. Mol Nutr Food Res. 2013;57(11):2049-2060. (PubMed)
37. Lee HH, Han MH, Hwang HJ, et al. Diallyl trisulfide exerts anti-inflammatory effects in lipopolysaccharide-stimulated RAW 264.7 macrophages by suppressing the Toll-like receptor 4/nuclear factor-κB pathway. Int J Mol Med. 2015;35(2):487-495. (PubMed)
38. Gebhardt R, Beck H. Differential inhibitory effects of garlic-derived organosulfur compounds on cholesterol biosynthesis in primary rat hepatocyte cultures. Lipids. 1996;31(12):1269-1276. (PubMed)
39. Ferri N, Yokoyama K, Sadilek M, et al. Ajoene, a garlic compound, inhibits protein prenylation and arterial smooth muscle cell proliferation. Br J Pharmacol. 2003;138(5):811-818. (PubMed)
40. Liu L, Yeh YY. S-alk(en)yl cysteines of garlic inhibit cholesterol synthesis by deactivating HMG-CoA reductase in cultured rat hepatocytes. J Nutr. 2002;132(6):1129-1134. (PubMed)
41. Singh DK, Porter TD. Inhibition of sterol 4α-methyl oxidase is the principal mechanism by which garlic decreases cholesterol synthesis. J Nutr. 2006;136(3 Suppl):759S-764S. (PubMed)
42. Allison GL, Lowe GM, Rahman K. Aged garlic extract may inhibit aggregation in human platelets by suppressing calcium mobilization. J Nutr. 2006;136(3 Suppl):789S-792S. (PubMed)
43. Chan KC, Hsu CC, Yin MC. Protective effect of three diallyl sulphides against glucose-induced erythrocyte and platelet oxidation, and ADP-induced platelet aggregation. Thromb Res. 2002;108(5-6):317-322. (PubMed)
44. Lawson LD, Ransom DK, Hughes BG. Inhibition of whole blood platelet-aggregation by compounds in garlic clove extracts and commercial garlic products. Thromb Res. 1992;65(2):141-156. (PubMed)
45. Allison GL, Lowe GM, Rahman K. Aged garlic extract inhibits platelet activation by increasing intracellular cAMP and reducing the interaction of GPIIb/IIIa receptor with fibrinogen. Life Sci. 2012;91(25-26):1275-1280. (PubMed)
46. Rahman K, Lowe GM, Smith S. Aged garlic extract inhibits human platelet aggregation by altering intracellular signaling and platelet shape change. J Nutr. 2016;146(2):410S-415S. (PubMed)
47. Hedin U, Roy J, Tran PK. Control of smooth muscle cell proliferation in vascular disease. Curr Opin Lipidol. 2004;15(5):559-565. (PubMed)
48. Campbell JH, Efendy JL, Smith NJ, Campbell GR. Molecular basis by which garlic suppresses atherosclerosis. J Nutr. 2001;131(3s):1006S-1009S. (PubMed)
49. Golovchenko I, Yang CH, Goalstone ML, Draznin B. Garlic extract methylallyl thiosulfinate blocks insulin potentiation of platelet-derived growth factor-stimulated migration of vascular smooth muscle cells. Metabolism. 2003;52(2):254-259. (PubMed)
50. Lei YP, Chen HW, Sheen LY, Lii CK. Diallyl disulfide and diallyl trisulfide suppress oxidized LDL-induced vascular cell adhesion molecule and E-selectin expression through protein kinase A- and B-dependent signaling pathways. J Nutr. 2008;138(6):996-1003. (PubMed)
51. Pryor WA, Houk KN, Foote CS, et al. Free radical biology and medicine: it's a gas, man! Am J Physiol Regul Integr Comp Physiol. 2006;291(3):R491-511. (PubMed)
52. Lefer DJ. A new gaseous signaling molecule emerges: cardioprotective role of hydrogen sulfide. Proc Natl Acad Sci U S A. 2007;104(46):17907-17908. (PubMed)
53. Ried K, Fakler P. Potential of garlic (Allium sativum) in lowering high blood pressure: mechanisms of action and clinical relevance. Integr Blood Press Control. 2014;7:71-82. (PubMed)
54. Benavides GA, Squadrito GL, Mills RW, et al. Hydrogen sulfide mediates the vasoactivity of garlic. Proc Natl Acad Sci U S A. 2007;104(46):17977-17982. (PubMed)
55. Yang CS, Chhabra SK, Hong JY, Smith TJ. Mechanisms of inhibition of chemical toxicity and carcinogenesis by diallyl sulfide (DAS) and related compounds from garlic. J Nutr. 2001;131(3s):1041S-1045S. (PubMed)
56. Brady JF, Ishizaki H, Fukuto JM, et al. Inhibition of cytochrome P-450 2E1 by diallyl sulfide and its metabolites. Chem Res Toxicol. 1991;4(6):642-647. (PubMed)
57. Taubert D, Glockner R, Muller D, Schomig E. The garlic ingredient diallyl sulfide inhibits cytochrome P450 2E1 dependent bioactivation of acrylamide to glycidamide. Toxicol Lett. 2006;164(1):1-5. (PubMed)
58. Jeong HG, Lee YW. Protective effects of diallyl sulfide on N-nitrosodimethylamine-induced immunosuppression in mice. Cancer Lett. 1998;134(1):73-79. (PubMed)
59. Park KA, Kweon S, Choi H. Anticarcinogenic effect and modification of cytochrome P450 2E1 by dietary garlic powder in diethylnitrosamine-initiated rat hepatocarcinogenesis. J Biochem Mol Biol. 2002;35(6):615-622. (PubMed)
60. Gurley BJ, Gardner SF, Hubbard MA, et al. Cytochrome P450 phenotypic ratios for predicting herb-drug interactions in humans. Clin Pharmacol Ther. 2002;72(3):276-287. (PubMed)
61. Gurley BJ, Gardner SF, Hubbard MA, et al. Clinical assessment of effects of botanical supplementation on cytochrome P450 phenotypes in the elderly: St John's wort, garlic oil, Panax ginseng and Ginkgo biloba. Drugs Aging. 2005;22(6):525-539. (PubMed)
62. Loizou GD, Cocker J. The effects of alcohol and diallyl sulphide on CYP2E1 activity in humans: a phenotyping study using chlorzoxazone. Hum Exp Toxicol. 2001;20(7):321-327. (PubMed)
63. Munday R, Munday CM. Induction of phase II enzymes by aliphatic sulfides derived from garlic and onions: an overview. Methods Enzymol. 2004;382:449-456. (PubMed)
64. Andorfer JH, Tchaikovskaya T, Listowsky I. Selective expression of glutathione S-transferase genes in the murine gastrointestinal tract in response to dietary organosulfur compounds. Carcinogenesis. 2004;25(3):359-367. (PubMed)
65. Hatono S, Jimenez A, Wargovich MJ. Chemopreventive effect of S-allylcysteine and its relationship to the detoxification enzyme glutathione S-transferase. Carcinogenesis. 1996;17(5):1041-1044. (PubMed)
66. Munday R, Munday CM. Relative activities of organosulfur compounds derived from onions and garlic in increasing tissue activities of quinone reductase and glutathione transferase in rat tissues. Nutr Cancer. 2001;40(2):205-210. (PubMed)
67. Lee IC, Kim SH, Baek HS, et al. The involvement of Nrf2 in the protective effects of diallyl disulfide on carbon tetrachloride-induced hepatic oxidative damage and inflammatory response in rats. Food Chem Toxicol. 2014;63:174-185. (PubMed)
68. Lee IC, Kim SH, Baek HS, et al. Protective effects of diallyl disulfide on carbon tetrachloride-induced hepatotoxicity through activation of Nrf2. Environ Toxicol. 2015;30(5):538-548. (PubMed)
69. Stewart ZA, Westfall MD, Pietenpol JA. Cell-cycle dysregulation and anticancer therapy. Trends Pharmacol Sci. 2003;24(3):139-145. (PubMed)
70. Powolny AA, Singh SV. Multitargeted prevention and therapy of cancer by diallyl trisulfide and related Allium vegetable-derived organosulfur compounds. Cancer Lett. 2008;269(2):305-314. (PubMed)
71. Singh SV, Powolny AA, Stan SD, et al. Garlic constituent diallyl trisulfide prevents development of poorly differentiated prostate cancer and pulmonary metastasis multiplicity in TRAMP mice. Cancer Res. 2008;68(22):9503-9511. (PubMed)
72. Jikihara H, Qi G, Nozoe K, et al. Aged garlic extract inhibits 1,2-dimethylhydrazine-induced colon tumor development by suppressing cell proliferation. Oncol Rep. 2015;33(3):1131-1140. (PubMed)
73. Wu X, Kassie F, Mersch-Sundermann V. Induction of apoptosis in tumor cells by naturally occurring sulfur-containing compounds. Mutat Res. 2005;589(2):81-102. (PubMed)
74. Balasenthil S, Rao KS, Nagini S. Apoptosis induction by S-allylcysteine, a garlic constituent, during 7,12-dimethylbenz[a]anthracene-induced hamster buccal pouch carcinogenesis. Cell Biochem Funct. 2002;20(3):263-268. (PubMed)
75. Balasenthil S, Rao KS, Nagini S. Garlic induces apoptosis during 7,12-dimethylbenz[a]anthracene-induced hamster buccal pouch carcinogenesis. Oral Oncol. 2002;38(5):431-436. (PubMed)
76. Zhang CL, Zeng T, Zhao XL, Yu LH, Zhu ZP, Xie KQ. Protective effects of garlic oil on hepatocarcinoma induced by N-nitrosodiethylamine in rats. Int J Biol Sci. 2012;8(3):363-374. (PubMed)
77. Bauer D, Redmon N, Mazzio E, et al. Diallyl disulfide inhibits TNFα induced CCL2 release through MAPK/ERK and NF-Kappa-B signaling. Cytokine. 2015;75(1):117-126. (PubMed)
78. Matsuura N, Miyamae Y, Yamane K, et al. Aged garlic extract inhibits angiogenesis and proliferation of colorectal carcinoma cells. J Nutr. 2006;136(3 Suppl):842S-846S. (PubMed)
79. Fenwick GR, Hanley AB. The genus Allium--Part 3. Crit Rev Food Sci Nutr. 1985;23(1):1-73. (PubMed)
80. Harris JC, Cottrell SL, Plummer S, Lloyd D. Antimicrobial properties of Allium sativum (garlic). Appl Microbiol Biotechnol. 2001;57(3):282-286. (PubMed)
81. Ankri S, Mirelman D. Antimicrobial properties of allicin from garlic. Microbes Infect. 1999;1(2):125-129. (PubMed)
82. Cavallito CJ, Bailey JH. Allicin, the antibacterial principle of Allium sativum. I. Isolation, physical properties and antibacterial action J Am Chem Soc. 1944;66(11):1950-1951.
83. Martin KW, Ernst E. Herbal medicines for treatment of bacterial infections: a review of controlled clinical trials. J Antimicrob Chemother. 2003;51(2):241-246. (PubMed)
84. Gail MH, Pfeiffer RM, Brown LM, et al. Garlic, vitamin, and antibiotic treatment for Helicobacter pylori: a randomized factorial controlled trial. Helicobacter. 2007;12(5):575-578. (PubMed)
85. You WC, Brown LM, Zhang L, et al. Randomized double-blind factorial trial of three treatments to reduce the prevalence of precancerous gastric lesions. J Natl Cancer Inst. 2006;98(14):974-983. (PubMed)
86. Ledezma E, DeSousa L, Jorquera A, et al. Efficacy of ajoene, an organosulphur derived from garlic, in the short-term therapy of tinea pedis. Mycoses. 1996;39(9-10):393-395. (PubMed)
87. Nantz MP, Rowe CA, Muller CE, Creasy RA, Stanilka JM, Percival SS. Supplementation with aged garlic extract improves both NK and γδ-T cell function and reduces the severity of cold and flu symptoms: a randomized, double-blind, placebo-controlled nutrition intervention. Clin Nutr. 2012;31(3):337-344. (PubMed)
88. Keys A. Wine, garlic, and CHD in seven countries. Lancet. 1980;1(8160):145-146. (PubMed)
89. Wirth J, di Giuseppe R, Boeing H, Weikert C. A Mediterranean-style diet, its components and the risk of heart failure: a prospective population-based study in a non-Mediterranean country. Eur J Clin Nutr. 2016;70(9):1015-1021. (PubMed)
90. Ackermann RT, Mulrow CD, Ramirez G, Gardner CD, Morbidoni L, Lawrence VA. Garlic shows promise for improving some cardiovascular risk factors. Arch Intern Med. 2001;161(6):813-824. (PubMed)
91. Wojcikowski K, Myers S, Brooks L. Effects of garlic oil on platelet aggregation: a double-blind placebo-controlled crossover study. Platelets. 2007;18(1):29-34. (PubMed)
92. Zeng T, Zhang CL, Zhao XL, Xie KQ. The roles of garlic on the lipid parameters: a systematic review of the literature. Crit Rev Food Sci Nutr. 2013;53(3):215-230. (PubMed)
93. Ried K, Toben C, Fakler P. Effect of garlic on serum lipids: an updated meta-analysis. Nutr Rev. 2013;71(5):282-299. (PubMed)
94. Koscielny J, Klussendorf D, Latza R, et al. The antiatherosclerotic effect of Allium sativum. Atherosclerosis. 1999;144(1):237-249. (PubMed)
95. Siegel G, Klussendorf D. The anti-atheroslerotic effect of Allium sativum: statistics re-evaluated. Atherosclerosis. 2000;150(2):437-438. (PubMed)
96. Almoudi M, Sun Z. Coronary artery calcium score: Re-evaluation of its predictive value for coronary artery disease. World J Cardiol. 2012;4(10):284-287. (PubMed)
97. Kwon SW, Kim YJ, Shim J, et al. Coronary artery calcium scoring does not add prognostic value to standard 64-section CT angiography protocol in low-risk patients suspected of having coronary artery disease. Radiology. 2011;259(1):92-99. (PubMed)
98. Matsumoto S, Nakanishi R, Li D, et al. Aged Garlic Extract Reduces Low Attenuation Plaque in Coronary Arteries of Patients with Metabolic Syndrome in a Prospective Randomized Double-Blind Study. J Nutr. 2016;146(2):427S-432S. (PubMed)
99. Hadamitzky M, Distler R, Meyer T, et al. Prognostic value of coronary computed tomographic angiography in comparison with calcium scoring and clinical risk scores. Circ Cardiovasc Imaging. 2011;4(1):16-23. (PubMed)
100. Nakanishi K, Fukuda S, Shimada K, et al. Non-obstructive low attenuation coronary plaque predicts three-year acute coronary syndrome events in patients with hypertension: multidetector computed tomographic study. J Cardiol. 2012;59(2):167-175. (PubMed)
101. Reinhart KM, Coleman CI, Teevan C, Vachhani P, White CM. Effects of garlic on blood pressure in patients with and without systolic hypertension: a meta-analysis. Ann Pharmacother. 2008;42(12):1766-1771. (PubMed)
102. Ried K. Garlic lowers blood pressure in hypertensive individuals, regulates serum cholesterol, and stimulates immunity: an updated meta-analysis and review. J Nutr. 2016;146(2):389S-396S. (PubMed)
103. Ried K, Frank OR, Stocks NP, Fakler P, Sullivan T. Effect of garlic on blood pressure: a systematic review and meta-analysis. BMC Cardiovasc Disord. 2008;8:13. (PubMed)
104. Silagy CA, Neil HA. A meta-analysis of the effect of garlic on blood pressure. J Hypertens. 1994;12(4):463-468. (PubMed)
105. Wang HP, Yang J, Qin LQ, Yang XJ. Effect of garlic on blood pressure: a meta-analysis. J Clin Hypertens (Greenwich). 2015;17(3):223-231. (PubMed)
106. Xiong XJ, Wang PQ, Li SJ, Li XK, Zhang YQ, Wang J. Garlic for hypertension: A systematic review and meta-analysis of randomized controlled trials. Phytomedicine. 2015;22(3):352-361. (PubMed)
107. Rohner A, Ried K, Sobenin IA, Bucher HC, Nordmann AJ. A systematic review and metaanalysis on the effects of garlic preparations on blood pressure in individuals with hypertension. Am J Hypertens. 2015;28(3):414-423. (PubMed)
108. Law MR, Morris JK, Wald NJ. Use of blood pressure lowering drugs in the prevention of cardiovascular disease: meta-analysis of 147 randomised trials in the context of expectations from prospective epidemiological studies. BMJ. 2009;338:b1665. (PubMed)
109. Stabler SN, Tejani AM, Huynh F, Fowkes C. Garlic for the prevention of cardiovascular morbidity and mortality in hypertensive patients. Cochrane Database Syst Rev. 2012(8):CD007653. (PubMed)
110. Ried K, Travica N, Sali A. The effect of aged garlic extract on blood pressure and other cardiovascular risk factors in uncontrolled hypertensives: the AGE at Heart trial. Integr Blood Press Control. 2016;9:9-21. (PubMed)
111. Kodali RT, Eslick GD. Meta-analysis: Does garlic intake reduce risk of gastric cancer? Nutr Cancer. 2015;67(1):1-11. (PubMed)
112. Kim JY, Kwon O. Garlic intake and cancer risk: an analysis using the Food and Drug Administration's evidence-based review system for the scientific evaluation of health claims. Am J Clin Nutr. 2009;89(1):257-264. (PubMed)
113. US Food and Drug Administration. Guidance for industry: evidence-based review system for the scientific evaluation of health claims - final. In: US Department of Health and Human Services, ed; 2009. http://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/LabelingNutrition/ucm073332.htm. Accessed 1/6/17.
114. Hansson LE, Nyren O, Bergstrom R, et al. Diet and risk of gastric cancer. A population-based case-control study in Sweden. Int J Cancer. 1993;55(2):181-189. (PubMed)
115. Kim HJ, Chang WK, Kim MK, Lee SS, Choi BY. Dietary factors and gastric cancer in Korea: a case-control study. Int J Cancer. 2002;97(4):531-535. (PubMed)
116. Dorant E, van den Brandt PA, Goldbohm RA. A prospective cohort study on the relationship between onion and leek consumption, garlic supplement use and the risk of colorectal carcinoma in The Netherlands. Carcinogenesis. 1996;17(3):477-484. (PubMed)
117. Ma JL, Zhang L, Brown LM, et al. Fifteen-year effects of Helicobacter pylori, garlic, and vitamin treatments on gastric cancer incidence and mortality. J Natl Cancer Inst. 2012;104(6):488-492. (PubMed)
118. Cañizares P, Gracia I, Gómez LA, et al. Allyl-thiosulfinates, the bacteriostatic compounds of garlic against Helicobacter pylori. Biotechnol Prog. 2004;20(1):397-401. (PubMed)
119. O'Gara EA, Hill DJ, Maslin DJ. Activities of garlic oil, garlic powder, and their diallyl constituents against Helicobacter pylori. Appl Environ Microbiol. 2000;66(5):2269-2273. (PubMed)
120. Shmuely H, Domniz N, Yahav J. Non-pharmacological treatment of Helicobacter pylori. World J Gastrointest Pharmacol Ther. 2016;7(2):171-178. (PubMed)
121. Salih BA, Abasiyanik FM. Does regular garlic intake affect the prevalence of Helicobacter pylori in asymptomatic subjects? Saudi Med J. 2003;24(8):842-845. (PubMed)
122. You WC, Zhang L, Gail MH, et al. Helicobacter pylori infection, garlic intake and precancerous lesions in a Chinese population at low risk of gastric cancer. Int J Epidemiol. 1998;27(6):941-944. (PubMed)
123. Graham DY, Anderson SY, Lang T. Garlic or jalapeno peppers for treatment of Helicobacter pylori infection. Am J Gastroenterol. 1999;94(5):1200-1202. (PubMed)
124. McNulty CA, Wilson MP, Havinga W, Johnston B, O'Gara EA, Maslin DJ. A pilot study to determine the effectiveness of garlic oil capsules in the treatment of dyspeptic patients with Helicobacter pylori. Helicobacter. 2001;6(3):249-253. (PubMed)
125. Aydin A, Ersoz G, Tekesin O, Akcicek E, Tuncyurek M. Garlic oil and Helicobacter pylori infection. Am J Gastroenterol. 2000;95(2):563-564. (PubMed)
126. Ernst E. Is garlic an effective treatment for Helicobacter pylori infection? Arch Intern Med. 1999;159(20):2484-2485. (PubMed)
127. Hu JY, Hu YW, Zhou JJ, Zhang MW, Li D, Zheng S. Consumption of garlic and risk of colorectal cancer: an updated meta-analysis of prospective studies. World J Gastroenterol. 2014;20(41):15413-15422. (PubMed)
128. Chiavarini M, Minelli L, Fabiani R. Garlic consumption and colorectal cancer risk in man: a systematic review and meta-analysis. Public Health Nutr. 2016;19(2):308-317. (PubMed)
129. Galeone C, Pelucchi C, Levi F, et al. Onion and garlic use and human cancer. Am J Clin Nutr. 2006;84(5):1027-1032. (PubMed)
130. Fleischauer AT, Poole C, Arab L. Garlic consumption and cancer prevention: meta-analyses of colorectal and stomach cancers. Am J Clin Nutr. 2000;72(4):1047-1052. (PubMed)
131. Tanaka S, Haruma K, Kunihiro M, et al. Effects of aged garlic extract (AGE) on colorectal adenomas: a double-blinded study. Hiroshima J Med Sci. 2004;53(3-4):39-45. (PubMed)
132. Tanaka S, Haruma K, Yoshihara M, et al. Aged garlic extract has potential suppressive effect on colorectal adenomas in humans. J Nutr. 2006;136(3 Suppl):821S-826S. (PubMed)
133. Ishikawa H, Saeki T, Otani T, et al. Aged garlic extract prevents a decline of NK cell number and activity in patients with advanced cancer. J Nutr. 2006;136(3 Suppl):816S-820S. (PubMed)
134. Bianchini F, Vainio H. Allium vegetables and organosulfur compounds: do they help prevent cancer? Environ Health Perspect. 2001;109(9):893-902. (PubMed)
135. Song K, Milner JA. The influence of heating on the anticancer properties of garlic. J Nutr. 2001;131(3s):1054S-1057S. (PubMed)
136. Cavagnaro PF, Camargo A, Galmarini CR, Simon PW. Effect of cooking on garlic (Allium sativum L.) antiplatelet activity and thiosulfinates content. J Agric Food Chem. 2007;55(4):1280-1288. (PubMed)
137. Song K, Milner JA. Heating garlic inhibits its ability to suppress 7, 12-dimethylbenz(a)anthracene-induced DNA adduct formation in rat mammary tissue. J Nutr. 1999;129(3):657-661. (PubMed)
138. Staba EJ, Lash L, Staba JE. A commentary on the effects of garlic extraction and formulation on product composition. J Nutr. 2001;131(3s):1118S-1119S. (PubMed)
139. Dietary Supplements: Garlic. The United States Pharmacopeia. Rockville, MD: United States Pharmacopeial Convention, Inc.; 2005:2087-2092.
140. Lawson LD, Wang ZJ. Low allicin release from garlic supplements: a major problem due to the sensitivities of alliinase activity. J Agric Food Chem. 2001;49(5):2592-2599. (PubMed)
141. Lawson LD, Wang ZJ, Papadimitriou D. Allicin release under simulated gastrointestinal conditions from garlic powder tablets employed in clinical trials on serum cholesterol. Planta Med. 2001;67(1):13-18. (PubMed)
142. Amagase H, Petesch BL, Matsuura H, Kasuga S, Itakura Y. Intake of garlic and its bioactive components. J Nutr. 2001;131(3s):955S-962S. (PubMed)
143. Ried K, Frank OR, Stocks NP. Aged garlic extract reduces blood pressure in hypertensives: a dose-response trial. Eur J Clin Nutr. 2013;67(1):64-70. (PubMed)
144. Steiner M, Li W. Aged garlic extract, a modulator of cardiovascular risk factors: a dose-finding study on the effects of AGE on platelet functions. J Nutr. 2001;131(3s):980S-984S. (PubMed)
145. Brace LD. Cardiovascular benefits of garlic (Allium sativum L). J Cardiovasc Nurs. 2002;16(4):33-49. (PubMed)
146. Borrelli F, Capasso R, Izzo AA. Garlic (Allium sativum L.): adverse effects and drug interactions in humans. Mol Nutr Food Res. 2007;51(11):1386-1397. (PubMed)
147. Burnham BE. Garlic as a possible risk for postoperative bleeding. Plast Reconstr Surg. 1995;95(1):213. (PubMed)
148. Carden SM, Good WV, Carden PA, Good RM. Garlic and the strabismus surgeon. Clin Experiment Ophthalmol. 2002;30(4):303-304. (PubMed)
149. German K, Kumar U, Blackford HN. Garlic and the risk of TURP bleeding. Br J Urol. 1995;76(4):518. (PubMed)
150. Rose KD, Croissant PD, Parliament CF, Levin MB. Spontaneous spinal epidural hematoma with associated platelet dysfunction from excessive garlic ingestion: a case report. Neurosurgery. 1990;26(5):880-882. (PubMed)
151. Anibarro B, Fontela JL, De La Hoz F. Occupational asthma induced by garlic dust. J Allergy Clin Immunol. 1997;100(6 Pt 1):734-738. (PubMed)
152. Jappe U, Bonnekoh B, Hausen BM, Gollnick H. Garlic-related dermatoses: case report and review of the literature. Am J Contact Dermat. 1999;10(1):37-39. (PubMed)
153. Ziaei S, Hantoshzadeh S, Rezasoltani P, Lamyian M. The effect of garlic tablet on plasma lipids and platelet aggregation in nulliparous pregnants at high risk of preeclampsia. Eur J Obstet Gynecol Reprod Biol. 2001;99(2):201-206. (PubMed)
154. Mennella JA, Beauchamp GK. Maternal diet alters the sensory qualities of human milk and the nursling's behavior. Pediatrics. 1991;88(4):737-744. (PubMed)
155. Sunter WH. Warfarin and garlic. Pharm J. 1991;246:722.
156. Macan H, Uykimpang R, Alconcel M, et al. Aged garlic extract may be safe for patients on warfarin therapy. J Nutr. 2006;136(3 Suppl):793S-795S. (PubMed)
157. Izzo AA, Ernst E. Interactions between herbal medicines and prescribed drugs: a systematic review. Drugs. 2001;61(15):2163-2175. (PubMed)
158. Piscitelli SC, Burstein AH, Welden N, Gallicano KD, Falloon J. The effect of garlic supplements on the pharmacokinetics of saquinavir. Clin Infect Dis. 2002;34(2):234-238. (PubMed)
159. Markowitz JS, Devane CL, Chavin KD, Taylor RM, Ruan Y, Donovan JL. Effects of garlic (Allium sativum L.) supplementation on cytochrome P450 2D6 and 3A4 activity in healthy volunteers. Clin Pharmacol Ther. 2003;74(2):170-177. (PubMed)
160. Gallicano K, Foster B, Choudhri S. Effect of short-term administration of garlic supplements on single-dose ritonavir pharmacokinetics in healthy volunteers. Br J Clin Pharmacol. 2003;55(2):199-202. (PubMed)
161. Berginc K, Kristl A. The mechanisms responsible for garlic - drug interactions and their in vivo relevance. Curr Drug Metab. 2013;14(1):90-101. (PubMed)
162. Berginc K, Milisav I, Kristl A. Garlic flavonoids and organosulfur compounds: impact on the hepatic pharmacokinetics of saquinavir and darunavir. Drug Metab Pharmacokinet. 2010;25(6):521-530. (PubMed)
Legumes
Contents
Summary
- Foods from the legume family include beans, peas, lentils, peanuts, and soybeans. (More information)
- Legumes are excellent sources of protein, low-glycemic index carbohydrates, essential micronutrients, and fiber. (More information)
- Substituting legumes for foods that are high in saturated fats or refined carbohydrates is likely to lower the risk of cardiovascular disease and type 2 diabetes mellitus. (More information)
- Regular consumption of legumes, both pulses (legumes excluding green peas, green beans, peanuts, and soybeans) and soy food, may result in favorable effects on blood lipid profiles by lowering LDL-cholesterol. (More information)
- Several observational studies have found non-soy legume intake to be inversely associated with incidence of prostate cancer. (More information)
- Lifelong exposure to soy food may be needed to lower risk of breast cancer. (More information)
- Short-term clinical trials have reported pulse consumption increases satiety; however, high-quality trials are needed to determine whether regular consumption of pulses results in weight loss in the long term. (More information)
- The most recent Dietary Guidelines for Americans include legumes in both the protein and vegetable groups, recommending a weekly intake of 1½ to 3 cup-equivalents of legumes for those who consume 2,000 calories/day. This recommended amount depends on the specific dietary pattern followed, and additionally, soy products are recommended for those who follow a vegetarian diet. (More information)
Introduction
Legumes are plants of the Leguminosae family with seed pods that split into two halves. Edible seeds from plants of this family include beans, peas, lentils, soybeans, and peanuts. The most recent Dietary Guidelines for Americans do not include green peas or green (string) beans as legumes due to a dissimilar nutritional profile when compared to the other legume family members (1). Peanuts are nutritionally similar to tree nuts; therefore, information on their health effects is presented in a separate article on Nuts and not discussed below. Many studies have examined how the consumption of pulses is related to health; pulses are legumes harvested as dry grain and thus exclude green peas, green beans, as well as crops used for oil extraction (soybeans, peanuts) (2).
Although legumes are an important part of traditional diets around the world, they are often neglected in typical Western diets. Legumes are inexpensive, nutrient-dense sources of protein that can be substituted for dietary animal protein (3). While sources of animal protein are often rich in saturated fats, the small quantities of fats in legumes are mostly unsaturated fats. Legumes are also good sources of several essential minerals, rich in dietary fiber (both soluble fiber and resistant starch), and considered to be low-glycemic index foods (4, 5). Moreover, legumes contain numerous phytochemicals that may affect health. Soybeans have attracted the most scientific interest, mainly because they are a unique source of phytoestrogens known as isoflavones (6). Overall, legumes represent unique packages of nutrients and phytochemicals that may work synergistically to reduce risk of chronic disease, as other edible seeds do (7).
Note: Research on the health effects of diets rich in legumes, including peas and soy foods, is summarized below. For a discussion of the potential health benefits and risks of soy isoflavones, see the separate article on Soy Isoflavones.
Disease Prevention
Type 2 diabetes mellitus
Consumption of legumes, which are rich in dietary fiber, other nutrients, and bioactive compounds, might help improve insulin sensitivity and regulate blood glucose concentrations, thereby influencing risk of type 2 diabetes mellitus.
Beans, peas, and lentils
The glycemic index is a measure of the potential for carbohydrates in different foods to raise blood glucose concentrations. In general, consuming foods with high-glycemic index values causes blood glucose concentrations to rise more rapidly, which results in greater insulin secretion by the pancreas, than after consuming foods with low-glycemic index values. Chronically elevated blood glucose concentrations and excessive insulin secretion are thought to play important roles in the development of type 2 diabetes mellitus (8). Low-glycemic load diets have been associated with reduced risk of developing type 2 diabetes in some, but not all, prospective cohort studies (see the article on Glycemic Index and Glycemic Load). High-fiber diets have also been associated with a decreased risk of type 2 diabetes (see the article on Fiber).
Together, the high-fiber content and low-glycemic index nature of legumes may contribute to help lower risk of type 2 diabetes in those who incorporate them into their daily diets. However, a 2017 meta-analysis of 12 prospective cohort studies found that higher legume intakes were not associated with risk of developing type 2 diabetes compared to lower intakes (range, 0-190 g/day; ~0-2 servings/day) (9), although a more recent prospective study not included in this pooled analysis found an inverse association (10).
Results of clinical trials of legume or pulse consumption have been more promising. A meta-analysis of 41 randomized controlled trials (some in individuals with normoglycemia and some in individuals with type 1 or type 2 diabetes) found that pulse consumption, either alone or in combination with a low-glycemic index diet or a high-fiber diet, improved some measures of glucose control, including fasting blood glucose and insulin concentrations, as well as markers of long-term glycemic control (11). The effect was stronger in patients with diagnosed diabetes compared to individuals with normoglycemia (11). In a three-month trial in 114 people with type 2 diabetes, those randomized to a low-glycemic diet that emphasized 1 cup/day of legumes had significant improvements in their hemoglobin A1C value — the main fraction of glycated hemoglobin that reflects glycemic control over the past four months — compared to those randomized to a high-wheat fiber diet (12).
Small studies in healthy individuals have found that consumption of legumes improved postprandial glucose response at the subsequent meal (called the 'subsequent meal effect' or 'second meal effect') (13-15), although this has not been extensively studied. In addition to improving glycemic and insulin control, consumption of legumes may increase satiety and decrease both food intake and body weight, indirectly improving glycemic control (see Obesity below).
Soy
Observational studies on the association of soy intake and type 2 diabetes mellitus have reported mixed results. A 2018 meta-analysis of eight observational studies (six prospective cohort and two cross sectional) found an inverse association between soy intake and risk of type 2 diabetes; however, there was a high degree of heterogeneity in the analysis (16). Moreover, when a subgroup analysis was done and only prospective cohort studies were examined, the association was no longer significant (16).
A meta-analysis of 19 randomized controlled trials, including 1,518 participants, found that soy consumption — as soy foods/whole soy, isolated soy protein, or soy isoflavone supplements — had no effect on fasting glucose or insulin concentrations (17). Yet, subgroup analyses reported the effect on hyperglycemia differed by type of soy intervention: adherence to a whole-soy diet lowered fasting glucose concentrations, while soy protein isolate or isoflavone supplementation had no effect on fasting glucose concentrations (17).
Cardiovascular disease
Several characteristics of legumes may contribute to protection against cardiovascular disease. They are rich in soluble fiber and phytosterols, which are known to have cholesterol-lowering effects, and their high folate content may help lower homocysteine concentrations. Additionally, legumes are good sources of magnesium and potassium, which may decrease cardiovascular disease risk by helping to lower blood pressure (18). The low-glycemic index values of beans mean that they are less likely to raise blood glucose and insulin concentrations (see Type 2 diabetes mellitus above), and this may also decrease cardiovascular disease risk. Further, the substitution of legumes for foods high in saturated fat or refined carbohydrates may offer some cardioprotection.
Beans, peas, and lentils
Some, but not all, observational studies have found regular legume intake to be associated with a lower risk of cardiovascular events, and overall, most clinical trials have found legume consumption decreases cardiometabolic risk factors, especially circulating LDL-cholesterol concentrations. In a meta-analysis of eight prospective cohort studies, legume intake as part of a Mediterranean diet was associated with a 9% lower risk of cardiovascular disease, although one of the included studies grouped legume consumption with nuts, which are known to be cardioprotective (see the article on Nuts) (19). In a systematic review and meta-analysis of five prospective cohort studies, consuming four 100-g servings of legumes (beans, peas, lentils, tofu) weekly was associated with a 14% lower risk of coronary heart disease (CHD) (20). This association was driven mainly by a prospective cohort study that followed participants for 19 years, finding those who ate dry beans, peas, or peanuts at least four times weekly had a 21% lower CHD risk compared to those who ate them less than once weekly (21). Most recently, in a meta-analysis that included both CHD incidence and CHD-related mortality together as outcomes (eight prospective cohort studies in total), the highest versus lowest legume intakes were associated with a 10% decreased risk (22). In contrast, legume intake has not been linked to risk of stroke in four separate meta-analyses (20, 22-24).
Serum lipid profiles: Some, but not all, clinical trials have found that regular legume consumption results in favorable changes in the lipid profile. A 2014 systematic review and meta-analysis of 26 randomized controlled trials reported that pulse intake (beans, lentils, peas; median intake of 130 g/day or ~1.3 servings/day) reduced LDL-cholesterol concentrations by 0.17 mmol/L (6.6 mg/dL) compared to an isocaloric diet without pulses; however, there was no effect of pulse intake on non-HDL cholesterol or apolipoprotein B concentrations (25). It is important to note that there was significant heterogeneity among included trials of this meta-analysis, and the baseline lipid status of participants varied among the trials: eight trials included hyperlipidemic subjects, three trials were done in participants with normal lipid profiles, and 15 trials had a combination of normal and high lipid levels (25). A previous meta-analysis of randomized controlled trials found that non-soy legume consumption decreased total and LDL-cholesterol concentrations by 11.76 mg/dL (10 studies) and 7.98 mg/dL (9 studies), respectively, when compared to a matched control (matched for total calories or macronutrients; baseline total cholesterol concentrations of participants ranged from 199 to 295 mg/dL among studies) (26). A trend for a reduction in serum triglycerides (-18.94 mg/dL, p=0.05) was observed in this meta-analysis, and no effect on HDL cholesterol was found (26).
Hypertension: Adherence to the Dietary Approaches to Stop Hypertension (DASH) eating plan, which includes dry beans and peas among many other components (27), has been shown to reduce blood pressure (28), but it is not known whether legumes contribute to this effect. A 2014 meta-analysis of eight isocaloric, controlled feeding trials (median of 10 weeks’ duration) — including both normotensive and hypertensive participants (n=554; median age, 49 years) — found that pulse consumption (median of 162 g/day or ~1.6 daily servings) decreased systolic blood pressure by 2.25 mm Hg but had no effect on diastolic blood pressure (29). It is important to note that there was significant heterogeneity among trials included in the meta-analysis (29).
Soy
Serum lipid profiles: In 1999, the US Food and Drug Administration (FDA) approved the following health claim: "Diets low in saturated fat and cholesterol that include 25 grams of soy protein a day may reduce the risk of heart disease" (30). Most of the evidence to support this health claim was included in the Anderson et al. meta-analysis of 38 controlled clinical trials that was published in 1995. This meta-analysis found that an average intake of 47 g/day of soy protein decreased serum total cholesterol concentrations by an average of 9% and LDL-cholesterol concentrations by an average of 13% (31). Hypocholesterolemic effects were primarily noted in individuals with high baseline cholesterol concentrations (31). A meta-analysis of 33 studies published since 1995 confirmed the hypocholesterolemic effect of soy protein reported in the Anderson et al. publication (32). Another meta-analysis of 30 studies in individuals with normal or mildly elevated cholesterol levels concluded that about 25 g/day of soy protein significantly lowers LDL-cholesterol concentrations by about 6% (33). Yet, a 2006 science advisory from the Nutrition Committee of the American Heart Association concluded that earlier research indicating that soy protein consumption results in clinically important reductions in LDL-cholesterol compared to other proteins has not been confirmed (34). A 2011 meta-analysis of randomized controlled trials, mostly in participants with hypercholesterolemia, found consumption of soy protein decreased LDL-cholesterol by 5.5% in parallel-design studies (20 trials; median soy protein intake of 31.5 g/day for a mean of 9.2 weeks) and 4.2% in cross-over studies (23 trials; median soy protein intake of 26.0 g/day for a mean of 6.0 weeks) — such decreases may translate to 6%-10% decreases in risk for coronary heart disease (35).
Consumption of isolated soy isoflavones (as supplements or extracts), however, does not appear to have favorable effects on serum lipid profiles (35-42).
In addition to possibly lowering cholesterol, incorporating soy foods into the diet may benefit overall cardiovascular health due to their relatively high content of polyunsaturated fat, fiber, and phytosterols compared to animal products (43).
Cancer
Legumes
Colorectal cancer: Legumes, including soybeans, contain fermentable dietary fiber and certain micronutrients and phytochemicals that may have anticancer effects. Some legumes contain serine protease inhibitors of the "Bowman-Birk family" (called Bowman-Birk inhibitors) that may have potential chemopreventive effects in the large intestine (44). Moreover, simply replacing red and processed meats, which are linked to a higher risk of colorectal cancer (45), with legumes in the diet might lower risk.
To date, observational studies of legume intake and colorectal cancer have reported conflicting findings. A meta-analysis of 14 prospective cohort studies found that higher legume intake was linked to a 9% lower risk of colorectal cancer, although there was significant heterogeneity among studies (46). Subgroup analyses revealed an inverse association between soybeans and colorectal cancer (RR, 0.85; 95% CI, 0.73-0.99) but not with dry beans and colorectal cancer (RR, 1.00; 95% CI, 0.89-1.13) (46). An earlier meta-analysis of cohort and case-control studies found that higher versus lower legume intake was associated with a 17% lower risk of colorectal adenoma (47).
Beans, peas, and lentils
Although beans, peas, and lentils are rich in a number of compounds that could potentially reduce the risk of certain cancers, the results of observational studies are too inconsistent to draw any firm conclusions regarding bean intake and cancer risk in general (48, 49).
Prostate cancer: Several prospective cohort studies have found non-soy legume intake to be inversely related to incidence of prostate cancer. In a six-year prospective study of more than 14,000 Seventh Day Adventist men living in the United States, those with the highest intakes of legumes (beans, lentils, or split peas) had a 47% lower risk of prostate cancer (50). In a prospective study of more than 58,000 men in the Netherlands, those with the highest intakes (median intake, 62 g/day) of legumes (including pulses and dried seeds) had a risk of prostate cancer that was 29% lower than those with the lowest intakes (median intake, 11 g/day) (51). Moreover, a prospective study in a multiethnic cohort of 82,483 men examined the risk of prostate cancer in men who consumed legumes excluding soy products. In this study, men who consumed the highest amount of non-soy legumes had a 10% lower risk of total prostate cancer and a 28% lower risk of nonlocalized or high-grade prostate cancer compared to those who consumed the least amount of non-soy legumes (52). However, another study found no association between dry bean intake and prostate cancer (53). Most recently, in a prospective study in 3,313 French men, non-soy legume consumption was linked to a 44% reduced risk of prostate cancer (54). A meta-analysis of these five prospective cohort studies found legume consumption was associated with a 15% lower risk of prostate cancer development (95% CI, 0.72-0.99) (55). Moreover, a pooled analysis of 13 prospective cohort studies found a similar association: a 14% lower risk of prostate cancer in men who consumed ≥100 g/day of mature beans (all beans excluding green beans and soybeans) compared to those who had intakes lower than 15 g/day (56).
Soy
Prostate cancer: Incidence rates of prostate cancer are much higher in North America, Northern and Western Europe, Australia, and New Zealand compared to Asian countries, such as Japan and China, where soy is common in the diet (57). Soy food consumption has been associated with a reduced risk of prostate cancer in pooled analyses of observational studies (58-62). In the most recent meta-analysis, total soy intake (16 studies) and unfermented soy intake (11 studies) were associated with a 29% and 35% lower risk of prostate cancer, respectively; no association was found between fermented soy intake and prostate cancer (eight studies) (62). Subgroup analyses revealed that the protective association of total soy intake was much stronger in case-control (39% lower risk) versus prospective cohort and nested case-control studies (10% lower risk) — unfermented soy intake was only associated with prostate cancer risk in case-control studies (45% lower risk) (62). Results of case-control studies are more likely to be distorted by bias (i.e., the selection bias with the selection of cases and controls, as well as dietary recall bias) than results of prospective cohort studies.
For a review of studies on soy isoflavone intake and prostate cancer, see the separate article on Soy Isoflavones.
Breast cancer: More than 25 observational studies have assessed the relationship between soy food intake and the risk of breast cancer. A 2008 meta-analysis of prospective cohort studies and case-control studies reported differential effects based on the typical level of soy consumption (63). In Asian populations, where soy intake is high, the authors found an inverse association between soy food intake and breast cancer; however, no association was observed in studies completed in Western populations, where soy food intake is much lower (63). A 2016 meta-analysis of 10 prospective cohort studies — four conducted in Japan, three in the US, two in China, and one in France — found that higher intake of soy food was associated with an 8% lower incidence of breast cancer compared to lower intake (64).
Age at exposure to soy might affect subsequent risk of developing breast cancer. For instance, a few case-control studies have reported that higher soy intake during childhood or adolescence was associated with a lower risk of developing breast cancer later in life (65-68). Soy intake later in life may not have as strong as an effect on breast cancer compared to exposure during childhood and adolescence (63), and lifelong exposure to soy food may be needed to lower risk of breast cancer (69).
Isoflavones are likely responsible for any protection of soy against the development of breast cancer; studies of soy isoflavone intake and breast cancer are reviewed in the separate article on Soy Isoflavones.
Endometrial cancer: Since soy isoflavones have estrogenic activity, studies have investigated whether isoflavone intake from soy food or supplements might be related to development of endometrial cancer. There is only limited evidence of an inverse association, primarily from case-control studies (70); see the separate article on Soy Isoflavones.
Gastrointestinal cancers: Results of observational studies examining soy intake and gastrointestinal cancers, including gastric cancer and colorectal cancer, have been mixed. A 2016 meta-analysis that included 18 case-control and 16 prospective cohort studies found soy consumption was associated with a 7% lower risk of gastrointestinal cancer; however, soy intake was not significantly linked to gastrointestinal cancer when the data from only cohort studies were considered (OR, 0.97; 95% CI, 0.90-1.03) (71). A separate meta-analysis reported soybean intake was associated with a 15% lower risk of colorectal cancer (95% CI, 0.73-0.99), but only three prospective cohort studies were included in this analysis (46). High-quality studies that control for potential confounders are needed to determine whether soy food intake — or legume intake in general — is linked to a lower risk of gastric or colorectal cancer.
Obesity
Numerous clinical trials have shown that consuming low-glycemic index food delays the return of hunger, decreases subsequent food intake, and increases the sensation of fullness compared to consuming high-glycemic index food (72, 73). Some trials have specifically examined the effect of legume or pulse consumption on food intake amount, satiety, or body weight. A 2016 meta-analysis that included 21 randomized controlled trials, mostly in overweight or obese subjects, reported an overall weight loss of 0.34 kg (0.75 lb) with diets that contained pulses (median intake 132 g/day for a median of six weeks; one serving ~100 g/day) (74). This analysis found a greater weight loss in negative-energy balance trials (i.e., energy-restricted diets; weight loss averaging 1.74 kg or 3.8 lb) than in neutral energy-balance trials (i.e., weight-maintenance diets; weight loss averaging 0.29 kg or 0.64 lb), although both effects were statistically significant (74). Six trials included in this meta-analysis examined the effect of pulse consumption (161 g/day; trial average duration of 10 weeks) on other measures of obesity: no effect was seen on waist circumference, but a trend for reduced body fat percentage was found with pulse consumption (p=0.07) (74). A meta-analysis of 21 controlled clinical trials (duration ranging from 4 weeks to 2 years) found that soy consumption — as whole soy or soy protein — had no effect on body weight, and a subgroup analysis showed that soy consumption actually increased body weight in obese subjects (75). Additionally, this meta-analysis found no effect of soy intake on fat mass (10 trials) or waist circumference (16 trials) (75).
A meta-analysis of nine acute, feeding trials in mostly normal-weight subjects reported pulse consumption, administered as a single bolus, increased measures of satiety by 31% compared to an isocaloric control; however, no effect on food intake at the subsequent/second meal was seen (seven trials) (76). Increases in acute satiety, likely due to the high protein and fiber content and the low-glycemic load of legumes, could lead to reduced food intake and the weight loss observed in some studies. Although currently available studies are limited, the effect on satiety appears to be following consumption of pulses and not soy food (reviewed in 77). High-quality trials are needed to determine whether regular pulse or legume consumption results in weight loss in the long term.
Cognitive health
Some studies have linked adherence to either the Dietary Approaches to Stop Hypertension eating pattern (78, 79) or a Mediterranean-style diet (78, 80-82) with better overall cognitive function in healthy older adults. Legumes are one of many components included in these dietary patterns that might contribute to this purported association; studies reporting specifically on legume intake and cognitive endpoints are limited and have reported mixed results. In a prospective cohort study of 2,613 middle-age and older adults (ages 43-70 years) residing in the Netherlands, followed for five years, legume intake was not associated with changes in global cognitive function (memory, speed of information processing, and cognitive flexibility) (83). In contrast, a prospective, nested case-control study of the Chinese Longitudinal Health Longevity Study found that older adults (≥65 years) who included legumes in their diets nearly every day had a 22% lower risk of cognitive decline over a three-year period compared to those who had lower intakes — this study included 5,691 illiterate older adults who had no evidence of cognitive decline at baseline (84). Most recently, a small study in 214 older, Italian adults (≥65 years) with normal cognitive function used the method of principal component analysis and identified legumes as one of the food categories linked to better cognitive scores at the end of a one-year period (85). Large-scale prospective cohort studies are needed to examine whether legume intake is associated with improvements in cognitive health or risk of dementia in older adults.
Mortality
A 2017 meta-analysis of six prospective cohort studies found a weak, inverse association between legume intake and risk of all-cause mortality (RR: 0.96; 95% CI: 0.94, 1.00) (86). In a large, international prospective cohort study (N=135,335) not included in the 2017 meta-analysis, stronger inverse associations were found with respect to all-cause mortality and non-cardiovascular mortality; however, legume consumption was not associated with cardiovascular-related mortality (87). In this study, consuming more than one serving of legumes daily was associated with a 42% lower risk of non-cardiovascular mortality and a 41% lower risk of all-cause mortality compared to consuming fewer than one legume serving per month (87). In a smaller prospective study (N=7,212) in Spain, conducted among older individuals at high risk for cardiovascular disease, non-soy legume intake was associated with a lower risk of cancer-related mortality but a higher risk of cardiovascular-related mortality (88). It is important to note that average intakes of legumes in this study were low, with those in the highest tertile consuming a mean 28 g/day of legumes — less than one-third of a serving (88). Moreover, a meta-analysis of prospective cohort studies did not find soy intake to be associated with all-cause (six studies), cardiovascular-related (six studies), or cancer-related (eight studies) mortality (89).
Intake Recommendations
Substituting beans, peas, and lentils for foods that are high in saturated fat or refined carbohydrates is likely to help lower the risk of type 2 diabetes mellitus and cardiovascular disease. Soybeans and foods made from soybeans (soy foods) are excellent sources of protein. In fact, soy protein is complete protein, meaning it provides all of the essential amino acids in adequate amounts for human health (6). Like beans, peas, and lentils, soy foods are also excellent substitutes for protein sources that are high in saturated fat like red meat or cheese. Legumes are not only good sources of protein and dietary fiber but also of vitamins, minerals, as well as phytochemicals that may benefit health (see Table 1).
The 2015-2020 Dietary Guidelines for Americans — healthy eating recommendations issued jointly by the US Department of Health and Human Services and the US Department of Agriculture — include legumes (beans, lentils, dried peas, and edamame [green soybeans]) in two different food groups: the protein group and the vegetable group (1). Recommended intake of legumes at the 2,000-calorie level translates to 1½ cup-equivalents weekly for those who follow a Healthy US-style or a Healthy Mediterranean-style eating pattern and 3 cup-equivalents weekly for those following a Healthy Vegetarian eating pattern (1). Current intakes of legumes among Americans are well below these recommendations (see Figure 2-4 in the Dietary Guidelines). The Dietary Guidelines also recommend 8 oz-equivalents/week of soy products for vegetarians consuming 2,000 calories/day and 5 oz-equivalents per week for those following other recommended eating patterns, although nuts and seeds can contribute to meet this recommendation. Moreover, fortified soy beverages are emphasized as alternative to dairy in the latest Dietary Guidelines (1).
| Macronutrients | Vitamins | Minerals | Phytochemicals |
|---|---|---|---|
| Essential Fatty Acids | Folate | Iron | Fiber |
| Niacin | Magnesium | Flavonoids | |
| Riboflavin | Potassium | Lignans | |
| Vitamin B6 | Zinc | Phytosterols | |
| Soy Isoflavones |
Authors and Reviewers
Originally written in 2004 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in December 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in April 2009 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in September 2019 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in December 2019 by:
Emilio Ros, M.D., Ph.D.
Endocrinology and Nutrition Service
Hospital Clinic
Barcelona, Spain
Copyright 2004-2024 Linus Pauling Institute
References
1. US Department of Health and Human Services and U.S. Department of Agriculture. 2015–2020 Dietary Guidelines for Americans. 8th ed. December 2015. Available at https://health.gov/dietaryguidelines/2015/guidelines/.
2. Food and Agriculture Organization of the United Nations. Definition and classification of commodities. 4. Pulses and derived products. Available at: http://www.fao.org/waicent/faoinfo/economic/faodef/fdef04e.htm. Accessed 3/8/18.
3. Anderson JW, Smith BM, Washnock CS. Cardiovascular and renal benefits of dry bean and soybean intake. Am J Clin Nutr. 1999;70(3 Suppl):464S-474S. (PubMed)
4. Mudryj AN, Yu N, Aukema HM. Nutritional and health benefits of pulses. Appl Physiol Nutr Metab. 2014;39(11):1197-1204. (PubMed)
5. Messina V. Nutritional and health benefits of dried beans. Am J Clin Nutr. 2014;100 Suppl 1:437S-442S. (PubMed)
6. Messina MJ. Legumes and soybeans: overview of their nutritional profiles and health effects. Am J Clin Nutr. 1999;70(3 Suppl):439S-450S. (PubMed)
7. Ros E, Hu FB. Consumption of plant seeds and cardiovascular health: epidemiological and clinical trial evidence. Circulation. 2013;128(5):553-565. (PubMed)
8. Willett W, Manson J, Liu S. Glycemic index, glycemic load, and risk of type 2 diabetes. Am J Clin Nutr. 2002;76(1):274S-280S. (PubMed)
9. Schwingshackl L, Hoffmann G, Lampousi AM, et al. Food groups and risk of type 2 diabetes mellitus: a systematic review and meta-analysis of prospective studies. Eur J Epidemiol. 2017;32(5):363-375. (PubMed)
10. Becerra-Tomas N, Diaz-Lopez A, Rosique-Esteban N, et al. Legume consumption is inversely associated with type 2 diabetes incidence in adults: A prospective assessment from the PREDIMED study. Clin Nutr. 2018;37(3):906-913. (PubMed)
11. Sievenpiper JL, Kendall CW, Esfahani A, et al. Effect of non-oil-seed pulses on glycaemic control: a systematic review and meta-analysis of randomised controlled experimental trials in people with and without diabetes. Diabetologia. 2009;52(8):1479-1495. (PubMed)
12. Jenkins DJ, Kendall CW, Augustin LS, et al. Effect of legumes as part of a low glycemic index diet on glycemic control and cardiovascular risk factors in type 2 diabetes mellitus: a randomized controlled trial. Arch Intern Med. 2012;172(21):1653-1660. (PubMed)
13. Jenkins DJ, Wolever TM, Taylor RH, et al. Slow release dietary carbohydrate improves second meal tolerance. Am J Clin Nutr. 1982;35(6):1339-1346. (PubMed)
14. Wolever TM, Jenkins DJ, Ocana AM, Rao VA, Collier GR. Second-meal effect: low-glycemic-index foods eaten at dinner improve subsequent breakfast glycemic response. Am J Clin Nutr. 1988;48(4):1041-1047. (PubMed)
15. Mollard RC, Zykus A, Luhovyy BL, Nunez MF, Wong CL, Anderson GH. The acute effects of a pulse-containing meal on glycaemic responses and measures of satiety and satiation within and at a later meal. Br J Nutr. 2012;108(3):509-517. (PubMed)
16. Li W, Ruan W, Peng Y, Wang D. Soy and the risk of type 2 diabetes mellitus: A systematic review and meta-analysis of observational studies. Diabetes Res Clin Pract. 2018;137:190-199. (PubMed)
17. Liu ZM, Chen YM, Ho SC. Effects of soy intake on glycemic control: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2011;93(5):1092-1101. (PubMed)
18. Anderson JW, Major AW. Pulses and lipaemia, short- and long-term effect: potential in the prevention of cardiovascular disease. Br J Nutr. 2002;88 Suppl 3:S263-271. (PubMed)
19. Grosso G, Marventano S, Yang J, et al. A comprehensive meta-analysis on evidence of Mediterranean diet and cardiovascular disease: Are individual components equal? Crit Rev Food Sci Nutr. 2017;57(15):3218-3232. (PubMed)
20. Afshin A, Micha R, Khatibzadeh S, Mozaffarian D. Consumption of nuts and legumes and risk of incident ischemic heart disease, stroke, and diabetes: a systematic review and meta-analysis. Am J Clin Nutr. 2014;100(1):278-288. (PubMed)
21. Bazzano LA, He J, Ogden LG, et al. Legume consumption and risk of coronary heart disease in US men and women: NHANES I Epidemiologic Follow-up Study. Arch Intern Med. 2001;161(21):2573-2578. (PubMed)
22. Marventano S, Izquierdo Pulido M, Sanchez-Gonzalez C, et al. Legume consumption and CVD risk: a systematic review and meta-analysis. Public Health Nutr. 2017;20(2):245-254. (PubMed)
23. Shi ZQ, Tang JJ, Wu H, Xie CY, He ZZ. Consumption of nuts and legumes and risk of stroke: a meta-analysis of prospective cohort studies. Nutr Metab Cardiovasc Dis. 2014;24(12):1262-1271. (PubMed)
24. Deng C, Lu Q, Gong B, et al. Stroke and food groups: an overview of systematic reviews and meta-analyses. Public Health Nutr. 2018;21(4):766-776. (PubMed)
25. Ha V, Sievenpiper JL, de Souza RJ, et al. Effect of dietary pulse intake on established therapeutic lipid targets for cardiovascular risk reduction: a systematic review and meta-analysis of randomized controlled trials. CMAJ. 2014;186(8):E252-262. (PubMed)
26. Bazzano LA, Thompson AM, Tees MT, Nguyen CH, Winham DM. Non-soy legume consumption lowers cholesterol levels: a meta-analysis of randomized controlled trials. Nutr Metab Cardiovasc Dis. 2011;21(2):94-103. (PubMed)
27. National Heart, Lung, and Blood Institute. US Department of Health & Human Services. DASH Eating Plan. Available at: https://www.nhlbi.nih.gov/health-topics/dash-eating-plan. Accessed 4/13/18.
28. Appel LJ, Moore TJ, Obarzanek E, et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N Engl J Med. 1997;336(16):1117-1124. (PubMed)
29. Jayalath VH, de Souza RJ, Sievenpiper JL, et al. Effect of dietary pulses on blood pressure: a systematic review and meta-analysis of controlled feeding trials. Am J Hypertens. 2014;27(1):56-64. (PubMed)
30. U.S. Food and Drug Administration. Final Rule: Food Labeling: Health Claims; Soy Protein and Coronary Heart Disease [Web site]. October 26, 1999. http://www.cfsan.fda.gov/~lrd/fr991026.html. Accessed 10/10/03.
31. Anderson JW, Johnstone BM, Cook-Newell ME. Meta-analysis of the effects of soy protein intake on serum lipids. N Engl J Med. 1995;333(5):276-282. (PubMed)
32. Sirtori CR, Eberini I, Arnoldi A. Hypocholesterolaemic effects of soya proteins: results of recent studies are predictable from the anderson meta-analysis data. Br J Nutr. 2007;97(5):816-822. (PubMed)
33. Harland JI, Haffner TA. Systematic review, meta-analysis and regression of randomised controlled trials reporting an association between an intake of circa 25 g soya protein per day and blood cholesterol. Atherosclerosis. 2008;200(1):13-27. (PubMed)
34. Sacks FM, Lichtenstein A, Van Horn L, Harris W, Kris-Etherton P, Winston M. Soy protein, isoflavones, and cardiovascular health. an American Heart Association Science Advisory for professionals from the Nutrition Committee. Circulation. 2006;113(7):1034-1044. (PubMed)
35. Anderson JW, Bush HM. Soy protein effects on serum lipoproteins: a quality assessment and meta-analysis of randomized, controlled studies. J Am Coll Nutr. 2011;30(2):79-91. (PubMed)
36. Lichtenstein AH, Jalbert SM, Adlercreutz H, et al. Lipoprotein response to diets high in soy or animal protein with and without isoflavones in moderately hypercholesterolemic subjects. Arterioscler Thromb Vasc Biol. 2002;22(11):1852-1858. (PubMed)
37. Nikander E, Tiitinen A, Laitinen K, Tikkanen M, Ylikorkala O. Effects of isolated isoflavonoids on lipids, lipoproteins, insulin sensitivity, and ghrelin in postmenopausal women. J Clin Endocrinol Metab. 2004;89(7):3567-3572. (PubMed)
38. Weggemans RM, Trautwein EA. Relation between soy-associated isoflavones and LDL and HDL cholesterol concentrations in humans: a meta-analysis. Eur J Clin Nutr. 2003;57(8):940-946. (PubMed)
39. Dewell A, Hollenbeck PL, Hollenbeck CB. Clinical review: a critical evaluation of the role of soy protein and isoflavone supplementation in the control of plasma cholesterol concentrations. J Clin Endocrinol Metab. 2006;91(3):772-780. (PubMed)
40. Liu ZM, Ho SC, Chen YM, et al. Whole soy, but not purified daidzein, had a favorable effect on improvement of cardiovascular risks: a 6-month randomized, double-blind, and placebo-controlled trial in equol-producing postmenopausal women. Mol Nutr Food Res. 2014;58(4):709-717. (PubMed)
41. Tokede OA, Onabanjo TA, Yansane A, Gaziano JM, Djousse L. Soya products and serum lipids: a meta-analysis of randomised controlled trials. Br J Nutr. 2015;114(6):831-843. (PubMed)
42. Qin Y, Niu K, Zeng Y, et al. Isoflavones for hypercholesterolaemia in adults. Cochrane Database Syst Rev. 2013(6):CD009518. (PubMed)
43. Kendall CW, Jenkins DJ. A dietary portfolio: maximal reduction of low-density lipoprotein cholesterol with diet. Curr Atheroscler Rep. 2004;6(6):492-498. (PubMed)
44. Clemente A, Arques Mdel C. Bowman-Birk inhibitors from legumes as colorectal chemopreventive agents. World J Gastroenterol. 2014;20(30):10305-10315. (PubMed)
45. Domingo JL, Nadal M. Carcinogenicity of consumption of red meat and processed meat: A review of scientific news since the IARC decision. Food Chem Toxicol. 2017;105:256-261. (PubMed)
46. Zhu B, Sun Y, Qi L, Zhong R, Miao X. Dietary legume consumption reduces risk of colorectal cancer: evidence from a meta-analysis of cohort studies. Sci Rep. 2015;5:8797. (PubMed)
47. Wang Y, Wang Z, Fu L, Chen Y, Fang J. Legume consumption and colorectal adenoma risk: a meta-analysis of observational studies. PLoS One. 2013;8(6):e67335. (PubMed)
48. Mathers JC. Pulses and carcinogenesis: potential for the prevention of colon, breast and other cancers. Br J Nutr. 2002;88 Suppl 3:S273-279. (PubMed)
49. World Cancer Research Fund. Food, Nutrition, and the Prevention of Cancer: a global perspective. Washington, D.C.: American Institute for Cancer Research; 1997.
50. Mills PK, Beeson WL, Phillips RL, Fraser GE. Cohort study of diet, lifestyle, and prostate cancer in Adventist men. Cancer. 1989;64(3):598-604. (PubMed)
51. Schuurman AG, Goldbohm RA, Dorant E, van den Brandt PA. Vegetable and fruit consumption and prostate cancer risk: a cohort study in The Netherlands. Cancer Epidemiol Biomarkers Prev. 1998;7(8):673-680. (PubMed)
52. Park SY, Murphy SP, Wilkens LR, Henderson BE, Kolonel LN. Legume and isoflavone intake and prostate cancer risk: The Multiethnic Cohort Study. Int J Cancer. 2008;123(4):927-932. (PubMed)
53. Kirsh VA, Peters U, Mayne ST, et al. Prospective study of fruit and vegetable intake and risk of prostate cancer. J Natl Cancer Inst. 2007;99(15):1200-1209. (PubMed)
54. Diallo A, Deschasaux M, Galan P, et al. Associations between fruit, vegetable and legume intakes and prostate cancer risk: results from the prospective Supplementation en Vitamines et Mineraux Antioxydants (SU.VI.MAX) cohort. Br J Nutr. 2016;115(9):1579-1585. (PubMed)
55. Li J, Mao QQ. Legume intake and risk of prostate cancer: a meta-analysis of prospective cohort studies. Oncotarget. 2017;8(27):44776-44784. (PubMed)
56. Petimar J, Wilson KM, Wu K, et al. A pooled analysis of 15 prospective cohort studies on the association between fruit, vegetable, and mature bean consumption and risk of prostate cancer. Cancer Epidemiol Biomarkers Prev. 2017;26(8):1276-1287. (PubMed)
57. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87-108. (PubMed)
58. Hwang YW, Kim SY, Jee SH, Kim YN, Nam CM. Soy food consumption and risk of prostate cancer: a meta-analysis of observational studies. Nutr Cancer. 2009;61(5):598-606. (PubMed)
59. Zhang M, Wang K, Chen L, Yin B, Song Y. Is phytoestrogen intake associated with decreased risk of prostate cancer? A systematic review of epidemiological studies based on 17,546 cases. Andrology. 2016;4(4):745-756. (PubMed)
60. Yan L, Spitznagel EL. Meta-analysis of soy food and risk of prostate cancer in men. Int J Cancer. 2005;117(4):667-669. (PubMed)
61. Yan L, Spitznagel EL. Soy consumption and prostate cancer risk in men: a revisit of a meta-analysis. Am J Clin Nutr. 2009;89(4):1155-1163. (PubMed)
62. Applegate CC, Rowles JL, Ranard KM, Jeon S, Erdman JW. Soy consumption and the risk of prostate cancer: an updated systematic review and meta-analysis. Nutrients. 2018;10(1). (PubMed)
63. Wu AH, Yu MC, Tseng CC, Pike MC. Epidemiology of soy exposures and breast cancer risk. Br J Cancer. 2008;98(1):9-14. (PubMed)
64. Wu J, Zeng R, Huang J, et al. Dietary protein sources and incidence of breast cancer: a dose-response meta-analysis of prospective studies. Nutrients. 2016;8(11). (PubMed)
65. Shu XO, Jin F, Dai Q, et al. Soyfood intake during adolescence and subsequent risk of breast cancer among Chinese women. Cancer Epidemiol Biomarkers Prev. 2001;10(5):483-488. (PubMed)
66. Wu AH, Wan P, Hankin J, Tseng CC, Yu MC, Pike MC. Adolescent and adult soy intake and risk of breast cancer in Asian-Americans. Carcinogenesis. 2002;23(9):1491-1496. (PubMed)
67. Korde LA, Wu AH, Fears T, et al. Childhood soy intake and breast cancer risk in Asian American women. Cancer Epidemiol Biomarkers Prev. 2009;18(4):1050-1059. (PubMed)
68. Thanos J, Cotterchio M, Boucher BA, Kreiger N, Thompson LU. Adolescent dietary phytoestrogen intake and breast cancer risk (Canada). Cancer Causes Control. 2006;17(10):1253-1261. (PubMed)
69. Messina M, Hilakivi-Clarke L. Early intake appears to be the key to the proposed protective effects of soy intake against breast cancer. Nutr Cancer. 2009;61(6):792-798. (PubMed)
70. Zhong XS, Ge J, Chen SW, Xiong YQ, Ma SJ, Chen Q. Association between dietary isoflavones in soy and legumes and endometrial cancer: a systematic review and meta-analysis. J Acad Nutr Diet. 2018;118(4):637-651. (PubMed)
71. Tse G, Eslick GD. Soy and isoflavone consumption and risk of gastrointestinal cancer: a systematic review and meta-analysis. Eur J Nutr. 2016;55(1):63-73. (PubMed)
72. Ludwig DS. Dietary glycemic index and the regulation of body weight. Lipids. 2003;38(2):117-121. (PubMed)
73. Bornet FR, Jardy-Gennetier AE, Jacquet N, Stowell J. Glycaemic response to foods: impact on satiety and long-term weight regulation. Appetite. 2007;49(3):535-553. (PubMed)
74. Kim SJ, de Souza RJ, Choo VL, et al. Effects of dietary pulse consumption on body weight: a systematic review and meta-analysis of randomized controlled trials. Am J Clin Nutr. 2016;103(5):1213-1223. (PubMed)
75. Akhlaghi M, Zare M, Nouripour F. Effect of soy and soy isoflavones on obesity-related anthropometric measures: a systematic review and meta-analysis of randomized controlled clinical trials. Adv Nutr. 2017;8(5):705-717. (PubMed)
76. Li SS, Kendall CW, de Souza RJ, et al. Dietary pulses, satiety and food intake: a systematic review and meta-analysis of acute feeding trials. Obesity (Silver Spring). 2014;22(8):1773-1780. (PubMed)
77. Ramdath DD, Padhi EM, Sarfaraz S, Renwick S, Duncan AM. Beyond the cholesterol-lowering effect of soy protein: a review of the effects of dietary soy and its constituents on risk factors for cardiovascular disease. Nutrients. 2017;9(4). (PubMed)
78. Wengreen H, Munger RG, Cutler A, et al. Prospective study of Dietary Approaches to Stop Hypertension- and Mediterranean-style dietary patterns and age-related cognitive change: the Cache County Study on Memory, Health and Aging. Am J Clin Nutr. 2013;98(5):1263-1271. (PubMed)
79. Berendsen AAM, Kang JH, van de Rest O, Feskens EJM, de Groot L, Grodstein F. The Dietary Approaches to Stop Hypertension diet, cognitive function, and cognitive decline in American older women. J Am Med Dir Assoc. 2017;18(5):427-432. (PubMed)
80. Aridi YS, Walker JL, Wright ORL. The association between the Mediterranean dietary pattern and cognitive health: a systematic review. Nutrients. 2017;9(7). (PubMed)
81. Knight A, Bryan J, Wilson C, Hodgson J, Murphy K. A randomised controlled intervention trial evaluating the efficacy of a Mediterranean dietary pattern on cognitive function and psychological wellbeing in healthy older adults: the MedLey study. BMC Geriatr. 2015;15:55. (PubMed)
82. Samieri C, Okereke OI, E ED, Grodstein F. Long-term adherence to the Mediterranean diet is associated with overall cognitive status, but not cognitive decline, in women. J Nutr. 2013;143(4):493-499. (PubMed)
83. Nooyens AC, Bueno-de-Mesquita HB, van Boxtel MP, van Gelder BM, Verhagen H, Verschuren WM. Fruit and vegetable intake and cognitive decline in middle-aged men and women: the Doetinchem Cohort Study. Br J Nutr. 2011;106(5):752-761. (PubMed)
84. Chen X, Huang Y, Cheng HG. Lower intake of vegetables and legumes associated with cognitive decline among illiterate elderly Chinese: a 3-year cohort study. J Nutr Health Aging. 2012;16(6):549-552. (PubMed)
85. Mazza E, Fava A, Ferro Y, et al. Impact of legumes and plant proteins consumption on cognitive performances in the elderly. J Transl Med. 2017;15(1):109. (PubMed)
86. Schwingshackl L, Schwedhelm C, Hoffmann G, et al. Food groups and risk of all-cause mortality: a systematic review and meta-analysis of prospective studies. Am J Clin Nutr. 2017;105(6):1462-1473. (PubMed)
87. Miller V, Mente A, Dehghan M, et al. Fruit, vegetable, and legume intake, and cardiovascular disease and deaths in 18 countries (PURE): a prospective cohort study. Lancet. 2017;390(10107):2037-2049. (PubMed)
88. Papandreou C, Becerra-Tomas N, Bullo M, et al. Legume consumption and risk of all-cause, cardiovascular, and cancer mortality in the PREDIMED study. Clin Nutr. 2019;38(1):348-356. (PubMed)
89. Namazi N, Saneei P, Larijani B, Esmaillzadeh A. Soy product consumption and the risk of all-cause, cardiovascular and cancer mortality: a systematic review and meta-analysis of cohort studies. Food Funct. 2018;9(5):2576-2588. (PubMed)
Nuts
Contents
Summary
- Nuts are good sources of several micronutrients, as well as unsaturated fatty acids, protein, fiber, and phytochemicals. (More information)
- Results from large prospective cohort studies show an association between regular nut consumption (≥5 ounces/week) and lower risk of coronary heart disease. There is strong evidence from randomized controlled trials that nut consumption lowers blood cholesterol concentrations. (More information)
- There is little evidence to support an association between regular nut consumption and a lower risk of developing type 2 diabetes mellitus. However, there is some evidence suggesting that nut consumption can improve glycemic control in individuals with type 2 diabetes mellitus. (More information)
- Current epidemiological data indicate that higher nut consumption does not result in greater weight gain; rather, incorporating nuts into the diet may benefit weight control and contribute to reductions in body weight and waist circumference in energy-restriction diets (i.e., weight-loss diets). (More information)
- Meta-analyses of prospective cohort studies show that nut consumption is associated with a reduced risk of all-cause mortality and mortality due to chronic conditions. (More information)
- Peanuts and tree nuts can cause life-threatening allergy reactions. Current guidelines for the primary prevention of allergy set by the National Institute of Allergy and Infectious Diseases discourage nut avoidance by non-allergic women during pregnancy and encourage the early introduction to peanuts in age-appropriate foods in infants with no peanut allergy. (More information)
- Substituting nuts for unhealthy snacks is a good strategy to avoid weight gain and improve the nutritional quality of one’s diet. (More information)
Introduction
In the not too distant past, nuts were considered unhealthy because of their relatively high fat content. Recent research, however, suggests that regular nut consumption is an important part of a healthful diet. Although the fat content of nuts is relatively high (13-20 grams (g)/ounce), most of the fats in nuts are monounsaturated and polyunsaturated fats rather than saturated fats (see Table 1). The term "nuts" refers to tree nuts like almonds, Brazil nuts, cashews, hazelnuts, macadamia nuts, pecans, pistachios, and walnuts. Despite their name, peanuts are legumes like peas and beans. However, because they are nutritionally similar to tree nuts, they may share some of the same beneficial properties. Studies mentioned in this article have examined the effects of tree nuts and peanuts either together or separately.
Disease Prevention
Cardiovascular disease
In large prospective cohort studies, regular nut consumption has been consistently associated with significant reductions in the risk of coronary heart disease (CHD). An early study that followed more than 30,000 Seventh Day Adventists over 12 years found that participants who consumed nuts at least five times weekly had a 48% lower risk of death from CHD and a 51% lower risk of nonfatal myocardial infarction (MI) compared to those who consumed nuts less than once weekly (1). In addition, the risk of death from CHD was 39% lower in Seventh Day Adventists older than 83 years who ate nuts at least five times weekly compared to those who consumed nuts less than once weekly (2). More recently, an analysis of data from the Nurses' Health Study I that followed 84,136 women (ages at enrollment, 30-55 years) for 26 years reported a 32% lower risk of CHD in those who ate an average of 2.8 servings/week compared to those who never ate nuts (3). A 2014 meta-analysis of 13 prospective cohort studies, including the Seventh Day Adventists and Nurses' Health Studies, found a 34% lower risk of CHD with the highest versus lowest level of nut consumption (4). A dose-response analysis indicated that every 1 serving/week increment in nut consumption was associated with a 5% reduction in CHD risk (4). In another meta-analysis that included data from over 200,000 participants enrolled in three large cohort studies, weekly intake of at least five servings of nuts was associated with a 20% lower risk of CHD when compared to no consumption (5). However, there was no evidence of an association between nut consumption and risk of stroke. In contrast, another recent meta-analysis of prospective cohort studies found a 12% lower risk of stroke (14 studies) and a 19% lower risk of stroke-related death (seven studies) with the highest versus lowest intake of nuts (6).
The PREDIMED (Prevención con Dieta Mediterránea) study that took place between 2003-2011 was a multicenter, randomized controlled trial that examined the effect of a Mediterranean diet, with either extra-virgin olive oil or nuts, compared to a control diet, in the primary prevention of cardiovascular events in 7,447 adults (≥55 years) at high risk of cardiovascular disease (7). Adherence to a Mediterranean diet, supplemented with olive oil or mixed nuts for nearly five years resulted in a 30% lower risk of cardiovascular events and no weight gain (7).
Finally, a modeling study estimated that an increase in daily nut intake from 5 g to 30 g could have prevented 7,680 incidental cardiovascular events and saved about 65,000 years of life that were lost to stroke or heart attack in a scenario based on data from the Swedish population in 2013 (8).
Risk factors
Blood lipids: In a cross-sectional analysis of a representative sample of the US population — the National Health and Nutrition Examination Survey [NHANES] 2005-2010 — tree nut consumers (≥¼ ounce/day) were found to have higher blood HDL-cholesterol concentrations and lower body mass index (BMI), waist circumference, and systolic blood pressure than subjects consuming <¼ ounce/day (9). However, these observations may be due to reverse causation, in particular because health-conscious people are more likely to consume healthy diets that include nuts. A 2015 meta-analysis assessed the evidence of the effect of tree nut consumption on blood lipid profile using findings from 42 randomized controlled studies in a total of 2,101 participants, among which 45% were at risk of cardiovascular disease and untreated. Results indicated lower concentrations of total and LDL-cholesterol concentrations but no effect on concentrations of HDL-cholesterol or triglycerides (10). Similar observations were made in meta-analyses of interventions examining the specific effect of walnut (11, 12), almond (13), hazelnut (14), or pistachio (15) consumption on blood lipid profile of people with normal or elevated blood cholesterol. Interestingly, in a small recent trial in 46 statin-treated participants, the daily consumption of 100 g (~3.5 ounces) of almonds for four weeks led to a 4.9% reduction in non-HDL-cholesterol (i.e., total cholesterol minus HDL-cholesterol) concentration (16).
Blood pressure: Adherence to a Mediterranean diet for nearly four years in the PREDIMED trial led to significant improvements in diastolic (but not systolic) blood pressure compared to a control diet. However, there were no differences in blood pressure changes whether the Mediterranean diet was supplemented with nuts or olive oil (17). A meta-analysis of 21 randomized controlled trials in 1,652 participants found little evidence for an effect of nut supplementation on either systolic or diastolic blood pressure. A blood pressure-lowering effect of nuts was reported when only subjects without type 2 diabetes mellitus were considered. Of note, this meta-analysis included four trials that used either peanuts or soy nuts, which are not tree nuts (18).
Endothelial function: Measures of brachial flow-mediated dilation (FMD), a surrogate marker of endothelial function, are inversely associated with risk of cardiovascular events. A 2017 meta-analysis of eight randomized controlled trials suggested that supplementation with walnuts (four studies), pistachios (three studies), or almonds (one study) for up to 12 weeks may help increase FMD in subjects at risk of cardiovascular disease (19). Similar observations were reported in another recent meta-analysis of randomized controlled trials (20).
Chronic low inflammation: A 2016 meta-analysis of 20 small randomized controlled trials conducted primarily in subjects at high risk for cardiovascular disease found no evidence of an effect of nut supplementation for up to 12 weeks (only two studies lasted longer) on markers of inflammation in blood (21). Of note, four of the trials included in this meta-analysis exclusively supplemented participants with either peanuts or soy nuts. Nonetheless, a similar conclusion was reached in another meta-analysis of 25 interventions (19).
Of note, a recent systematic review of meta-analysis corroborated the account of the cardiovascular health benefits of nut consumption presented above (22).
Cardioprotective compounds in nuts
Nuts are energy-dense in particular because of their high fat content; yet, most of their fat is in the form of monounsaturated fatty acids (MUFAs) and polyunsaturated fatty acids (PUFAs). MUFAs and PUFAs are fatty acids that respectively contain one or more carbon-carbon double bonds (C=C) in their chemical structure, as opposed to saturated fatty acids that have none. Tree nuts contain more MUFAs than PUFAs with the exception of walnuts and pine nuts, which have more PUFAs than MUFAs, and Brazil nuts, which contain equivalent amounts of MUFAs and PUFAs (Table 1). Walnuts are especially rich in α-linolenic acid (~0.8 g/ounce), an omega-3 fatty acid with cardioprotective properties (see the article on Essential Fatty Acids). Other bioactive compounds, including micronutrients, phytosterols, and fiber, may also contribute to improving the cardiometabolic profile (Table 1 and Figure 1). Some nuts (pecans, pistachios, almonds, and hazelnuts) are a source of flavonoids that may contribute to cardiovascular health (see the article on Flavonoids) (23). For more information on the nutrient content of nuts, search USDA's FoodData Central.
The US Food and Drug Administration (FDA) has acknowledged the current evidence for a relationship between nut consumption and cardiovascular disease by approving the following qualified health claim for nuts (24): "Scientific evidence suggests but does not prove that eating 1.5 ounces per day of most nuts (such as macadamia nuts (25)) as part of a diet low in saturated fat and cholesterol may reduce the risk of heart disease" (24).
Type 2 diabetes mellitus
Early results from the Nurses' Health Study I suggested that nut and peanut butter consumption might be inversely associated with the risk of type 2 diabetes mellitus in women (26). However, two independent meta-analyses of more recent prospective cohort reported no evidence of an association between nut consumption and risk of type 2 diabetes (27, 28).
Nonetheless, a meta-analysis of 12 randomized controlled trials in 450 participants with type 2 diabetes showed that supplementation with tree nuts for a median of eight weeks could reduce fasting glucose concentration and glycated hemoglobin (HbA1c) concentration, a measure of glycemic control (29). Although there were no significant effects on fasting insulin concentration or a measure of insulin resistance (HOMA-IR) (29), these findings suggest that nuts might be part of a healthful diet for the management of hyperglycemia in individuals with type 2 diabetes.
Weight control
Cancer
Cognitive function
Mortality
Safety
Nut allergies
Peanuts and tree nuts are among the most common foods to trigger allergic reactions, potentially severe (anaphylaxis) and fatal (48). Such reactions can be triggered by a primary antibody response against some nut proteins or by antibodies raised against protein in pollen but cross-reacting with structurally similar proteins in nuts. Mixed method-based estimates of peanut allergy in US children suggest that the condition is increasingly prevalent and ranges between 2 and 5% (49). Estimates based solely on self reports suggest a prevalence of tree nut allergy <1% in US adults and <2% in US children (50). Individuals with peanut or tree nut allergies need to take special precautions to avoid inadvertently consuming peanuts or tree nuts by checking labels and avoiding unlabeled snacks, candies, and desserts (50). See the Food Allergy Research and Education website for additional tips to avoiding unintentional peanut or tree nut exposure.
Nut consumption during pregnancy and lactation
In the 2010 'Guidelines for the Diagnosis and Management of Food Allergy in the United States,' the National Institute of Allergy and Infectious Diseases discourages nut avoidance during pregnancy or breastfeeding as a way of preventing food intolerance in the offspring (51). Results from two birth cohort studies suggested an inverse association between maternal peanut or tree nut consumption during, shortly before, or just after their pregnancy and the risk of food allergy (including nut allergy) in the offspring (52, 53), supporting the current recommendations. Yet, prior studies found higher peanut consumption in mothers of children with peanut allergy (54). Additional studies are needed to clarify the effect of maternal nut intake on food tolerance in the offspring.
Introduction to peanuts during infancy
A 2017 addendum to the 2010 'Guidelines for the Diagnosis and Management of Food Allergy in the United States' included recommendations for the prevention of peanut allergy through the early introduction of peanuts in infants' diet (55) (Table 2).
| Infant Criteria | Earliest Age for Peanut Introduction | Recommendations |
|---|---|---|
| Severe eczema and/or egg allergy | 4-6 months |
|
| Mild-to-moderate eczema | ~6 months |
|
| No eczema or food allergies | Age-appropriate and in accordance with family preferences |
|
Adverse effects
Brazil nuts grown in areas of Brazil with selenium-rich soil may provide more than 100 µg of selenium in one nut, while those grown in selenium-poor soil may provide 10 times less (56). For information regarding toxicity of selenium, see the article on Selenium.
Intake Recommendations
Regular nut consumption, equivalent to 1 ounce of nuts five times weekly, has been consistently associated with significant reductions in risk of coronary heart disease in epidemiological studies. Consuming nuts daily as part of a diet that is low in saturated fat has been found to lower serum total and LDL-cholesterol in a number of controlled clinical trials. Since an ounce of most nuts provides at least 160 calories (kcal), simply adding an ounce of nuts daily to one's habitual diet without eliminating other foods may result in weight gain. Substituting unsalted nuts for less healthy snacks or for meat in main dishes are two ways to make nuts part of a healthful diet. A modeling study that used 2009-2012 NHANES data from 17,444 individuals (≥1 year old) estimated that substituting tree nuts for between-meal snacks on a calorie-per-calorie basis would improve the nutritional quality of dietary intakes through a shift in fatty acid intake (less saturated and more unsaturated fatty acids), a reduction in added sugar and sodium (salt), and an increase in potassium, magnesium, and fiber (57).
The 2015-2020 Dietary Guidelines for Americans encourage the consumption of nuts, seeds, and soy products as part of a healthy diet. The recommendations are presented in Table 3.
| Life Stage | Age | Mediterranean-style or US-style Eating Patternsc | Vegetarian Eating Patternd |
|---|---|---|---|
| Children | 2-3 years | 2-3 | 2-3 |
| Children | 4-8 years | 2-4 | 2-7 |
| Children | 9-13 years | 4-5 | 5-9 |
| Adolescents | 14-18 years | 4-6 | 6-13 |
| Adults | 19 years and older | 5-6 | 7-10 |
|
aDietary guidelines apply when no quantitative Dietary Reference Intake (DRI) value is available. |
|||
Authors and Reviewers
Originally written in 2003 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in December 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in March 2018 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in September 2018 by:
Emilio Ros, M.D., Ph.D.
Former Director
Lipid Clinic, Endocrinology & Nutrition Service, Hospital Clínic
University of Barcelona
Barcelona, Spain
Copyright 2003-2024 Linus Pauling Institute
References
1. Fraser GE, Sabate J, Beeson WL, Strahan TM. A possible protective effect of nut consumption on risk of coronary heart disease. The Adventist Health Study. Arch Intern Med. 1992;152(7):1416-1424. (PubMed)
2. Fraser GE, Shavlik DJ. Risk factors for all-cause and coronary heart disease mortality in the oldest-old. The Adventist Health Study. Arch Intern Med. 1997;157(19):2249-2258. (PubMed)
3. Bernstein AM, Sun Q, Hu FB, Stampfer MJ, Manson JE, Willett WC. Major dietary protein sources and risk of coronary heart disease in women. Circulation. 2010;122(9):876-883. (PubMed)
4. Ma L, Wang F, Guo W, Yang H, Liu Y, Zhang W. Nut consumption and the risk of coronary artery disease: a dose-response meta-analysis of 13 prospective studies. Thromb Res. 2014;134(4):790-794. (PubMed)
5. Guasch-Ferre M, Liu X, Malik VS, et al. Nut consumption and risk of cardiovascular disease. J Am Coll Cardiol. 2017;70(20):2519-2532. (PubMed)
6. Shao C, Tang H, Zhao W, He J. Nut intake and stroke risk: A dose-response meta-analysis of prospective cohort studies. Sci Rep. 2016;6:30394. (PubMed)
7. Ros E, Martinez-Gonzalez MA, Estruch R, et al. Mediterranean diet and cardiovascular health: Teachings of the PREDIMED study. Adv Nutr. 2014;5(3):330s-336s. (PubMed)
8. Eneroth H, Wallin S, Leander K, Nilsson Sommar J, Akesson A. Risks and benefits of increased nut consumption: cardiovascular health benefits outweigh the burden of carcinogenic effects attributed to aflatoxin B(1) exposure. Nutrients. 2017;9(12). (PubMed)
9. O'Neil CE, Fulgoni VL, 3rd, Nicklas TA. Tree nut consumption is associated with better adiposity measures and cardiovascular and metabolic syndrome health risk factors in U.S. Adults: NHANES 2005-2010. Nutr J. 2015;14:64. (PubMed)
10. Del Gobbo LC, Falk MC, Feldman R, Lewis K, Mozaffarian D. Effects of tree nuts on blood lipids, apolipoproteins, and blood pressure: systematic review, meta-analysis, and dose-response of 61 controlled intervention trials. Am J Clin Nutr. 2015;102(6):1347-1356. (PubMed)
11. Banel DK, Hu FB. Effects of walnut consumption on blood lipids and other cardiovascular risk factors: a meta-analysis and systematic review. Am J Clin Nutr. 2009;90(1):56-63. (PubMed)
12. Guasch-Ferre M, Li J, Hu FB, Salas-Salvado J, Tobias DK. Effects of walnut consumption on blood lipids and other cardiovascular risk factors: an updated meta-analysis and systematic review of controlled trials. Am J Clin Nutr. 2018;108(1):174-187. (PubMed)
13. Musa-Veloso K, Paulionis L, Poon T, Lee HY. The effects of almond consumption on fasting blood lipid levels: a systematic review and meta-analysis of randomised controlled trials. J Nutr Sci. 2016;5:e34. (PubMed)
14. Perna S, Giacosa A, Bonitta G, et al. Effects of hazelnut consumption on bood lipids and body weight: a systematic review and Bayesian meta-analysis. Nutrients. 2016;8(12). (PubMed)
15. Lippi G, Cervellin G, Mattiuzzi C. More pistachio nuts for improving the blood lipid profile. Systematic review of epidemiological evidence. Acta Biomed. 2016;87(1):5-12. (PubMed)
16. Ruisinger JF, Gibson CA, Backes JM, et al. Statins and almonds to lower lipoproteins (the STALL Study). J Clin Lipidol. 2015;9(1):58-64. (PubMed)
17. Toledo E, Hu FB, Estruch R, et al. Effect of the Mediterranean diet on blood pressure in the PREDIMED trial: results from a randomized controlled trial. BMC Med. 2013;11:207. (PubMed)
18. Mohammadifard N, Salehi-Abargouei A, Salas-Salvado J, Guasch-Ferre M, Humphries K, Sarrafzadegan N. The effect of tree nut, peanut, and soy nut consumption on blood pressure: a systematic review and meta-analysis of randomized controlled clinical trials. Am J Clin Nutr. 2015;101(5):966-982. (PubMed)
19. Neale EP, Tapsell LC, Guan V, Batterham MJ. The effect of nut consumption on markers of inflammation and endothelial function: a systematic review and meta-analysis of randomised controlled trials. BMJ Open. 2017;7(11):e016863. (PubMed)
20. Xiao Y, Huang W, Peng C, et al. Effect of nut consumption on vascular endothelial function: A systematic review and meta-analysis of randomized controlled trials. Clin Nutr. 2018;37(3):831-839. (PubMed)
21. Mazidi M, Rezaie P, Ferns GA, Gao HK. Impact of different types of tree nut, peanut, and soy nut consumption on serum C-reactive protein (CRP): A systematic review and meta-analysis of randomized controlled clinical trials. Medicine (Baltimore). 2016;95(44):e5165. (PubMed)
22. Schwingshackl L, Hoffmann G, Missbach B, Stelmach-Mardas M, Boeing H. An umbrella review of nuts intake and risk of cardiovascular disease. Curr Pharm Des. 2017;23(7):1016-1027. (PubMed)
23. Bolling BW, Chen CY, McKay DL, Blumberg JB. Tree nut phytochemicals: composition, antioxidant capacity, bioactivity, impact factors. A systematic review of almonds, Brazils, cashews, hazelnuts, macadamias, pecans, pine nuts, pistachios and walnuts. Nutr Res Rev. 2011;24(2):244-275. (PubMed)
24. US Food and Drug Administration. Qualified Health Claims: Letter of Enforcement Discretion - Nuts and Coronary Heart Disease (Docket No 02P-0505). 07/09/2015. Available at: http://wayback.archive-it.org/7993/20171114183724/https://www.fda.gov/Food/IngredientsPackagingLabeling/LabelingNutrition/ucm072926.htm. Accessed 2/17/18.
25. US Food and Drug Administration. FDA Completes Review of Qualified Health Claim Petition for Macadamia Nuts and the Risk of Coronary Heart Disease. 12/07/2017. Available at: https://www.fda.gov/Food/NewsEvents/ConstituentUpdates/ucm568052.htm. Accessed 2/17/18.
26. Jiang R, Manson JE, Stampfer MJ, Liu S, Willett WC, Hu FB. Nut and peanut butter consumption and risk of type 2 diabetes in women. JAMA. 2002;288(20):2554-2560. (PubMed)
27. Luo C, Zhang Y, Ding Y, et al. Nut consumption and risk of type 2 diabetes, cardiovascular disease, and all-cause mortality: a systematic review and meta-analysis. Am J Clin Nutr. 2014;100(1):256-269. (PubMed)
28. Zhou D, Yu H, He F, et al. Nut consumption in relation to cardiovascular disease risk and type 2 diabetes: a systematic review and meta-analysis of prospective studies. Am J Clin Nutr. 2014;100(1):270-277. (PubMed)
29. Viguiliouk E, Kendall CW, Blanco Mejia S, et al. Effect of tree nuts on glycemic control in diabetes: a systematic review and meta-analysis of randomized controlled dietary trials. PLoS One. 2014;9(7):e103376. (PubMed)
30. Freisling H, Noh H, Slimani N, et al. Nut intake and 5-year changes in body weight and obesity risk in adults: results from the EPIC-PANACEA study. Eur J Nutr. 2018;57(7):2399-2408. (PubMed)
31. Flores-Mateo G, Rojas-Rueda D, Basora J, Ros E, Salas-Salvado J. Nut intake and adiposity: meta-analysis of clinical trials. Am J Clin Nutr. 2013;97(6):1346-1355. (PubMed)
32. Mattes RD, Dreher ML. Nuts and healthy body weight maintenance mechanisms. Asia Pac J Clin Nutr. 2010;19(1):137-141. (PubMed)
33. Rock CL, Flatt SW, Barkai HS, Pakiz B, Heath DD. Walnut consumption in a weight reduction intervention: effects on body weight, biological measures, blood pressure and satiety. Nutr J. 2017;16(1):76. (PubMed)
34. Nieuwenhuis L, van den Brandt PA. Total nut, tree nut, peanut, and peanut butter consumption and the risk of pancreatic cancer in the Netherlands Cohort Study. Cancer Epidemiol Biomarkers Prev. 2018;27(3):274-284. (PubMed)
35. van den Brandt PA, Nieuwenhuis L. Tree nut, peanut, and peanut butter intake and risk of postmenopausal breast cancer: The Netherlands Cohort Study. Cancer Causes Control. 2018;29(1):63-75. (PubMed)
36. Hashemian M, Murphy G, Etemadi A, Dawsey SM, Liao LM, Abnet CC. Nut and peanut butter consumption and the risk of esophageal and gastric cancer subtypes. Am J Clin Nutr. 2017;106(3):858-864. (PubMed)
37. Arab L, Ang A. A cross sectional study of the association between walnut consumption and cognitive function among adult US populations represented in NHANES. J Nutr Health Aging. 2015;19(3):284-290. (PubMed)
38. O'Brien J, Okereke O, Devore E, Rosner B, Breteler M, Grodstein F. Long-term intake of nuts in relation to cognitive function in older women. J Nutr Health Aging. 2014;18(5):496-502. (PubMed)
39. Martinez-Lapiscina EH, Clavero P, Toledo E, et al. Mediterranean diet improves cognition: the PREDIMED-NAVARRA randomised trial. J Neurol Neurosurg Psychiatry. 2013;84(12):1318-1325. (PubMed)
40. Valls-Pedret C, Sala-Vila A, Serra-Mir M, et al. Mediterranean diet and age-related cognitive decline: a randomized clinical trial. JAMA Intern Med. 2015;175(7):1094-1103. (PubMed)
41. Aune D, Keum N, Giovannucci E, et al. Nut consumption and risk of cardiovascular disease, total cancer, all-cause and cause-specific mortality: a systematic review and dose-response meta-analysis of prospective studies. BMC Med. 2016;14(1):207. (PubMed)
42. Grosso G, Yang J, Marventano S, Micek A, Galvano F, Kales SN. Nut consumption on all-cause, cardiovascular, and cancer mortality risk: a systematic review and meta-analysis of epidemiologic studies. Am J Clin Nutr. 2015;101(4):783-793. (PubMed)
43. Mayhew AJ, de Souza RJ, Meyre D, Anand SS, Mente A. A systematic review and meta-analysis of nut consumption and incident risk of CVD and all-cause mortality. Br J Nutr. 2016;115(2):212-225. (PubMed)
44. Karimi B, Nabizadeh R, Yunesian M, Mehdipour P, Rastkari N, Aghaie A. Foods, dietary patterns and occupational class and leukocyte telomere length in the male population. Am J Mens Health. 2018;12(2):479-492. (PubMed)
45. Nettleton JA, Diez-Roux A, Jenny NS, Fitzpatrick AL, Jacobs DR, Jr. Dietary patterns, food groups, and telomere length in the Multi-Ethnic Study of Atherosclerosis (MESA). Am J Clin Nutr. 2008;88(5):1405-1412. (PubMed)
46. Tucker LA. Consumption of nuts and seeds and telomere length in 5,582 men and women of the National Health and Nutrition Examination Survey (NHANES). J Nutr Health Aging. 2017;21(3):233-240. (PubMed)
47. Zhou M, Zhu L, Cui X, et al. Influence of diet on leukocyte telomere length, markers of inflammation and oxidative stress in individuals with varied glucose tolerance: a Chinese population study. Nutr J. 2016;15:39. (PubMed)
48. Al-Muhsen S, Clarke AE, Kagan RS. Peanut allergy: an overview. CMAJ. 2003;168(10):1279-1285. (PubMed)
49. Bunyavanich S, Rifas-Shiman SL, Platts-Mills TA, et al. Peanut allergy prevalence among school-age children in a US cohort not selected for any disease. J Allergy Clin Immunol. 2014;134(3):753-755. (PubMed)
50. McWilliam V, Koplin J, Lodge C, Tang M, Dharmage S, Allen K. The prevalence of tree nut allergy: a systematic review. Curr Allergy Asthma Rep. 2015;15(9):54. (PubMed)
51. Boyce JA, Assa'ad A, Burks AW, et al. Guidelines for the diagnosis and management of food allergy in the United States: report of the NIAID-sponsored expert panel. J Allergy Clin Immunol. 2010;126(6 Suppl):S1-58. (PubMed)
52. Frazier AL, Camargo CA, Jr., Malspeis S, Willett WC, Young MC. Prospective study of peripregnancy consumption of peanuts or tree nuts by mothers and the risk of peanut or tree nut allergy in their offspring. JAMA Pediatr. 2014;168(2):156-162. (PubMed)
53. Maslova E, Granstrom C, Hansen S, et al. Peanut and tree nut consumption during pregnancy and allergic disease in children-should mothers decrease their intake? Longitudinal evidence from the Danish National Birth Cohort. J Allergy Clin Immunol. 2012;130(3):724-732. (PubMed)
54. Thompson RL, Miles LM, Lunn J, et al. Peanut sensitisation and allergy: influence of early life exposure to peanuts. Br J Nutr. 2010;103(9):1278-1286. (PubMed)
55. Togias A, Cooper SF, Acebal ML, et al. Addendum guidelines for the prevention of peanut allergy in the United States: Report of the National Institute of Allergy and Infectious Diseases-sponsored expert panel. J Allergy Clin Immunol. 2017;139(1):29-44. (PubMed)
56. Chang JC. Selenium content of Brazil nuts from two geographic locations in Brazil. Chemosphere. 1995(30):801-802. (PubMed)
57. Rehm CD, Drewnowski A. Replacing American snacks with tree nuts increases consumption of key nutrients among US children and adults: results of an NHANES modeling study. Nutr J. 2017;16(1):17. (PubMed)
Whole Grains
Contents
Summary
- Grains are the edible seeds of specific grasses of the Poaceae family and include wheat, rice, maize (corn), barley, oats, rye, triticale, millet, bulgur, and sorghum. Quinoa, amaranth, and buckwheat are pseudo-grains that are nutritionally similar to true grains. (More information)
- Whole grains are defined as intact or cracked, crushed, and flaked grain seeds in which all the components of the kernel, i.e., the bran, the endosperm, and the germ, are retained in the same relative proportions as in the intact grain. (More information)
- Bran and germ, which are lost during the refining (milling) process, are rich in minerals, vitamins, phytochemicals, and dietary fiber that play important roles in the health benefits associated with whole-grain consumption. (More information)
- There is no consensus as to what constitutes a whole-grain food. Products that bear the FDA health claims for whole grains contain at least 51% of whole-grain ingredients by weight. (More information)
- Observational studies have found that diets rich in whole grains are associated with reduced risks of type 2 diabetes mellitus and cardiovascular disease compared to diets high in refined grains. (More information)
- Although the protective effects of whole grains against cancer are not as well established as those against cardiovascular disease and type 2 diabetes mellitus, some prospective cohort studies have found whole-grain intake to be associated with a decreased risk of esophageal and colorectal cancers. (More information)
- Results from large prospective cohort studies showed that whole-grain consumption was inversely correlated with all-cause mortality and mortality from several conditions, including cardiovascular disease, cancer, type 2 diabetes mellitus, respiratory disease, and infections. (More information)
- Diets rich in whole grains and fiber may help prevent constipation in healthy people and the formation of pouches (diverticula) in the wall of the colon. In a recent prospective cohort study, higher intakes of dietary fiber, especially from cereal and fruit, was associated with a significantly lower risk of diverticular disease. (More information)
- The 2015-2020 Dietary Guidelines for Americans recommend consuming a minimum of three servings (about 90 g) of whole-grain products daily. (More information)
Introduction
Grains are seeds of plants belonging to the Poaceae family (also called Gramineae or true grasses). Some examples of edible grains include wheat, rice, maize (corn), barley, oats, rye, triticale (wheat-rye hybrid), millet, bulgur, and sorghum (1). Although they are not members of the Poaceae family, whole-grain ingredients also include pseudo-grains like quinoa, amaranth, and buckwheat. A whole grain has an outer layer of bran, a carbohydrate-rich middle layer called the endosperm, and an inner germ layer (Figure 1). Whole-grain foods contain entire grain seeds either intact, cracked, crushed, or flaked, as long as the bran, endosperm, and germ are retained in the same proportions as they exist in the intact kernel (1). Whole grains are rich in potentially beneficial compounds, including vitamins, minerals, fiber, and phytochemicals, such as lignans and phytosterols (2). Most of these compounds are located in the bran or the germ of the grain, both of which are lost during the refining (milling) process, leaving only the starchy endosperm (1). Compared to diets high in refined grains, diets rich in whole grains are associated with reduced risks of several chronic diseases. The health benefits of whole grains are not entirely explained by the individual contributions of the nutrients and phytochemicals they contain. Whole grains represent a unique package of energy, micronutrients, and phytochemicals that work synergistically to promote health and prevent disease (3).
Disease Prevention
Because there is no globally accepted definition as to what constitutes a whole-grain food, it is difficult to compare studies that examined the effects of whole-grain consumption on markers of health and disease outcomes. The US Food and Drug Administration (FDA) approved health claims for whole grains (see Finding whole grain foods) to foods containing ≥51% of whole-grain ingredients by weight or ≥8 g of whole grains per one ounce (~30 g)-serving size (4). An international multidisciplinary expert group recently proposed to label "whole grain" a food with a whole-grain content of ≥8 g per ounce (5). Yet, to date, most epidemiological studies that examined the health impact of consuming whole-grain foods have included foods containing ≥25% of whole grains and added brans by weight (6).
Type 2 diabetes mellitus
A recent meta-analysis of eight large prospective cohort studies, including 385,868 participants, have found that high versus low intakes of total grains and whole grains were associated with a significant reduction in the risk of developing type 2 diabetes mellitus (7). On the other hand, no relationship was found between refined-grain intake and diabetes in a meta-analysis of six prospective studies of 258,078 subjects (7). Specifically, the consumption of three daily servings of whole-grain foods was associated with a 32% lower risk of diabetes (see below for Examples of one serving of whole grains). Further analyses showed a significantly lower risk of diabetes with high versus low consumption of whole grains as a single food (i.e., brown rice, wheat bran) or as an ingredient in food (i.e., whole-grain bread, whole-grain breakfast cereal) but not with refined grains like white rice and wheat germ (7). In addition, a pooled analysis of three large prospective cohorts — the Health Professionals Follow-up Study [HPFS] and the Nurses’ Health Studies [NHS I and II] — reported a 17% increased risk of type 2 diabetes in participants in the highest (≥5 servings/week) versus lowest (<1 serving/month) quintile of white rice intake, while brown rice consumption (≥2 servings/week versus <1 serving/month) was associated with an 11% reduction in risk (8). Interestingly, substituting 50 g/day (⅔ serving/day) of brown rice or other whole grains for the same amount of white rice could be associated with a predicted diabetes risk reduction of 16% or more (8).
Whole grains and glucose control
Whole grains have been hypothesized to reduce the risk of type 2 diabetes mellitus by improving postprandial glycemia. Immediately after a meal, blood glucose and lipid concentrations are increased, and secretion of insulin by the pancreas stimulates glucose and lipid storage into tissues. Prolonged postprandial hyperglycemia and hyperlipidemia have been associated with oxidative stress, inflammation, insulin resistance, and endothelial dysfunction, all contributing to the development of chronic diseases like type 2 diabetes mellitus (9). The refining process that removes bran and germ facilitates the digestion of the carbohydrate-rich endosperm such that carbohydrates from refined grains were thought to elicit a higher and more rapid elevation in blood glucose, as well as a greater demand for insulin, than whole grains (10). However, compared with foods made from refined grains, whole-grain products do not necessarily have a lower glucose-raising potential, i.e., a lower glycemic index (GI) (11). The GI concept is based on the idea that foods containing carbohydrates that are easily digested, absorbed, and metabolized have a high GI (GI ≥70 on the glucose scale); in contrast, foods containing slowly digestible carbohydrates that elicit a reduced postprandial glucose response are considered to have a low GI (GI ≤55 on the glucose scale) (see also the article on Glycemic Index and Glycemic Load) (12). Bread, breakfast cereal, rice, and snack products have been attributed either a low or high GI, whether or not they include whole grains (11), suggesting that the type of food rather than its whole-grain content affects postprandial blood glucose concentrations.
In some observational studies, higher whole-grain intakes have been associated with decreased insulin resistance (13) and increased insulin sensitivity (14) in healthy individuals. In a controlled cross-over trial in 11 overweight or obese adults, consumption of a diet rich in whole grains for six weeks lowered several clinical measures of insulin resistance compared with a diet high in refined grains (15). However, in a recent randomized controlled study of 61 adults with metabolic syndrome, the consumption of a diet based on several whole-grain cereal products for 12 weeks had no effect on fasting plasma concentrations of glucose, insulin, lipids, or on insulin resistance compared with a refined grain-based diet. Yet, postprandial plasma insulin and triglycerides — but not postprandial plasma glucose — were significantly reduced with the whole grain-based diet (16). A decreased postprandial insulin response may be associated with an increase in tissue sensitivity to insulin (3). In another intervention trial in 20 healthy volunteers, three-day consumption of whole barley-based bread induced a lower insulin peak value following a standard breakfast than the same course with refined wheat bread. Whole barley-based bread consumption was also associated with an increase in circulating concentrations of gut-related hormones (e.g., peptide YY, glucagon-like peptide) and a higher gut fermentation activity. This suggested improvements in hormonal control of digestion and in colonic fermentation of resistant starch (indigestible fiber) (17), possibly promoting the feeling of satiety (18) and increasing insulin sensitivity (19).
Whole-grain consumption might be improving insulin sensitivity rather than blunting postprandial hyperglycemia; however, well-designed, large randomized controlled trials are necessary to provide further insight into how whole-grain consumption may protect against type 2 diabetes.
Cardiovascular disease
A meta-analysis of 10 large prospective cohort studies published between 1998 and 2010 found that the highest intake of whole grains (about three servings daily) was associated with an overall 21% reduced risk of cardiovascular disease (CVD), including coronary heart disease (CHD), ischemic heart disease, heart failure, and ischemic stroke, when compared to the lowest intake of whole grains and after adjustment for several CVD risk factors (20). Further, although evidence is currently limited, whole-grain intake may be associated with a reduced risk of hypertension, a risk factor for cardiovascular disease (21, 22).
Compared to refined grains, whole grains are rich in nutrients associated with cardiovascular risk reduction. In the Health Professionals Follow-up Study (HPFS) in 42,850 men, the top versus bottom quintile (49.6 g/day vs. 3.3 g/day) of whole-grain intake was associated with a 16% reduced risk of CHD after multiple adjustments for age, gender, and CHD risk factors (23). Further adjustments for whole-grain constituents, such as fiber, folate, magnesium, manganese, vitamin B6 and vitamin E, attenuated the association such that it was no longer statistically significant, suggesting that the micronutrient and fiber content may explain the cardiovascular benefits of consuming whole grains. Protective cardiovascular effects associated with higher intakes of whole grains and lower intakes of refined grains have included improvements in blood lipid profiles and reductions in markers of subclinical inflammation.
Whole grains and cardiometabolic markers
A meta-analysis of 21 randomized controlled trials indicated that whole-grain interventions for 4 to 16 weeks could improve an individual’s blood concentrations of fasting glucose, insulin, total and LDL-cholesterol, as well as reduce diastolic and systolic blood pressure (20). Consistent with this, a recently updated meta-analysis of 23 randomized controlled trials published between 1988 and 2015 indicated that consumption of whole grains (28 g/day-213 g/day for 2 to 16 weeks), especially whole-grain oats in cereal and other products, for a couple of weeks resulted in significant reductions in blood concentrations of triglycerides and total and LDL-cholesterol when compared to control diets with refined grain (24). Interventions that included mixed whole-grain products (bread, muesli, ready-to-eat cereal, pasta, rice, crisps, muffins, cookies) also improved blood HDL-cholesterol concentrations (24). In addition, although wheat fiber has not been found to lower serum cholesterol concentrations, numerous clinical studies have demonstrated that increasing intakes of oat fiber and soluble fiber from barley resulted in modest reductions in total and LDL-cholesterol (25-27). In light of such findings, the US Food and Drug Administration (FDA) approved claims regarding whole grains and reduction in risk of CHD that apply to diets low in saturated fat and cholesterol providing at least 3 g/day of β-glucan soluble fiber from oats (oat bran, rolled oats [oatmeal], whole oat flour) or whole-grain barley (28). Whole grains are also sources of phytosterols — compounds that can decrease serum cholesterol by interfering with its intestinal absorption (2).
Whole grains and inflammation markers
Evidence from observational studies suggested an inverse association between whole-grain intake and chronic low-grade inflammation that characterizes cardiovascular and metabolic diseases (29). However, intervention studies have provided mixed results. In a recent cross-over trial in healthy low whole-grain consumers, the effect of increased consumption of mixed whole grains (mean intake, 168 g/day) for six weeks was compared to whole-grain consumption of less than 16 g/day. Increasing whole-grain intake had no effect on absolute counts of immune cells in blood (leukocytes, lymphocytes, natural killer cells), on ex vivo phagocytic activities of these cells, or on markers of inflammation (e.g., IL-10, TNF-α, C-reactive protein [CRP]) in blood (30). Previous randomized controlled studies in healthy normal weight, overweight, or obese subjects have also failed to demonstrate any benefits of whole-grain intake on markers of inflammation (31-35). One eight-week dietary intervention study in 80 overweight or obese subjects found that replacement of refined products in the habitual diet by whole-grain wheat products resulted in a significant reduction in pro-inflammatory cytokine TNF-α, a transient increase in anti-inflammatory IL-10, and no change in CRP compared to intake of refined wheat (36). In another randomized cross-over intervention study, overweight/obese children (ages, 8-15 years) were provided with a list of whole-grain products and asked to either obtain half of their grain servings from whole-grain foods every day for six weeks (whole-grain group) or abstain from consuming any of these foods (control group). Mean daily consumption of 98 g of whole-grain products (compared to 11 g/day) resulted in reductions in serum concentrations of CRP, sICAM-1 (soluble intercellular adhesion molecule-1), acute phase protein SAA (serum amyloid A), and leptin (37). An increased whole-grain intake to about five daily servings (compared to <1 serving/day) also reduced blood concentrations of CRP but had no effect on IL-10 and TNF-α concentrations in obese adults with metabolic syndrome following a hypocaloric diet (38). Inconsistency among studies may be attributed to differences in the health status of participants, the duration of interventions, and/or the types of whole grains selected. In particular, if foods with a low glycemic index (GI) can lower cardiometabolic and inflammation markers (39), substituting refined grain products by whole grains with high GI may not demonstrate any benefits regarding the risk of heart disease.
Cancer
Although the protective effects of whole grains against various types of cancer are not as well established as those against type 2 diabetes mellitus and cardiovascular disease, numerous case-control studies have found inverse associations between whole-grain intake and cancer risk (40-42). An early meta-analysis of 40 case-control studies examining 20 different types of cancer found that people with higher whole-grain intakes had an overall risk of cancer that was 34% lower than those with lesser whole-grain intakes (40). Higher intakes of whole grains were most consistently associated with decreased risk of gastrointestinal tract cancers, including cancers of the mouth, throat, esophagus, stomach, colon, and rectum. A prospective cohort study that followed more than 61,000 Swedish women for 15 years found that those who consumed more than 4.5 servings of whole grains daily had a 35% lower risk of colon cancer than those who consumed less than 1.5 servings of whole grains daily (43). In the large National Institutes of Health (NIH)-AARP Diet and Health prospective study in 291,988 men and 197,623 women, mean whole-grain intakes — much lower than in the above-mentioned Swedish cohort — were also inversely associated with risk of colorectal cancer, especially rectal cancer (44). Specifically, the highest versus lowest quintile of whole-grain intake (2.6 servings/day vs. 0.4 servings/day) was associated with a 36% lower risk of developing rectal cancer (44). In a nested case-control study, including participants of the multicenter European Prospective Investigation into Cancer and Nutrition (EPIC), the top versus bottom quartile of plasma alkylresorcinol concentrations, used as a surrogate marker of whole-grain wheat and rye intakes, was found to be associated with a 52% lower incidence of distal colon cancer. No correlations were reported with the incidence of rectal cancer, colon cancer, and proximal colon cancer, or with the overall incidence of colorectal cancers (45). Not all cohort studies have suggested that whole grains might protect against intestinal cancers (46, 47). However, a dose-response analysis based on the results of six cohort studies found a 17% reduction in colorectal cancer risk with an increment of three servings (three oz-eq or 90 g) of whole grains daily (48). Of note, a recent analysis of three Scandinavian cohorts that are also part of the EPIC study and include over 110,000 participants showed an inverse correlation between total whole-grain intake and esophageal cancer risk. Each 10 g-increase in whole-grain wheat intake was found to be associated with a 50% lower risk of esophageal cancer. Such an association was not observed with whole-grain rye or with whole-grain oats (49).
In contrast to refined-grain products, whole grains are rich in numerous compounds that may be protective against cancer, particularly cancers of the gastrointestinal tract (50). Whole grains are a major source of fiber, and high-fiber intakes are thought to speed up the passage of stool through the colon, allowing less time for potentially carcinogenic compounds to stay in contact with cells that line the inner surface of the colon (51). Dietary fiber can also exert chemopreventive effects via short-chain fatty acids that are generated when fiber is fermented by the colonic microbiota (52). Whole grains also contain compounds such as phenolic acids, lignans, phytoestrogens, flavonoids, and vitamin E, that may modify signal transduction pathways that promote the development of cancer or bind potentially damaging free metal ions in the gastrointestinal tract (53, 54).
Mortality
Recent large prospective cohort studies have investigated the relationship between whole-grain consumption and the risk of all-cause and cause-specific mortality. Higher versus lower intakes of whole grains (1.20 oz-eq/day versus 0.13 oz-eq/day) have been associated with a 17% lower risk of all-cause mortality in the NIH-AARP Diet and Health Study of 367,442 older adults (55). Higher whole-grain intakes were significantly associated with a decreased risk of mortality from cardiovascular disease (-17%), cancer (-15%), type 2 diabetes mellitus (-48%), respiratory disease (-11%), and infections (-23%). These associations were largely attenuated after adjustments for cereal fiber intakes, suggesting a major role for fiber in the protective effects of whole grains on mortality (55). Another recent analysis of two US prospective cohort studies, the Nurses’ Health Study (NHS) in 74,341 women and the Health Professionals Follow-up Study (HPFS) in 43,744 men, reported a 9% lower risk of all-cause mortality in individuals in the highest versus lowest quintile of whole-grain intake (56). Higher whole-grain consumption was associated with a 15% lower risk of cardiovascular disease-related mortality, but no correlation was found with cancer-related mortality. Finally, the association of whole-grain intake with mortality was also examined in over 110,000 participants of the Scandinavian HELGA cohort (57). In this cohort, a doubling of the consumption of whole-grain products or that of specific whole-grain wheat, rye, or oats was associated with a reduced risk of all-cause and cause-specific mortality.
These results from cohort studies in the US and northern Europe consistently suggested a role of whole-grain consumption in the prevention of early death.
Intestinal health
Diets rich in whole grains and fiber may help prevent or improve constipation symptoms by softening and adding bulk to stool and by speeding its passage through the colon (58, 59). Such diets are also associated with decreased risk of diverticulosis, a condition characterized by the formation of small pouches (diverticula) in the colon. Although most people with diverticulosis experience no symptoms, about 10%-25% may develop pain or inflammation, known as diverticulitis (58). Diverticulitis was virtually unheard of before the practice of milling (refining) flour began in industrialized countries, and the role of a low-fiber diet in the development of diverticular disease is well established (60). If high-fiber diets reduce the risk of diverticular disease (61, 62), then the source of fiber (e.g., from cereal, fruit, vegetables) may be important. Interestingly, a 5 g-increase in intake of fiber from cereal was found to be associated with a 14% reduced risk of diverticular disease in a UK-based cohort of 690,075 women (mean age, 60 years) followed for up to six years; the risk of diverticular disease was decreased by 15% and 5% with a 5 g-increase in the consumption of fruit fiber and vegetable fiber, respectively (62). High-fiber diets are also recommended for people with diverticulosis in order to prevent the formation of additional diverticula rather than to resolve formed diverticula (58). People with diverticulosis are sometimes advised to avoid eating small seeds and husks to prevent them from becoming lodged in diverticula and causing diverticulitis, especially if they do not consume a high-fiber diet (58). However, it should be noted that no study has ever shown that avoiding seeds or popcorn reduces the risk of diverticulitis in an individual with diverticulosis (60).
Body weight management
Prospective cohort studies have consistently suggested that whole-grain consumption is associated with lower body mass index (BMI) and lower risks of weight gain and obesity (6, 20). However, a recent meta-analysis of randomized controlled trials published between 1988 and 2012 reported no significant effects of whole-grain intakes (from 18.2 g/day-150 g/day for 2 to 16 weeks) on body weight (26 trials), body fat (7 studies), and waist circumference (9 studies) in up to 2,060 normal-weight or overweight/obese adults without chronic health conditions (63). In a recent randomized, open-label, controlled trial in 60 overweight/obese individuals with metabolic syndrome, the consumption of whole grains (about 6-12 servings/day) was compared to that of refined grains during a 12-week intervention period that included a weight-maintenance diet for the first six weeks followed by six weeks of a hypocaloric diet (64). Increased whole-grain intake failed to lower body weight, BMI, percentage of body fat, or waist circumference beyond reductions also observed with consumption of refined grains. Of note, individuals who consumed whole grains showed an improved fasting glycemia compared to those fed refined grains, but other cardiometabolic variables remained unchanged (64). These results contrast with other energy-restricted dietary interventions showing a more favorable effect of whole grains on percentage of body fat compared to refined grains (38, 65). Further investigation is warranted to clarify whether whole-grain consumption could play a role in body weight regulation.
Intake Recommendations
Whole-grain intakes approaching three servings daily are associated with significant reductions in chronic disease risk in populations with relatively low whole-grain intakes. The 2015-2020 Dietary Guidelines for Americans — issued jointly by the US Department of Health and Human Services and US Department of Agriculture — recommend that at least half of all grains consumed be whole grains and to increase whole-grain intake by replacing refined grains with whole grains (66). In the 2015-2020 Dietary Guidelines for Americans, the unit of measure of a whole-grain serving size is the ounce-equivalent (oz-eq). A whole-grain serving size corresponds to (1) one ounce (~30 g) of a 100% whole-grain food in its ready-to-eat form, (2) two ounces of partly whole-grain products, or (3) the amount of food containing 16 g of whole-grain ingredients (67). Table 1 summarizes the 2015-2020 US Dietary Guidelines for whole-grain intakes.
| Life Stage | Age | Daily Intake (oz-eq/day)3 | Daily Intake (g/day)4 |
|---|---|---|---|
| Children | 2-3 years | 1.5-2.5 | 24-40 |
| Children | 4-8 years | 2-3 | 32-48 |
| Children | 9-13 years | 2.5-4.5 | 40-72 |
| Adolescents | 14-18 years | 3-5 | 48-80 |
| Adults | 19 years and older | 3-5 | 48-80 |
|
1Dietary guidelines apply when no quantitative Dietary Reference Intake (DRI) value is available. |
|||
The US National Health and Nutrition Examination Survey (NHANES) 2009-2010 reported mean whole-grain intakes of 0.57 oz-eq/day in children and adolescents and 0.82 oz-eq/day in adults (68). Approximately 40% of Americans consume no whole grains, and only 2.9% of children/adolescents and 7.7% of adults consume ≥3 oz-eq/day of whole grains (68). In view of the potential health benefits of increasing whole-grain intake, three daily servings of whole-grain foods should be seen as a minimum amount, and whole-grain foods should be substituted for refined carbohydrates whenever possible.
Examples of one serving of whole grains
- 1 slice of whole-grain bread
- ½ whole-grain English muffin, bagel, or bun
- 1 ounce of ready to eat whole-grain cereal
- ½ cup of oatmeal, brown rice, or whole-wheat pasta (cooked)
- 5-6 whole-grain crackers
- 1 tortilla (6" diameter)
- 1 pancake (5" diameter)
Increasing whole-grain intake
Finding whole-grain foods
Whole-grain foods may contain amaranth, whole-grain barley, brown and wild rice, buckwheat (kasha), millet, oats, popcorn, quinoa, whole rye, triticale, whole wheat (wheat berries) with various wheat species (including common wheat, emmer, spelt, and khorasan) (69). Unfortunately, it is not always clear from the label whether a product is made mostly from whole grains or refined grains. Some strategies to use when shopping for whole-grain foods include:
- Look for products that list whole grain(s) as the first ingredient(s).
- Look for whole-grain products that contain at least 2 grams of fiber per serving, since whole-grain foods are usually rich in fiber.
- Look for products that display the following health claim: "Diets rich in whole grain foods and other plant foods and low in total fat, saturated fat, and cholesterol may help reduce the risk of heart disease and certain cancers." Products displaying this health claim must contain at least 51% whole grain by weight or at least 8 grams of whole grains per ounce-equivalent (4).
- Look for whole-wheat pasta that lists whole-wheat flour as the first ingredient. Most pasta is made from refined semolina or durum wheat flour.
- Be aware that foods labeled with words like "multi-grain," "stone-ground," "100% wheat," "seven-grain," "cracked wheat," or "bran" are usually not 100% whole-grain products or can even be completely devoid of any whole grains (1).
Some strategies for increasing whole-grain intake
- Eat whole-grain breakfast cereal, such as wheat flakes, shredded wheat, muesli (rolled oats), and oatmeal. Bran cereal is not actually whole-grain cereal, but the high-fiber content makes it a good breakfast choice.
- Substitute whole-grain bread, rolls, tortillas, and crackers for those made from refined grains.
- Substitute whole-wheat pasta or pasta made from 50% whole wheat and 50% white flour for conventional pastas.
- Substitute brown rice for white rice.
- Add whole-grain barley to soups and stews.
- When baking, substitute whole-wheat flour (100% whole-wheat flour, white whole-wheat flour, or whole-wheat pastry flour) for white or unbleached flour.
Bioactive Components in Whole Grains
Whole grains are a source of numerous biologically active components; some are listed in Table 2.
| Macronutrients | Vitamins | Minerals | Phytochemicals |
|---|---|---|---|
| Unsaturated Fats | Folate | Magnesium | Fiber |
| Vitamin E | Potassium | Flavonoids | |
| Selenium | Lignans | ||
| Phytosterols |
Authors and Reviewers
Originally written in 2003 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in December 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in May 2009 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in January 2016 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in January 2016 by:
Simin Liu, M.D., M.S., M.P.H., Sc.D.
Professor of Epidemiology, Professor of Medicine
Brown University
Copyright 2003-2024 Linus Pauling Institute
References
1. US Department of Agriculture and US Department of Health and Human Services. Dietary Guidelines for Americans 2010; 2010.
2. Bartlomiej S, Justyna RK, Ewa N. Bioactive compounds in cereal grains - occurrence, structure, technological significance and nutritional benefits - a review. Food Sci Technol Int. 2012;18(6):559-568. (PubMed)
3. Seal CJ, Brownlee IA. Whole-grain foods and chronic disease: evidence from epidemiological and intervention studies. Proc Nutr Soc. 2015;74(3):313-319. (PubMed)
4. US Food and Drug Administration. Guidance for industry: a food labeling guide. Center for Food Safety and Applied Nutrition, Office of Food Labeling [Web page]. August 2015. http://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/LabelingNutrition/ucm064919.htm. Accessed 11/25/15.
5. Ferruzzi MG, Jonnalagadda SS, Liu S, et al. Developing a standard definition of whole-grain foods for dietary recommendations: summary report of a multidisciplinary expert roundtable discussion. Adv Nutr. 2014;5(2):164-176. (PubMed)
6. Cho SS, Qi L, Fahey GC, Jr., Klurfeld DM. Consumption of cereal fiber, mixtures of whole grains and bran, and whole grains and risk reduction in type 2 diabetes, obesity, and cardiovascular disease. Am J Clin Nutr. 2013;98(2):594-619. (PubMed)
7. Aune D, Norat T, Romundstad P, Vatten LJ. Whole grain and refined grain consumption and the risk of type 2 diabetes: a systematic review and dose-response meta-analysis of cohort studies. Eur J Epidemiol. 2013;28(11):845-858. (PubMed)
8. Sun Q, Spiegelman D, van Dam RM, et al. White rice, brown rice, and risk of type 2 diabetes in US men and women. Arch Intern Med. 2010;170(11):961-969. (PubMed)
9. Blaak EE, Antoine JM, Benton D, et al. Impact of postprandial glycaemia on health and prevention of disease. Obes Rev. 2012;13(10):923-984. (PubMed)
10. Liu S. Intake of refined carbohydrates and whole grain foods in relation to risk of type 2 diabetes mellitus and coronary heart disease. J Am Coll Nutr. 2002;21(4):298-306. (PubMed)
11. Atkinson FS, Foster-Powell K, Brand-Miller JC. International tables of glycemic index and glycemic load values: 2008. Diabetes Care. 2008;31(12):2281-2283. (PubMed)
12. Augustin LS, Kendall CW, Jenkins DJ, et al. Glycemic index, glycemic load and glycemic response: An International Scientific Consensus Summit from the International Carbohydrate Quality Consortium (ICQC). Nutr Metab Cardiovasc Dis. 2015;25(9):795-815. (PubMed)
13. McKeown NM, Meigs JB, Liu S, Wilson PW, Jacques PF. Whole-grain intake is favorably associated with metabolic risk factors for type 2 diabetes and cardiovascular disease in the Framingham Offspring Study. Am J Clin Nutr. 2002;76(2):390-398. (PubMed)
14. Liese AD, Roach AK, Sparks KC, Marquart L, D'Agostino RB, Jr., Mayer-Davis EJ. Whole-grain intake and insulin sensitivity: the Insulin Resistance Atherosclerosis Study. Am J Clin Nutr. 2003;78(5):965-971. (PubMed)
15. Pereira MA, Jacobs DR, Jr., Pins JJ, et al. Effect of whole grains on insulin sensitivity in overweight hyperinsulinemic adults. Am J Clin Nutr. 2002;75(5):848-855. (PubMed)
16. Giacco R, Costabile G, Della Pepa G, et al. A whole-grain cereal-based diet lowers postprandial plasma insulin and triglyceride levels in individuals with metabolic syndrome. Nutr Metab Cardiovasc Dis. 2014;24(8):837-844. (PubMed)
17. Nilsson AC, Johansson-Boll EV, Bjorck IM. Increased gut hormones and insulin sensitivity index following a 3-d intervention with a barley kernel-based product: a randomised cross-over study in healthy middle-aged subjects. Br J Nutr. 2015;114(6):899-907. (PubMed)
18. Rosen LA, Ostman EM, Bjorck IM. Effects of cereal breakfasts on postprandial glucose, appetite regulation and voluntary energy intake at a subsequent standardized lunch; focusing on rye products. Nutr J. 2011;10:7. (PubMed)
19. Robertson MD, Bickerton AS, Dennis AL, Vidal H, Frayn KN. Insulin-sensitizing effects of dietary resistant starch and effects on skeletal muscle and adipose tissue metabolism. Am J Clin Nutr. 2005;82(3):559-567. (PubMed)
20. Ye EQ, Chacko SA, Chou EL, Kugizaki M, Liu S. Greater whole-grain intake is associated with lower risk of type 2 diabetes, cardiovascular disease, and weight gain. J Nutr. 2012;142(7):1304-1313. (PubMed)
21. Wang L, Gaziano JM, Liu S, Manson JE, Buring JE, Sesso HD. Whole- and refined-grain intakes and the risk of hypertension in women. Am J Clin Nutr. 2007;86(2):472-479. (PubMed)
22. Flint AJ, Hu FB, Glynn RJ, et al. Whole grains and incident hypertension in men. Am J Clin Nutr. 2009;90(3):493-498. (PubMed)
23. Jensen MK, Koh-Banerjee P, Hu FB, et al. Intakes of whole grains, bran, and germ and the risk of coronary heart disease in men. Am J Clin Nutr. 2004;80(6):1492-1499. (PubMed)
24. Hollaender PL, Ross AB, Kristensen M. Whole-grain and blood lipid changes in apparently healthy adults: a systematic review and meta-analysis of randomized controlled studies. Am J Clin Nutr. 2015;102(3):556-572. (PubMed)
25. Ames NP, Rhymer CR. Issues surrounding health claims for barley. J Nutr. 2008;138(6):1237S-1243S. (PubMed)
26. Behall KM, Scholfield DJ, Hallfrisch J. Diets containing barley significantly reduce lipids in mildly hypercholesterolemic men and women. Am J Clin Nutr. 2004;80(5):1185-1193. (PubMed)
27. Truswell AS. Cereal grains and coronary heart disease. Eur J Clin Nutr. 2002;56(1):1-14. (PubMed)
28. U.S. Food and Drug Administration. CFR - Code of Federal Regulations Title 21, Vol. 2. Part 101: food labeling. Subpart E: specific requirements for health claims.
29. Lefevre M, Jonnalagadda S. Effect of whole grains on markers of subclinical inflammation. Nutr Rev. 2012;70(7):387-396. (PubMed)
30. Ampatzoglou A, Williams CL, Atwal KK, et al. Effects of increased wholegrain consumption on immune and inflammatory markers in healthy low habitual wholegrain consumers. Eur J Nutr. 2015. Jan 25. [Epub ahead of print]. (PubMed)
31. Andersson A, Tengblad S, Karlstrom B, et al. Whole-grain foods do not affect insulin sensitivity or markers of lipid peroxidation and inflammation in healthy, moderately overweight subjects. J Nutr. 2007;137(6):1401-1407. (PubMed)
32. Brownlee IA, Moore C, Chatfield M, et al. Markers of cardiovascular risk are not changed by increased whole-grain intake: the WHOLEheart study, a randomised, controlled dietary intervention. Br J Nutr. 2010;104(1):125-134. (PubMed)
33. Giacco R, Clemente G, Cipriano D, et al. Effects of the regular consumption of wholemeal wheat foods on cardiovascular risk factors in healthy people. Nutr Metab Cardiovasc Dis. 2010;20(3):186-194. (PubMed)
34. Nelson K, Mathai ML, Ashton JF, et al. Effects of malted and non-malted whole-grain wheat on metabolic and inflammatory biomarkers in overweight/obese adults: A randomised crossover pilot study. Food Chem. 2016;194:495-502. (PubMed)
35. Tighe P, Duthie G, Vaughan N, et al. Effect of increased consumption of whole-grain foods on blood pressure and other cardiovascular risk markers in healthy middle-aged persons: a randomized controlled trial. Am J Clin Nutr. 2010;92(4):733-740. (PubMed)
36. Vitaglione P, Mennella I, Ferracane R, et al. Whole-grain wheat consumption reduces inflammation in a randomized controlled trial on overweight and obese subjects with unhealthy dietary and lifestyle behaviors: role of polyphenols bound to cereal dietary fiber. Am J Clin Nutr. 2015;101(2):251-261. (PubMed)
37. Hajihashemi P, Azadbakht L, Hashemipor M, Kelishadi R, Esmaillzadeh A. Whole-grain intake favorably affects markers of systemic inflammation in obese children: a randomized controlled crossover clinical trial. Mol Nutr Food Res. 2014;58(6):1301-1308. (PubMed)
38. Katcher HI, Legro RS, Kunselman AR, et al. The effects of a whole grain-enriched hypocaloric diet on cardiovascular disease risk factors in men and women with metabolic syndrome. Am J Clin Nutr. 2008;87(1):79-90. (PubMed)
39. Feliciano Pereira P, das Gracas de Almeida C, Alfenas Rde C. Glycemic index role on visceral obesity, subclinical inflammation and associated chronic diseases. Nutr Hosp. 2014;30(2):237-243. (PubMed)
40. Jacobs DR, Jr., Marquart L, Slavin J, Kushi LH. Whole-grain intake and cancer: an expanded review and meta-analysis. Nutr Cancer. 1998;30(2):85-96. (PubMed)
41. La Vecchia C, Chatenoud L, Negri E, Franceschi S. Session: Whole cereal grains, fibre and human cancer Wholegrain cereals and cancer in Italy. Proc Nutr Soc. 2003;62(1):45-49. (PubMed)
42. Chan JM, Wang F, Holly EA. Whole grains and risk of pancreatic cancer in a large population-based case-control study in the San Francisco Bay Area, California. Am J Epidemiol. 2007;166(10):1174-1185. (PubMed)
43. Larsson SC, Giovannucci E, Bergkvist L, Wolk A. Whole grain consumption and risk of colorectal cancer: a population-based cohort of 60,000 women. Br J Cancer. 2005;92(9):1803-1807. (PubMed)
44. Schatzkin A, Mouw T, Park Y, et al. Dietary fiber and whole-grain consumption in relation to colorectal cancer in the NIH-AARP Diet and Health Study. Am J Clin Nutr. 2007;85(5):1353-1360. (PubMed)
45. Kyro C, Olsen A, Landberg R, et al. Plasma alkylresorcinols, biomarkers of whole-grain wheat and rye intake, and incidence of colorectal cancer. J Natl Cancer Inst. 2014;106(1):djt352. (PubMed)
46. McCullough ML, Robertson AS, Chao A, et al. A prospective study of whole grains, fruits, vegetables and colon cancer risk. Cancer Causes Control. 2003;14(10):959-970. (PubMed)
47. Pietinen P, Malila N, Virtanen M, et al. Diet and risk of colorectal cancer in a cohort of Finnish men. Cancer Causes Control. 1999;10(5):387-396. (PubMed)
48. Aune D, Chan DS, Lau R, et al. Dietary fibre, whole grains, and risk of colorectal cancer: systematic review and dose-response meta-analysis of prospective studies. BMJ. 2011;343:d6617. (PubMed)
49. Skeie G, Braaten T, Olsen A, et al. Intake of whole grains and incidence of oesophageal cancer in the HELGA Cohort. Eur J Epidemiol. 2015. Jun 20. [Epub ahead of print]. (PubMed)
50. Slavin JL. Mechanisms for the impact of whole grain foods on cancer risk. J Am Coll Nutr. 2000;19(3 Suppl):300S-307S. (PubMed)
51. Lipkin M, Reddy B, Newmark H, Lamprecht SA. Dietary factors in human colorectal cancer. Annu Rev Nutr. 1999;19:545-586. (PubMed)
52. Scharlau D, Borowicki A, Habermann N, et al. Mechanisms of primary cancer prevention by butyrate and other products formed during gut flora-mediated fermentation of dietary fibre. Mutat Res. 2009;682(1):39-53. (PubMed)
53. Kuijsten A, Arts IC, Hollman PC, van't Veer P, Kampman E. Plasma enterolignans are associated with lower colorectal adenoma risk. Cancer Epidemiol Biomarkers Prev. 2006;15(6):1132-1136. (PubMed)
54. Van Hung P. Phenolic Compounds of Cereals and Their Antioxidant Capacity. Crit Rev Food Sci Nutr. 2016;56(1):25-35. (PubMed)
55. Huang T, Xu M, Lee A, Cho S, Qi L. Consumption of whole grains and cereal fiber and total and cause-specific mortality: prospective analysis of 367,442 individuals. BMC Med. 2015;13:59. (PubMed)
56. Wu H, Flint AJ, Qi Q, et al. Association between dietary whole grain intake and risk of mortality: two large prospective studies in US men and women. JAMA Intern Med. 2015;175(3):373-384. (PubMed)
57. Johnsen NF, Frederiksen K, Christensen J, et al. Whole-grain products and whole-grain types are associated with lower all-cause and cause-specific mortality in the Scandinavian HELGA cohort. Br J Nutr. 2015;114(4):608-623. (PubMed)
58. Slavin JL. Position of the American Dietetic Association: health implications of dietary fiber. J Am Diet Assoc. 2008;108(10):1716-1731. (PubMed)
59. Woo HI, Kwak SH, Lee Y, Choi JH, Cho YM, Om AS. A Controlled, Randomized, Double-blind Trial to Evaluate the Effect of Vegetables and Whole Grain Powder That Is Rich in Dietary Fibers on Bowel Functions and Defecation in Constipated Young Adults. J Cancer Prev. 2015;20(1):64-69. (PubMed)
60. Farrell RJ, Farrell JJ, Morrin MM. Diverticular disease in the elderly. Gastroenterol Clin North Am. 2001;30(2):475-496. (PubMed)
61. Crowe FL, Appleby PN, Allen NE, Key TJ. Diet and risk of diverticular disease in Oxford cohort of European Prospective Investigation into Cancer and Nutrition (EPIC): prospective study of British vegetarians and non-vegetarians. BMJ. 2011;343:d4131. (PubMed)
62. Crowe FL, Balkwill A, Cairns BJ, et al. Source of dietary fibre and diverticular disease incidence: a prospective study of UK women. Gut. 2014;63(9):1450-1456. (PubMed)
63. Pol K, Christensen R, Bartels EM, Raben A, Tetens I, Kristensen M. Whole grain and body weight changes in apparently healthy adults: a systematic review and meta-analysis of randomized controlled studies. Am J Clin Nutr. 2013;98(4):872-884. (PubMed)
64. Harris Jackson K, West SG, Vanden Heuvel JP, et al. Effects of whole and refined grains in a weight-loss diet on markers of metabolic syndrome in individuals with increased waist circumference: a randomized controlled-feeding trial. Am J Clin Nutr. 2014;100(2):577-586. (PubMed)
65. Kristensen M, Toubro S, Jensen MG, et al. Whole grain compared with refined wheat decreases the percentage of body fat following a 12-week, energy-restricted dietary intervention in postmenopausal women. J Nutr. 2012;142(4):710-716. (PubMed)
66. US Department of Health and Human Services and US Department of Agriculture. 2015 – 2020 Dietary Guidelines for Americans. 8th ed; 2015. Available at: http://health.gov/dietaryguidelines/2015/guidelines/.
67. Whole Grains Council. What is an Ounce Equivalent? Available at: http://wholegrainscouncil.org/whole-grains-101/what-is-an-ounce-equivalent. Accessed 11/27/15.
68. Reicks M, Jonnalagadda S, Albertson AM, Joshi N. Total dietary fiber intakes in the US population are related to whole grain consumption: results from the National Health and Nutrition Examination Survey 2009 to 2010. Nutr Res. 2014;34(3):226-234. (PubMed)
69. Willett WC. Eat, Drink, and be Healthy: The Harvard Medical School Guide to Healthy Eating. New York: Simon & Schuster; 2001.
Glycemic Index and Glycemic Load
Contents
Summary
- The glycemic index (GI) is a measure of the blood glucose-raising potential of the carbohydrate content of a food compared to a reference food (generally pure glucose). Carbohydrate-containing foods can be classified as high- (≥70), moderate- (56-69), or low-GI (≤55) relative to pure glucose (GI=100). (More information)
- Consumption of high-GI foods causes a sharp increase in postprandial blood glucose concentration that declines rapidly, whereas consumption of low-GI foods results in a lower blood glucose concentration that declines gradually. (More information)
- The glycemic load (GL) is obtained by multiplying the quality of carbohydrate in a given food (GI) by the amount of carbohydrate in a serving of that food. (More information)
- Prospective cohort studies found high-GI or -GL diets to be associated with a higher risk of adverse health outcomes, including type 2 diabetes mellitus and cardiovascular disease. (More information)
- Meta-analyses of observational studies have found little-to-no evidence of an association between high dietary GI and GL and risk of cancer. (More information)
- Lowering the GL of the diet may be an effective method to improve glycemic control in individuals with type 2 diabetes mellitus. This approach is not currently included in the overall strategy of diabetes management in the US. (More information)
- Several dietary intervention studies found that low-GI/GL diets were as effective as conventional, low-fat diets in reducing body weight. Both types of diets resulted in beneficial effects on metabolic markers associated with the risk of type 2 diabetes mellitus and cardiovascular disease. (More information)
- Lowering dietary GL can be achieved by increasing the consumption of whole grains, nuts, legumes, fruit, and non-starchy vegetables, and decreasing intakes of moderate- and high-GI foods like potatoes, white rice, white bread, and sugary foods. (More information)
Glycemic Index
Glycemic index of individual foods
In the past, carbohydrates were classified as simple or complex based on the number of simple sugars in the molecule. Carbohydrates composed of one or two simple sugars like fructose or sucrose (table sugar; a disaccharide composed of one molecule of glucose and one molecule of fructose) were labeled simple, while starchy foods were labeled complex because starch is composed of long chains of the simple sugar, glucose. Advice to eat less simple and more complex carbohydrates (i.e., polysaccharides) was based on the assumption that consuming starchy foods would lead to smaller increases in blood glucose than sugary foods (1). This assumption turned out to be too simplistic since the blood glucose (glycemic) response to complex carbohydrates has been found to vary considerably. The concept of glycemic index (GI) has thus been developed in order to rank dietary carbohydrates based on their overall effect on postprandial blood glucose concentration relative to a referent carbohydrate, generally pure glucose (2). The GI is meant to represent the relative quality of a carbohydrate-containing food. Foods containing carbohydrates that are easily digested, absorbed, and metabolized have a high GI (GI≥70 on the glucose scale), while low-GI foods (GI≤55 on the glucose scale) have slowly digestible carbohydrates that elicit a reduced postprandial glucose response. Intermediate-GI foods have a GI between 56 and 69 (3). The GI of selected carbohydrate-containing foods can be found in Table 1.
Measuring the glycemic index of foods
To determine the glycemic index (GI) of a food, healthy volunteers are typically given a test food that provides 50 grams (g) of carbohydrate and a control food (white, wheat bread or pure glucose) that provides the same amount of carbohydrate, on different days (4). Blood samples for the determination of glucose concentrations are taken prior to eating, and at regular intervals for a few hours after eating. The changes in blood glucose concentration over time are plotted as a curve. The GI is calculated as the incremental area under the glucose curve (iAUC) after the test food is eaten, divided by the corresponding iAUC after the control food (pure glucose) is eaten. The value is multiplied by 100 to represent a percentage of the control food (5):
GI = (iAUCtest food/iAUCglucose) x 100
For example, a boiled white potato has an average GI of 82 relative to glucose and 116 relative to white bread, which means that the blood glucose response to the carbohydrate in a baked potato is 82% of the blood glucose response to the same amount of carbohydrate in pure glucose and 116% of the blood glucose response to the same amount of carbohydrate in white bread. In contrast, cooked brown rice has an average GI of 50 relative to glucose and 69 relative to white bread. In the traditional system of classifying carbohydrates, both brown rice and potato would be classified as complex carbohydrates despite the difference in their effects on blood glucose concentrations.
While the GI should preferably be expressed relative to glucose, other reference foods (e.g., white bread) can be used for practical reasons as long as their preparation has been standardized and they have been calibrated against glucose (2). Additional recommendations have been suggested to improve the reliability of GI values for research, public health, and commercial application purposes (2, 6).
Physiological responses to high- versus low-glycemic index foods
By definition, the consumption of high-GI foods results in higher and more rapid increases in blood glucose concentrations than the consumption of low-GI foods. Rapid increases in blood glucose (resulting in hyperglycemia) are potent signals to the β-cells of the pancreas to increase insulin secretion (7). Over the next few hours, the increase in blood insulin concentration (hyperinsulinemia) induced by the consumption of high-GI foods may cause a sharp decrease in the concentration of glucose in blood (resulting in hypoglycemia). In contrast, the consumption of low-GI foods results in lower but more sustained increases in blood glucose and lower insulin demands on pancreatic β-cells (8).
Glycemic index of a mixed meal or diet
Many observational studies have examined the association between GI and risk of chronic disease, relying on published GI values of individual foods and using the following formula to calculate meal (or diet) GI (9):
Meal GI = [(GI x amount of available carbohydrate)Food A + (GI x amount of available carbohydrate)Food B +…]/ total amount of available carbohydrate
Yet, the use of published GI values of individual foods to estimate the average GI value of a meal or diet may be inappropriate because factors such as food variety, ripeness, processing, and cooking are known to modify GI values. In a study by Dodd et al., the estimation of meal GIs using published GI values of individual foods was overestimated by 22 to 50% compared to direct measures of meal GIs (9).
Besides the GI of individual foods, various food factors are known to influence the postprandial glucose and insulin responses to a carbohydrate-containing mixed diet. A recent cross-over, randomized trial in 14 subjects with type 2 diabetes mellitus examined the acute effects of four types of breakfasts with high- or low-GI and high- or low-fiber content on postprandial glucose concentrations. Plasma glucose was found to be significantly higher following consumption of a high-GI and low-fiber breakfast than following a low-GI and high-fiber breakfast. However, there was no significant difference in postprandial glycemic responses between high-GI and low-GI breakfasts of similar fiber content (10). In this study, meal GI values (derived from published data) failed to correctly predict postprandial glucose response, which appeared to be essentially influenced by the fiber content of meals. Since the amounts and types of carbohydrate, fat, protein, and other dietary factors in a mixed meal modify the glycemic impact of carbohydrate GI values, the GI of a mixed meal calculated using the above-mentioned formula is unlikely to accurately predict the postprandial glucose response to this meal (3). Moreover, the GI is a property of a given food carbohydrate such that it does not take into account individuals’ characteristics like ethnicity, metabolic status, or eating habits (e.g., the degree to which we masticate) which might, to a limited extent, also influence the glycemic response to a given carbohydrate-containing meal (11-14).
Using direct measures of meal GIs in future trials — rather than estimates derived from GI tables — would increase the accuracy and predictive value of the GI method (2, 6). In addition, in a recent meta-analysis of 28 studies examining the effect of low- versus high-GI diets on serum lipids, Goff et al. indicated that the mean GI of low-GI diets varied from 21 to 57 across studies, while the mean GI of high-GI diets ranged from 51 to 75 (15). Therefore, a stricter use of GI cutoff values may also be warranted to provide more reliable information about carbohydrate-containing foods.
Glycemic Load
The glycemic index (GI) compares the potential of foods containing the same amount of carbohydrate to raise blood glucose. However, the amount of carbohydrate contained in a food serving also affects blood glucose concentrations and insulin responses. For example, the mean GI of watermelon is 76, which is as high as the GI of a doughnut (see Table 1). Yet, one serving of watermelon provides 11 g of available carbohydrate, while a medium doughnut provides 23 g of available carbohydrate.
The concept of glycemic load (GL) was developed by scientists to simultaneously describe the quality (GI) and quantity of carbohydrate in a food serving, meal, or diet. The GL of a single food is calculated by multiplying the GI by the amount of carbohydrate in grams (g) provided by a food serving and then dividing the total by 100 (4):
GLFood = (GIFood x amount (g) of available carbohydrateFood per serving)/100
For a typical serving of a food, GL would be considered high with GL≥20, intermediate with GL of 11-19, and low with GL≤10. Using the above-mentioned example, despite similar GIs, one serving of watermelon has a GL of 8, while a medium-sized doughnut has a GL of 17. Dietary GL is the sum of the GLs for all foods consumed in the diet.
It should be noted that while healthy food choices generally include low-GI foods, this is not always the case. For example, intermediate-to-high-GI foods like parsnip, watermelon, banana, and pineapple, have low-to-intermediate GLs (see Table 1).
Disease Prevention
Type 2 diabetes mellitus
The consumption of high-GI and -GL diets for several years might result in higher postprandial blood glucose concentration and excessive insulin secretion. This might contribute to the loss of the insulin-secreting function of pancreatic β-cells and lead to irreversible type 2 diabetes mellitus (16).
A US ecologic study of national data from 1909 to 1997 found that the increased consumption of refined carbohydrates in the form of corn syrup, coupled with the declining intake of dietary fiber, has paralleled the increased prevalence of type 2 diabetes (17). In addition, high-GI and -GL diets have been associated with an increased risk of type 2 diabetes in several large prospective cohort studies. A recent updated analysis of three large US cohorts indicated consumption of foods with the highest versus lowest GI was associated with a risk of developing type 2 diabetes that was increased by 44% in the Nurses’ Health Study (NHS) I, 20% in the NHS II, and 30% in the Health Professionals Follow-up Study (HPFS). High-GL diets were associated with an increased risk of type 2 diabetes (+18%) only in the NHS I and in the pooled analysis of the three studies (+10%) (18). Additionally, the consumption of high-GI foods that are low in cereal fiber was associated with a 59% increase in diabetes risk compared to low-GI and high-cereal-fiber foods. High-GL and low-cereal-fiber diets were associated with a 47% increase in risk compared to low-GL and high-cereal-fiber diets. Moreover, obese participants who consumed foods with high-GI or -GL values had a risk of developing type 2 diabetes that was more than 10-fold greater than lean subjects consuming low-GI or -GL diets (18).
However, a number of prospective cohort studies have reported a lack of association between GI or GL and type 2 diabetes (19-24). The use of GI food classification tables based predominantly on Australian and American food products might be a source of GI value misassignment and partly explain null associations reported in many prospective studies of European and Asian cohorts.
Nevertheless, conclusions from several recent meta-analyses of prospective studies (including the above-mentioned studies) suggest that low-GI and -GL diets might have a modest but significant effect in the prevention of type 2 diabetes (18, 25, 26). Organizations like Diabetes UK (27) and the European Association for the Study of Diabetes (28) have included the use of diets of low GI/GL and high in dietary fiber and whole grains in their recommendations for diabetes prevention in high-risk individuals. The use of GI and GL is currently not implemented in US dietary guidelines (29).
Cardiovascular disease
Observational studies
Numerous observational studies have examined the relationship between dietary GI/GL and the incidence of cardiovascular events, especially coronary heart disease (CHD) and stroke. A meta-analysis of 14 prospective cohort studies (229,213 participants; mean follow-up of 11.5 years) found a 13% and 23% increased risk of cardiovascular disease (CVD) with high versus low dietary GI and GL, respectively (30). Three independent meta-analyses of prospective studies also reported that higher GI or GL was associated with increased risk of CHD in women but not in men (31-33). A recent analysis of the European Prospective Investigation into Cancer and Nutrition (EPIC) study in 20,275 Greek participants, followed for a median of 10.4 years, showed a significant increase in CHD incidence and mortality with high dietary GL specifically in those with high BMI (≥28 kg/m2) (34). This is in line with earlier findings in the Nurses’ Health Study (NHS) showing that a high dietary GL was associated with a doubling of the risk of CHD over 10 years in women with higher (≥23 kg/m2) vs. lower BMI (35). A similar finding was reported in a cohort of middle-aged Dutch women followed for nine years (36).
Additionally, high dietary GL (but not GI) was associated with a 19% increased risk of stroke in pooled analyses of prospective cohort studies (32, 37). A meta-analysis of seven prospective studies (242,132 participants; 3,255 stroke cases) found that dietary GL was associated with an overall 23% increase in risk of stroke and a specific 35% increase in risk of ischemic stroke; GL was not found to be related to hemorrhagic stroke (38).
Overall, observational studies have found that higher glycemic load diets are associated with increased risk of cardiovascular disease, especially in women and in those with higher BMIs.
GI/GL and cardiometabolic markers
The GI/GL of carbohydrate foods may modify cardiometabolic markers associated with CVD risk. A meta-analysis of 27 randomized controlled trials (published between 1991 and 2008) examining the effect of low-GI diets on serum lipid profile reported a significant reduction in total and LDL-cholesterol independent of weight loss (15). Yet, further analysis suggested significant reductions in serum lipids only with the consumption of low-GI diets with high fiber content. In a three-month, randomized controlled study, an increase in the values of flow-mediated dilation (FMD) of the brachial artery, a surrogate marker of vascular health, was observed following the consumption of a low- versus high-GI hypocaloric diet in obese subjects (39).
High dietary GLs have been associated with increased concentrations of markers of systemic inflammation, such as C-reactive protein (CRP), interleukin-6, and tumor necrosis factor-α (TNF-α) (40, 41). In a small 12-week dietary intervention study, the consumption of a Mediterranean-style, low-GL diet (without caloric restriction) significantly reduced waist circumference, insulin resistance, systolic blood pressure, as well as plasma fasting insulin, triglycerides, LDL-cholesterol, and TNF-α in women with metabolic syndrome. A reduction in the expression of the gene coding for 3-hydroxy-3-methylglutaryl (HMG)-CoA reductase, the rate-limiting enzyme in cholesterol synthesis, in blood cells further confirmed an effect for the low-GI diet on cholesterol homeostasis (42). Well-controlled, long-term intervention studies are needed to confirm the potential cardiometabolic benefits of low GI/GL diets in people at risk for CVD.
Cancer
Evidence that high-GI or -GL diets are related to cancer is inconsistent. A recent meta-analysis of 32 case-control studies and 20 prospective cohort studies found modest and nonsignificant increased risks of hormone-related cancers (breast, prostate, ovarian, and endometrial cancers) and digestive tract cancers (esophageal, gastric, pancreas, and liver cancers) with high versus low dietary GI and GL (43). A significant positive association was found only between a high dietary GI and colorectal cancer (43). Yet, earlier meta-analyses of prospective cohort studies failed to find a link between high-GI or -GL diets and colorectal cancer (44-46). Another recent meta-analysis of prospective studies suggested a borderline increase in breast cancer risk with high dietary GI and GL. Adjustment for confounding factors across studies found no modification of menopausal status or BMI on the association (47). Further investigations are needed to verify whether GI and GL are associated with various cancers.
Gallbladder disease
Results of two studies indicate GI and GL may be related to gallbladder disease: a higher dietary GI and GL were associated with significantly increased risks of developing gallstones in a cohort of men participating in the Health Professionals Follow-up Study (48) and in a cohort of women participating in the Nurses’ Health Study (49). However, more epidemiological research is needed to determine an association between dietary glycemic index/load and gallbladder disease.
Disease Treatment
Diabetes mellitus
Whether low-GI foods could improve overall blood glucose control in people with type 1 or type 2 diabetes mellitus has been investigated in a number of intervention studies. A meta-analysis of 19 randomized controlled trials that included 840 diabetic patients (191 with type 1 diabetes and 649 with type 2 diabetes) found that consumption of low-GI foods improved short-term and long-term control of blood glucose concentrations, reflected by significant decreases in fructosamine and glycated hemoglobin (HbA1c) levels (50). However, these results need to be cautiously interpreted because of significant heterogeneity among the included studies. The American Diabetes Association has rated poorly the current evidence supporting the substitution of low-GL foods for high-GL foods to improve glycemic control in adults with type 1 or type 2 diabetes (51, 52). Well-controlled studies are needed to further assess whether the use of low-GI/GL diets could significantly improve long-term glycemic control and the quality of life of subjects with diabetes.
A randomized controlled study in 92 pregnant women (20-32 weeks) diagnosed with gestational diabetes found no significant effects of a low-GI diet on maternal metabolic profile (e.g., blood concentrations of glucose, insulin, fructosamine, HbA1c; insulin resistance) and pregnancy outcomes (i.e., maternal weight gain and neonatal anthropometric measures) compared to a conventional high-fiber, moderate-GI diet (53). The low-GI diet consumed during the pregnancy also failed to improve maternal glucose tolerance, insulin sensitivity, and other cardiovascular risk factors, or maternal and infant anthropometric data in a three-month postpartum follow-up study of 55 of the mother-infant pairs (54). In addition, another trial in 139 pregnant women (12-20 weeks’ gestation) at high risk for gestational diabetes showed no statistical differences regarding the diagnosis of gestational diabetes during the second and third trimester of pregnancy, the requirement for insulin therapy, and pregnancy outcomes and neonatal anthropometry whether women followed a low-GI diet or a high-fiber, moderate-GI diet (55). At present, there is no evidence that a low-GI diet provides benefits beyond those of a healthy, moderate-GI diet in women at high risk or affected by gestational diabetes.
Obesity
Obesity is often associated with metabolic disorders, such as hyperglycemia, insulin resistance, dyslipidemia, and hypertension, which place individuals at increased risk for type 2 diabetes mellitus, cardiovascular disease, and early death (56, 57). Traditionally, weight-loss strategies have included energy-restricted, low-fat, high-carbohydrate diets with >50% of calories from carbohydrates, ≤30% from fat, and the remainder from protein. However, a recent meta-analysis of randomized controlled intervention studies (≥6 months’ duration) has reported that low- or moderate-carbohydrate diets (4%-45% carbohydrate) and low-fat diets (10%-30% fat) were equally effective at reducing body weight and waist circumference in overweight or obese subjects (58).
Low-GI/GL diet versus moderate-GI/GL, low-fat diet
Several dietary intervention studies have examined how low-GI/GL diets compared with conventional low-fat diets to promote weight loss. Lowering the GI of conventional energy-restricted, low-fat diets was proven to be more effective to reduce postpartum body weight and waist and hip circumferences and prevent type 2 diabetes mellitus in women with prior gestational diabetes mellitus (59). In a six-month dietary intervention study in 73 obese adults, no differences in weight loss were reported in subjects following either a low-GL diet (40% carbohydrate and 35% fat) or a low-fat diet (55% carbohydrate and 20% fat). Yet, the consumption of a low-GL diet increased HDL-cholesterol and decreased triglyceride concentrations significantly more than the low-fat diet, but LDL-cholesterol concentration was significantly more reduced with the low-fat than low-GI diet (60).
A one-year randomized controlled study of 202 individuals with a body mass index (BMI) ≥28 and at least another metabolic disorder compared the effect of two dietary counseling-based interventions advocating either for a low-GL diet (30%-35% of calories from low-GI carbohydrates) or a low-fat diet (<30% of calories from fat) (61). Weight loss with each diet was equivalent (~4 kg). Both interventions similarly reduced triglycerides, C-reactive protein (CRP), and fasting insulin, and increased HDL-cholesterol. Yet, the reduction in waist and hip circumferences was greater with the low-fat diet, while blood pressure was significantly more reduced with the low-GL diet (61). In the GLYNDIET study, a six-month randomized dietary intervention trial, the comparison of two moderate-carbohydrate diets (42% of calories from carbohydrates) with different GIs (GI of 34 or GI of 62) and a low-fat diet (30% of calories from fat; GI of 65) on weight loss indicated that the low-GI diet reduced body weight more effectively than the low-fat diet. Additionally, the low-GI diet improved fasting insulin concentration, β-cell function, and insulin resistance better than the low-fat diet. None of the diets modulated hunger or satiety or affected biomarkers of endothelial function or inflammation. Finally, no significant differences were observed in low- compared to high-GL diets regarding weight loss and insulin metabolism (62).
Low-GI/GL diet versus high-GI/GL diet
In a meta-analysis of 14 randomized controlled trials published between 2005 and 2011, neither high- nor low-GI/GL dietary interventions conducted for 6 to 17 months had any significant effect on body weight and waist circumference in a total of 2,344 overweight and obese subjects (63). Low-GI/GL diets were found to significantly reduce C-reactive protein and fasting insulin but had no effect on blood lipid profile, fasting glucose concentration, or HbA1c concentration compared to high-GI/GL diets.
It has been suggested that the consumption of low-GI foods delayed the return of hunger, decreased subsequent food intake, and increased satiety when compared to high-GI foods (64). The effect of isocaloric low- and high-GI test meals on the activity of brain regions controlling appetite and eating behavior was evaluated in a small randomized, blinded, cross-over study in 12 overweight or obese men (65). During the postprandial period, blood glucose and insulin rose higher after the high-GI meal than after the low-GI meal. In addition, in response to the excess insulin secretion, blood glucose dropped below fasting concentrations three to five hours after high-GI meal consumption. Cerebral blood flow was significantly higher four hours after ingestion of the high-GI meal (compared to a low-GI meal) in a specific region of the striatum (right nucleus accumbens) associated with food intake reward and craving. If the data suggested that consuming low- rather than high-GI foods may help restrain overeating and protect against weight gain, this has not yet been confirmed in long-term randomized controlled trials. In the recent multicenter, randomized controlled Diet, Obesity, and Genes (DiOGenes) study in 256 overweight and obese individuals who lost ≥8% of body weight following an eight-week calorie-restricted diet, consumption of ad libitum diets with different protein and GI content for 12 months showed that only high-protein diets — regardless of their GI — could mitigate weight regain (66). However, the dietary interventions only achieved a modest difference in GI (~5 units) between high- and low-GI diets such that the effect of GI in weight maintenance remained unknown.
Lifestyle modification programs do not currently include the reduction of calories from carbohydrate as an alternative to standard prescription of low-fat diets, nor do they suggest the use of GI/GL as a guide to healthier dietary choices (67).
Lowering Dietary Glycemic Load
Some strategies for lowering dietary GL include:
• Increasing the consumption of whole grains, nuts, legumes, fruit, and non-starchy vegetables
• Decreasing the consumption of starchy, moderate- and high-GI foods like potatoes, white rice, and white bread
• Decreasing the consumption of sugary foods like cookies, cakes, candy, and soft drinks
Table 1 includes GI and GL values of selected foods relative to pure glucose (68). Foods are ranked in descending order of their GI values, with high-GI foods (GI≥70) at the top and foods with low-GI values (≤55) at the bottom of the table. To look up the GI values for other foods, visit the University of Sydney’s GI website.
Authors and Reviewers
Originally written in 2003 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in December 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in February 2009 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in March 2016 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in March 2016 by:
Simin Liu, M.D., M.S., M.P.H., Sc.D.
Professor of Epidemiology, Professor of Medicine
Brown University
Copyright 2003-2024 Linus Pauling Institute
References
1. Liu S, Willett WC. Dietary glycemic load and atherothrombotic risk. Curr Atheroscler Rep. 2002;4(6):454-461.
2. Brouns F, Bjorck I, Frayn KN, et al. Glycaemic index methodology. Nutr Res Rev. 2005;18(1):145-171. (PubMed)
3. Augustin LS, Kendall CW, Jenkins DJ, et al. Glycemic index, glycemic load and glycemic response: An International Scientific Consensus Summit from the International Carbohydrate Quality Consortium (ICQC). Nutr Metab Cardiovasc Dis. 2015;25(9):795-815. (PubMed)
4. Monro JA, Shaw M. Glycemic impact, glycemic glucose equivalents, glycemic index, and glycemic load: definitions, distinctions, and implications. Am J Clin Nutr. 2008;87(1):237S-243S. (PubMed)
5. The University of Sydney. About Glycemic Index. Available at: https://glycemicindex.com/about-gi/. Accessed 1/4/22.
6. The International Organization for Standardization. Food products - Determination of the glycaemic index (GI) and recommendation for food classification. 2016. Available at: https://www.iso.org/obp/ui/#iso:std:iso:26642:ed-1:v1:en. Accessed 2/22/16.
7. Ludwig DS. The glycemic index: physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA. 2002;287(18):2414-2423. (PubMed)
8. Willett WC. Eat, Drink, and be Healthy: The Harvard Medical School Guide to Healthy Eating. New York: Simon & Schuster; 2001.
9. Dodd H, Williams S, Brown R, Venn B. Calculating meal glycemic index by using measured and published food values compared with directly measured meal glycemic index. Am J Clin Nutr. 2011;94(4):992-996. (PubMed)
10. Silva FM, Kramer CK, Crispim D, Azevedo MJ. A high-glycemic index, low-fiber breakfast affects the postprandial plasma glucose, insulin, and ghrelin responses of patients with type 2 diabetes in a randomized clinical trial. J Nutr. 2015;145(4):736-741. (PubMed)
11. Ranawana V, Leow MK, Henry CJ. Mastication effects on the glycaemic index: impact on variability and practical implications. Eur J Clin Nutr. 2014;68(1):137-139. (PubMed)
12. Sun L, Ranawana DV, Tan WJ, Quek YC, Henry CJ. The impact of eating methods on eating rate and glycemic response in healthy adults. Physiol Behav. 2015;139:505-510. (PubMed)
13. Venn BS, Williams SM, Mann JI. Comparison of postprandial glycaemia in Asians and Caucasians. Diabet Med. 2010;27(10):1205-1208. (PubMed)
14. Wolever TM, Jenkins AL, Vuksan V, Campbell J. The glycaemic index values of foods containing fructose are affected by metabolic differences between subjects. Eur J Clin Nutr. 2009;63(9):1106-1114. (PubMed)
15. Goff LM, Cowland DE, Hooper L, Frost GS. Low glycaemic index diets and blood lipids: a systematic review and meta-analysis of randomised controlled trials. Nutr Metab Cardiovasc Dis. 2013;23(1):1-10. (PubMed)
16. Willett W, Manson J, Liu S. Glycemic index, glycemic load, and risk of type 2 diabetes. Am J Clin Nutr. 2002;76(1):274S-280S. (PubMed)
17. Gross LS, Li L, Ford ES, Liu S. Increased consumption of refined carbohydrates and the epidemic of type 2 diabetes in the United States: an ecologic assessment. Am J Clin Nutr. 2004;79(5):774-779. (PubMed)
18. Bhupathiraju SN, Tobias DK, Malik VS, et al. Glycemic index, glycemic load, and risk of type 2 diabetes: results from 3 large US cohorts and an updated meta-analysis. Am J Clin Nutr. 2014;100(1):218-232. (PubMed)
19. Mosdol A, Witte DR, Frost G, Marmot MG, Brunner EJ. Dietary glycemic index and glycemic load are associated with high-density-lipoprotein cholesterol at baseline but not with increased risk of diabetes in the Whitehall II study. Am J Clin Nutr. 2007;86(4):988-994. (PubMed)
20. Sahyoun NR, Anderson AL, Tylavsky FA, et al. Dietary glycemic index and glycemic load and the risk of type 2 diabetes in older adults. Am J Clin Nutr. 2008;87(1):126-131. (PubMed)
21. Sakurai M, Nakamura K, Miura K, et al. Dietary glycemic index and risk of type 2 diabetes mellitus in middle-aged Japanese men. Metabolism. 2012;61(1):47-55. (PubMed)
22. Sluijs I, Beulens JW, van der Schouw YT, et al. Dietary glycemic index, glycemic load, and digestible carbohydrate intake are not associated with risk of type 2 diabetes in eight European countries. J Nutr. 2013;143(1):93-99. (PubMed)
23. van Woudenbergh GJ, Kuijsten A, Sijbrands EJ, Hofman A, Witteman JC, Feskens EJ. Glycemic index and glycemic load and their association with C-reactive protein and incident type 2 diabetes. J Nutr Metab. 2011;2011:623076. (PubMed)
24. Villegas R, Liu S, Gao YT, et al. Prospective study of dietary carbohydrates, glycemic index, glycemic load, and incidence of type 2 diabetes mellitus in middle-aged Chinese women. Arch Intern Med. 2007;167(21):2310-2316. (PubMed)
25. Greenwood DC, Threapleton DE, Evans CE, et al. Glycemic index, glycemic load, carbohydrates, and type 2 diabetes: systematic review and dose-response meta-analysis of prospective studies. Diabetes Care. 2013;36(12):4166-4171. (PubMed)
26. Livesey G, Taylor R, Livesey H, Liu S. Is there a dose-response relation of dietary glycemic load to risk of type 2 diabetes? Meta-analysis of prospective cohort studies. Am J Clin Nutr. 2013;97(3):584-596. (PubMed)
27. Dyson PA, Kelly T, Deakin T, et al. Diabetes UK evidence-based nutrition guidelines for the prevention and management of diabetes. Diabet Med. 2011;28(11):1282-1288. (PubMed)
28. Mann JI, De Leeuw I, Hermansen K, et al. Evidence-based nutritional approaches to the treatment and prevention of diabetes mellitus. Nutr Metab Cardiovasc Dis. 2004;14(6):373-394. (PubMed)
29. American Diabetes Association. 4. Prevention or delay of type 2 diabetes. Diabetes Care. 2016;39 Suppl 1:S36-38. (PubMed)
30. Ma XY, Liu JP, Song ZY. Glycemic load, glycemic index and risk of cardiovascular diseases: meta-analyses of prospective studies. Atherosclerosis. 2012;223(2):491-496. (PubMed)
31. Dong JY, Zhang YH, Wang P, Qin LQ. Meta-analysis of dietary glycemic load and glycemic index in relation to risk of coronary heart disease. Am J Cardiol. 2012;109(11):1608-1613. (PubMed)
32. Fan J, Song Y, Wang Y, Hui R, Zhang W. Dietary glycemic index, glycemic load, and risk of coronary heart disease, stroke, and stroke mortality: a systematic review with meta-analysis. PLoS One. 2012;7(12):e52182. (PubMed)
33. Mirrahimi A, de Souza RJ, Chiavaroli L, et al. Associations of glycemic index and load with coronary heart disease events: a systematic review and meta-analysis of prospective cohorts. J Am Heart Assoc. 2012;1(5):e000752. (PubMed)
34. Turati F, Dilis V, Rossi M, et al. Glycemic load and coronary heart disease in a Mediterranean population: the EPIC Greek cohort study. Nutr Metab Cardiovasc Dis. 2015;25(3):336-342. (PubMed)
35. Liu S, Willett WC, Stampfer MJ, et al. A prospective study of dietary glycemic load, carbohydrate intake, and risk of coronary heart disease in US women. Am J Clin Nutr. 2000;71(6):1455-1461. (PubMed)
36. Beulens JW, de Bruijne LM, Stolk RP, et al. High dietary glycemic load and glycemic index increase risk of cardiovascular disease among middle-aged women: a population-based follow-up study. J Am Coll Cardiol. 2007;50(1):14-21. (PubMed)
37. Cai X, Wang C, Wang S, et al. Carbohydrate intake, glycemic index, glycemic load, and stroke: a meta-analysis of prospective cohort studies. Asia Pac J Public Health. 2015;27(5):486-496. (PubMed)
38. Rossi M, Turati F, Lagiou P, Trichopoulos D, La Vecchia C, Trichopoulou A. Relation of dietary glycemic load with ischemic and hemorrhagic stroke: a cohort study in Greece and a meta-analysis. Eur J Nutr. 2015;54(2):215-222. (PubMed)
39. Buscemi S, Cosentino L, Rosafio G, et al. Effects of hypocaloric diets with different glycemic indexes on endothelial function and glycemic variability in overweight and in obese adult patients at increased cardiovascular risk. Clin Nutr. 2013;32(3):346-352. (PubMed)
40. Bullo M, Casas R, Portillo MP, et al. Dietary glycemic index/load and peripheral adipokines and inflammatory markers in elderly subjects at high cardiovascular risk. Nutr Metab Cardiovasc Dis. 2013;23(5):443-450. (PubMed)
41. Liu S, Manson JE, Buring JE, Stampfer MJ, Willett WC, Ridker PM. Relation between a diet with a high glycemic load and plasma concentrations of high-sensitivity C-reactive protein in middle-aged women. Am J Clin Nutr. 2002;75(3):492-498. (PubMed)
42. Jones JL, Park Y, Lee J, Lerman RH, Fernandez ML. A Mediterranean-style, low-glycemic-load diet reduces the expression of 3-hydroxy-3-methylglutaryl-coenzyme A reductase in mononuclear cells and plasma insulin in women with metabolic syndrome. Nutr Res. 2011;31(9):659-664. (PubMed)
43. Turati F, Galeone C, Gandini S, et al. High glycemic index and glycemic load are associated with moderately increased cancer risk. Mol Nutr Food Res. 2015;59(7):1384-1394. (PubMed)
44. Aune D, Chan DS, Lau R, et al. Carbohydrates, glycemic index, glycemic load, and colorectal cancer risk: a systematic review and meta-analysis of cohort studies. Cancer Causes Control. 2012;23(4):521-535. (PubMed)
45. Choi Y, Giovannucci E, Lee JE. Glycaemic index and glycaemic load in relation to risk of diabetes-related cancers: a meta-analysis. Br J Nutr. 2012;108(11):1934-1947. (PubMed)
46. Mulholland HG, Murray LJ, Cardwell CR, Cantwell MM. Glycemic index, glycemic load, and risk of digestive tract neoplasms: a systematic review and meta-analysis. Am J Clin Nutr. 2009;89(2):568-576. (PubMed)
47. Mullie P, Koechlin A, Boniol M, Autier P, Boyle P. Relation between breast cancer and high glycemic index or glycemic load: a meta-analysis of prospective cohort studies. Crit Rev Food Sci Nutr. 2016;56(1):152-159. (PubMed)
48. Tsai CJ, Leitzmann MF, Willett WC, Giovannucci EL. Dietary carbohydrates and glycaemic load and the incidence of symptomatic gall stone disease in men. Gut. 2005;54(6):823-828. (PubMed)
49. Tsai CJ, Leitzmann MF, Willett WC, Giovannucci EL. Glycemic load, glycemic index, and carbohydrate intake in relation to risk of cholecystectomy in women. Gastroenterology. 2005;129(1):105-112. (PubMed)
50. Wang Q, Xia W, Zhao Z, Zhang H. Effects comparison between low glycemic index diets and high glycemic index diets on HbA1c and fructosamine for patients with diabetes: A systematic review and meta-analysis. Prim Care Diabetes. 2015;9(5):362-369. (PubMed)
51. Evert AB, Boucher JL. New diabetes nutrition therapy recommendations: what you need to know. Diabetes Spectr. 2014;27(2):121-130. (PubMed)
52. Evert AB, Boucher JL, Cypress M, et al. Nutrition therapy recommendations for the management of adults with diabetes. Diabetes Care. 2014;37 Suppl 1:S120-143. (PubMed)
53. Louie JC, Markovic TP, Perera N, et al. A randomized controlled trial investigating the effects of a low-glycemic index diet on pregnancy outcomes in gestational diabetes mellitus. Diabetes Care. 2011;34(11):2341-2346. (PubMed)
54. Louie JC, Markovic TP, Ross GP, Foote D, Brand-Miller JC. Effect of a low glycaemic index diet in gestational diabetes mellitus on post-natal outcomes after 3 months of birth: a pilot follow-up study. Matern Child Nutr. 2015;11(3):409-414. (PubMed)
55. Markovic TP, Muirhead R, Overs S, et al. Randomized controlled trial investigating the effects of a low-glycemic index diet on pregnancy outcomes in women at high risk of gestational diabetes mellitus: The GI Baby 3 Study. Diabetes Care. 2016;39(1):31-38. (PubMed)
56. Flegal KM, Kit BK, Orpana H, Graubard BI. Association of all-cause mortality with overweight and obesity using standard body mass index categories: a systematic review and meta-analysis. JAMA. 2013;309(1):71-82. (PubMed)
57. Kopelman P. Health risks associated with overweight and obesity. Obes Rev. 2007;8 Suppl 1:13-17. (PubMed)
58. Hu T, Mills KT, Yao L, et al. Effects of low-carbohydrate diets versus low-fat diets on metabolic risk factors: a meta-analysis of randomized controlled clinical trials. Am J Epidemiol. 2012;176 Suppl 7:S44-54. (PubMed)
59. Shyam S, Arshad F, Abdul Ghani R, Wahab NA. Low glycaemic index diets improve glucose tolerance and body weight in women with previous history of gestational diabetes: a six months randomized trial. 2013;12:68. (PubMed)
60. Ebbeling CB, Leidig MM, Feldman HA, Lovesky MM, Ludwig DS. Effects of a low-glycemic load vs low-fat diet in obese young adults: a randomized trial. JAMA. 2007;297(19):2092-2102. (PubMed)
61. Klemsdal TO, Holme I, Nerland H, Pedersen TR, Tonstad S. Effects of a low glycemic load diet versus a low-fat diet in subjects with and without the metabolic syndrome. Nutr Metab Cardiovasc Dis. 2010;20(3):195-201. (PubMed)
62. Juanola-Falgarona M, Salas-Salvado J, Ibarrola-Jurado N, et al. Effect of the glycemic index of the diet on weight loss, modulation of satiety, inflammation, and other metabolic risk factors: a randomized controlled trial. Am J Clin Nutr. 2014;100(1):27-35. (PubMed)
63. Schwingshackl L, Hoffmann G. Long-term effects of low glycemic index/load vs. high glycemic index/load diets on parameters of obesity and obesity-associated risks: a systematic review and meta-analysis. Nutr Metab Cardiovasc Dis. 2013;23(8):699-706. (PubMed)
64. Ludwig DS. Dietary glycemic index and the regulation of body weight. Lipids. 2003;38(2):117-121. (PubMed)
65. Lennerz BS, Alsop DC, Holsen LM, et al. Effects of dietary glycemic index on brain regions related to reward and craving in men. Am J Clin Nutr. 2013;98(3):641-647. (PubMed)
66. Aller EE, Larsen TM, Claus H, et al. Weight loss maintenance in overweight subjects on ad libitum diets with high or low protein content and glycemic index: the DIOGENES trial 12-month results. Int J Obes (Lond). 2014;38(12):1511-1517. (PubMed)
67. Wadden TA, Webb VL, Moran CH, Bailer BA. Lifestyle modification for obesity: new developments in diet, physical activity, and behavior therapy. Circulation. 2012;125(9):1157-1170. (PubMed)
68. Atkinson FS, Foster-Powell K, Brand-Miller JC. International tables of glycemic index and glycemic load values: 2008. Diabetes Care. 2008;31(12):2281-2283. (PubMed)
Coffee
Contents
Summary
- Coffee is a complex mixture of chemicals that provides significant amounts of chlorogenic acid and caffeine. (More information)
- Unfiltered coffee is a significant source of diterpenes (mainly cafestol and kahweol) that appear to raise serum total and LDL-cholesterol concentrations in humans. (More information)
- The results of observational studies suggest that coffee consumption is associated with lower risks of type 2 diabetes mellitus, Parkinson’s disease, liver disease, and mortality. However, it is premature to recommend coffee consumption for disease prevention based on this evidence. (More information)
- Coffee consumption has been associated with lower risks of cirrhosis, cirrhosis-related mortality, and liver cancer. Coffee consumption was also found to be inversely associated with the risk of oral/pharyngeal cancer, colon cancer, prostate cancer, endometrial cancer, and melanoma. Evidence also suggests that coffee consumption is not a risk factor for lung cancer. (More information)
- Despite evidence from clinical trials that caffeine in coffee can increase blood pressure, most prospective cohort studies have not found moderate coffee consumption to be associated with increased risk of cardiovascular disease. (More information)
- Caffeine intake comparable to the amount in 2-3 cups of coffee may raise blood pressure, especially in people with borderline or high blood pressure. However, regular coffee consumption in hypertensive subjects has not been associated with an increased risk of cardiovascular disease. (More information)
- Current evidence from dose-response meta-analyses of observational studies does not exclude that moderate maternal coffee consumption could adversely affect fetal growth during pregnancy. Limiting intakes of caffeinated coffee to ≤1 cup/day during pregnancy and 2-3 cups/day during breast-feeding is recommended. (More information)
- Ensuring adequate calcium and vitamin D intakes and limiting coffee consumption to 3 cups/day (~300 mg/day of caffeine) are unlikely to cause any adverse effects on calcium absorption and bone health. (More information)
- Overall, there is little evidence of health risks and some evidence of health benefits for adults consuming moderate amounts of filtered coffee (3-4 cups/day providing ~300-400 mg/day of caffeine). (More information)
Introduction
Coffee, an infusion of ground, roasted coffee beans, is among the most widely consumed beverages in the world. The main types of coffee consumed are (1) boiled unfiltered coffee, (2) filtered coffee, and (3) decaffeinated coffee (1). Although caffeine has received the most attention from scientists, coffee is a complex mixture of many chemicals, including carbohydrates, lipids (fats), amino acids, vitamins, minerals, alkaloids, and phenolic compounds (2). The composition of coffee varies with the source of coffee beans (Coffea arabica or Coffea canephora var. robusta) (3), as well as with the method of preparation (i.e., filtration methods, boiling, steeping, or brewing under pressure) (1, 4).
Some Bioactive Compounds in Coffee
Chlorogenic acids
Chlorogenic acids are a family of esters formed between quinic acid and phenolic compounds known as cinnamic acids (mostly caffeic acid and ferulic acid) (1). The most abundant chlorogenic acid in coffee is 5-O-caffeoylquinic acid, an ester formed between quinic acid and caffeic acid (Figure 1). Coffee represents one of the richest dietary sources of chlorogenic acids. The chlorogenic acid content of a 200 mL (7-oz) cup of coffee has been reported to range from 70 to 350 mg, which would provide about 35 to 175 mg of caffeic acid. Although chlorogenic and caffeic acids have demonstrated antioxidant activities in vitro (5), it is unclear how much antioxidant activity they contribute in vivo because they are extensively metabolized, and the metabolites often have lower antioxidant activity than the parent compounds (6, 7). Additionally, the antioxidant capacity of coffee is attenuated by the decaffeination process, which decreases total polyphenol content (8). Other phenolic compounds in coffee, though less abundant than chlorogenic acids, include tannins, lignans, and anthocyanine (1).
Caffeine
Caffeine (1,3,7-trimethylxanthine) is a purine alkaloid that occurs naturally in coffee beans (Figure 2). At intake levels associated with coffee consumption, caffeine appears to exert most of its biological effects through antagonism of the A1 and A2A subtypes of the adenosine receptor (1). Adenosine is an endogenous compound that modulates the response of neurons to neurotransmitters. Adenosine has mostly inhibitory effects in the central nervous system, so the effects of adenosine antagonism by caffeine are generally stimulatory. Caffeine is rapidly and almost completely absorbed in the stomach and small intestine and then distributed to all tissues, including the brain. Caffeine concentrations in coffee beverages can be quite variable. A standard cup of coffee is often assumed to provide 100 mg of caffeine, but an analysis of 14 different specialty coffees purchased at coffee shops in the US found that the amount of caffeine in 8 oz (~240 mL) of brewed coffee ranged from 72 to 130 mg (9). Caffeine in espresso coffees ranged from 58 to 76 mg in a single shot (~35 to 50 mL). In countries other than the US, coffee is often stronger, but the volume per cup is smaller, making 100 mg of caffeine/cup a reasonable estimate.
Diterpenes
Cafestol and kahweol are fat-soluble compounds known as diterpenes (Figure 3), which have been found to raise serum total and LDL-cholesterol concentrations in humans (10). Some cafestol and kahweol are extracted from ground coffee during brewing, but are largely removed from coffee by paper filters. Scandinavian boiled coffee, Turkish coffee, and French press (cafetiere) coffee contain relatively high levels of cafestol and kahweol (6 to 12 mg/cup), while filtered coffee, percolated coffee, and instant coffee contain low levels of cafestol and kahweol (0.2 to 0.6 mg/cup) (11). Although diterpene concentrations are relatively high in espresso coffee, the small serving size makes it an intermediate source of cafestol and kahweol (4 mg/cup). Since coffee beans are high in cafestol and kahweol, ingestion of coffee beans or grounds on a regular basis may also raise serum total and LDL-cholesterol.
Trigonelline
Trigonelline (N-methylnicotinic acid) is a plant alkaloid derived from niacin (vitamin B3) (Figure 4). Trigonelline is largely broken down to nicotinic acid during the roasting process, although some intact molecules remain in roasted beans. Trigonelline has been found to exert antioxidant, hypoglycemic, and hypolipidemic activities (reviewed in 1).
Disease Prevention
Type 2 diabetes mellitus
Observational studies
The three largest prospective cohort studies in the US to examine the relationship between caffeinated coffee consumption and type 2 diabetes mellitus were the Health Professionals Follow-up Study ([HPFS]; 41,934 men), the Nurses’ Health Study ([NHS I]; 84,276 women), and the NHS II (88,259 women). Men who drank at least six cups of coffee daily had a 54% lower risk of developing type 2 diabetes than men who did not drink coffee. In the NHS I cohort, women who drank at least six cups of coffee daily had a 29% lower risk of type 2 diabetes than women who did not drink coffee (12). In the NHS II cohort, women who consumed ≥4 cups of coffee daily had a 39% lower risk of developing type 2 diabetes; similar results were found in women who drank 2 to 3 cups/day of coffee (13). In a pooled analysis of all three cohorts, an increase in caffeinated coffee intake by >1 cup/day over a four-year period was associated with a 13% decreased risk of type 2 diabetes in the subsequent four years; those who decreased their intake of caffeinated coffee by >1 cup/day had a 20% increased risk of type 2 diabetes over the four-year period (14).
Several other cohort studies have found higher coffee intakes to be associated with significant reductions in the risk of type 2 diabetes. A systematic review and meta-analysis of 18 prospective cohort studies (published between 1966 and 2009), including more than 450,000 men and women, found that the risk of developing type 2 diabetes was 24% lower in those consuming 3 to 4 cups/day of coffee compared to those consuming ≤2 cups/day or none (15). Two additional meta-analyses that included data from more recent prospective cohort studies were published concomitantly and found similar results (16, 17). The data analysis of 28 prospective studies in over 1 million participants with 45,335 incident cases of diabetes reported a 30% decreased risk of type 2 diabetes with coffee intake of 5 cups/day versus 0 cups/day (16). In addition, a 9% reduction in the incidence of type 2 diabetes was estimated for every one cup per day increase in total coffee intake. Likewise, a dose-response analysis of 11 studies found a 6% reduction in type 2 diabetes incidence for every one cup per day increase in decaffeinated coffee intake. Every 140 mg/day (~1 cup/day of coffee) increment in caffeine intake was also associated with an 8% reduction in the risk of type 2 diabetes (16). Although decaffeinated coffee consumption is associated with a more modest decrease in the risk of type 2 diabetes, it is likely that compounds other than caffeine contribute to the reduction in diabetes risk.
Intervention studies
The mechanisms that might contribute to the association between coffee consumption and lower risk of type 2 diabetes in prospective cohort studies are unclear. Bioactive compounds other than caffeine appear to show temporary hypoglycemic effects. For example, the acute ingestion of chlorogenic acid (1 g) and trigonelline (0.5 g) transiently lowered blood glucose concentration shortly after the administration of 75 g of glucose in an oral glucose tolerance test (18). Conversely, short-term clinical trials have found that acute administration of caffeine (3 to 6 mg/kg) impaired glucose tolerance and decreased insulin sensitivity in healthy participants (19). In addition, incremental doses of decaffeinated coffee (1, 2, and 4 servings) failed to lower postprandial blood glucose in the presence of 100 mg of caffeine (20). However, despite the deleterious effect of caffeine on glucose homeostasis, caffeinated coffee consumption may favorably affect other metabolic pathways. In a single-blinded clinical trial, subjects at risk for type 2 diabetes abstained from drinking caffeinated coffee for one month, then consumed 4 cups/day of coffee for another month, and finally consumed 8 cups/day for a third month. Compared to one month of coffee abstinence, the consumption of 8 cups/day of coffee for one month appeared to increase antioxidant capacity and reduce subclinical inflammation, as indicated by changes in plasma markers of oxidative stress and inflammation (21).
Until the relationship between long-term coffee consumption and type 2 diabetes risk is better understood, it is premature to recommend coffee consumption as a means of preventing type 2 diabetes (12, 22).
Parkinson’s disease
Studies in animal models of Parkinson’s disease suggest that caffeine may protect dopaminergic neurons by acting as an adenosine A2A-receptor antagonist in the brain (23). Several large prospective cohort studies have examined coffee and/or caffeine intakes in association with Parkinson’s disease risk. A meta-analysis of nine prospective cohort studies found higher caffeine intake to be associated with significant reductions in Parkinson’s disease risk in both men (-39%) and women (-29%) (24). In another meta-analysis of six case-control studies and seven prospective studies, including 901,764 participants and 3,954 cases, an inverse association between coffee intake and Parkinson’s disease risk — only significant in men — was found to be nonlinear, with no further risk reduction beyond 3 to 4 cups/day of coffee. This meta-analysis also reported significant reductions in Parkinson’s disease risk in men (-43%) and women (-36%) with the highest (700 mg/day) versus lowest (100 mg/day) intake of caffeine (25).
However, while prospective studies have consistently found a lower risk of developing Parkinson’s disease with higher coffee and caffeine intakes in men, such an association has not always been observed in women (26-28). It is hypothesized that estrogen replacement therapy may modify the interaction between caffeine and risk of Parkinson’s disease in postmenopausal women. Indeed, because estrogen and caffeine are metabolized by hepatic cytochrome P450 (CYP) 1A2 in the body, estrogen might compete for CYP1A2 activity and hinder the metabolism of caffeine in estrogen users (29). An analysis of data from more than 77,000 female nurses, followed for 18 years in NHS I, revealed that coffee consumption was inversely associated with Parkinson’s disease risk in women who had never used postmenopausal estrogen (30). However, drinking ≥6 cups of coffee was associated with an increased risk or Parkinson’s disease in women who had used postmenopausal estrogen (30). In a prospective cohort study that included more than 238,000 women, a significant inverse association between coffee consumption and Parkinson’s disease mortality was also observed in women who had never used postmenopausal estrogen, but not in those who had used postmenopausal estrogen (31). Yet, in a recent analysis of the National Institutes of Health (NIH)-AARP Diet and Health Study, which included 303,880 participants and 1,100 cases, the highest versus lowest intake of caffeine was associated with a reduced risk of Parkinson’s disease in postmenopausal women who ever used hormones but not among never users (24).
At present, it remains unclear whether caffeine consumption can prevent Parkinson’s disease, particularly in women taking estrogen. Of note, whether caffeine could help reduce some common symptoms associated with Parkinson’s disease (e.g., sleepiness, freezing of gait) is under investigation (32, 33).
Cognitive decline and dementia
Results from observational studies regarding a possible link between coffee consumption and cognitive disorders are inconclusive. A recent meta-analysis of nine prospective cohort studies in 34,282 older adults reported an 18% reduced risk of cognitive disorders with 1 to 2 cups/day of coffee compared to <1 cup/day (34). Yet, there was no difference in risk of cognitive disorders between daily coffee intakes >3 cups and <1 cup (34). Two other meta-analyses of prospective studies failed to find an association between high versus low intakes of coffee and risk of cognitive disorders (35, 36). No dose-response relationship was reported between coffee intake and risk of cognitive disorders (36).
Whether moderate coffee intake may reduce the risk of cognitive decline and dementia later in life is still not known.
Cirrhosis and liver cancer
Chronic inflammation-inducing liver injury may result in cirrhosis. In cirrhosis, the formation of fibrotic scar tissue leads to the progressive deterioration of liver function and other complications, including liver cancer (primarily hepatocellular carcinoma [HCC]) (37). The most common causes of cirrhosis in developed countries are alcohol abuse and chronic infections with hepatitis B and C viruses. Often associated with metabolic disorders, nonalcoholic fatty liver disease (NAFLD) is a liver condition that can progress to nonalcoholic steatohepatitis (NASH) in about one-third of NAFLD patients, thereby increasing the risk of cirrhosis and HCC (38).
A cross-sectional study in 177 subjects with chronic liver disease (especially chronic hepatitis B or C and NASH) found an association between daily caffeine intake >308 mg — equivalent to >2 cups/day of coffee — and a lower risk of having advanced liver fibrosis (39). Of note, no association was reported with non-coffee sources of caffeine like caffeinated soda or green and black tea (39). Recent cross-sectional studies also suggested a protective association of coffee intake against fibrosis development in patients with hepatitis C (40, 41). Additional studies have suggested that consumption of coffee, but not of caffeine, was inversely associated with the risk of advanced liver fibrosis in patients with NAFLD or NASH (42-44).
A recent meta-analysis of four case-control and three prospective cohort studies reported an inverse association between coffee consumption and risk of cirrhosis (45). In addition, a few large prospective cohort studies found that coffee drinking was associated with reduced mortality from alcoholic cirrhosis (46-49). A 17-year study of more than 51,000 men and women in Norway found that those who consumed ≥2 cups/day of coffee had a risk of cirrhosis-related death that was 40% lower than those who never consumed coffee (49). A 22-year prospective cohort study in 125,580 US adults found that coffee drinking was protective against alcoholic cirrhosis but not nonalcoholic cirrhosis (48). Specifically, the risk of developing alcoholic cirrhosis was 40% lower in those who drank 1 to 3 cups/day of coffee and 80% lower in those who drank ≥4 cups/day (48). Recent data from 63,275 Chinese participants of the Singapore Chinese Health Study showed that consumption of ≥2 cups/day of coffee was associated with a 66% lower risk of death from non-viral hepatitis-related cirrhosis — no such association was found with mortality from cirrhosis due to viral hepatitis (46).
Several case-control and prospective cohort studies have found significant inverse associations between coffee consumption and the risk of HCC (reviewed in 50). In a recent 18-year prospective cohort study of 162,022 US adults — comprising Japanese Americans, Caucasians, Mexican Americans, African Americans, and Native Hawaiians — consumption of coffee, but not decaffeinated coffee, was inversely associated with risk of developing HCC (51). Specifically, drinking 2 to 3 cups/day of coffee was associated with a 38% reduced risk of HCC compared to no coffee drinking (51). In addition, the risk of chronic liver disease-related mortality was 71% lower in individuals consuming ≥4 cups/day of regular coffee and 46% lower in those consuming ≥2 cups/day of decaffeinated coffee compared to non-consumers (51). A pooled analysis of this study with 10 other prospective cohort studies found an overall 46% lower risk of liver cancer with coffee consumption (see also Cancer) (52).
Cancer
Numerous observational studies have examined the relationship between coffee consumption and cancer risk (53, 54). Results from recently published meta-analyses of observational studies are reported in Table 1. In addition, a recent meta-analysis of prospective cohort studies by Wang et al. (54) investigated the relationships between the highest versus lowest categories of coffee intake and the risk of most cancer types. Coffee consumption was found to be associated with reduced risks of oral/pharyngeal cancer (6 studies; -31%), colon cancer (10 studies; -13%), liver cancer (9 studies; -54%), prostate cancer (14 studies; -11%), endometrial cancer (12 studies; -27%), and melanoma (6 studies; -11%) (54). Overall, these results are in agreement with those from other pooled analyses of prospective studies presented in Table 1. However, unlike the results reported by Wang et al. (54), coffee consumption has been inversely associated with the risk of colon cancer in case-control studies, but not in prospective cohort studies (see Table 1). Also, in the case of prostate cancer, several meta-analyses suggested a reduced risk with increased coffee intake in prospective but not case-control studies (54-56). However, whether this association exists only in certain study populations or at specific cancer stages remains unclear (57, 58). There seems to be little evidence of associations between coffee consumption and breast cancer, esophageal cancer, glioma, laryngeal cancer, pancreatic cancer, rectal cancer, stomach cancer, and thyroid cancer (Table 1) (54).
| Type of Cancer | Type of Observational Studies | Relative Risk [RR] or Odds Ratio [OR]# (95% Confidence Interval) | Relative Risk [RR] or Odds Ratio [OR] in Subgroup Analyses (e.g., by study types) | References |
|---|---|---|---|---|
| Breast cancer | 10 case-control and 16 prospective cohort studies |
RR: 0.96 (0.93-1.00) |
RR: 0.93 (0.86-1.00) for case-control studies RR: 0.98 (0.93-1.02) for cohort studies RR: 0.81 (0.67-0.97) for ER-negative cancer RR: 1.01 (0.93-1.09) for ER-positive cancer |
Li et al. (2013; 59) |
| 20 case-control and 17 prospective cohort studies | RR: 0.97 (0.93-1.00) |
RR: 0.94 (0.89-1.00) for case-control studies |
Jiang et al. (2013; 60) | |
|
Colorectal cancer |
25 case-control studies | OR: 0.85 (0.75-0.97) |
OR: 0.68 (0.57-0.81) for colon cancer OR: 0.95 (0.79-1.15) for rectal cancer |
Li et al. (2013; 61) |
| 16 prospective cohort studies | RR: 0.94 (0.88-1.01) |
RR: 0.93 (0.86-1.01) for colon cancer RR: 0.98 (0.88-1.09) for rectal cancer |
Li et al. (2013; 61) | |
| 7 case-control and 5 prospective cohort studies | - |
RR: 0.78 (0.65-0.95) for case-control studies RR: 0.82 (0.65-1.02) for cohort studies |
Akter et al. (2016; 62) | |
| 19 prospective cohort studies | RR: 0.98 (0.90-1.06) |
RR: 0.92 (0.83-1.02) for colon cancer RR: 1.06 (0.95-1.19) for rectal cancer |
Gan et al. (2017; 63) | |
|
Endometrial cancer |
10 case-control and 6 prospective cohort studies | RR: 0.71 (0.62-0.81) |
RR: 0.69 (0.55-0.87) for case-control studies RR: 0.70 (0.61-0.80) for cohort studies |
Je et al. (2012; 64) |
| 13 prospective cohort studies | RR: 0.80 (0.74-0.86) |
RR: 0.66 (0.52-0.84) for caffeinated coffee RR: 0.77 (0.63-0.94) for decaffeinated coffee |
Zhou et al. (2015; 65) | |
| Esophageal cancer | 10 case-control and 4 prospective cohort studies | RR: 0.88 (0.76-1.01) | Zheng et al. (2013; 66) | |
| Glioma | 2 case-control and 4 prospective cohort studies | RR: 1.01 (0.83-1.22) | Malerba et al. (2013; 67) | |
| Laryngeal cancer | 5 case-control studies and 1 prospective cohort study | RR: 1.47 (1.03-2.11) | Chen et al. (2014; 68) | |
| 7 case-control studies and 1 prospective cohort study | RR: 1.22 (0.92-1.62) | Ouyang et al. (2014; 69) | ||
| Liver cancer | 10 prospective cohort studies | RR: 0.55 (0.44-0.67) |
RR: 0.57 (0.42-0.79) for women RR: 0.58 (0.40-0.83) for men |
Yu et al. (2016; 52) |
| Lung cancer | 12 case-control and 5 prospective cohort studies | OR: 1.31 (1.11-1.55) |
OR: 1.36 (1.10-1.69) for hospital-based case-control studies OR: 0.99 (0.77-1.28) for community-based case-control studies OR: 1.59 (1.26-2.00) for cohort studies OR: 1.41 (1.21-1.63) for men OR:1.16 (0.86-1.56) for women OR: 1.24 (1.00-1.54) for smokers OR: 0.85 (0.64-1.11) for non-smokers |
Xie et al. (2016; 70) |
| Melanoma | 4 case-control and 8 prospective cohort studies | RR: 0.80 (0.69-0.93) |
RR: 0.85 (0.71-1.01) for caffeinated coffee RR: 0.92 (0.81-1.05) for decaffeinated coffee |
Wang et al. (2016; 71) |
| Oral cancer | 9 case-control and 3 prospective cohort studies | RR: 0.69 (0.54-0.89) |
RR: 0.65 (0.46-0.92) for case-control studies RR: 0.81 (0.62-1.05) for cohort studies RR: 0.81 (0.58-1.13) for studies in the US RR: 0.57 (0.38-0.86) for studies in Europe |
Zhang et al. (2015; 72) |
|
Pancreatic cancer |
22 case-control and 15 prospective cohort studies | RR: 1.08 (0.94-1.25) |
RR: 1.10 (0.92-1.31) for case-control studies RR: 1.04 (0.80-1.36) for cohort studies |
Turati et al. (2012; 73) |
| 20 prospective cohort studies | RR: 0.88 (0.64-1.12) | Ran et al. (2016; 74) | ||
| 21 prospective cohort studies | RR: 0.99 (0.81-1.21) | Nie et al. (2016; 75) | ||
| Prostate cancer | 12 case-control and 12 prospective cohort studies | RR: 0.94 (0.85-1.05) |
RR: 1.36 (1.06-1.75) for studies in the US RR: 1.08 (0.80-1.45) for studies in Europe RR: 0.92 (0.66-1.28) for studies in Asia RR: 0.61 (0.42-0.90) for fatal cancer RR: 0.70 (0.52-0.94) for high-grade tumors RR: 1.07 (0.89-1.29) for low-grade tumors |
Zhong et al. (2014; 58) |
| 12 case-control and 9 prospective cohort studies | RR: 0.91 (0.86-0.97) |
RR: 0.91 (0.95-1.26) for case-control studies RR: 0.89 (0.84-0.95) for cohort studies |
Lu et al. (2014; 56) | |
| 13 prospective cohort studies | RR: 0.90 (0.85-0.95) |
RR: 0.93 (0.87-1.00) for studies in the US RR: 0.83 (0.75-0.92) for studies in Europe RR: 0.82 (0.51-1.31) for studies in Asia RR: 0.76 (0.55-1.06) for fatal cancer RR: 0.82 (0.61-1.10) for advanced tumors RR: 0.89 (0.83-0.96) for non-advanced tumors |
Liu et al. (2015; 57) | |
|
Stomach cancer |
9 prospective cohort studies | RR: 1.18 (0.90-1.55) | Zeng et al. (2015; 76) | |
| 13 prospective cohort studies | RR: 1.13 (0.94-1.35) |
RR: 1.36 (1.06-1.75) for studies in the US RR: 1.08 (0.80-1.45) for studies in Europe RR: 0.92 (0.66-1.28) for studies in Asia |
Li et al. (2015; 77) | |
| 13 case-control and 9 prospective cohort studies | RR: 0.96 (0.82-1.12) |
RR: 0.85 (0.77-0.95) for case-control studies RR: 1.12 (0.94-1.33) for cohort studies |
Xie et al. (2016; 78) | |
| Thyroid cancer | 5 case-control and 2 prospective cohort studies | OR: 0.88 (0.71-1.07) | Han et al. (2017; 79) | |
| #Odds ratio or relative risk of cancer for the highest vs. lowest categories of coffee intake. | ||||
Finally, there is some evidence suggesting a potential increase in the risk of lung cancer with the highest versus lowest levels of coffee intakes. Yet, cigarette smoking has a major confounding effect on this association (see Health risks associated with coffee consumption) (54, 70).
Mortality
Three large US prospective cohort studies, namely NHS I (74,890 women), NHS II (93,054 women), and HPFS (40,557 men), examined whether coffee drinking was associated with all-cause, cardiovascular disease-related, or cancer-related mortality (80). Compared to no coffee consumption, the consumption of coffee, whether caffeinated or decaffeinated, up to 5 cups/day, was inversely associated with all-cause mortality. There was no difference in risk of death between non-consumers and consumers of >5 cups/day of coffee. Coffee consumption was also found to be inversely associated with mortality related to cardiovascular disease, neurological disease, and suicide (80). Other large cohort studies have reported habitual consumption of any coffee being inversely associated with all-cause and cardiovascular disease-related mortality, but generally not with cancer-related mortality (81-86). Moreover, the associations have not always been consistent among women and men, especially regarding cancer-related mortality (85, 86). A dose-response meta-analysis of 21 prospective studies found a nonlinear inverse association between coffee consumption and all-cause and cardiovascular disease-related mortality (87). Consumption of only 1 cup/day of coffee was significantly associated with lower risk of all-cause (-8%) and cardiovascular disease-related mortality (-11%). The largest risk reductions for all-cause (-16%) and cardiovascular disease-related mortality (-21%) were found to be associated with the consumption of 4 cups/day and 3 cups/day of coffee, respectively (87).
Safety
Adverse effects
Most adverse effects attributed to coffee consumption are related to caffeine. In healthy adults, daily caffeine consumption ≤400 mg — corresponding to 6.5 mg/kg body weight/day for a 70-kg person — is usually not associated with adverse effects (88). Caffeine intakes of less than 300 mg/day in women of childbearing age (equivalent to 4.3 mg/kg body weight/day for a 70-kg woman) and less than 2.5 mg/kg body weight/day in children are unlikely to cause adverse effects (88).
Adverse reactions to caffeine may include tachycardia (rapid heart rate), palpitations, insomnia, restlessness, nervousness, tremor, headache, abdominal pain, nausea, vomiting, diarrhea, and diuresis (increased urination) (89). Very high caffeine intakes, not usually from coffee, may induce hypokalemia (abnormally low serum potassium) (90). Sudden cessation of caffeine consumption after long-term use may result in caffeine withdrawal symptoms (91). Gradual withdrawal from caffeine appears less likely to result in withdrawal symptoms than abrupt withdrawal (92). Commonly reported caffeine withdrawal symptoms include headache, fatigue, drowsiness, irritability, difficulty concentrating, and depressed mood.
Potential health risks associated with regular coffee consumption
Cardiovascular disease
Serum lipids: An early meta-analysis of nine randomized controlled trials found that the consumption of unfiltered, boiled coffee dose-dependently increased serum total and LDL-cholesterol concentrations, while the consumption of filtered coffee resulted in very little change (93). A more recent meta-analysis of 12 randomized controlled trials reported that the consumption of coffee increased serum total cholesterol by 8.1 mg/dL, LDL-cholesterol by 5.1 mg/dL, and triglycerides by 12.6 mg/dL (94). The consumption of filtered coffee raised total cholesterol by only 3.6 mg/dL compared to an increase of 12.9 mg/dL with unfiltered coffee consumption. Unlike filtered coffee, consumption of unfiltered coffee significantly increased LDL-cholesterol and triglycerides by 11.9 mg/dL and 18.8 mg/dL, respectively (94). The cholesterol-raising factors in unfiltered coffee have been identified as cafestol and kahweol, two diterpenes that are largely removed from coffee by paper filters (see Diterpenes) (10).
Homocysteine: An elevated plasma total homocysteine concentration has been associated with increased risk of cardiovascular disease, including coronary heart disease, stroke, and peripheral vascular disease, but the relationship may not be causal (95). Higher coffee intakes have been associated with increased plasma homocysteine concentrations in cross-sectional studies conducted in Europe, Scandinavia, and the US (96-100). Controlled clinical trials have confirmed the homocysteine-raising effect of coffee at intakes of about 4 cups/day (101-103).
Hypertension: Hypertension is a well-recognized risk factor for cardiovascular disease. Two meta-analyses of randomized controlled trials showed that high intakes of coffee (3 to 5 cups/day) for <85 days significantly increased systolic/diastolic blood pressure by 1.2/0.5 mm Hg (104) or 2.4/1.2 mm Hg (105). Although the increases in blood pressure seem modest by individual standards, it has been estimated that an average systolic blood pressure reduction of 2 mm Hg in a population may result in 10% lower mortality from stroke and 7% lower mortality from coronary heart disease (106). However, a more recent pooled analysis excluding the trials that used decaffeinated coffee in control groups — and thus assessed the effect of caffeine rather than that of coffee — found no significant changes in systolic blood pressure and diastolic blood pressure with high coffee intakes (107). Additionally, two meta-analyses combined the data from observational studies that examined prospectively the association between habitual coffee consumption and risk of hypertension (107, 108). One meta-analysis of six prospective cohort studies included 172,567 non-hypertensive participants (one study enrolled pre-hypertensive subjects) of which 37,135 cases of incident hypertension were reported over follow-up periods spanning from 6.4 years to 33 years (108). Compared to coffee intakes of <1 cup/day, intakes of 1 to 3 cups/day were found to be associated with a 9% increased risk of hypertension. However, no such association could be observed for coffee intakes >3 cups/day (108). Another meta-analysis of four prospective cohort studies found no association between coffee consumption and risk of hypertension (107). Also, a recent analysis of data from 29,985 postmenopausal women followed for nearly four years in the Women’s Health Initiative Observational Study found no increased risk of hypertension with intakes of caffeinated coffee, decaffeinated coffee, or caffeine (109).
While there is little evidence of an association between long-term coffee consumption and risk of hypertension, the available evidence from trials suggests that consumption of caffeine modestly raises systolic blood pressure. Whether this may result in increased risk of stroke and coronary heart disease in the population, particularly in those with hypertension, is still uncertain. Yet, to date, regular coffee consumption in hypertensive subjects has not been associated with an increased risk of cardiovascular disease (110).
Coronary heart disease: A meta-analysis of 20 prospective cohort studies, including 407,806 participants and 15,599 incident coronary heart disease (CHD) cases, found no significant association between coffee consumption and CHD risk (111). Yet, a more recent meta-analysis of 22 prospective cohort studies reported a modest reduction in CHD risk with moderate (3-5 cups/day) — but not high (≥6 cups/day) — coffee intakes compared to low intakes (<1 cup/day) (112).
In addition, cross-sectional studies have provided little evidence that the formation of atherosclerotic plaques, an early event in the development of CHD, is more prevalent in regular coffee drinkers than in non-drinkers (113-116). Coffee consumption has not been linked to the development of atherosclerosis in two prospective cohort studies. In the Coronary Artery Risk Development in Young Adults (CARDIA) study that followed approximately 4,000 young US adults for 15 to 20 years, there was no evidence of an association between regular coffee intake and progression of coronary artery calcification, a measure of subclinical atherosclerosis (117). Moreover, in the Multi-Ethnic Study of Atherosclerosis (MESA) that followed about 6,500 US adults over a median period of 11.1 years, occasional but not regular drinking of coffee was associated with a 28% increased risk of cardiovascular disease compared with no drinking (114). This study found no association between coffee intake and progression of coronary artery calcification (114).
Cardiac arrhythmias: Early clinical trials found coffee or caffeine intake equivalent to 5 to 6 cups/day did not increas the frequency or severity of cardiac arrhythmias in healthy people or in people with coronary heart disease (118, 119). A meta-analysis of six prospective cohort studies in 214,316 participants found no association between coffee consumption and risk of atrial fibrillation, the most common type of cardiac arrhythmias. In addition, a recent meta-analysis of 11 short-term intervention studies (single dose to two-week trials) found that caffeine consumption did not increase the occurrence of ventricular arrhythmias (120). Finally, two meta-analyses of observational studies found no evidence to suggest that caffeine consumption was associated with an increased risk of atrial fibrillation and even reported a modest reduction in risk with moderate intakes (121, 122).
Thus, consumption of coffee or caffeine at usual intakes does not appear to increase the risk of cardiac arrhythmias. The current evidence does not support clinical recommendations that discourage moderate consumption of coffee in patients at risk or with suspicions of cardiac arrhythmia (120, 123).
Stroke: A 2011 meta-analysis that included eight prospective cohort studies — all following participants who were free of cardiovascular disease and diabetes mellitus at baseline — found that consumption of 3 to 4 cups/day of coffee was associated with an 18% lower risk of stroke compared with no consumption and that higher intakes were not associated with an increased risk (124). Since this meta-analysis, a few large prospective studies have reported mixed results on the association between coffee consumption and stroke incidence or stroke-related mortality. Results from 42,659 participants in the German cohort of the European Prospective Investigation in Cancer and Nutrition (EPIC-Germany) reported no association between coffee consumption and stroke incidence over an 8.9 year-period (125). Compared to no intake, consumption of ≥3-6 cups/week of coffee was associated with a reduced risk of stroke in a prospective study of 82,369 Japanese participants (126). Also, the consumption of 4 to 5 cups/day of coffee was associated with a reduced risk of stroke-related mortality among men, but not women, followed in the large prospective NIH-AARP Diet and Health Study (81).
Lung cancer
Several observational studies have examined the relationship between coffee intake and lung cancer risk in humans. A recent meta-analysis of 12 case-control and 5 prospective cohort studies, including a total of 12,276 cases and 102,516 controls, found an overall 31% increased risk of lung cancer with the highest versus lowest levels of coffee intake (see Table 1). Subgroup analyses outlined a significant increase in lung cancer risk with coffee intake in hospital-based case-control (+36%) and prospective cohort studies (+59%), in studies conducted in American (+34%) and Asian (+49%) populations, in men (+41%), and in smokers (+24%); conversely, no significant association between coffee intake and lung cancer risk was found in community-based case-control studies, in studies conducted in European populations, in women, and in non-smokers (70). Another meta-analysis of 13 case-control and 8 prospective cohort studies, including 19,892 cases and 623,645 controls, found a 9% increased risk of lung cancer in coffee drinkers compared to non-drinkers (127). However, a pooled analysis restricted to the 16 (out of 21) studies that adjusted for smoking found no significant association between coffee intake and risk of lung cancer [RR: 1.03 (0.95-1.12)].
Cigarette smoking is a major confounding factor in the association between coffee consumption and lung cancer risk, and the evidence suggests that coffee intake is unlikely to be a risk factor for lung cancer. Of note, residual confounding by smoking remains a concern when a slight increase in lung cancer risk is still observed in studies even after adjustment for tobacco smoking (127).
Adverse pregnancy outcomes
It has been suggested that in utero exposure to caffeine through maternal coffee consumption might have adverse effects on the embryo/fetus during pregnancy and the offspring.
Miscarriage: The results of observational studies that have examined the relationship between maternal coffee or caffeine intake and the risk of miscarriage (spontaneous abortion) have been conflicting. While some prospective cohort studies have observed significant associations between high caffeine intakes, particularly from coffee, and the risk of spontaneous abortion (128-132), other studies have not (133-136). The most recent meta-analysis of 14 prospective cohort studies in 130,456 pregnant women and 3,429 cases of miscarriage found risk of miscarriage increased by 40% and 72% with maternal caffeine intakes of 350 to 699 mg/day and ≥700 mg/day during pregnancy, respectively (137). No significant associations were found for daily doses of caffeine less than 350 mg. A dose-response analysis found a 7% increase in the risk of miscarriage per 100 mg/day-increment in caffeine intake during pregnancy (137). Of note, one prospective cohort study that assessed caffeine intake by measuring serum concentrations of paraxanthine, a caffeine metabolite, found that the risk of spontaneous abortion was only elevated in women with paraxanthine concentrations that suggested caffeine intakes of ≥600 mg/day (138).
It has been proposed that an association between caffeine consumption and the risk of spontaneous abortion could be explained by the relationship between nausea and fetal viability (139). Nausea is more common in women with viable pregnancies than nonviable pregnancies, such that women with viable pregnancies may be more likely avoid or limit caffeine consumption due to nausea (140). However, at least one study found that the significant increase in risk of spontaneous abortion observed in women with caffeine intakes >300 mg/day was independent of nausea in pregnancy (141). Additionally, two other studies found that caffeine consumption was associated with an increased risk of spontaneous abortion in women who experienced nausea or aversion to coffee during pregnancy (131, 142). Nonetheless, this does not exclude the possibility of reverse causality, when the loss of fetal viability results in reduction of pregnancy symptoms, like nausea and aversion to coffee, and may be followed by an increase in coffee intake (137).
Of note, consumption of <400 mg/day of caffeine or <4 cups/day of caffeinated coffee by women prior to pregnancy has not been linked to the risk of miscarriage in an analysis of a large prospective study (NHS I) that followed 11,072 women and a total of 15,590 pregnancies (143).
Although the topic remains unsettled, the American College of Obstetricians and Gynecologists recommends that women limit their daily caffeine intake to <200 mg during pregnancy (144).
Intrauterine growth restriction and low birth weight: Observational studies examining the effects of maternal caffeine and coffee consumption on fetal growth have assessed intrauterine growth restriction (IUGR; also known as small-for-gestational age; defined as fetal weight <10th percentile for gestational age), and/or incidence of low birth weight (defined as weight at birth <2,500 g [5.5 pounds]).
A recent meta-analysis of eight prospective cohort and four case-control studies reported a 38% increased risk of low birth weight with the highest versus lowest intakes of caffeine during pregnancy (145). This risk appeared to increase linearly with incremental doses of caffeine (145, 146). A dose-response meta-analysis of six prospective cohort and five case-control studies found a 7% increased risk of low-birth-weight infants per 100 mg of caffeine consumed daily during pregnancy (146). Likewise, a 100 mg-increment in maternal caffeine intakes has been associated with a 10% increased risk of small-for-gestational age infants in the dose-response analysis of 10 prospective cohort and 5 case-control studies (146).
At present, only one study has examined the impact of limiting caffeine intake during pregnancy on birth weight. In a double-blind, intervention trial that randomized 1,197 regular coffee drinkers (≥3 cups/day of coffee) to drink decaffeinated (median caffeine intake of 117 mg/day) or caffeinated coffee (median caffeine intake of 317 mg/day) throughout the second half of their pregnancy, no differences in length of gestation or infant birth weight were found between the two groups (147).
Although the relationship between maternal caffeine consumption and fetal growth requires further clarification, it appears that even moderate caffeine intakes might adversely affect fetal growth in non-smoking women (145, 146, 148). Limiting caffeine intake to ≤100 mg/day (≤1 cup/day of coffee) during pregnancy may be recommended to avoid any adverse effect, assuming that the associations of caffeine intake with the risk of IUGR and low birth weight are causal (145, 146).
Birth defects: Potential relationships between coffee consumption during pregnancy and congenital birth defects have been investigated in the US population-based National Birth Defects Prevention Study (NBDPS), an ongoing multi-site case-control study. In an analysis that included mothers of 1,531 infants with cleft lips (with or without cleft palates), 813 infants with cleft palates only, and 5,711 control infants, no association was found between the highest versus lowest intakes of coffee and caffeine and the risk of orofacial clefts (149). Another analysis, including mothers of 3,346 cases and 6,642 control infants, suggested an increased risk of anotia/microtia and craniosynostosis with the consumption of coffee or caffeine. However, no dose-response could be detected (150). Further, an analysis of the NBDPS in mothers of 844 infants with limb deficiencies and 8,069 control infants found no increased risk associated with coffee and/or caffeine intake during pregnancy (151). There was no association between maternal coffee or caffeine intake during pregnancy and risk of congenital talipes equinovarus (known as "clubfoot") in another US population-based case-control study of mothers of 646 infants with isolated clubfoot and 2,037 control infants (152). Finally, a recent meta-analysis that combined data from one prospective cohort study and six case-control studies found no association between maternal coffee consumption during pregnancy and risk of neural tube defects (153).
At present, there is no convincing evidence that maternal consumption of 3 cups/day of coffee or 300 mg/day of caffeine during pregnancy increases the risk of congenital malformations in humans.
Childhood acute leukemia: The etiology of acute lymphoblastic leukemia (ALL) and acute myeloblastic leukemia (AML), which primarily affect children, is unclear. It has been suggested that exposure to caffeine during pregnancy might have long-lasting adverse effects on the health of the offspring. A meta-analysis of seven case-control studies that examined maternal coffee consumption during pregnancy in relation to the incidence of childhood acute leukemia found coffee consumption was associated with increased risks of overall acute leukemia (+72%), ALL (+65%), and AML (+58%) (154). Another meta-analysis of eight case-control studies found an increased risk of ALL (+43%), but not AML, with the highest versus lowest intakes of coffee during pregnancy (155). High versus low consumption of other sources of caffeine during pregnancy (tea, cola beverages) and childhood (cola beverages) were not found to be associated with childhood acute leukemia (155).
The evidence of a positive association between maternal coffee intake and childhood acute leukemia is currently limited to case-control studies. Case-control studies usually include more cancer cases than prospective cohort studies, but they are subject to recall bias with respect to coffee consumption and selection bias with respect to the control group (156). Further studies with a prospective design are needed to confirm the possible link between coffee intake during pregnancy and childhood acute leukemia.
Lactation
The American Academy of Pediatrics categorizes caffeine as a maternal medication that is usually compatible with breast-feeding (157). Although high maternal caffeine intakes have been reported to cause irritability and poor sleeping patterns in infants, no adverse effects have been reported with moderate maternal intake of caffeinated beverages equivalent to 2 to 3 cups of coffee daily.
Nutrient interactions
Calcium, osteoporosis, and risk of fracture
Osteoporosis is a multifactorial bone disorder that compromises bone mass and strength and increases the risk of fracture. The results of early controlled studies in humans indicated that coffee and caffeine consumption decreased the efficiency of calcium absorption resulting in a loss of about 4 to 6 mg of calcium per cup of coffee (158, 159). However, there is little evidence to suggest detrimental effects of coffee on bone health in populations with adequate calcium intakes (160). To date, results from observational studies that examined associations between coffee intakes and measures of bone mineral density (BMD) loss — generally used to diagnose osteoporosis — have been mixed (161-164). Further, two meta-analyses of observational studies reported no significant associations between coffee intake and risk of hip fracture (165, 166). A third meta-analysis of six case-control and nine prospective cohort studies found no overall association between coffee intake and total fracture (167). Yet, a gender subgroup analysis of eight studies showed a 14% increased risk of fracture in women — but not in men — with the highest versus lowest intakes of coffee. Another subgroup analysis of six studies found a 35% increased risk of osteoporotic fracture in participants with the highest versus lowest intakes of coffee (167).
Current evidence is scarce to suggest that coffee consumption could increase the risk of bone loss and fracture. Limiting coffee consumption to ≤3 cups/day while ensuring adequate calcium and vitamin D intakes should prevent any potential adverse effects on calcium absorption and bone health.
Nonheme iron
Phenolic compounds in coffee can bind nonheme iron and inhibit its intestinal absorption (168). Drinking 150 to 250 mL of coffee with a test meal has been found to inhibit iron absorption by 24%-73% (169, 170). To maximize iron absorption from a meal or supplements, people with poor iron status should not consume coffee at the same time.
Drug interactions
Habitual caffeine consumption increases hepatic cytochrome P450 (CYP) 1A2 activity, which has implications for the metabolism for a number of medications (171). Conversely, drugs that inhibit the activity of CYP1A2 interfere with the metabolism and elimination of caffeine, thereby increasing the risk of adverse effects (172).
Drugs that alter caffeine metabolism
The following medications may impair the hepatic metabolism of caffeine, delaying its excretion and potentially increasing the risk of caffeine-related side effects: cimetidine (Tagamet), disulfiram (Antabuse), estrogens, fluconazole (Diflucan), fluvoxamine (Luvox), mexiletine (Mexitil), quinolone class antibiotics (Cipro, Avelox), riluzole (Rilutek), terbinafine (Lamisil), and Verapamil (Calan) (173). Concomitant use of ephedrine and caffeine can lead to life-threatening adverse effects, including heart attack, stroke, seizures, and death (173). Use of the drug phenytoin (Dilantin) or cigarette smoking increases the hepatic metabolism of caffeine, resulting in increased elimination and decreased plasma caffeine concentrations (89).
Caffeine effects on other drugs
Caffeine and other methylxanthines may enhance the effects and side effects of β-adrenergic stimulating agents, such as epinephrine and albuterol (89, 171). Caffeine doses of 400 to 1,000 mg may inhibit the hepatic metabolism of the antipsychotic medication, clozapine (Clozaril), potentially elevating serum clozapine concentration and increasing the risk of toxicity. Those taking levothyroxine are advised to avoid drinking coffee at the same time they take their medication because coffee may reduce the absorption of levothyroxine in some patients. Caffeine consumption can decrease the elimination of theophylline, potentially increasing serum theophylline levels. Caffeine has been also found to decrease the systemic elimination of acetaminophen (i.e., paracetamol) and to increase the bioavailability of aspirin, which may partially explain its efficacy in enhancing their analgesic effects. This is important because many pain-relievers on the market today combine caffeine with aspirin and/or acetaminophen. Further, caffeine may decrease the bioavailability of lithium and alendronate (Fosamax) by enhancing their elimination (173).
Authors and Reviewers
Originally written in 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in September 2008 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in April 2017 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in June 2017 by:
Rob van Dam, Ph.D.
Adjunct Associate Professor of Nutrition and Epidemiology
Harvard T.H. Chan School of Public Health
Copyright 2005-2024 Linus Pauling Institute
References
1. Godos J, Pluchinotta FR, Marventano S, et al. Coffee components and cardiovascular risk: beneficial and detrimental effects. Int J Food Sci Nutr. 2014;65(8):925-936. (PubMed)
2. Spiller MA. The chemical components of coffee. In: Spiller GA, ed. Caffeine. Boca Raton: CRC Press; 1998:97-161.
3. Cagliani LR, Pellegrino G, Giugno G, Consonni R. Quantification of Coffea arabica and Coffea canephora var. robusta in roasted and ground coffee blends. Talanta. 2013;106:169-173. (PubMed)
4. Caprioli G, Cortese M, Sagratini G, Vittori S. The influence of different types of preparation (espresso and brew) on coffee aroma and main bioactive constituents. Int J Food Sci Nutr. 2015;66(5):505-513. (PubMed)
5. Ozgen M, Reese RN, Tulio AZ, Jr., Scheerens JC, Miller AR. Modified 2,2-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (abts) method to measure antioxidant capacity of Selected small fruits and comparison to ferric reducing antioxidant power (FRAP) and 2,2'-diphenyl-1-picrylhydrazyl (DPPH) methods. J Agric Food Chem. 2006;54(4):1151-1157. (PubMed)
6. Olthof MR, Hollman PC, Buijsman MN, van Amelsvoort JM, Katan MB. Chlorogenic acid, quercetin-3-rutinoside and black tea phenols are extensively metabolized in humans. J Nutr. 2003;133(6):1806-1814. (PubMed)
7. Piazzon A, Vrhovsek U, Masuero D, Mattivi F, Mandoj F, Nardini M. Antioxidant activity of phenolic acids and their metabolites: synthesis and antioxidant properties of the sulfate derivatives of ferulic and caffeic acids and of the acyl glucuronide of ferulic acid. J Agric Food Chem. 2012;60(50):12312-12323. (PubMed)
8. Niseteo T, Komes D, Belscak-Cvitanovic A, Horzic D, Budec M. Bioactive composition and antioxidant potential of different commonly consumed coffee brews affected by their preparation technique and milk addition. Food Chem. 2012;134(4):1870-1877. (PubMed)
9. McCusker RR, Goldberger BA, Cone EJ. Caffeine content of specialty coffees. J Anal Toxicol. 2003;27(7):520-522. (PubMed)
10. Urgert R, Katan MB. The cholesterol-raising factor from coffee beans. Annu Rev Nutr. 1997;17:305-324. (PubMed)
11. Naidoo N, Chen C, Rebello SA, et al. Cholesterol-raising diterpenes in types of coffee commonly consumed in Singapore, Indonesia and India and associations with blood lipids: a survey and cross sectional study. Nutr J. 2011;10:48. (PubMed)
12. Salazar-Martinez E, Willett WC, Ascherio A, et al. Coffee consumption and risk for type 2 diabetes mellitus. Ann Intern Med. 2004;140(1):1-8. (PubMed)
13. van Dam RM, Willett WC, Manson JE, Hu FB. Coffee, caffeine, and risk of type 2 diabetes: a prospective cohort study in younger and middle-aged U.S. women. Diabetes Care. 2006;29(2):398-403. (PubMed)
14. Bhupathiraju SN, Pan A, Manson JE, Willett WC, van Dam RM, Hu FB. Changes in coffee intake and subsequent risk of type 2 diabetes: three large cohorts of US men and women. Diabetologia. 2014;57(7):1346-1354. (PubMed)
15. Huxley R, Lee CM, Barzi F, et al. Coffee, decaffeinated coffee, and tea consumption in relation to incident type 2 diabetes mellitus: a systematic review with meta-analysis. Arch Intern Med. 2009;169(22):2053-2063. (PubMed)
16. Ding M, Bhupathiraju SN, Chen M, van Dam RM, Hu FB. Caffeinated and decaffeinated coffee consumption and risk of type 2 diabetes: a systematic review and a dose-response meta-analysis. Diabetes Care. 2014;37(2):569-586. (PubMed)
17. Jiang X, Zhang D, Jiang W. Coffee and caffeine intake and incidence of type 2 diabetes mellitus: a meta-analysis of prospective studies. Eur J Nutr. 2014;53(1):25-38. (PubMed)
18. van Dijk AE, Olthof MR, Meeuse JC, Seebus E, Heine RJ, van Dam RM. Acute effects of decaffeinated coffee and the major coffee components chlorogenic acid and trigonelline on glucose tolerance. Diabetes Care. 2009;32(6):1023-1025. (PubMed)
19. Shi X, Xue W, Liang S, Zhao J, Zhang X. Acute caffeine ingestion reduces insulin sensitivity in healthy subjects: a systematic review and meta-analysis. Nutr J. 2016;15(1):103. (PubMed)
20. Robertson TM, Clifford MN, Penson S, Chope G, Robertson MD. A single serving of caffeinated coffee impairs postprandial glucose metabolism in overweight men. Br J Nutr. 2015;114(8):1218-1225. (PubMed)
21. Kempf K, Herder C, Erlund I, et al. Effects of coffee consumption on subclinical inflammation and other risk factors for type 2 diabetes: a clinical trial. Am J Clin Nutr. 2010;91(4):950-957. (PubMed)
22. van Dam RM, Hu FB. Coffee consumption and risk of type 2 diabetes: a systematic review. JAMA. 2005;294(1):97-104. (PubMed)
23. Kolahdouzan M, Hamadeh MJ. The neuroprotective effects of caffeine in neurodegenerative diseases. CNS Neurosci Ther. 2017;23(4):272-290. (PubMed)
24. Liu R, Guo X, Park Y, et al. Caffeine intake, smoking, and risk of Parkinson disease in men and women. Am J Epidemiol. 2012;175(11):1200-1207. (PubMed)
25. Qi H, Li S. Dose-response meta-analysis on coffee, tea and caffeine consumption with risk of Parkinson's disease. Geriatr Gerontol Int. 2014;14(2):430-439. (PubMed)
26. Ascherio A, Zhang SM, Hernan MA, et al. Prospective study of caffeine consumption and risk of Parkinson's disease in men and women. Ann Neurol. 2001;50(1):56-63. (PubMed)
27. Hu G, Bidel S, Jousilahti P, Antikainen R, Tuomilehto J. Coffee and tea consumption and the risk of Parkinson's disease. Mov Disord. 2007;22(15):2242-2248. (PubMed)
28. Palacios N, Gao X, McCullough ML, et al. Caffeine and risk of Parkinson's disease in a large cohort of men and women. Mov Disord. 2012;27(10):1276-1282. (PubMed)
29. Pollock BG, Wylie M, Stack JA, et al. Inhibition of caffeine metabolism by estrogen replacement therapy in postmenopausal women. J Clin Pharmacol. 1999;39(9):936-940. (PubMed)
30. Ascherio A, Chen H, Schwarzschild MA, Zhang SM, Colditz GA, Speizer FE. Caffeine, postmenopausal estrogen, and risk of Parkinson's disease. Neurology. 2003;60(5):790-795. (PubMed)
31. Ascherio A, Weisskopf MG, O'Reilly EJ, et al. Coffee consumption, gender, and Parkinson's disease mortality in the cancer prevention study II cohort: the modifying effects of estrogen. Am J Epidemiol. 2004;160(10):977-984. (PubMed)
32. Kitagawa M, Houzen H, Tashiro K. Effects of caffeine on the freezing of gait in Parkinson's disease. Mov Disord. 2007;22(5):710-712. (PubMed)
33. Postuma RB, Lang AE, Munhoz RP, et al. Caffeine for treatment of Parkinson disease: a randomized controlled trial. Neurology. 2012;79(7):651-658. (PubMed)
34. Wu L, Sun D, He Y. Coffee intake and the incident risk of cognitive disorders: A dose-response meta-analysis of nine prospective cohort studies. Clin Nutr. 2016;36(3):730-736. (PubMed)
35. Kim YS, Kwak SM, Myung SK. Caffeine intake from coffee or tea and cognitive disorders: a meta-analysis of observational studies. Neuroepidemiology. 2015;44(1):51-63. (PubMed)
36. Liu QP, Wu YF, Cheng HY, et al. Habitual coffee consumption and risk of cognitive decline/dementia: A systematic review and meta-analysis of prospective cohort studies. Nutrition. 2016;32(6):628-636. (PubMed)
37. Friedman SL, Schiano TD. Cirrhosis and its sequelae. In: Goldman L, Ausiello D, eds. Cecil Textbook of Medicine. 22nd ed. St. Louis: W. B. Saunders; 2004:940-944.
38. Michelotti GA, Machado MV, Diehl AM. NAFLD, NASH and liver cancer. Nat Rev Gastroenterol Hepatol. 2013;10(11):656-665. (PubMed)
39. Modi AA, Feld JJ, Park Y, et al. Increased caffeine consumption is associated with reduced hepatic fibrosis. Hepatology. 2010;51(1):201-209. (PubMed)
40. Khalaf N, White D, Kanwal F, et al. Coffee and caffeine are associated with decreased risk of advanced hepatic fibrosis among patients with hepatitis C. Clin Gastroenterol Hepatol. 2015;13(8):1521-1531.e1523. (PubMed)
41. Machado SR, Parise ER, Carvalho L. Coffee has hepatoprotective benefits in Brazilian patients with chronic hepatitis C even in lower daily consumption than in American and European populations. Braz J Infect Dis. 2014;18(2):170-176. (PubMed)
42. Anty R, Marjoux S, Iannelli A, et al. Regular coffee but not espresso drinking is protective against fibrosis in a cohort mainly composed of morbidly obese European women with NAFLD undergoing bariatric surgery. J Hepatol. 2012;57(5):1090-1096. (PubMed)
43. Molloy JW, Calcagno CJ, Williams CD, Jones FJ, Torres DM, Harrison SA. Association of coffee and caffeine consumption with fatty liver disease, nonalcoholic steatohepatitis, and degree of hepatic fibrosis. Hepatology. 2012;55(2):429-436. (PubMed)
44. Shen H, Rodriguez AC, Shiani A, et al. Association between caffeine consumption and nonalcoholic fatty liver disease: a systemic review and meta-analysis. Therap Adv Gastroenterol. 2016;9(1):113-120. (PubMed)
45. Liu F, Wang X, Wu G, et al. Coffee consumption decreases risks for hepatic fibrosis and cirrhosis: a meta-analysis. PLoS One. 2015;10(11):e0142457. (PubMed)
46. Goh GB, Chow WC, Wang R, Yuan JM, Koh WP. Coffee, alcohol and other beverages in relation to cirrhosis mortality: the Singapore Chinese Health Study. Hepatology. 2014;60(2):661-669. (PubMed)
47. Klatsky AL, Armstrong MA, Friedman GD. Coffee, tea, and mortality. Ann Epidemiol. 1993;3(4):375-381. (PubMed)
48. Klatsky AL, Morton C, Udaltsova N, Friedman GD. Coffee, cirrhosis, and transaminase enzymes. Arch Intern Med. 2006;166(11):1190-1195. (PubMed)
49. Tverdal A, Skurtveit S. Coffee intake and mortality from liver cirrhosis. Ann Epidemiol. 2003;13(6):419-423. (PubMed)
50. Saab S, Mallam D, Cox GA, 2nd, Tong MJ. Impact of coffee on liver diseases: a systematic review. Liver Int. 2014;34(4):495-504. (PubMed)
51. Setiawan VW, Wilkens LR, Lu SC, Hernandez BY, Le Marchand L, Henderson BE. Association of coffee intake with reduced incidence of liver cancer and death from chronic liver disease in the US multiethnic cohort. Gastroenterology. 2015;148(1):118-125; quiz e115. (PubMed)
52. Yu C, Cao Q, Chen P, et al. An updated dose-response meta-analysis of coffee consumption and liver cancer risk. Sci Rep. 2016;6:37488. (PubMed)
53. Alicandro G, Tavani A, La Vecchia C. Coffee and cancer risk: a summary overview. Eur J Cancer Prev. 2017. (PubMed)
54. Wang A, Wang S, Zhu C, et al. Coffee and cancer risk: A meta-analysis of prospective observational studies. Sci Rep. 2016;6:33711. (PubMed)
55. Cao S, Liu L, Yin X, Wang Y, Liu J, Lu Z. Coffee consumption and risk of prostate cancer: a meta-analysis of prospective cohort studies. Carcinogenesis. 2014;35(2):256-261. (PubMed)
56. Lu Y, Zhai L, Zeng J, et al. Coffee consumption and prostate cancer risk: an updated meta-analysis. Cancer Causes Control. 2014;25(5):591-604. (PubMed)
57. Liu H, Hu GH, Wang XC, et al. Coffee consumption and prostate cancer risk: a meta-analysis of cohort studies. Nutr Cancer. 2015;67(3):392-400. (PubMed)
58. Zhong S, Chen W, Yu X, Chen Z, Hu Q, Zhao J. Coffee consumption and risk of prostate cancer: an up-to-date meta-analysis. Eur J Clin Nutr. 2014;68(3):330-337. (PubMed)
59. Li XJ, Ren ZJ, Qin JW, et al. Coffee consumption and risk of breast cancer: an up-to-date meta-analysis. PLoS One. 2013;8(1):e52681. (PubMed)
60. Jiang W, Wu Y, Jiang X. Coffee and caffeine intake and breast cancer risk: an updated dose-response meta-analysis of 37 published studies. Gynecol Oncol. 2013;129(3):620-629. (PubMed)
61. Li G, Ma D, Zhang Y, Zheng W, Wang P. Coffee consumption and risk of colorectal cancer: a meta-analysis of observational studies. Public Health Nutr. 2013;16(2):346-357. (PubMed)
62. Akter S, Kashino I, Mizoue T, et al. Coffee drinking and colorectal cancer risk: an evaluation based on a systematic review and meta-analysis among the Japanese population. Jpn J Clin Oncol. 2016;46(8):781-787. (PubMed)
63. Gan Y, Wu J, Zhang S, et al. Association of coffee consumption with risk of colorectal cancer: a meta-analysis of prospective cohort studies. Oncotarget. 2016; 8(12):18699-18711. (PubMed)
64. Je Y, Giovannucci E. Coffee consumption and risk of endometrial cancer: findings from a large up-to-date meta-analysis. Int J Cancer. 2012;131(7):1700-1710. (PubMed)
65. Zhou Q, Luo ML, Li H, Li M, Zhou JG. Coffee consumption and risk of endometrial cancer: a dose-response meta-analysis of prospective cohort studies. Sci Rep. 2015;5:13410. (PubMed)
66. Zheng JS, Yang J, Fu YQ, Huang T, Huang YJ, Li D. Effects of green tea, black tea, and coffee consumption on the risk of esophageal cancer: a systematic review and meta-analysis of observational studies. Nutr Cancer. 2013;65(1):1-16. (PubMed)
67. Malerba S, Galeone C, Pelucchi C, et al. A meta-analysis of coffee and tea consumption and the risk of glioma in adults. Cancer Causes Control. 2013;24(2):267-276. (PubMed)
68. Chen J, Long S. Tea and coffee consumption and risk of laryngeal cancer: a systematic review meta-analysis. PLoS One. 2014;9(12):e112006. (PubMed)
69. Ouyang Z, Wang Z, Jin J. Association between tea and coffee consumption and risk of laryngeal cancer: a meta-analysis. Int J Clin Exp Med. 2014;7(12):5192-5200. (PubMed)
70. Xie Y, Qin J, Nan G, Huang S, Wang Z, Su Y. Coffee consumption and the risk of lung cancer: an updated meta-analysis of epidemiological studies. Eur J Clin Nutr. 2016;70(2):199-206. (PubMed)
71. Wang J, Li X, Zhang D. Coffee consumption and the risk of cutaneous melanoma: a meta-analysis. Eur J Nutr. 2016;55(4):1317-1329. (PubMed)
72. Zhang Y, Wang X, Cui D. Association between coffee consumption and the risk of oral cancer: a meta-analysis of observational studies. Int J Clin Exp Med. 2015;8(7):11657-11665. (PubMed)
73. Turati F, Galeone C, Edefonti V, et al. A meta-analysis of coffee consumption and pancreatic cancer. Ann Oncol. 2012;23(2):311-318. (PubMed)
74. Ran HQ, Wang JZ, Sun CQ. Coffee consumption and pancreatic cancer risk: an update meta-analysis of cohort studies. Pak J Med Sci. 2016;32(1):253-259. (PubMed)
75. Nie K, Xing Z, Huang W, Wang W, Liu W. Coffee intake and risk of pancreatic cancer: an updated meta-analysis of prospective studies. Minerva Med. 2016;107(4):270-278. (PubMed)
76. Zeng SB, Weng H, Zhou M, Duan XL, Shen XF, Zeng XT. Long-term coffee consumption and risk of gastric cancer: a PRISMA-compliant dose-response meta-analysis of prospective cohort studies. Medicine (Baltimore). 2015;94(38):e1640. (PubMed)
77. Li L, Gan Y, Wu C, Qu X, Sun G, Lu Z. Coffee consumption and the risk of gastric cancer: a meta-analysis of prospective cohort studies. BMC Cancer. 2015;15:733. (PubMed)
78. Xie Y, Huang S, He T, Su Y. Coffee consumption and risk of gastric cancer: an updated meta-analysis. Asia Pac J Clin Nutr. 2016;25(3):578-588. (PubMed)
79. Han MA, Kim JH. Coffee consumption and the risk of thyroid cancer: a systematic review and meta-analysis. Int J Environ Res Public Health. 2017;14(2). (PubMed)
80. Ding M, Satija A, Bhupathiraju SN, et al. Association of coffee consumption with total and cause-specific mortality in 3 large prospective cohorts. Circulation. 2015;132(24):2305-2315. (PubMed)
81. Freedman ND, Park Y, Abnet CC, Hollenbeck AR, Sinha R. Association of coffee drinking with total and cause-specific mortality. N Engl J Med. 2012;366(20):1891-1904. (PubMed)
82. Lof M, Sandin S, Yin L, Adami HO, Weiderpass E. Prospective study of coffee consumption and all-cause, cancer, and cardiovascular mortality in Swedish women. Eur J Epidemiol. 2015;30(9):1027-1034. (PubMed)
83. Loftfield E, Freedman ND, Graubard BI, et al. Association of coffee consumption with overall and cause-specific mortality in a large US prospective cohort study. Am J Epidemiol. 2015;182(12):1010-1022. (PubMed)
84. Saito E, Inoue M, Sawada N, et al. Association of coffee intake with total and cause-specific mortality in a Japanese population: the Japan Public Health Center-based Prospective Study. Am J Clin Nutr. 2015;101(5):1029-1037. (PubMed)
85. Sugiyama K, Kuriyama S, Akhter M, et al. Coffee consumption and mortality due to all causes, cardiovascular disease, and cancer in Japanese women. J Nutr. 2010;140(5):1007-1013. (PubMed)
86. Tamakoshi A, Lin Y, Kawado M, Yagyu K, Kikuchi S, Iso H. Effect of coffee consumption on all-cause and total cancer mortality: findings from the JACC study. Eur J Epidemiol. 2011;26(4):285-293. (PubMed)
87. Crippa A, Discacciati A, Larsson SC, Wolk A, Orsini N. Coffee consumption and mortality from all causes, cardiovascular disease, and cancer: a dose-response meta-analysis. Am J Epidemiol. 2014;180(8):763-775. (PubMed)
88. Nawrot P, Jordan S, Eastwood J, Rotstein J, Hugenholtz A, Feeley M. Effects of caffeine on human health. Food Addit Contam. 2003;20(1):1-30. (PubMed)
89. CNS Stimulants: Caffeine. In: Novak K, ed. Drug Facts and Comparisons. St. Louis: Wolters Kluwer Health; 2005:917-919.
90. Engebretsen KM, Harris CR. Caffeine and Related Nonprescription Sympathomimetics. In: Ford MD, Delaney KA, Ling LJ, Erickson T, eds. Clinical Toxicology. Philadelphia: W. B. Saunders; 2001:310-315.
91. Juliano LM, Griffiths RR. A critical review of caffeine withdrawal: empirical validation of symptoms and signs, incidence, severity, and associated features. Psychopharmacology (Berl). 2004;176(1):1-29. (PubMed)
92. Dews PB, Curtis GL, Hanford KJ, O'Brien CP. The frequency of caffeine withdrawal in a population-based survey and in a controlled, blinded pilot experiment. J Clin Pharmacol. 1999;39(12):1221-1232. (PubMed)
93. Jee SH, He J, Appel LJ, Whelton PK, Suh I, Klag MJ. Coffee consumption and serum lipids: a meta-analysis of randomized controlled clinical trials. Am J Epidemiol. 2001;153(4):353-362. (PubMed)
94. Cai L, Ma D, Zhang Y, Liu Z, Wang P. The effect of coffee consumption on serum lipids: a meta-analysis of randomized controlled trials. Eur J Clin Nutr. 2012;66(8):872-877. (PubMed)
95. Splaver A, Lamas GA, Hennekens CH. Homocysteine and cardiovascular disease: biological mechanisms, observational epidemiology, and the need for randomized trials. Am Heart J. 2004;148(1):34-40. (PubMed)
96. Husemoen LL, Thomsen TF, Fenger M, Jorgensen T. Effect of lifestyle factors on plasma total homocysteine concentrations in relation to MTHFR(C677T) genotype. Inter99 (7). Eur J Clin Nutr. 2004;58(8):1142-1150. (PubMed)
97. Mennen LI, de Courcy GP, Guilland JC, et al. Homocysteine, cardiovascular disease risk factors, and habitual diet in the French Supplementation with Antioxidant Vitamins and Minerals Study. Am J Clin Nutr. 2002;76(6):1279-1289. (PubMed)
98. de Bree A, Verschuren WM, Blom HJ, Kromhout D. Lifestyle factors and plasma homocysteine concentrations in a general population sample. Am J Epidemiol. 2001;154(2):150-154. (PubMed)
99. Stolzenberg-Solomon RZ, Miller ER, 3rd, Maguire MG, Selhub J, Appel LJ. Association of dietary protein intake and coffee consumption with serum homocysteine concentrations in an older population. Am J Clin Nutr. 1999;69(3):467-475. (PubMed)
100. Nygard O, Refsum H, Ueland PM, et al. Coffee consumption and plasma total homocysteine: The Hordaland Homocysteine Study. Am J Clin Nutr. 1997;65(1):136-143. (PubMed)
101. Christensen B, Mosdol A, Retterstol L, Landaas S, Thelle DS. Abstention from filtered coffee reduces the concentrations of plasma homocysteine and serum cholesterol--a randomized controlled trial. Am J Clin Nutr. 2001;74(3):302-307. (PubMed)
102. Urgert R, van Vliet T, Zock PL, Katan MB. Heavy coffee consumption and plasma homocysteine: a randomized controlled trial in healthy volunteers. Am J Clin Nutr. 2000;72(5):1107-1110. (PubMed)
103. Grubben MJ, Boers GH, Blom HJ, et al. Unfiltered coffee increases plasma homocysteine concentrations in healthy volunteers: a randomized trial. Am J Clin Nutr. 2000;71(2):480-484. (PubMed)
104. Noordzij M, Uiterwaal CS, Arends LR, Kok FJ, Grobbee DE, Geleijnse JM. Blood pressure response to chronic intake of coffee and caffeine: a meta-analysis of randomized controlled trials. J Hypertens. 2005;23(5):921-928. (PubMed)
105. Jee SH, He J, Whelton PK, Suh I, Klag MJ. The effect of chronic coffee drinking on blood pressure: a meta-analysis of controlled clinical trials. Hypertension. 1999;33(2):647-652. (PubMed)
106. Lewington S, Clarke R, Qizilbash N, Peto R, Collins R. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet. 2002;360(9349):1903-1913. (PubMed)
107. Steffen M, Kuhle C, Hensrud D, Erwin PJ, Murad MH. The effect of coffee consumption on blood pressure and the development of hypertension: a systematic review and meta-analysis. J Hypertens. 2012;30(12):2245-2254. (PubMed)
108. Zhang Z, Hu G, Caballero B, Appel L, Chen L. Habitual coffee consumption and risk of hypertension: a systematic review and meta-analysis of prospective observational studies. Am J Clin Nutr. 2011;93(6):1212-1219. (PubMed)
109. Rhee JJ, Qin F, Hedlin HK, et al. Coffee and caffeine consumption and the risk of hypertension in postmenopausal women. Am J Clin Nutr. 2016;103(1):210-217. (PubMed)
110. Mesas AE, Leon-Munoz LM, Rodriguez-Artalejo F, Lopez-Garcia E. The effect of coffee on blood pressure and cardiovascular disease in hypertensive individuals: a systematic review and meta-analysis. Am J Clin Nutr. 2011;94(4):1113-1126. (PubMed)
111. Wu JN, Ho SC, Zhou C, et al. Coffee consumption and risk of coronary heart diseases: a meta-analysis of 21 prospective cohort studies. Int J Cardiol. 2009;137(3):216-225. (PubMed)
112. Ding M, Bhupathiraju SN, Satija A, van Dam RM, Hu FB. Long-term coffee consumption and risk of cardiovascular disease: a systematic review and a dose-response meta-analysis of prospective cohort studies. Circulation. 2014;129(6):643-659. (PubMed)
113. Choi Y, Chang Y, Ryu S, et al. Coffee consumption and coronary artery calcium in young and middle-aged asymptomatic adults. Heart. 2015;101(9):686-691. (PubMed)
114. Miller PE, Zhao D, Frazier-Wood AC, et al. Associations of Coffee, Tea, and Caffeine Intake with Coronary Artery Calcification and Cardiovascular Events. Am J Med. 2017;130(2):188-197.e185. (PubMed)
115. Patel YR, Gadiraju TV, Ellison RC, et al. Coffee consumption and calcified atherosclerotic plaques in the coronary arteries: The NHLBI Family Heart Study. Clin Nutr ESPEN. 2017;17:18-21. (PubMed)
116. van Woudenbergh GJ, Vliegenthart R, van Rooij FJ, et al. Coffee consumption and coronary calcification: the Rotterdam Coronary Calcification Study. Arterioscler Thromb Vasc Biol. 2008;28(5):1018-1023. (PubMed)
117. Reis JP, Loria CM, Steffen LM, et al. Coffee, decaffeinated coffee, caffeine, and tea consumption in young adulthood and atherosclerosis later in life: the CARDIA study. Arterioscler Thromb Vasc Biol. 2010;30(10):2059-2066. (PubMed)
118. Chelsky LB, Cutler JE, Griffith K, Kron J, McClelland JH, McAnulty JH. Caffeine and ventricular arrhythmias. An electrophysiological approach. JAMA. 1990;264(17):2236-2240. (PubMed)
119. Myers MG. Caffeine and cardiac arrhythmias. Ann Intern Med. 1991;114(2):147-150. (PubMed)
120. Zuchinali P, Ribeiro PA, Pimentel M, da Rosa PR, Zimerman LI, Rohde LE. Effect of caffeine on ventricular arrhythmia: a systematic review and meta-analysis of experimental and clinical studies. Europace. 2016;18(2):257-266. (PubMed)
121. Caldeira D, Martins C, Alves LB, Pereira H, Ferreira JJ, Costa J. Caffeine does not increase the risk of atrial fibrillation: a systematic review and meta-analysis of observational studies. Heart. 2013;99(19):1383-1389. (PubMed)
122. Cheng M, Hu Z, Lu X, Huang J, Gu D. Caffeine intake and atrial fibrillation incidence: dose response meta-analysis of prospective cohort studies. Can J Cardiol. 2014;30(4):448-454. (PubMed)
123. Glatter KA, Myers R, Chiamvimonvat N. Recommendations regarding dietary intake and caffeine and alcohol consumption in patients with cardiac arrhythmias: what do you tell your patients to do or not to do? Curr Treat Options Cardiovasc Med. 2012;14(5):529-535. (PubMed)
124. Larsson SC, Orsini N. Coffee consumption and risk of stroke: a dose-response meta-analysis of prospective studies. Am J Epidemiol. 2011;174(9):993-1001. (PubMed)
125. Floegel A, Pischon T, Bergmann MM, Teucher B, Kaaks R, Boeing H. Coffee consumption and risk of chronic disease in the European Prospective Investigation into Cancer and Nutrition (EPIC)-Germany study. Am J Clin Nutr. 2012;95(4):901-908. (PubMed)
126. Kokubo Y, Iso H, Saito I, et al. The impact of green tea and coffee consumption on the reduced risk of stroke incidence in Japanese population: the Japan public health center-based study cohort. Stroke. 2013;44(5):1369-1374. (PubMed)
127. Galarraga V, Boffetta P. Coffee Drinking and Risk of Lung Cancer-A Meta-Analysis. Cancer Epidemiol Biomarkers Prev. 2016;25(6):951-957. (PubMed)
128. Bech BH, Nohr EA, Vaeth M, Henriksen TB, Olsen J. Coffee and fetal death: a cohort study with prospective data. Am J Epidemiol. 2005;162(10):983-990. (PubMed)
129. Greenwood DC, Alwan N, Boylan S, et al. Caffeine intake during pregnancy, late miscarriage and stillbirth. Eur J Epidemiol. 2010;25(4):275-280. (PubMed)
130. Tolstrup JS, Kjaer SK, Munk C, et al. Does caffeine and alcohol intake before pregnancy predict the occurrence of spontaneous abortion? Hum Reprod. 2003;18(12):2704-2710. (PubMed)
131. Wen W, Shu XO, Jacobs DR, Jr., Brown JE. The associations of maternal caffeine consumption and nausea with spontaneous abortion. Epidemiology. 2001;12(1):38-42. (PubMed)
132. Weng X, Odouli R, Li DK. Maternal caffeine consumption during pregnancy and the risk of miscarriage: a prospective cohort study. Am J Obstet Gynecol. 2008;198(3):279 e271-278. (PubMed)
133. Fenster L, Hubbard AE, Swan SH, et al. Caffeinated beverages, decaffeinated coffee, and spontaneous abortion. Epidemiology. 1997;8(5):515-523. (PubMed)
134. Mills JL, Holmes LB, Aarons JH, et al. Moderate caffeine use and the risk of spontaneous abortion and intrauterine growth retardation. JAMA. 1993;269(5):593-597. (PubMed)
135. Pollack AZ, Buck Louis GM, Sundaram R, Lum KJ. Caffeine consumption and miscarriage: a prospective cohort study. Fertil Steril. 2010;93(1):304-306. (PubMed)
136. Savitz DA, Chan RL, Herring AH, Howards PP, Hartmann KE. Caffeine and miscarriage risk. Epidemiology. 2008;19(1):55-62. (PubMed)
137. Chen LW, Wu Y, Neelakantan N, Chong MF, Pan A, van Dam RM. Maternal caffeine intake during pregnancy and risk of pregnancy loss: a categorical and dose-response meta-analysis of prospective studies. Public Health Nutr. 2016;19(7):1233-1244. (PubMed)
138. Klebanoff MA, Levine RJ, DerSimonian R, Clemens JD, Wilkins DG. Maternal serum paraxanthine, a caffeine metabolite, and the risk of spontaneous abortion. N Engl J Med. 1999;341(22):1639-1644. (PubMed)
139. Leviton A, Cowan L. A review of the literature relating caffeine consumption by women to their risk of reproductive hazards. Food Chem Toxicol. 2002;40(9):1271-1310. (PubMed)
140. Peck JD, Leviton A, Cowan LD. A review of the epidemiologic evidence concerning the reproductive health effects of caffeine consumption: a 2000-2009 update. Food Chem Toxicol. 2010;48(10):2549-2576. (PubMed)
141. Giannelli M, Doyle P, Roman E, Pelerin M, Hermon C. The effect of caffeine consumption and nausea on the risk of miscarriage. Paediatr Perinat Epidemiol. 2003;17(4):316-323. (PubMed)
142. Cnattingius S, Signorello LB, Anneren G, et al. Caffeine intake and the risk of first-trimester spontaneous abortion. N Engl J Med. 2000;343(25):1839-1845. (PubMed)
143. Gaskins AJ, Rich-Edwards JW, Williams PL, Toth TL, Missmer SA, Chavarro JE. Pre-pregnancy caffeine and caffeinated beverage intake and risk of spontaneous abortion. Eur J Nutr. 2016. [Epub ahead of print]. (PubMed)
144. American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 462: Moderate caffeine consumption during pregnancy. Obstet Gynecol. 2010 (Reaffirmed 2016);116(2 Pt 1):467-468.
145. Rhee J, Kim R, Kim Y, et al. Maternal caffeine consumption during pregnancy and risk of low birth weight: a dose-response meta-analysis of observational studies. PLoS One. 2015;10(7):e0132334. (PubMed)
146. Greenwood DC, Thatcher NJ, Ye J, et al. Caffeine intake during pregnancy and adverse birth outcomes: a systematic review and dose-response meta-analysis. Eur J Epidemiol. 2014;29(10):725-734. (PubMed)
147. Bech BH, Obel C, Henriksen TB, Olsen J. Effect of reducing caffeine intake on birth weight and length of gestation: randomised controlled trial. BMJ. 2007;334(7590):409. (PubMed)
148. Chen LW, Wu Y, Neelakantan N, Chong MF, Pan A, van Dam RM. Maternal caffeine intake during pregnancy is associated with risk of low birth weight: a systematic review and dose-response meta-analysis. BMC Med. 2014;12:174. (PubMed)
149. Collier SA, Browne ML, Rasmussen SA, Honein MA. Maternal caffeine intake during pregnancy and orofacial clefts. Birth Defects Res A Clin Mol Teratol. 2009;85(10):842-849. (PubMed)
150. Browne ML, Hoyt AT, Feldkamp ML, et al. Maternal caffeine intake and risk of selected birth defects in the National Birth Defects Prevention Study. Birth Defects Res A Clin Mol Teratol. 2011;91(2):93-101. (PubMed)
151. Chen L, Bell EM, Browne ML, et al. Maternal caffeine consumption and risk of congenital limb deficiencies. Birth Defects Res A Clin Mol Teratol. 2012;94(12):1033-1043. (PubMed)
152. Werler MM, Yazdy MM, Kasser JR, et al. Maternal cigarette, alcohol, and coffee consumption in relation to risk of clubfoot. Paediatr Perinat Epidemiol. 2015;29(1):3-10. (PubMed)
153. Li ZX, Gao ZL, Wang JN, Guo QH. Maternal coffee consumption during pregnancy and neural tube defects in offspring: a meta-analysis. Fetal Pediatr Pathol. 2016;35(1):1-9. (PubMed)
154. Cheng J, Su H, Zhu R, et al. Maternal coffee consumption during pregnancy and risk of childhood acute leukemia: a metaanalysis. Am J Obstet Gynecol. 2014;210(2):151.e151-151.e110. (PubMed)
155. Thomopoulos TP, Ntouvelis E, Diamantaras AA, et al. Maternal and childhood consumption of coffee, tea and cola beverages in association with childhood leukemia: a meta-analysis. Cancer Epidemiol. 2015;39(6):1047-1059. (PubMed)
156. Song JW, Chung KC. Observational studies: cohort and case-control studies. Plast Reconstr Surg. 2010;126(6):2234-2242. (PubMed)
157. The American Academy of Pediatrics. Things to Avoid When Breastfeeding. 21 November 2015 Available at: https://www.healthychildren.org/English/ages-stages/baby/breastfeeding/Pages/Things-to-Avoid-When-Breastfeeding.aspx. Accessed 4/11/17.
158. Barger-Lux MJ, Heaney RP. Caffeine and the calcium economy revisited. Osteoporos Int. 1995;5(2):97-102. (PubMed)
159. Hasling C, Sondergaard K, Charles P, Mosekilde L. Calcium metabolism in postmenopausal osteoporotic women is determined by dietary calcium and coffee intake. J Nutr. 1992;122(5):1119-1126. (PubMed)
160. Heaney RP. Effects of caffeine on bone and the calcium economy. Food Chem Toxicol. 2002;40(9):1263-1270. (PubMed)
161. Choi E, Choi KH, Park SM, Shin D, Joh HK, Cho E. The benefit of bone health by drinking coffee among Korean postmenopausal women: a cross-sectional analysis of the fourth & fifth Korea National Health and Nutrition Examination Surveys. PLoS One. 2016;11(1):e0147762. (PubMed)
162. Hallstrom H, Byberg L, Glynn A, Lemming EW, Wolk A, Michaelsson K. Long-term coffee consumption in relation to fracture risk and bone mineral density in women. Am J Epidemiol. 2013;178(6):898-909. (PubMed)
163. Harris SS, Dawson-Hughes B. Caffeine and bone loss in healthy postmenopausal women. Am J Clin Nutr. 1994;60(4):573-578. (PubMed)
164. Rapuri PB, Gallagher JC, Kinyamu HK, Ryschon KL. Caffeine intake increases the rate of bone loss in elderly women and interacts with vitamin D receptor genotypes. Am J Clin Nutr. 2001;74(5):694-700. (PubMed)
165. Li S, Dai Z, Wu Q. Effect of coffee intake on hip fracture: a meta-analysis of prospective cohort studies. Nutr J. 2015;14:38. (PubMed)
166. Sheng J, Qu X, Zhang X, et al. Coffee, tea, and the risk of hip fracture: a meta-analysis. Osteoporos Int. 2014;25(1):141-150. (PubMed)
167. Lee DR, Lee J, Rota M, et al. Coffee consumption and risk of fractures: a systematic review and dose-response meta-analysis. Bone. 2014;63:20-28. (PubMed)
168. Fairweather-Tait SJ. Iron nutrition in the UK: getting the balance right. Proc Nutr Soc. 2004;63(4):519-528. (PubMed)
169. Morck TA, Lynch SR, Cook JD. Inhibition of food iron absorption by coffee. Am J Clin Nutr. 1983;37(3):416-420. (PubMed)
170. Hallberg L, Rossander L. Effect of different drinks on the absorption of non-heme iron from composite meals. Hum Nutr Appl Nutr. 1982;36(2):116-123. (PubMed)
171. Carrillo JA, Benitez J. Clinically significant pharmacokinetic interactions between dietary caffeine and medications. Clin Pharmacokinet. 2000;39(2):127-153. (PubMed)
172. Faber MS, Fuhr U. Time response of cytochrome P450 1A2 activity on cessation of heavy smoking. Clin Pharmacol Ther. 2004;76(2):178-184. (PubMed)
173. Natural Medicines. Coffee - professional handout: drug interactions. © 2017 Therapeutic Research Center 27 May 2015. Available at: http://naturaldatabase.therapeuticresearch.com/. Accessed 4/11/17.
Tea
Contents
Summary
- Tea is an infusion of the leaves of the Camellia sinensis plant, which is not to be confused with herbal teas. (More information)
- All the tea types, including white, green, oolong, black, and Pu-erh tea, are produced from the leaves of the Camellia sinensis plant. Different processing methods yield the various types of tea. (More information)
- Biologically active chemicals in tea include flavonoids, caffeine, fluoride, and theanine. Green teas are especially rich in a group of flavonoids called flavan-3-ol monomers or catechins. Black teas contain more complex chemicals — theaflavins and thearubigins — derived from catechins. (More information)
- Observational studies in humans suggest that daily consumption of tea may be associated with a reduced risk of cardiovascular disease (CVD). Intervention studies showed that tea exhibited cholesterol-lowering, anti-inflammatory, antioxidant, and anti-hypertensive properties, which might be beneficial in the prevention of CVD. (More information)
- Tea consumption has been associated with reduced risk of developing type 2 diabetes mellitus in large prospective cohort studies. Yet, the mechanisms behind this association are complex, possibly involving a role for tea bioactive compounds in the regulation of energy balance, lipid and glucose metabolism, insulin sensitivity, body composition, and/or body temperature. (More information)
- Despite promising results from animal studies, current evidence does not support a role for tea consumption in the prevention of most cancers in humans. (More information)
- It is still unclear whether tea consumption is associated with increased bone mineral density and/or reduced risk of osteoporotic fractures. (More information)
- Limited research suggests an inverse association between tea consumption and risk of tooth cavities. The incorporation of green tea extract in mouthwashes and/or toothpastes may also help reduce dental plaque and gum inflammation in patients. (More information)
- The pooled analysis of three large US prospective cohort studies found an 11% lower risk of kidney stones with the highest versus lowest level of tea intake. More research is needed to assess whether the oxalate content in tea may affect subjects with a history of kidney stones. (More information)
- The mounting evidence for a role of tea consumption in the prevention of cognitive decline comes from observational studies. Clinical studies would be needed to establish whether tea or its bioactive constituents could limit cognitive decline and/or improve cognitive dysfunction in older individuals. (More information)
- The use of green tea extracts in clinical trials was found to cause gastrointestinal disorders and liver toxicity. Tea consumption may also potentially interfere with certain medications, including the anticoagulant, warfarin, and some cardiovascular drugs. (More information)
Introduction
Tea is an infusion of the leaves of the Camellia sinensis plant and, aside from water, is the most widely consumed beverage in the world (1). Different processing methods of tea leaves involve variable degrees of oxidation yielding different types of tea (green, oolong, or black tea). In 2014, Americans consumed 3.6 billion gallons of tea, of which 84% was black tea, 15% was green tea, and the remaining was white, oolong, and dark tea (2). Herbal teas are infusions of herbs or plants other than Camellia sinensis and will not be discussed in this article. Although tea contains a number of bioactive chemicals, including caffeine and fluoride, most research has focused on the potential health benefits of a class of compounds in tea known as flavonoids. In many cultures, tea is an important source of dietary flavonoids.
Definitions
Types of tea
All teas are derived from the leaves of the tea plant Camellia sinensis, but different processing methods produce different types of tea. Fresh tea leaves are rich in polyphenolic compounds known as flavonoids (see the article on Flavonoids). Flavonoids are divided in six subclasses: flavan-3-ols, anthocyanidins, flavanones, flavonols, flavones, and isoflavones (Figure 1). Tea leaves contain a polyphenol oxidase (PPO) enzyme in separate compartments from flavan-3-ol monomers or catechins (Figure 2) (3). When tea leaves are intentionally broken or rolled during processing, cell compartmentalization is disrupted and PPO comes into contact with catechins. This causes catechins to condense (join together) forming dimers and polymers known as theaflavins (Figure 3) and thearubigins, respectively (4). This oxidation process is often described as "fermentation" in the tea industry. Steaming, firing, or baking tea leaves inactivates PPO and stops the oxidation process (5).
The two prominent varieties of Camellia sinensis used in tea cultivation are Camellia sinensis var. sinensis and Camellia sinensis var. assamica. The former is native of China and usually used to make white and green tea. The latter originates from the Assam region of India, as well as regions of Southeast Asia, and is often used to make black teas, including pu-erh tea in the Yunnan province of China.
Although there are thousands of tea cultivars derived from the principal Camellia sinensis tea varieties, teas are usually divided into five types based on the extent of oxidation they undergo during processing. The withering method (the process of allowing the fresh leaves to dry) and the process of deactivating PPO may also differ among tea preparations (1).
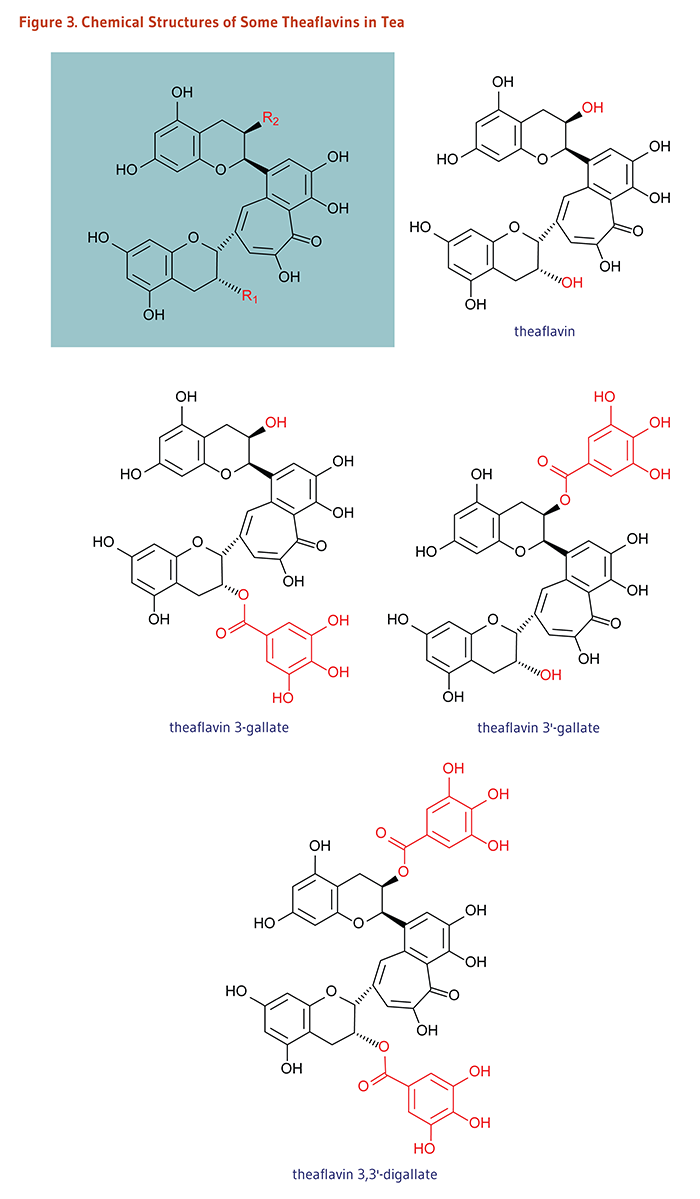
White tea
White tea is made from unopened buds and immature leaves, which are steamed or fired to inactivate polyphenol oxidase, and then dried. Thus, due to minimal oxidation, white tea retains the high concentrations of catechins present in fresh tea leaves (see Flavonoids below).
Green tea
Green tea is made from more mature tea leaves than white tea, and tea leaves may be withered prior to steaming or firing, and then rolled and dried. Like white teas, green teas are high in catechins, but the total content and composition of catechins may vary depending on the cultivar and the commercial source (6). Of note, green teas and white teas may sometimes contain similar amounts of catechins but still exhibit different antioxidant capacities; this is due to the presence of other non-catechin antioxidants in teas (6).
Oolong (Wulong) tea
Tea leaves destined to become oolong teas are "bruised" to allow the release of some of the polyphenol oxidase present in the leaves. Oolong teas are allowed to oxidize to a greater extent than for white or green teas, but for less time than black teas, before they are heated and dried. Consequently, the catechin, theaflavin, and thearubigin levels in oolong teas are generally between those of unfermented green and white teas and completely oxidized black teas (1).
Black tea
Tea leaves destined to become black tea are fully rolled or broken to maximize the interaction between catechins and polyphenol oxidase. Because they are allowed to oxidize completely before drying, most black teas are relatively low in monomeric flavan-3-ols, like (-)-epigallocatechin gallate (EGCG), and rich in theaflavins (2%-6% of extracted solids) and thearubigins (>20% of extracted solids) (see Table 1 below). Some theaflavins have shown greater antioxidant activities than EGCG (7).
Pu-erh tea (also pu’erh tea, pu’er tea, or Chinese black tea)
Most pu-erh tea is produced in the Yunnan province of China from the larger leaves of the assamica variety of Camellia sinensis. The making process may include both enzymatic oxidation and fungus-led fermentation. In the case of "raw (aged) pu-erh tea," the initial preparation resembles that used to make green tea. The leaves are heated, dried, and then dampened before being pan-fired and compressed; the preparation is then carefully stored in a controlled environment and left to age for decades. A faster aging process, combining oxidation and fermentation by the fungus Aspergillus niger for several months, can also be used to produce "ripened pu-erh tea."
Cup sizes
The definition of a cup of tea varies in different countries or regions. In Japan, a typical cup of green tea may contain only 100 mL (3.5 ounces). A traditional European teacup holds approximately 125 to 150 mL (5 ounces), while a mug of tea may contain about 240 mL (8 ounces).
Bioactive Compounds in Tea
Tea contains over 2,000 components, including polyphenols (flavonoids), pigments (carotenoids and chlorophyll), alkaloids (caffeine, theophylline, theobromine), lignans, carbohydrates, lipids, proteins, amino acids (including L-theanine), vitamins (vitamin C, vitamin E, riboflavin), and various minerals and trace elements (8). Only some of them are described below.
Flavonoids
Dietary flavonoids are divided in six subclasses: flavan-3-ols, anthocyanidins, flavanones, flavonols, flavones, and isoflavones (see the article on Flavonoids). Total flavonoid content in green tea and black tea is of about 138 mg and 118 mg per 100 mL, respectively (9). A major subclass of flavonoids in tea is that of flavan-3-ols. Flavan-3-ol monomers, also known as catechins, constitute 30%-42% of the solid weight of brewed green tea. The principal catechins found in tea are (-)-epicatechin (EC), (-)-epigallocatechin (EGC), (-)-epicatechin gallate (ECG), and (-)-epigallocatechin gallate (EGCG) (see Figure 2). When catechins are enzymatically oxidized by polyphenol oxidase during the oxidation process that yields black tea, they form low molecular weight dimers known as theaflavins (see Figure 3) and complex polymers (of mostly unknown structures) called thearubigins. Non-oxidized teas are rich in catechins, while fully oxidized teas are rich in theaflavins and thearubigins (Table 1) (5).
| Type of Tea1 | EC | ECG | EGC | EGCG | Thearubigins |
|---|---|---|---|---|---|
| Tea, white, brewed | -2 | 8.3 | 18.6 | 42.4 | - |
| Tea, green, brewed | 8.3 | 17.9 | 29.2 | 70.2 | 1.1 |
| Tea, oolong, brewed | 2.5 | 6.3 | 6.1 | 34.5 | - |
| Tea, black, brewed | 2.1 | 5.9 | 8.0 | 9.4 | 81.3 |
|
11 g of tea leaves infused in 100 mL of boiling water (1% weight/volume) 2The lack of a value for a particular flavonoid in a food in the database does not imply a zero value, but only that data were unavailable. |
|||||
Tea is also a good source of another class of flavonoids called flavonols. Flavonols found in tea include kaempferol, quercetin, and myricetin. The flavonol content of tea is minimally affected by processing, and flavonols are present in comparable quantities in oxidized and non-oxidized teas. Unlike flavan-3-ols, flavonols are usually present in tea as glycosides, i.e., bound to a sugar molecule (Figure 4). Despite their poor bioavailability, flavonoids are thought to contribute substantially to the health benefits associated with daily tea consumption (10). For more detailed information, see the article on Flavonoids.

Caffeine
All teas contain caffeine, unless they are deliberately decaffeinated during processing. The caffeine content of different varieties of tea may vary considerably and is influenced by factors like brewing time, the amount of tea and water used for brewing, and whether the tea is loose or in teabags. In general, a mug of tea contains about half as much caffeine as a mug of coffee (11). The caffeine contents of more than 20 green and black teas prepared according to package directions are presented in Table 2 (12). The caffeine content of oolong teas is comparable to green teas (13). There is little information on the caffeine content of white teas, since they are often grouped together with green teas (14). Buds and young tea leaves have been found to contain higher levels of caffeine than older leaves (15), suggesting that the caffeine content of some white teas may be slightly higher than that of green teas (16).
| Type of Tea | Caffeine (mg/L) | Caffeine (mg/8 ounces) |
|---|---|---|
| Green | 40-234 | 9-63 |
| Black | 177-333 | 42-79 |
| Coffee, brewed | 306-553 | 72-130 |
Caffeine is a known stimulant of the central nervous system, thought to protect dopaminergic neurons by antagonizing adenosine A2A receptors (Figure 5) (18). Because adenosine has mostly inhibitory effects in the central nervous system, the effects of adenosine antagonism by caffeine are generally stimulatory.
Fluoride
Tea plants accumulate fluoride in their leaves. In general, the oldest tea leaves contain the most fluoride (19). Most high-quality teas are made from the bud or the first two to four leaves — the youngest leaves on the plant. Fluoride levels in green, oolong, and black teas are generally comparable to those recommended for the prevention of dental caries (cavities). Thus, daily consumption of up to one liter of green, oolong, black, or pu-erh tea would be unlikely to result in fluoride intakes higher than those recommended for dental health (20, 21). The fluoride content of white tea is likely to be less than other teas, since white teas are made from the buds and youngest leaves of the tea plant. A comparative study of green, oolong, and black teas from six provinces of China found that fluoride content was inversely correlated with the quality level of tea sensory attributes (i.e., appearance, taste, flavor) (22). The fluoride content of 17 brands of green, oolong, and black teas is presented in Table 3 (20). These values do not include the fluoride content of the water used to make the tea. For more information, see the article on Fluoride.
| Type of Tea | Fluoride (mg/liter)1 | Fluoride (mg/8 ounces) |
|---|---|---|
| Green | 1.2-1.7 | 0.3-0.4 |
| Oolong | 0.6-1.0 | 0.1-0.2 |
| Black | 1.0-1.9 | 0.2-0.5 |
| Pu-erh tea | 0.9-1.6 | 0.2-0.4 |
| 1Fluoride in 1% weight/volume tea prepared by continuous infusion from 5 minutes (number before hyphen) to 360 minutes (number after hyphen). | ||
L-theanine
L-theanine (also L-g-glutamylethylamide) is a non-protein amino acid that constitutes about 1%-2% (w/w) of Camellia sinensis dry leaves (23). L-theanine is rapidly absorbed in the small intestine and has a bioavailability close to 100% (24). L-theanine can cross the blood-brain barrier and exert neuroprotective effects (25). Because its chemical structure resembles that of glutamate, a neurotransmitter critically involved in memory, theanine may compete with glutamate for binding to glutamate receptors (Figure 6). This glutamate antagonism has been associated with the prevention of neuronal death by theanine after brain ischemia (reviewed in 25).
Disease Prevention
Cardiovascular disease
Many epidemiological studies have considered the relationship between tea consumption and manifestations of cardiovascular disease (CVD), including coronary heart disease (CHD) and stroke (reviewed in 26). A recent meta-analysis by Zhang et al. included the results of prospective observational studies (cohort or nested case-control studies) that examined the association between tea consumption and cardiovascular morbidity and mortality (27). The results showed that a three-cup (125 mL/cup) increase in daily tea intake was associated with a 27% lower risk of CHD (seven studies), an 18% lower risk of total stroke (eight studies), a 16% lower risk of ischemic stroke (four studies), a 21% lower risk of intracerebral hemorrhage (a type of hemorrhagic stroke), a 26% lower risk of cardiac deaths (12 studies), and a 24% lower risk of total deaths (seven studies). No associations were found between tea consumption and stroke mortality (five studies) or risk of subarachnoid hemorrhage (a subtype of stroke; two studies). Further subgroup analyses indicated that green tea consumption was specifically linked to a reduced risk of stroke, cardiac mortality, and all-cause mortality, while a lower CHD risk was associated with black tea consumption (27). Another recent meta-analysis of prospective cohort studies found that the highest versus lowest level of green tea consumption was associated with a 33% lower risk of cardiovascular mortality (five studies) and a 20% lower risk of all-cause mortality (five studies) (28). Black tea consumption was linked to a 10% reduced risk of all-cause mortality, but not specifically to cardiovascular-related mortality (28).
Tea is a major source of flavonoids in US and European diets (29, 30). The results of several epidemiological studies suggest that dietary flavonoids might influence cardiovascular health. A recent meta-analysis of 14 prospective cohort studies reported that highest versus lowest quantiles of flavonol, flavan-3-ol, flavanone, flavone, anthocyanidin, and proanthocyanidin intake were associated with modest reductions (~10%) in cardiovascular risk (31). A dose-response analysis based on the results of 13 studies in nearly 350,000 individuals and 12,445 CVD cases found a 5% risk reduction with an average 10 mg-incremental increase in daily flavonol intakes (31). It is not clear whether the antioxidant, anti-inflammatory, and/or vasodilatory properties of flavonoids are responsible for some of the cardiovascular benefits associated with tea consumption (see also the article on Flavonoids).
Intervention studies
A number of intervention trials have investigated the effects of tea consumption on markers of cardiovascular health, including biological parameters related to lipid and glucose metabolism, inflammation, blood coagulation, endothelial health, and body composition.
Metabolic markers of cardiovascular disease: Clinical trials examining the effects of green and/or black tea beverages or extracts have been relatively heterogeneous, especially regarding concentrations of active substances, duration of interventions, and included populations. Pooled analyses of mostly short-term interventions (<3 months) have suggested a reduction in total and LDL cholesterol concentrations with green tea catechin consumption, but the data regarding a potential lipid-lowering effect of black tea are inconsistent (32-34).The studies mentioned below investigated the effect of tea/tea extracts given for at least three months.
Black tea: The Tea’s Effect on Atherosclerosis (TEA) pilot study in 28 older adults at increased risk for cardiovascular disease (CVD) did not find any effect of a six-month black tea consumption intervention (three glasses per day equivalent to 318 mg/day of black tea catechins) on specific biomarkers, including circulating lipoproteins, inflammation markers, homocysteine concentration, adhesion molecules, and hemostatic factors (35). Yet, in a randomized, placebo-controlled study in 47 individuals with borderline-to-moderate hypercholesterolemia, the daily consumption of 1 g of pu-erh tea (Chinese black tea) extract for three months reduced the blood concentrations of total cholesterol, LDL-cholesterol, and triglycerides (36). A three-month, placebo-controlled trial in 77 healthy subjects who drank 9 g of black tea infused in 600 mL of boiling water per day (about 740 mg/day of black tea polyphenols) had improvements in lipoprotein and triglyceride profiles, fasting serum glucose concentrations, and measures of antioxidant activities in the plasma (37). The same protocol was found to also reduce markers of oxidative stress and inflammation in the plasma of individuals at risk for CVD (38). In contrast, a recent randomized, double-blind, placebo-controlled trial in 77 regular tea drinkers (35 to 75 years old) found that the daily consumption of 429 mg of black tea polyphenols for six months had no effect on fasting blood glucose and serum lipids (39).
Green tea: Daily supplementation with a capsule containing 150 mg of green tea catechins, 75 mg of black tea theaflavins, and 150 mg of other polyphenols, for six months significantly lowered plasma LDL-cholesterol concentrations and the ratio total cholesterol:HDL in a randomized, double-blind, placebo-controlled study in 220 individuals with mild-to-moderate hypercholesterolemia (40). In a pilot study in 74 overweight/obese breast cancer survivors, the daily consumption of green tea (containing 26.7 mg of caffeine and 235.6 mg of catechins with 128.8 mg as EGCG) for six months increased HDL concentration compared to a citrus-based herbal placebo but had no effect on LDL concentration and markers of glycemic control (41). Another placebo-controlled study conducted in obese subjects with controlled hypertension found that the daily ingestion of one capsule of green tea extract (379 mg/capsule containing 208 mg of EGCG) for three months significantly lowered systolic and diastolic blood pressure and improved blood lipid profile, antioxidant status, and measures of glycemic control and inflammation (42).
Endothelial dysfunction: The vascular endothelial cells that line the inner surface of all blood vessels synthesize an enzyme, endothelial nitric oxide synthase (eNOS), which plays a critical role in maintaining cardiovascular health. Specifically, eNOS utilizes L-arginine to produce nitric oxide (NO), a compound that regulates vascular tone and blood flow by promoting the relaxation (vasodilation) of all types of blood vessels, including arteries (26). NO also regulates vascular homeostasis and protects the integrity of the endothelium by inhibiting vascular inflammation, leukocyte adhesion, platelet adhesion and aggregation, and proliferation of vascular smooth muscle cells (43). In the presence of cardiovascular risk factors (e.g., hypertension, hypercholesterolemia, hyperglycemia), early alterations in the structure and function of the vascular endothelium are associated with the loss of normal NO-mediated endothelium-dependent vasodilation. Endothelial dysfunction results in widespread vasoconstriction and coagulation abnormalities and is considered to be an early step in the development of atherosclerosis. The measurement of brachial flow-mediated dilation (FMD) is often used as a surrogate marker of endothelial function; FMD values are inversely correlated with the risk of future cardiovascular events (44).
Black tea: Two small controlled clinical trials found that daily consumption of 900 to 1,250 mL of black tea for four weeks significantly improved endothelium-dependent FMD in patients with coronary heart disease (45) and in patients with mildly elevated serum cholesterol concentrations (46). Improvements were noted in comparison to an equivalent amount of hot water. Incremental doses of black tea flavonoids (0, 100, 200, 400, and 800 mg/day; each dose being given for one week) have been associated with dose-dependent increases in brachial FMD in 19 healthy volunteers. Specifically, FMD values went from 7.8% at baseline (no flavonoids) to 10.3% with 800 mg/day of flavonoids (47). Of note, in this study, the ingestion of black tea flavonoids significantly lowered systolic and diastolic blood pressure in a non dose-dependent manner, while other variables, including markers of arterial stiffness, glucose metabolism, inflammation, endothelial activation, and lipid profile, remain largely unchanged (47). In a recent randomized trial, seven days of black tea consumption (450 mL/day) followed by the ingestion of two cups (300 mL) 20 minutes before an experimental ischemia-reperfusion (IR) injury procedure on healthy participants failed to prevent IR injury-associated FMD reduction. Even if tea flavonoids were able to limit the impact of IR injury on FMD — for example by counteracting the production of reactive oxygen species — the presence of caffeine in black tea (and the lack of control for it) could have confounded this effect (48).
Green tea: In a small study conducted in 14 healthy young adults (50% smokers), a significant increase in brachial FMD was reported 30 to 120 minutes after the consumption of 450 mL of green tea (6 g of green tea, including 125 mg of caffeine) compared to caffeine alone or hot water (49). Another study that compared the acute effect of black and green tea on brachial FMD in 21 postmenopausal women found a similar increase in FMD two hours after the ingestion of either tea preparation (50). Both black and green teas have been found to be equally able to increase eNOS activity and NO production in cultured endothelial cells (50, 51). Specifically, the prominent green tea catechin, EGCG, and black tea polyphenols, theaflavins and thearubigins, are thought to contribute to the protective effect of drinking tea by promoting antioxidant activity and endothelium-dependent vasodilation (51). Other flavonoids, like (-)-epicatechin and quercetin glucoside have recently failed to show an effect on FMD (and blood pressure) in hypertensive adults (52). For more information, see the article on Flavonoids.
A meta-analysis of nine human intervention studies estimated that the acute and/or short-term (up to four weeks), daily ingestion of 500 mL of tea — containing about 248 mg of flavonoids in green tea and 415 mg in black tea — significantly increased brachial FMD (53). Yet, the clinical relevance of these FMD improvements is unclear. It is also not clear whether the chronic consumption of tea might benefit vascular endothelial function and eventually lower the risk of cardiovascular disease.
Hypertension: Hypertension (high blood pressure) is a risk factor for CVD morbidity and mortality.
Black tea: A recent meta-analysis of 11 small randomized controlled trials in either healthy or at-risk individuals found a significant reduction of 2 mm Hg in systolic and 1 mm Hg in diastolic blood pressure with the daily consumption of at least 400 mL (13 oz) of black tea for one week to six months, providing a minimum of 240 mg/day of flavonoids (54). Two recent trials also reported that black tea lowered the rate of circadian variations in blood pressure at nighttime and variations after a dietary fat challenge. A six-month intervention in 76 participants from the general population (most with moderate hypertension) showed that the consumption of three cups per day of black tea, supplying a daily total of 1.29 g of polyphenols and 288 mg of caffeine, lowered the rate of nighttime blood pressure variations compared to a polyphenol-free caffeine-matched placebo (55). Two cups of black tea per day, equivalent to 300 mg of polyphenols, also limited blood pressure variations after a fat-rich meal in 19 patients with primary (idiopathic) hypertension (56). Mechanisms underlying the blood pressure-lowering properties of black tea may involve the inhibition of angiotensin-II synthesis by flavonoids (see below).
Green tea: Several recent meta-analyses of randomized controlled trials indicated that the consumption of green tea or green tea extracts could significantly lower blood pressure (57-60). In one of them, the pooled analysis of 13 trials in 1,367 subjects found a 2.0 mm Hg reduction in systolic blood pressure and a 1.9 mm Hg reduction in diastolic blood pressure with green tea polyphenols (208 mg/day-1,207 mg/day) for a median period of 12 weeks (60). Subgroup analyses suggested greater blood pressure lowering effect with polyphenol intake levels lower than 582.8 mg/day and with adjustment for the confounding effect of caffeine. Another meta-analysis included randomized controlled trials that specifically explored the effect of green tea or green tea extracts on blood pressure in overweight or obese subjects. The pooled analysis of 14 trials showed significant reductions of 1.4 mm Hg in systolic blood pressure and 1.3 mm Hg in diastolic blood pressure (58). The anti-hypertensive effect of green tea may be mediated by a number of mechanisms. For example, pharmacological concentrations of several catechins have been shown to inhibit the activity of a key regulator of arterial blood pressure, angiotensin-converting enzyme (ACE), in vitro (61). ACE catalyzes the conversion of angiotensin-I into angiotensin-II, a potent inducer of vasoconstriction. In addition, studies in rats showed that chronic treatment with epicatechin prevented salt-induced hypertension, partly through inhibition of endothelin-1 expression and NADPH oxidase (NOX) activity (62). Other potential benefits of green tea consumption, including improvements in blood lipid profile, insulin sensitivity, and endothelial function, may also contribute to its blood pressure-lowering effects.
Type 2 diabetes mellitus
Impaired glucose tolerance in patients with prediabetes is often associated with loss of insulin sensitivity, impaired lipid metabolism, low-grade inflammation, and endothelial dysfunction (63). Without changes in lifestyle behavior (especially regarding dietary habits and physical activity), individuals with prediabetes will eventually progress to develop overt type 2 diabetes mellitus (64).
In this context, the association between tea consumption and risk of type 2 diabetes has been examined in a recent European, multicenter, nested case-control study — the “EPIC-InterAct” project — that included 16,835 diabetes-free participants and 12,043 individuals with diabetes (65). The results showed that tea consumption was inversely associated with diabetes incidence. The consumption of four cups per day rather than none was found to be associated with a 16% lower risk of diabetes (65). Of note, in a meta-analysis of 15 prospective cohort studies, including the EPIC-InterAct study, an incremental increase of two cups per day in tea consumption was found to be associated with an estimated 4.6% risk reduction (66). The EPIC-InterAct study also found that participants in the highest quintile (>608.1 mg/day) of total flavonoid intake had a 10% lower risk of diabetes than those in the lowest quintile (<178.2 mg/day) (67). Specifically, the risk of diabetes was inversely correlated with the consumption of flavan-3-ols (catechins, proanthocyanidins, and theaflavins) and flavonols (67, 68). The intakes of other flavonoid subclasses that are less abundant in tea, namely anthocyanidins, flavanones, flavones, and isoflavones, were not associated with a reduced risk of diabetes (67).
Recent meta-analyses of randomized controlled trials that examined the possible health benefits of green tea catechins on glucose metabolism have provided conflicting results. A meta-analysis of seven trials in prediabetic and diabetic patients found no effect of green tea or green tea extracts on fasting plasma glucose, fasting serum insulin, or measures of glycemic control (HbA1c) and insulin sensitivity (HOMA-IR) (69). Conversely, another meta-analysis of 17 trials in prediabetic, diabetic, or overweight/obese subjects found that administration of green tea extracts for 4 to 16 weeks improved fasting plasma glucose and HbA1c levels (70). The effect on fasting glucose was observed only with high doses of catechins (≥457 mg/day) and when the confounding effect of caffeine was removed. However, a third meta-analysis of 25 trials found that ingestion of green tea extracts for at least two weeks could lower fasting blood glucose in both the presence or absence of caffeine (71).
Overweight and obesity
A recent meta-analysis of five small, randomized controlled trials (<100 participants per study) found that regular consumption of green or pu-erh tea extracts reduced body weight and body mass index (BMI) in overweight/obese participants with metabolic syndrome (72). The influence of green tea on body composition may be attributed to the regulation of appetite, fat absorption, fatty acid oxidation, and thermogenesis by catechins and caffeine (73). Yet, studies in overweight/obese people who are otherwise healthy have provided mixed results (reviewed in 74). Intervention studies in Caucasian populations have shown a less favorable effect of green tea catechins on body weight and energy expenditure compared to those conducted in Asian subjects. These discrepancies suggested that differences in genetic background, body composition, and dietary habits (including caffeine consumption) might interfere with the possible anti-obesity effect of green tea consumption. Large-scale, intervention trials that control for energy intake and physical activity are needed to determine if tea or tea extracts promote weight loss or improve weight maintenance in different populations with obesity and/or metabolic syndrome (74).
Cancer
Tea and tea constituents have been found to have cancer preventive activities in a variety of animal models of cancer, such as cancer of lung, mouth, esophagus, stomach, colon, and prostate (75). However, the results of epidemiological studies in humans have been mostly inconclusive.
Breast cancer
An early meta-analysis of prospective cohort studies had reported that black tea intake (five cohorts) — but not green tea (three cohorts) — was associated with a 15% higher risk of breast cancer (76). The relationship between tea consumption and breast cancer has been recently examined in the Swedish Women’s Lifestyle and Health prospective cohort Study (WLHS), which followed 42,099 women for 20 years and documented 1,395 breast cancer cases (77). The results indicated a 14% higher risk of breast cancer with each cup (200 mL) of tea consumed daily. The risk was specifically increased in postmenopausal women rather than in premenopausal women. Similarly, in a recent case-control study in Chinese women in Hong Kong, regular tea consumption was inversely correlated with breast cancer risk in premenopausal women but associated with an increased risk in postmenopausal women (78). The risk associated with consuming tea was also significantly higher for estrogen- and progesterone-receptor-positive (ER+/PR+) breast cancer type in the Swedish cohort, while the highest risk was found in women with ER-negative tumors in the Chinese study (77, 78). Yet, in the European Prospective Investigation into Nutrition and Cancer Study (EPIC) in 335,060 women followed for 11 years (10,198 incidental breast cancer cases), tea intake was not associated with breast cancer overall or when the data were analyzed for menopausal status or breast cancer type (79). The most recent meta-analysis of prospective cohort studies found no association between consumption of green or black tea and breast cancer (80). Thus, current epidemiological evidence does not suggest a benefit of tea in breast cancer prevention despite promising data from cultured cells and rodent models (81).
However, some observational studies found that consumption of certain subclasses of flavonoids might have the potential to reduce the incidence of breast cancer in postmenopausal women (82). Recently, the effects of decaffeinated green tea extracts on biomarkers of breast cancer risk were examined in the Minnesota Green Tea Trial (MGTT) in 1,075 high-risk postmenopausal women randomized to receive the equivalent of four 8-ounce mugs/day (960 mL/day) in green tea extracts (1,315±116 mg/day of catechins) or a placebo for one year (83). The results have yet to be published.
Mouth, throat, and esophageal cancers
In the large, prospective NIH-AARP Diet and Health study (1995-2006) in 481,563 US adults, 1,305 cases of oral (392), pharyngeal (178), laryngeal (307), and esophageal (428) cancers have been identified during the follow-up period (84). The highest versus lowest level of tea intake (≥1 cup/day vs. non-consumption) was correlated with a 63% lower risk of pharyngeal cancer but with no other above-cited cancer types (84). Observational studies do not currently provide clear evidence for an association between tea consumption and laryngeal (85) or esophageal (86) cancers. In the case of esophageal cancer, the consumption of high-temperature beverages (including very hot tea) might even damage the epithelium and increase the risk of cancer (87). High temperature may act as a confounding factor that complicates the interaction between tea consumption and esophageal cancer (88). In 2009, a phase II, randomized, double-blind trial was conducted in 41 patients with high-risk oral premalignant lesions (OPLs). Participants were randomly assigned to orally receive 0.5, 0.75, or 1 g of green tea extract per m2 (body surface area) or a placebo, thrice a day for three months. While the results suggested that OPLs might be clinically responsive to green tea extract treatment, larger trial populations are needed to confirm these preliminary data (89).
Gastric cancer
Several prospective cohort studies reported no association between tea consumption and risk of gastric cancer (90-92), including the US NIH-AARP Diet and Health study (84). Tea consumption also failed to predict gastric cancer cases in the EPIC study, which followed 477,312 participants and identified 683 cases during a median 11.6 years of follow-up (93). Yet, a decreased risk of intestinal type gastric cancer was observed in women in the highest versus lowest quartile of tea consumption (≥475 mL/day vs. ≤21 mL/day). Interestingly, women (but not men) in the highest versus lowest quartile of flavonol, flavanol, theaflavin, or total flavonoid intakes had a significantly reduced risk of developing gastric cancer (94). A pooled analysis of six Japanese cohort studies, including 219,080 total participants and 3,577 cases, found an inverse association between green tea consumption and gastric cancer in women but not in men: daily consumption of at least five cups of tea was associated with a 21% lower risk of gastric cancer in women compared to low intakes (<1 cup/day) (95).
Gynecologic cancers
Meta-analyses of case-control studies have found a significant 34% reduction in risk of ovarian cancer for highest versus lowest intake of green tea (four studies) (96), but no association was observed for black tea (six studies) (97). Yet, a meta-analysis of six prospective studies showed an inverse relationship between black tea consumption and ovarian cancer (98). A recent analysis of the Nurses’ Health Studies (NHS I and II) showed a 31% lower risk of ovarian cancer in women consuming at least 1 cup/day compared to rare/non-black tea drinkers (99). In a prospective study in 244 women diagnosed with ovarian cancer and followed for over three years, green tea consumption was associated with a mean survival time greater for consumers (5.39 years) than for non-consumers (4.19 years) (100). However, in a single-arm, phase II trial in 16 women in complete remission from advanced stage ovarian cancer, the daily intake of 500 mL of green tea (containing 319.8 mg of EGCG) failed to effectively prevent cancer recurrence within the 18-month follow-up period (101). Further, a recent systematic review and meta-analysis of observational studies suggested a possible benefit of green tea — but not black tea — for endometrial cancer (102).
Additional studies have examined the association between tea consumption and risk of lung, prostate, liver, or colorectal cancer in humans, providing mixed results (reviewed in 103).
Bone health and osteoporosis
The etiology of osteoporosis is complex, involving factors such as aging, decreased sex hormones, inadequate nutrition, physical inactivity, genetic predisposition, as well as socioeconomic determinants. Tea bioactive components, including flavonoids, caffeine, and fluoride, have the potential to influence health and the risk of osteoporosis and fracture (104, 105). A small prospective study in 164 elderly women found that consumption of tea limited the age-related loss in total hip bone mineral density (BMD) over a four-year follow-up period (106). Also, in a six-month randomized, placebo-controlled trial in 171 postmenopausal women with osteopenia, the daily consumption of green tea catechins (500 mg) alone or combined with Tai Chi exercise (3 hours/week) improved bone turnover by stimulating bone formation (107).
Hip fracture is one of the most serious consequences of osteoporosis. A recent prospective study in 1,188 elderly women (mean age, 80 years) followed for 10 years found that participants in the highest versus lowest tertile of tea consumption (≥3 cups/day vs. ≤1 cup/week) had a 30% lower risk of any osteoporotic fracture. However, no interaction was found when the analysis was conducted on major fractures (hip, spine, humerus, and wrist) or hip fractures only (108). Yet, a meta-analysis based on 147,488 individuals from 11 observational studies published between 1990 and 2010 suggested that the consumption of 1-4 cups/day was associated with a significantly lower risk of hip fracture (109). The results of another recent meta-analysis of mostly case-control studies did not suggest any interaction between tea consumption and risk of any fracture or hip fracture (110). Additional studies are required to determine whether tea consumption affects the development of osteoporosis or the risk of osteoporotic fracture in a meaningful way.
Dental health
The cross-sectional analysis of the Ohsaki prospective cohort study that included data from 25,078 Japanese participants found an inverse association between the daily consumption of at least one cup of tea and the risk of tooth loss (111). Specifically, the risk of tooth loss was 11% lower in women and 23% lower in men who consumed at least five cups per day of tea compared to those drinking less than one cup per day. An earlier cross-sectional study of more than 6,000 14-year old children in the UK found that those who drank tea had significantly fewer dental caries than coffee drinkers; results were independent of the amount of beverage consumed or whether sugar was added (112). Although tea is a good source of fluoride — a recognized anticaries agent — both flavonoids and tannins in tea have been shown to have antimicrobial properties (reviewed in 113). Oral bacteria like Streptococcus mutans and Porphyromonas gingivalis have been associated with plaque formation, dental caries, and periodontal (gum) diseases. Untreated caries and gum inflammation can lead to severe pain, local infection, tooth loss or extraction, nutritional problems, and serious systemic infections in susceptible individuals. A pilot study in 25 adults suggested that mouthwash with a 2% green tea solution could lower acidity level and Streptococcus mutans count in saliva and plaque and improve measures of gum bleeding after exposure to sugar (114). A small, randomized study in 66 young volunteers (12-18 years old) also reported a significant antibacterial effect of a mouth rinse made with pulverized tea leaves compared to a placebo solution (115). Recent randomized, double-blind, controlled studies demonstrated further that tea extract-containing mouthwashes could benefit dental health and offer a possible alternative to current chlorhexidine- and fluoride-containing rinsing solutions (116-118). Finally, the incorporation of tea extract in toothpastes was found to be as effective — if not better — than regular pastes (containing fluoride and triclosan) to reduce dental plaque and gum inflammation in patients with mild to moderate periodontitis (119).
For more information on dental caries, see the article on Fluoride.
Kidney stones
The formation of kidney stones, usually composed of calcium oxalate or calcium phosphate, is a common condition that affects 7% of US women and 11% of US men during their lifetime (120). A pooled analysis of three ongoing prospective cohort studies — the Health Professionals Follow-up Study and the Nurses’ Health Studies I and II, including a total of 194,095 participants — found that the risk of developing symptomatic kidney stones was 11% lower in individuals consuming at least one 8-ounce mug of tea per day compared to those consuming less than one cup per week (121). High fluid intake, including tea intake, is generally considered the most effective and economical means of preventing kidney stones (122). However, the finding that black tea may contain high amounts of oxalate (48 to 92 mg/100 mL) suggested that black tea consumption may increase urinary oxalate concentrations, a risk factor for calcium oxalate stone formation (123). The Academy of Nutrition and Dietetics recommends that kidney stone patients restrict oxalate intake to 40 mg/day-50 mg/day, and some experts advise those with a history of calcium oxalate stones to limit the consumption of oxalate-rich food, including black (but not green) tea (123, 124). Yet, recent studies have reported amounts of oxalate in different samples of green teas (0.8 to 14 mg/100 mL) (125) and black teas (1 to 2.6 mg/100 mL) (126, 127) much lower than previously published, suggesting that tea consumption would not increase kidney stone incidence or recurrence.
Mood
The term "mood" refers to an emotional state of mind that includes aspects like contentedness, relaxation, alertness, energy, and relief from depression, anxiety, and feelings of guilt and failure (128). Clinical depression is described as a mood disorder. An analysis of the NIH-AARP Diet and Health Study (1995-2006) in 263,923 participants — of which 11,311 self-reported depression — found that consumption of decaffeinated (but not caffeinated) hot tea was associated with an increased risk for depression (129). However, smaller cohort studies had previously recorded significantly less depressive symptoms in participants with higher versus lower intakes of tea (130, 131).
Tea consumption may have short-term effects on mood. In a recent cross-sectional study in 95 university staff members, consumption of tea recorded during 10 working days was associated with self reports of feeling less tired and performing better at work (132). Tea was also found to increase the positive valence of mood immediately after consumption in a small randomized controlled study in 150 participants (133).
Cognitive function
Cognitive function includes the domains of attention, memory, processing speed, and executive function, which decline gradually with increasing age.
Cognitive performance
A few studies investigated whether tea consumption was associated with cognitive benefits, especially in the domain of attention (reviewed in 134). Two cross-over, randomized, double-blind, placebo-controlled studies evaluated the effects of two servings of black tea over the course of 60 min (study 1; 26 volunteers) or three servings of black tea over the course of 90 min (study 2; 32 volunteers) on measures of attention and alertness (135). Both studies reported improved performance on objective attention tests and self-reported alertness with black tea compared to placebo. In a small open-label study, 19 participants were asked to consume either black tea (with/without caffeine), coffee (with caffeine), or water (with/without caffeine) before undergoing a battery of psychometric tests (136). Most of the improvements in cognitive function (measured with the Critical Flicker Fusion Threshold [CFFT] task) and subjective alertness were attributed to caffeine in the beverages. In addition, CFFT task scores were greater after consumption of caffeinated tea compared to caffeinated water (136). In a follow-up study, caffeinated tea outperformed caffeinated coffee in the CFFT test, suggesting that tea ingredients other than caffeine might have acute effects on cognitive function (137). A recent meta-analysis of small, randomized controlled trials (<50 participants/trial) that measured the acute effect of L-theanine (36 mg-250 mg) with or without caffeine (40 mg-250 mg) suggested an increase in alertness and attention-switching accuracy but no change regarding other parameters, such as calmness, contentedness, or anxiety (138).
Cognitive decline
The cross-sectional data analysis of 2,501 participants (≥55 years old) in the Singapore Longitudinal Ageing Study (SLAS) indicated that higher intakes of tea correlated with better global cognitive function, as assessed by Mini-Mental State Examination (MMSE) scores (139). Conversely, lower levels of tea consumption were associated with a higher prevalence of cognitive impairments, defined as MMSE scores ≤23. Similar observations have been reported in several other cross-sectional studies (140-143). In the SLAS study, the follow-up of 1,438 cognitively healthy people for one to two years showed that the risk of cognitive decline (defined as a drop of ≥1 point in the MMSE score) was up to 43% lower in tea drinkers compared to non-drinkers (139). Further research in 716 SLAS participants (mean age, 64.5 years) with normal cognitive function confirmed that those consuming tea scored higher in the MMSE global cognition test than non-consumers. Tea consumption was also correlated with higher cognitive test scores regarding memory, executive function, and information-processing speed (144). Tea consumption was also modestly associated with a reduced risk of cognitive decline in 2,722 women (but not in men) followed for a median 7.9 years in the US population-based Cardiovascular Health Study, despite a much lower frequency of tea consumption (up to 5 cups/week) than that observed in the SLAS (over 10 cups/day) (145). In a prospective cohort study in Japanese older people (>60 years old) followed for nearly five years, daily green tea consumers were shown to have lower risks of mild cognitive impairments (MCI) and dementia compared to non-consumers (146). In a recent pilot trial, the consumption of green tea extracts (2 g/day, of which 227 mg of catechins and 42 mg of theanine) for three months resulted in higher MMSE scores (compared to baseline) due to improved short-term memory scores in 12 elderly nursing home residents (ages, 70 to 98 years) with symptoms from MCI to severe dementia (147).
Long-term, large randomized controlled trials are needed to establish whether tea or its bioactive components could limit cognitive decline and/or improve cognitive dysfunction in older individuals.
Parkinson’s disease
Parkinson’s disease (PD) is a neurodegenerative disease characterized by the selective death of dopaminergic brain cells in the substantia nigra. PD is estimated to affect 0.5%-4% of older people (≥65 years old) worldwide (148). A retrospective study in 279 subjects with PD suggested that the onset of motor symptoms in those drinking more than 3 cups/day of tea was delayed by several years compared to nondrinkers (149); yet, after disease onset, a similar rate of disease progression was observed among tea drinkers and non-drinkers (150). A meta-analysis of eight case-control studies, including 4,250 controls and 1,418 PD cases, found a 15% reduced risk of PD with higher versus lower intake of tea (151). Another meta-analysis of four case-control studies and four prospective cohort studies published between 1999 and 2012 indicated that individuals in the highest category of tea consumption had a 37% lower risk of PD compared to those in the lowest category (152). The authors estimated that each 2 cups/day-increase in tea consumption was associated with a 26% lower risk of developing PD (152). If a protective effect of tea consumption can be further demonstrated, several bioactive compounds, especially caffeine (153) and flavonoids (154), could be responsible for the tea benefits in PD prevention.
Safety
Adverse effects
Tea
Tea is generally considered to be safe, even in large amounts. However, two cases of hypokalemia (abnormally low serum potassium concentrations) in the elderly have been attributed to excessive consumption of black and oolong tea (3 L/day-14 L/day) (155, 156). Hypokalemia is a potentially life-threatening condition that has been associated with caffeine toxicity (157, 158). Case reports of stomach cramps (159), kidney stones (160), and skeletal fluorosis (161-163) due to excessive tea consumption have also been published.
Tea extracts
In clinical trials employing caffeinated green tea extracts, cancer patients who took 6 g/day, in three to six divided doses, experienced mild-to-moderate gastrointestinal side effects, including nausea, vomiting, abdominal pain, and diarrhea (164, 165). Central nervous system symptoms, including agitation, restlessness, insomnia, tremors, dizziness, and confusion, have also been reported. In one case, confusion was severe enough to require hospitalization (164). In a systematic review published in 2008, the US Pharmacopeia (USP) Dietary Supplement Information Expert Committee identified 34 adverse event reports implicating the use of green tea extract products (containing 25%-97% of polyphenols) as the likely cause of liver damage (hepatotoxicity) in humans (166). Nineteen additional cases of hepatotoxicity associated with the consumption of herbal products containing green tea have been reported for the period 2008-2015 (167). In a four-week clinical trial that assessed the safety of decaffeinated green tea extracts (800 mg/day of EGCG) in healthy individuals, a few of the participants reported mild nausea, stomach upset, dizziness, or muscle pain (168). In the Minnesota Green Tea Trial (MGTT), 1,075 postmenopausal women were randomized to receive green tea extracts (1,315±116 mg/day of catechins; the equivalent of four 8-ounce mugs of brewed decaffeinated green tea) or a placebo for one year. The total number of adverse events and the number of serious adverse events were not different between the treatment and placebo groups (169). However, the use of green tea extracts was directly associated with abnormally high liver enzyme levels in 7 out of the 12 women who experienced serious adverse events. Also, the incidence of nausea was twice as high in the green tea arm as in the placebo group (169).
Pregnancy and lactation
The safety of tea extracts or supplements for pregnant or breast-feeding women has not been established. Some organizations, like the American College of Obstetricians and Gynecologists, suggest to limit caffeine consumption during pregnancy to less than 200 mg/day (170), because higher caffeine intakes have been associated with increased risk of miscarriage and low birth weight in some epidemiological studies (171, 172).
Drug interactions
Green tea
Excessive green tea consumption may decrease the therapeutic effects of the anticoagulant, warfarin (Coumadin, Jantoven). Such an effect was documented in only one patient who began drinking one-half gallon to one gallon of green tea daily (173). It is probably not necessary for people on warfarin therapy to avoid green tea entirely; however, large quantities of green tea may increase the risk of bleeding in warfarin-treated patients (174). Green tea extracts may also reduce the efficacy or increase the toxicity of at least two other cardiovascular drugs, namely simvastatin (Zocor) and nadolol (Corgard) (175). Preclinical studies suggested that green tea extracts may interfere with drug metabolism by affecting the activity of cytochrome P450 3A4 (CYP3A4), which catalyzes the metabolism of about one-half of all marketed drugs in the US and Canada (176). Additional information on drug interactions is available in the article on Flavonoids.
Caffeine
A number of drugs can impair the metabolism of caffeine, increasing the potential for adverse effects from caffeine (177). Such drugs include cimetidine (Tagamet), disulfiram (Antabuse), estrogens, fluoroquinolone antibiotics (e.g., ciprofloxacin, enoxacin, norfloxacin), fluconazole (Diflucan), fluvoxamine (Luvox), mexiletine (Mexitil), riluzole (Rilutek), terbinafine (Lamisil), and verapamil (Calan). High caffeine intakes may increase the risk of toxicity of some drugs, including albuterol (Ventolin), metaproterenol (Alupent), clozapine (Clozaril), ephedrine, stimulant drugs (e.g., epinephrine), monoamine oxidase inhibitors, phenylpropanolamine, and theophylline. High caffeine intakes may also reduce the bioavailability and/or the efficacy of drugs like carbamazepine, valproate, dipyridamole (Persantine), pentobarbital (Nembutal), and phenobarbital (Luminal). Abrupt caffeine withdrawal has been found to increase serum lithium levels in people taking lithium, potentially increasing the risk of lithium toxicity.
Nutrient interactions
Iron
Flavonoids in tea can bind nonheme iron, inhibiting its intestinal absorption (178, 179). Nonheme iron is the principal form of iron in plant foods, dairy products, and iron supplements. The consumption of one cup of tea with a meal has been found to decrease the absorption of nonheme iron in that meal by about 70% (180, 181). Flavonoids can also inhibit intestinal heme iron absorption (182). Interestingly, ascorbic acid (vitamin C) greatly enhances the absorption of iron and is able to counteract the inhibitory effect of flavonoids on nonheme and heme iron absorption (179, 182, 183). To maximize iron absorption from a meal or iron supplements, subjects with poor iron status should not consumed tea at the same time (184). In addition, healthy individuals at no risk of iron deficiency do not need to restrict their consumption of tea (184, 185).
Authors and Reviewers
Written in January 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in January 2008 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in October 2015 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in January 2016 by:
Richard Draijer, Ph.D.
Lead Scientist, Unilever R&D
Vlaardingen, The Netherlands
Reviewed in January 2016 by:
Guus Duchateau, Ph.D.
Science Leader, Unilever R&D
Vlaardingen, The Netherlands
Reviewed in January 2016 by:
Suzanne Einöther
Scientist, Unilever R&D
Vlaardingen, The Netherlands
Copyright 2002-2024 Linus Pauling Institute
References
1. Vuong QV. Epidemiological evidence linking tea consumption to human health: a review. Crit Rev Food Sci Nutr. 2014;54(4):523-536. (PubMed)
2. Tea Association of the USA. Tea Factsheet 2014. Available at: http://www.teausa.com/14655/tea-fact-sheet. Accessed 1/19/16.
3. Drynan JW, Clifford MN, Obuchowicz J, Kuhnert N. The chemistry of low molecular weight black tea polyphenols. Nat Prod Rep. 2010;27(3):417-462. (PubMed)
4. Butt MS, Imran A, Sharif MK, et al. Black tea polyphenols: a mechanistic treatise. Crit Rev Food Sci Nutr. 2014;54(8):1002-1011. (PubMed)
5. Sang S, Lambert JD, Ho CT, Yang CS. The chemistry and biotransformation of tea constituents. Pharmacol Res. 2011;64(2):87-99. (PubMed)
6. Unachukwu UJ, Ahmed S, Kavalier A, Lyles JT, Kennelly EJ. White and green teas (Camellia sinensis var. sinensis): variation in phenolic, methylxanthine, and antioxidant profiles. J Food Sci. 2010;75(6):C541-548. (PubMed)
7. Yang Z, Jie G, Dong F, Xu Y, Watanabe N, Tu Y. Radical-scavenging abilities and antioxidant properties of theaflavins and their gallate esters in H2O2-mediated oxidative damage system in the HPF-1 cells. Toxicol In Vitro. 2008;22(5):1250-1256. (PubMed)
8. Yashin AY, Nemzer BV, Combet E, Yashin YI. Determination of the Chemical Composition of Tea by Chromatographic Methods: A Review. J Food Res. 2015;4(3):56-88.
9. Bhagwat, S., Haytowitz, D.B. Holden, J.M. (Ret.). 2014. USDA Database for the Flavonoid Content of Selected Foods, Release 3.1. US Department of Agriculture, Agricultural Research Service. Nutrient Data Laboratory Home Page: http://www.ars.usda.gov/nutrientdata/flav. Accessed 1/19/15.
10. van Duynhoven J, Vaughan EE, van Dorsten F, et al. Interactions of black tea polyphenols with human gut microbiota: implications for gut and cardiovascular health. Am J Clin Nutr. 2013;98(6 Suppl):1631S-1641S. (PubMed)
11. Lakenbrink C, Lapczynski S, Maiwald B, Engelhardt UH. Flavonoids and other polyphenols in consumer brews of tea and other caffeinated beverages. J Agric Food Chem. 2000;48(7):2848-2852. (PubMed)
12. Astill C, Birch MR, Dacombe C, Humphrey PG, Martin PT. Factors affecting the caffeine and polyphenol contents of black and green tea infusions. J Agric Food Chem. 2001;49(11):5340-5347. (PubMed)
13. Lin JK, Lin CL, Liang YC, Lin-Shiau SY, Juan IM. Survey of catechins, gallic acid, and methylxanthines in green, oolong, pu-erh, and black teas. J Agric Food Chem. 1998;46(9):3635-3642.
14. Chin JM, Merves ML, Goldberger BA, Sampson-Cone A, Cone EJ. Caffeine content of brewed teas. J Anal Toxicol. 2008;32(8):702-704. (PubMed)
15. Lin YS, Tsai YJ, Tsay JS, Lin JK. Factors affecting the levels of tea polyphenols and caffeine in tea leaves. J Agric Food Chem. 2003;51(7):1864-1873. (PubMed)
16. Santana-Rios G, Orner GA, Amantana A, Provost C, Wu SY, Dashwood RH. Potent antimutagenic activity of white tea in comparison with green tea in the Salmonella assay. Mutat Res. 2001;495(1-2):61-74. (PubMed)
17. McCusker RR, Goldberger BA, Cone EJ. Caffeine content of specialty coffees. J Anal Toxicol. 2003;27(7):520-522. (PubMed)
18. Nehlig A. Effects of coffee/caffeine on brain health and disease: What should I tell my patients? Pract Neurol. 2015; pii: practneurol-2015-001162. doi: 10.1136/practneurol-2015-001162. [Epub ahead of print]. (PubMed)
19. Wong MH, Fung KF, Carr HP. Aluminium and fluoride contents of tea, with emphasis on brick tea and their health implications. Toxicol Lett. 2003;137(1-2):111-120. (PubMed)
20. Fung KF, Zhang ZQ, Wong JWC, Wong MH. Fluoride contents in tea and soil from tea plantations and the release of fluoride into tea liquor during infusion. Environmental Pollution. 1999;104(2):197-205.
21. Cao J, Luo SF, Liu JW, Li YH. Safety evaluation on fluoride content in black tea. Food Chemistry. 2004;88(2):233-236.
22. Lu Y, Guo WF, Yang XQ. Fluoride content in tea and its relationship with tea quality. J Agric Food Chem. 2004;52(14):4472-4476. (PubMed)
23. Nobre AC, Rao A, Owen GN. L-theanine, a natural constituent in tea, and its effect on mental state. Asia Pac J Clin Nutr. 2008;17 Suppl 1:167-168. (PubMed)
24. van der Pijla PC, Chenb L, Mulder TPJ. Human disposition of L-theanine in tea or aqueous solution. Journal of functional foods. 2010;2(4):239-244.
25. Kakuda T. Neuroprotective effects of theanine and its preventive effects on cognitive dysfunction. Pharmacol Res. 2011;64(2):162-168. (PubMed)
26. Grassi D, Desideri G, Di Giosia P, et al. Tea, flavonoids, and cardiovascular health: endothelial protection. Am J Clin Nutr. 2013;98(6 Suppl):1660S-1666S. (PubMed)
27. Zhang C, Qin YY, Wei X, Yu FF, Zhou YH, He J. Tea consumption and risk of cardiovascular outcomes and total mortality: a systematic review and meta-analysis of prospective observational studies. Eur J Epidemiol. 2015;30(2):103-113. (PubMed)
28. Tang J, Zheng JS, Fang L, Jin Y, Cai W, Li D. Tea consumption and mortality of all cancers, CVD and all causes: a meta-analysis of eighteen prospective cohort studies. Br J Nutr. 2015:1-11. (PubMed)
29. Kim K, Vance TM, Chun OK. Estimated intake and major food sources of flavonoids among US adults: changes between 1999-2002 and 2007-2010 in NHANES. Eur J Nutr. 2015. [Epub ahead of print]. (PubMed)
30. Vogiatzoglou A, Mulligan AA, Lentjes MA, et al. Flavonoid intake in European adults (18 to 64 years). PLoS One. 2015;10(5):e0128132. (PubMed)
31. Wang X, Ouyang YY, Liu J, Zhao G. Flavonoid intake and risk of CVD: a systematic review and meta-analysis of prospective cohort studies. Br J Nutr. 2014;111(1):1-11. (PubMed)
32. Kim A, Chiu A, Barone MK, et al. Green tea catechins decrease total and low-density lipoprotein cholesterol: a systematic review and meta-analysis. J Am Diet Assoc. 2011;111(11):1720-1729. (PubMed)
33. Wang D, Chen C, Wang Y, Liu J, Lin R. Effect of black tea consumption on blood cholesterol: a meta-analysis of 15 randomized controlled trials. PLoS One. 2014;9(9):e107711. (PubMed)
34. Zhao Y, Asimi S, Wu K, Zheng J, Li D. Black tea consumption and serum cholesterol concentration: Systematic review and meta-analysis of randomized controlled trials. Clin Nutr. 2015;34(4):612-619. (PubMed)
35. Mukamal KJ, MacDermott K, Vinson JA, Oyama N, Manning WJ, Mittleman MA. A 6-month randomized pilot study of black tea and cardiovascular risk factors. Am Heart J. 2007;154(4):724 e721-726. (PubMed)
36. Fujita H, Yamagami T. Antihypercholesterolemic effect of Chinese black tea extract in human subjects with borderline hypercholesterolemia. Nutr Res. 2008;28(7):450-456. (PubMed)
37. Bahorun T, Luximon-Ramma A, Neergheen-Bhujun VS, et al. The effect of black tea on risk factors of cardiovascular disease in a normal population. Prev Med. 2012;54 Suppl:S98-102. (PubMed)
38. Bahorun T, Luximon-Ramma A, Gunness TK, et al. Black tea reduces uric acid and C-reactive protein levels in humans susceptible to cardiovascular diseases. Toxicology. 2010;278(1):68-74. (PubMed)
39. Bohn SK, Croft KD, Burrows S, et al. Effects of black tea on body composition and metabolic outcomes related to cardiovascular disease risk: a randomized controlled trial. Food Funct. 2014;5(7):1613-1620. (PubMed)
40. Maron DJ, Lu GP, Cai NS, et al. Cholesterol-lowering effect of a theaflavin-enriched green tea extract: a randomized controlled trial. Arch Intern Med. 2003;163(12):1448-1453. (PubMed)
41. Stendell-Hollis NR, Thomson CA, Thompson PA, Bea JW, Cussler EC, Hakim IA. Green tea improves metabolic biomarkers, not weight or body composition: a pilot study in overweight breast cancer survivors. J Hum Nutr Diet. 2010;23(6):590-600. (PubMed)
42. Bogdanski P, Suliburska J, Szulinska M, Stepien M, Pupek-Musialik D, Jablecka A. Green tea extract reduces blood pressure, inflammatory biomarkers, and oxidative stress and improves parameters associated with insulin resistance in obese, hypertensive patients. Nutr Res. 2012;32(6):421-427. (PubMed)
43. Forstermann U, Sessa WC. Nitric oxide synthases: regulation and function. Eur Heart J. 2012;33(7):829-837, 837a-837d. (PubMed)
44. Ras RT, Streppel MT, Draijer R, Zock PL. Flow-mediated dilation and cardiovascular risk prediction: a systematic review with meta-analysis. Int J Cardiol. 2013;168(1):344-351. (PubMed)
45. Duffy SJ, Keaney JF, Jr., Holbrook M, et al. Short- and long-term black tea consumption reverses endothelial dysfunction in patients with coronary artery disease. Circulation. 2001;104(2):151-156. (PubMed)
46. Hodgson JM, Puddey IB, Burke V, Watts GF, Beilin LJ. Regular ingestion of black tea improves brachial artery vasodilator function. Clin Sci (Lond). 2002;102(2):195-201.
47. Grassi D, Mulder TP, Draijer R, Desideri G, Molhuizen HO, Ferri C. Black tea consumption dose-dependently improves flow-mediated dilation in healthy males. J Hypertens. 2009;27(4):774-781. (PubMed)
48. Schreuder TH, Eijsvogels TM, Greyling A, Draijer R, Hopman MT, Thijssen DH. Effect of black tea consumption on brachial artery flow-mediated dilation and ischaemia-reperfusion in humans. Appl Physiol Nutr Metab. 2014;39(2):145-151. (PubMed)
49. Alexopoulos N, Vlachopoulos C, Aznaouridis K, et al. The acute effect of green tea consumption on endothelial function in healthy individuals. Eur J Cardiovasc Prev Rehabil. 2008;15(3):300-305. (PubMed)
50. Jochmann N, Baumann G, Stangl V. Green tea and cardiovascular disease: from molecular targets towards human health. Curr Opin Clin Nutr Metab Care. 2008;11(6):758-765. (PubMed)
51. Lorenz M, Urban J, Engelhardt U, Baumann G, Stangl K, Stangl V. Green and black tea are equally potent stimuli of NO production and vasodilation: new insights into tea ingredients involved. Basic Res Cardiol. 2009;104(1):100-110. (PubMed)
52. Dower JI, Geleijnse JM, Gijsbers L, Zock PL, Kromhout D, Hollman PC. Effects of the pure flavonoids epicatechin and quercetin on vascular function and cardiometabolic health: a randomized, double-blind, placebo-controlled, crossover trial. Am J Clin Nutr. 2015;101(5):914-921. (PubMed)
53. Ras RT, Zock PL, Draijer R. Tea consumption enhances endothelial-dependent vasodilation; a meta-analysis. PLoS One. 2011;6(3):e16974. (PubMed)
54. Greyling A, Ras RT, Zock PL, et al. The effect of black tea on blood pressure: a systematic review with meta-analysis of randomized controlled trials. PLoS One. 2014;9(7):e103247. (PubMed)
55. Hodgson JM, Croft KD, Woodman RJ, et al. Black tea lowers the rate of blood pressure variation: a randomized controlled trial. Am J Clin Nutr. 2013;97(5):943-950. (PubMed)
56. Grassi D, Draijer R, Desideri G, Mulder T, Ferri C. Black tea lowers blood pressure and wave reflections in fasted and postprandial conditions in hypertensive patients: a randomised study. Nutrients. 2015;7(2):1037-1051. (PubMed)
57. Khalesi S, Sun J, Buys N, Jamshidi A, Nikbakht-Nasrabadi E, Khosravi-Boroujeni H. Green tea catechins and blood pressure: a systematic review and meta-analysis of randomised controlled trials. Eur J Nutr. 2014;53(6):1299-1311. (PubMed)
58. Li G, Zhang Y, Thabane L, et al. Effect of green tea supplementation on blood pressure among overweight and obese adults: a systematic review and meta-analysis. J Hypertens. 2015;33(2):243-254. (PubMed)
59. Onakpoya I, Spencer E, Heneghan C, Thompson M. The effect of green tea on blood pressure and lipid profile: a systematic review and meta-analysis of randomized clinical trials. Nutr Metab Cardiovasc Dis. 2014;24(8):823-836. (PubMed)
60. Peng X, Zhou R, Wang B, et al. Effect of green tea consumption on blood pressure: a meta-analysis of 13 randomized controlled trials. Sci Rep. 2014;4:6251. (PubMed)
61. Guerrero L, Castillo J, Quinones M, et al. Inhibition of angiotensin-converting enzyme activity by flavonoids: structure-activity relationship studies. PLoS One. 2012;7(11):e49493. (PubMed)
62. Gomez-Guzman M, Jimenez R, Sanchez M, et al. Epicatechin lowers blood pressure, restores endothelial function, and decreases oxidative stress and endothelin-1 and NADPH oxidase activity in DOCA-salt hypertension. Free Radic Biol Med. 2012;52(1):70-79. (PubMed)
63. Laakso M. Cardiovascular disease in type 2 diabetes from population to man to mechanisms: the Kelly West Award Lecture 2008. Diabetes Care. 2010;33(2):442-449. (PubMed)
64. Tuso P. Prediabetes and lifestyle modification: time to prevent a preventable disease. Perm J. 2014;18(3):88-93. (PubMed)
65. InterAct Consortium, van Woudenbergh GJ, Kuijsten A, et al. Tea consumption and incidence of type 2 diabetes in Europe: the EPIC-InterAct case-cohort study. PLoS One. 2012;7(5):e36910. (PubMed)
66. Yang WS, Wang WY, Fan WY, Deng Q, Wang X. Tea consumption and risk of type 2 diabetes: a dose-response meta-analysis of cohort studies. Br J Nutr. 2014;111(8):1329-1339. (PubMed)
67. Zamora-Ros R, Forouhi NG, Sharp SJ, et al. The association between dietary flavonoid and lignan intakes and incident type 2 diabetes in European populations: the EPIC-InterAct study. Diabetes Care. 2013;36(12):3961-3970. (PubMed)
68. Zamora-Ros R, Forouhi NG, Sharp SJ, et al. Dietary intakes of individual flavanols and flavonols are inversely associated with incident type 2 diabetes in European populations. J Nutr. 2014;144(3):335-343. (PubMed)
69. Wang X, Tian J, Jiang J, et al. Effects of green tea or green tea extract on insulin sensitivity and glycaemic control in populations at risk of type 2 diabetes mellitus: a systematic review and meta-analysis of randomised controlled trials. J Hum Nutr Diet. 2014;27(5):501-512. (PubMed)
70. Liu K, Zhou R, Wang B, et al. Effect of green tea on glucose control and insulin sensitivity: a meta-analysis of 17 randomized controlled trials. Am J Clin Nutr. 2013;98(2):340-348. (PubMed)
71. Zheng XX, Xu YL, Li SH, Hui R, Wu YJ, Huang XH. Effects of green tea catechins with or without caffeine on glycemic control in adults: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2013;97(4):750-762. (PubMed)
72. Zhong X, Zhang T, Liu Y, et al. Short-term weight-centric effects of tea or tea extract in patients with metabolic syndrome: a meta-analysis of randomized controlled trials. Nutr Diabetes. 2015;5:e160. (PubMed)
73. Rains TM, Agarwal S, Maki KC. Antiobesity effects of green tea catechins: a mechanistic review. J Nutr Biochem. 2011;22(1):1-7. (PubMed)
74. Huang J, Wang Y, Xie Z, Zhou Y, Zhang Y, Wan X. The anti-obesity effects of green tea in human intervention and basic molecular studies. Eur J Clin Nutr. 2014;68(10):1075-1087. (PubMed)
75. Yang CS, Wang X, Lu G, Picinich SC. Cancer prevention by tea: animal studies, molecular mechanisms and human relevance. Nat Rev Cancer. 2009;9(6):429-439. (PubMed)
76. Sun CL, Yuan JM, Koh WP, Yu MC. Green tea, black tea and breast cancer risk: a meta-analysis of epidemiological studies. Carcinogenesis. 2006;27(7):1310-1315. (PubMed)
77. Oh JK, Sandin S, Strom P, Lof M, Adami HO, Weiderpass E. Prospective study of breast cancer in relation to coffee, tea and caffeine in Sweden. Int J Cancer. 2015;137(8):1979-1989. (PubMed)
78. Li M, Tse LA, Chan WC, et al. Evaluation of breast cancer risk associated with tea consumption by menopausal and estrogen receptor status among Chinese women in Hong Kong. Cancer Epidemiol. 2015;40:73-78. (PubMed)
79. Bhoo-Pathy N, Peeters PH, Uiterwaal CS, et al. Coffee and tea consumption and risk of pre- and postmenopausal breast cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort study. Breast Cancer Res. 2015;17:15. (PubMed)
80. Wu Y, Zhang D, Kang S. Black tea, green tea and risk of breast cancer: an update. Springerplus. 2013;2(1):240. (PubMed)
81. Li MJ, Yin YC, Wang J, Jiang YF. Green tea compounds in breast cancer prevention and treatment. World J Clin Oncol. 2014;5(3):520-528. (PubMed)
82. Hui C, Qi X, Qianyong Z, Xiaoli P, Jundong Z, Mantian M. Flavonoids, flavonoid subclasses and breast cancer risk: a meta-analysis of epidemiologic studies. PLoS One. 2013;8(1):e54318. (PubMed)
83. Samavat H, Dostal AM, Wang R, et al. The Minnesota Green Tea Trial (MGTT), a randomized controlled trial of the efficacy of green tea extract on biomarkers of breast cancer risk: study rationale, design, methods, and participant characteristics. Cancer Causes Control. 2015;26(10):1405-1419. (PubMed)
84. Ren JS, Freedman ND, Kamangar F, et al. Tea, coffee, carbonated soft drinks and upper gastrointestinal tract cancer risk in a large United States prospective cohort study. Eur J Cancer. 2010;46(10):1873-1881. (PubMed)
85. Chen J, Long S. Tea and coffee consumption and risk of laryngeal cancer: a systematic review meta-analysis. PLoS One. 2014;9(12):e112006. (PubMed)
86. Zheng JS, Yang J, Fu YQ, Huang T, Huang YJ, Li D. Effects of green tea, black tea, and coffee consumption on the risk of esophageal cancer: a systematic review and meta-analysis of observational studies. Nutr Cancer. 2013;65(1):1-16. (PubMed)
87. Islami F, Boffetta P, Ren JS, Pedoeim L, Khatib D, Kamangar F. High-temperature beverages and foods and esophageal cancer risk--a systematic review. Int J Cancer. 2009;125(3):491-524. (PubMed)
88. Yuan JM. Green tea and prevention of esophageal and lung cancers. Mol Nutr Food Res. 2011;55(6):886-904. (PubMed)
89. Tsao AS, Liu D, Martin J, et al. Phase II randomized, placebo-controlled trial of green tea extract in patients with high-risk oral premalignant lesions. Cancer Prev Res (Phila). 2009;2(11):931-941. (PubMed)
90. Goldbohm RA, Hertog MG, Brants HA, van Poppel G, van den Brandt PA. Consumption of black tea and cancer risk: a prospective cohort study. J Natl Cancer Inst. 1996;88(2):93-100. (PubMed)
91. Heilbrun LK, Nomura A, Stemmermann GN. Black tea consumption and cancer risk: a prospective study. Br J Cancer. 1986;54(4):677-683. (PubMed)
92. Tsubono Y, Nishino Y, Komatsu S, et al. Green tea and the risk of gastric cancer in Japan. N Engl J Med. 2001;344(9):632-636. (PubMed)
93. Sanikini H, Dik VK, Siersema PD, et al. Total, caffeinated and decaffeinated coffee and tea intake and gastric cancer risk: results from the EPIC cohort study. Int J Cancer. 2015;136(6):E720-730. (PubMed)
94. Zamora-Ros R, Agudo A, Lujan-Barroso L, et al. Dietary flavonoid and lignan intake and gastric adenocarcinoma risk in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Am J Clin Nutr. 2012;96(6):1398-1408. (PubMed)
95. Sasazuki S, Tamakoshi A, Matsuo K, et al. Green tea consumption and gastric cancer risk: an evaluation based on a systematic review of epidemiologic evidence among the Japanese population. Jpn J Clin Oncol. 2012;42(4):335-346. (PubMed)
96. Butler LM, Wu AH. Green and black tea in relation to gynecologic cancers. Mol Nutr Food Res. 2011;55(6):931-940. (PubMed)
97. Zhou B, Yang L, Wang L, et al. The association of tea consumption with ovarian cancer risk: A metaanalysis. Am J Obstet Gynecol. 2007;197(6):594 e591-596. (PubMed)
98. Steevens J, Schouten LJ, Verhage BA, Goldbohm RA, van den Brandt PA. Tea and coffee drinking and ovarian cancer risk: results from the Netherlands Cohort Study and a meta-analysis. Br J Cancer. 2007;97(9):1291-1294. (PubMed)
99. Cassidy A, Huang T, Rice MS, Rimm EB, Tworoger SS. Intake of dietary flavonoids and risk of epithelial ovarian cancer. Am J Clin Nutr. 2014;100(5):1344-1351. (PubMed)
100. Zhang M, Lee AH, Binns CW, Xie X. Green tea consumption enhances survival of epithelial ovarian cancer. Int J Cancer. 2004;112(3):465-469. (PubMed)
101. Trudel D, Labbe DP, Araya-Farias M, et al. A two-stage, single-arm, phase II study of EGCG-enriched green tea drink as a maintenance therapy in women with advanced stage ovarian cancer. Gynecol Oncol. 2013;131(2):357-361. (PubMed)
102. Zhou Q, Li H, Zhou JG, Ma Y, Wu T, Ma H. Green tea, black tea consumption and risk of endometrial cancer: a systematic review and meta-analysis. Arch Gynecol Obstet. 2015;293(1):143-155. (PubMed)
103. Yuan JM. Cancer prevention by green tea: evidence from epidemiologic studies. Am J Clin Nutr. 2013;98(6 Suppl):1676S-1681S. (PubMed)
104. Dew TP, Day AJ, Morgan MR. Bone mineral density, polyphenols and caffeine: a reassessment. Nutr Res Rev. 2007;20(1):89-105. (PubMed)
105. Nash LA, Ward WE. Tea and Bone Health: Findings from Human Studies, Potential Mechanisms, and Identification of Knowledge Gaps. Crit Rev Food Sci Nutr. 2015:0. [Epub ahead of print]. (PubMed)
106. Devine A, Hodgson JM, Dick IM, Prince RL. Tea drinking is associated with benefits on bone density in older women. Am J Clin Nutr. 2007;86(4):1243-1247. (PubMed)
107. Shen CL, Chyu MC, Yeh JK, et al. Effect of green tea and Tai Chi on bone health in postmenopausal osteopenic women: a 6-month randomized placebo-controlled trial. Osteoporos Int. 2012;23(5):1541-1552. (PubMed)
108. Myers G, Prince RL, Kerr DA, et al. Tea and flavonoid intake predict osteoporotic fracture risk in elderly Australian women: a prospective study. Am J Clin Nutr. 2015;102(4):958-965. (PubMed)
109. Sheng J, Qu X, Zhang X, et al. Coffee, tea, and the risk of hip fracture: a meta-analysis. Osteoporos Int. 2014;25(1):141-150. (PubMed)
110. Chen IJ, Liu CY, Chiu JP, Hsu CH. Therapeutic effect of high-dose green tea extract on weight reduction: A randomized, double-blind, placebo-controlled clinical trial. Clin Nutr. 2015; pii: S0261-5614(15)00134-X. doi: 10.1016/j.clnu.2015.05.003. [Epub ahead of print]. (PubMed)
111. Koyama Y, Kuriyama S, Aida J, et al. Association between green tea consumption and tooth loss: cross-sectional results from the Ohsaki Cohort 2006 Study. Prev Med. 2010;50(4):173-179. (PubMed)
112. Jones C, Woods K, Whittle G, Worthington H, Taylor G. Sugar, drinks, deprivation and dental caries in 14-year-old children in the north west of England in 1995. Community Dent Health. 1999;16(2):68-71. (PubMed)
113. Goenka P, Sarawgi A, Karun V, Nigam AG, Dutta S, Marwah N. Camellia sinensis (Tea): Implications and role in preventing dental decay. Pharmacogn Rev. 2013;7(14):152-156. (PubMed)
114. Awadalla HI, Ragab MH, Bassuoni MW, Fayed MT, Abbas MO. A pilot study of the role of green tea use on oral health. Int J Dent Hyg. 2011;9(2):110-116. (PubMed)
115. Ferrazzano GF, Roberto L, Amato I, Cantile T, Sangianantoni G, Ingenito A. Antimicrobial properties of green tea extract against cariogenic microflora: an in vivo study. J Med Food. 2011;14(9):907-911. (PubMed)
116. Hambire CU, Jawade R, Patil A, Wani VR, Kulkarni AA, Nehete PB. Comparing the antiplaque efficacy of 0.5% Camellia sinensis extract, 0.05% sodium fluoride, and 0.2% chlorhexidine gluconate mouthwash in children. J Int Soc Prev Community Dent. 2015;5(3):218-226. (PubMed)
117. Radafshar G, Ghotbizadeh M, Saadat F, Mirfarhadi N. Effects of green tea (Camellia sinensis) mouthwash containing 1% tannin on dental plaque and chronic gingivitis: a double-blinded, randomized, controlled trial. J Investig Clin Dent. 2015 Aug 14. doi: 10.1111/jicd.12184. [Epub ahead of print]. (PubMed)
118. Sarin S, Marya C, Nagpal R, Oberoi SS, Rekhi A. Preliminary Clinical Evidence of the Antiplaque, Antigingivitis Efficacy of a Mouthwash Containing 2% Green Tea - A Randomised Clinical Trial. Oral Health Prev Dent. 2015;13(3):197-203. (PubMed)
119. Hrishi T, Kundapur P, Naha A, Thomas B, Kamath S, Bhat G. Effect of adjunctive use of green tea dentifrice in periodontitis patients - A Randomized Controlled Pilot Study. Int J Dent Hyg. 2015. (PubMed)
120. Scales CD, Jr., Smith AC, Hanley JM, Saigal CS, Urologic Diseases in America P. Prevalence of kidney stones in the United States. Eur Urol. 2012;62(1):160-165. (PubMed)
121. Ferraro PM, Taylor EN, Gambaro G, Curhan GC. Soda and other beverages and the risk of kidney stones. Clin J Am Soc Nephrol. 2013;8(8):1389-1395. (PubMed)
122. Xu C, Zhang C, Wang XL, et al. Self-Fluid Management in Prevention of Kidney Stones: A PRISMA-Compliant Systematic Review and Dose-Response Meta-Analysis of Observational Studies. Medicine (Baltimore). 2015;94(27):e1042. (PubMed)
123. Massey LK. Food oxalate: factors affecting measurement, biological variation, and bioavailability. J Am Diet Assoc. 2007;107(7):1191-1194; quiz 1195-1196. (PubMed)
124. Komaroff AL. By the way, doctor. Two of my friends suffer from kidney stones. Each was advised to give up tea. I drink a lot of tea. Am I in danger of getting kidney stones? Harv Health Lett. 2007;32(3):8. (PubMed)
125. Honow R, Gu KL, Hesse A, Siener R. Oxalate content of green tea of different origin, quality, preparation and time of harvest. Urol Res. 2010;38(5):377-381. (PubMed)
126. Lotfi Yagin N, Mahdavi R, Nikniaz Z. Oxalate content of different drinkable dilutions of tea infusions after different brewing times. Health Promot Perspect. 2012;2(2):218-222. (PubMed)
127. Mahdavi R, Lotfi Yagin N, Liebman M, Nikniaz Z. Effect of different brewing times on soluble oxalate content of loose-packed black teas and tea bags. Urolithiasis. 2013;41(1):15-19. (PubMed)
128. Appleton M, Rogers PJ. Food and mood. Womens Health Med. 2004;1:4-6.
129. Guo X, Park Y, Freedman ND, et al. Sweetened beverages, coffee, and tea and depression risk among older US adults. PLoS One. 2014;9(4):e94715. (PubMed)
130. Chen X, Lu W, Zheng Y, et al. Exercise, tea consumption, and depression among breast cancer survivors. J Clin Oncol. 2010;28(6):991-998. (PubMed)
131. Hintikka J, Tolmunen T, Honkalampi K, et al. Daily tea drinking is associated with a low level of depressive symptoms in the Finnish general population. Eur J Epidemiol. 2005;20(4):359-363. (PubMed)
132. Bryan J, Tuckey M, Einöther SJ, Garczarek U, Garrick A, De Bruin EA. Relationships between tea and other beverage consumption to work performance and mood. Appetite. 2012;58(1):339-346. (PubMed)
133. Einöther SJL, Baas M, Rowson M, Giesbrecht T. Investigating the effects of tea, water and a positive affect induction on mood and creativity. Food Quality and Preference. 2015;39:56-61.
134. Einöther SJ, Martens VE. Acute effects of tea consumption on attention and mood. Am J Clin Nutr. 2013;98(6 Suppl):1700S-1708S. (PubMed)
135. De Bruin EA, Rowson MJ, Van Buren L, Rycroft JA, Owen GN. Black tea improves attention and self-reported alertness. Appetite. 2011;56(2):235-240. (PubMed)
136. Hindmarch I, Quinlan PT, Moore KL, Parkin C. The effects of black tea and other beverages on aspects of cognition and psychomotor performance. Psychopharmacology (Berl). 1998;139(3):230-238. (PubMed)
137. Hindmarch I, Rigney U, Stanley N, Quinlan P, Rycroft J, Lane J. A naturalistic investigation of the effects of day-long consumption of tea, coffee and water on alertness, sleep onset and sleep quality. Psychopharmacology (Berl). 2000;149(3):203-216. (PubMed)
138. Camfield DA, Stough C, Farrimond J, Scholey AB. Acute effects of tea constituents L-theanine, caffeine, and epigallocatechin gallate on cognitive function and mood: a systematic review and meta-analysis. Nutr Rev. 2014;72(8):507-522. (PubMed)
139. Ng TP, Feng L, Niti M, Kua EH, Yap KB. Tea consumption and cognitive impairment and decline in older Chinese adults. Am J Clin Nutr. 2008;88(1):224-231. (PubMed)
140. Chin AV, Robinson DJ, O'Connell H, et al. Vascular biomarkers of cognitive performance in a community-based elderly population: the Dublin Healthy Ageing study. Age Ageing. 2008;37(5):559-564. (PubMed)
141. Kuriyama S, Hozawa A, Ohmori K, et al. Green tea consumption and cognitive function: a cross-sectional study from the Tsurugaya Project 1. Am J Clin Nutr. 2006;83(2):355-361. (PubMed)
142. Nurk E, Refsum H, Drevon CA, et al. Intake of flavonoid-rich wine, tea, and chocolate by elderly men and women is associated with better cognitive test performance. J Nutr. 2009;139(1):120-127. (PubMed)
143. Shen W, Xiao Y, Ying X, et al. Tea Consumption and Cognitive Impairment: A Cross-Sectional Study among Chinese Elderly. PLoS One. 2015;10(9):e0137781. (PubMed)
144. Feng L, Gwee X, Kua EH, Ng TP. Cognitive function and tea consumption in community dwelling older Chinese in Singapore. J Nutr Health Aging. 2010;14(6):433-438. (PubMed)
145. Arab L, Biggs ML, O'Meara ES, Longstreth WT, Crane PK, Fitzpatrick AL. Gender differences in tea, coffee, and cognitive decline in the elderly: the Cardiovascular Health Study. J Alzheimers Dis. 2011;27(3):553-566. (PubMed)
146. Noguchi-Shinohara M, Yuki S, Dohmoto C, et al. Consumption of green tea, but not black tea or coffee, is associated with reduced risk of cognitive decline. PLoS One. 2014;9(5):e96013. (PubMed)
147. Ide K, Yamada H, Takuma N, et al. Green tea consumption affects cognitive dysfunction in the elderly: a pilot study. Nutrients. 2014;6(10):4032-4042. (PubMed)
148. de Lau LM, Breteler MM. Epidemiology of Parkinson's disease. Lancet Neurol. 2006;5(6):525-535. (PubMed)
149. Kandinov B, Giladi N, Korczyn AD. Smoking and tea consumption delay onset of Parkinson's disease. Parkinsonism Relat Disord. 2009;15(1):41-46. (PubMed)
150. Kandinov B, Giladi N, Korczyn AD. The effect of cigarette smoking, tea, and coffee consumption on the progression of Parkinson's disease. Parkinsonism Relat Disord. 2007;13(4):243-245. (PubMed)
151. Li FJ, Ji HF, Shen L. A meta-analysis of tea drinking and risk of Parkinson's disease. ScientificWorldJournal. 2012;2012:923464. (PubMed)
152. Qi H, Li S. Dose-response meta-analysis on coffee, tea and caffeine consumption with risk of Parkinson's disease. Geriatr Gerontol Int. 2014;14(2):430-439. (PubMed)
153. Liu R, Guo X, Park Y, et al. Caffeine intake, smoking, and risk of Parkinson disease in men and women. Am J Epidemiol. 2012;175(11):1200-1207. (PubMed)
154. Gao X, Cassidy A, Schwarzschild MA, Rimm EB, Ascherio A. Habitual intake of dietary flavonoids and risk of Parkinson disease. Neurology. 2012;78(15):1138-1145. (PubMed)
155. Aizaki T, Osaka M, Hara H, et al. Hypokalemia with syncope caused by habitual drinking of oolong tea. Intern Med. 1999;38(3):252-256. (PubMed)
156. Trewby PN, Rutter MD, Earl UM, Sattar MA. Teapot myositis. Lancet. 1998;351(9111):1248. (PubMed)
157. Bioh G, Gallagher MM, Prasad U. Survival of a highly toxic dose of caffeine. BMJ Case Rep. 2013; pii: bcr2012007454. doi: 10.1136/bcr-2012-007454. (PubMed)
158. Rudolph T, Knudsen K. A case of fatal caffeine poisoning. Acta Anaesthesiol Scand. 2010;54(4):521-523. (PubMed)
159. Finsterer J. Earl Grey tea intoxication. Lancet. 2002;359(9316):1484. (PubMed)
160. Syed F, Mena-Gutierrez A, Ghaffar U. A case of iced-tea nephropathy. N Engl J Med. 2015;372(14):1377-1378. (PubMed)
161. Izuora K, Twombly JG, Whitford GM, Demertzis J, Pacifici R, Whyte MP. Skeletal fluorosis from brewed tea. J Clin Endocrinol Metab. 2011;96(8):2318-2324. (PubMed)
162. Kakumanu N, Rao SD. Images in clinical medicine. Skeletal fluorosis due to excessive tea drinking. N Engl J Med. 2013;368(12):1140. (PubMed)
163. Whyte MP, Totty WG, Lim VT, Whitford GM. Skeletal fluorosis from instant tea. J Bone Miner Res. 2008;23(5):759-769. (PubMed)
164. Jatoi A, Ellison N, Burch PA, et al. A phase II trial of green tea in the treatment of patients with androgen independent metastatic prostate carcinoma. Cancer. 2003;97(6):1442-1446. (PubMed)
165. Pisters KM, Newman RA, Coldman B, et al. Phase I trial of oral green tea extract in adult patients with solid tumors. J Clin Oncol. 2001;19(6):1830-1838. (PubMed)
166. Sarma DN, Barrett ML, Chavez ML, et al. Safety of green tea extracts : a systematic review by the US Pharmacopeia. Drug Saf. 2008;31(6):469-484. (PubMed)
167. Mazzanti G, Di Sotto A, Vitalone A. Hepatotoxicity of green tea: an update. Arch Toxicol. 2015;89(8):1175-1191. (PubMed)
168. Chow HH, Cai Y, Hakim IA, et al. Pharmacokinetics and safety of green tea polyphenols after multiple-dose administration of epigallocatechin gallate and polyphenon E in healthy individuals. Clin Cancer Res. 2003;9(9):3312-3319. (PubMed)
169. Dostal AM, Samavat H, Bedell S, et al. The safety of green tea extract supplementation in postmenopausal women at risk for breast cancer: results of the Minnesota Green Tea Trial. Food Chem Toxicol. 2015;83:26-35. (PubMed)
170. American College of Obstetricians and Gynecologists. ACOG CommitteeOpinion No. 462: Moderate caffeine consumption during pregnancy. Obstet Gynecol. 2010;116(2 Pt 1):467-468. (PubMed)
171. Li J, Zhao H, Song JM, Zhang J, Tang YL, Xin CM. A meta-analysis of risk of pregnancy loss and caffeine and coffee consumption during pregnancy. Int J Gynaecol Obstet. 2015;130(2):116-122. (PubMed)
172. Rhee J, Kim R, Kim Y, et al. Maternal Caffeine Consumption during Pregnancy and Risk of Low Birth Weight: A Dose-Response Meta-Analysis of Observational Studies. PLoS One. 2015;10(7):e0132334. (PubMed)
173. Taylor JR, Wilt VM. Probable antagonism of warfarin by green tea. Ann Pharmacother. 1999;33(4):426-428. (PubMed)
174. Heck AM, DeWitt BA, Lukes AL. Potential interactions between alternative therapies and warfarin. Am J Health Syst Pharm. 2000;57(13):1221-1227; quiz 1228-1230. (PubMed)
175. Werba JP, Misaka S, Giroli MG, et al. Overview of green tea interaction with cardiovascular drugs. Curr Pharm Des. 2015;21(9):1213-1219. (PubMed)
176. An G, Mukker JK, Derendorf H, Frye RF. Enzyme- and transporter-mediated beverage-drug interactions: An update on fruit juices and green tea. J Clin Pharmacol. 2015;5(12):1313-1331. (PubMed)
177. Carrillo JA, Benitez J. Clinically significant pharmacokinetic interactions between dietary caffeine and medications. Clin Pharmacokinet. 2000;39(2):127-153. (PubMed)
178. Kim EY, Ham SK, Shigenaga MK, Han O. Bioactive dietary polyphenolic compounds reduce nonheme iron transport across human intestinal cell monolayers. J Nutr. 2008;138(9):1647-1651. (PubMed)
179. Thankachan P, Walczyk T, Muthayya S, Kurpad AV, Hurrell RF. Iron absorption in young Indian women: the interaction of iron status with the influence of tea and ascorbic acid. Am J Clin Nutr. 2008;87(4):881-886. (PubMed)
180. Hurrell RF, Reddy M, Cook JD. Inhibition of non-haem iron absorption in man by polyphenolic-containing beverages. Br J Nutr. 1999;81(4):289-295. (PubMed)
181. Zijp IM, Korver O, Tijburg LB. Effect of tea and other dietary factors on iron absorption. Crit Rev Food Sci Nutr. 2000;40(5):371-398. (PubMed)
182. Ma Q, Kim EY, Lindsay EA, Han O. Bioactive dietary polyphenols inhibit heme iron absorption in a dose-dependent manner in human intestinal Caco-2 cells. J Food Sci. 2011;76(5):H143-150. (PubMed)
183. Kim EY, Ham SK, Bradke D, Ma Q, Han O. Ascorbic acid offsets the inhibitory effect of bioactive dietary polyphenolic compounds on transepithelial iron transport in Caco-2 intestinal cells. J Nutr. 2011;141(5):828-834. (PubMed)
184. Nelson M, Poulter J. Impact of tea drinking on iron status in the UK: a review. J Hum Nutr Diet. 2004;17(1):43-54. (PubMed)
185. Mennen L, Hirvonen T, Arnault N, Bertrais S, Galan P, Hercberg S. Consumption of black, green and herbal tea and iron status in French adults. Eur J Clin Nutr. 2007;61(10):1174-1179. (PubMed)
Alcoholic Beverages
Contents
Summary
- Observational studies have consistently found that moderate alcohol consumption (no more than two alcoholic drinks/day for men and no more than one alcoholic drink/day for women) is associated with a decreased risk of cardiovascular and all-cause mortality. (More information)
- Moderate alcohol consumption is associated with lowered risks of coronary heart disease (CHD) and ischemic stroke. (More information)
- Evidence from observational studies suggests that moderate alcohol intake may be associated with reduced risk of type 2 diabetes, dementia, and gallstones, as well as with improved bone mineral density. (More information)
- Even moderate alcohol consumption may increase the risk of female breast cancer, alcohol-related birth defects, and progression to heavy alcohol consumption in some people. (More information)
- Heavy alcohol consumption is associated with increased risks of hypertension, stroke, heart rhythm disturbances, dementia, accidents, injury, violence, and damage to the heart, liver, and pancreas. (More information)
- Heavy alcohol consumption is associated with increased risk of many cancers, including cancers of the mouth, pharynx, larynx, esophagus, liver, breast, colon and rectum. The combined use of alcohol and tobacco greatly increases the risk of oral and esophageal cancers. (More information)
- Those who consume more than minimal amounts of alcohol should make sure they also consume adequate folate by taking a daily multivitamin that provides 400 μg of folic acid. (More information)
- There is consensus that the health risks of moderate alcohol consumption outweigh the health benefits for some people. People who should abstain from alcohol include (1, 2): children and adolescents; pregnant women and women who may become pregnant; anyone who has trouble limiting his or her alcohol consumption to moderate levels, particularly recovering alcoholics and those with a family history of alcoholism or alcohol problems; and anyone with chronic liver disease or alcohol-related disease or organ damage.
- Anyone planning to drive, operate heavy machinery, or perform other potentially hazardous activities requiring coordination and skill should not consume alcohol.
- People who would benefit from individualized advice regarding potential health risks and benefits of moderate alcohol consumption include: anyone taking medications (over-the-counter or prescription) with the potential for adverse interactions with alcohol; and anyone with a personal or family history (e.g., parent or sibling) of breast cancer, coronary heart disease, or other conditions related positively or inversely to moderate drinking.
Introduction
While excessive alcohol consumption has been linked to a number of serious health and social problems, observational studies have associated moderate alcohol consumption with some important health benefits. The relationship between alcohol consumption and mortality is often described as J-shaped, meaning that when graphed from alcohol abstinence on the left to heavy drinking on the right, light-to-moderate alcohol consumption (≤2 drinks/day) is associated with lower rates of mortality — mostly from cardiovascular disease — than abstention, while heavy alcohol consumption (>3-4 drinks/day) is associated with higher rates of mortality from a number of causes (3-5). Because the consumption of alcohol can be viewed as a “double-edged sword,” individual decisions regarding alcohol use should take into consideration scientific evidence regarding potential health benefits and risks, as well as personal and family histories of health problems and addictions.
It is important to note the data on alcohol-disease relationships come from only observational studies, not randomized controlled trials, and observational data cannot establish causation. In observational research, potential confounding variables should be adequately adjusted for using statistical techniques. For instance, nondrinkers have been shown to differ from those who consume alcohol in ways that might affect the disease outcome of interest (6). Even when controlling for many potential confounders, residual confounding may still occur.
Definitions (7)
Standard alcoholic drink (8)
A standard alcoholic drink contains approximately 14 grams of alcohol, which is equivalent to 12 ounces of beer (~5% alcohol), 8.5 ounces of malt liquor (~9% alcohol), 5 ounces of wine (~12% alcohol), 3.5 ounces of fortified wine (e.g., sherry or port), or 1.5 ounces of liquor (distilled spirits; ~40% alcohol).
Moderate alcohol consumption
- Men: No more than two standard alcoholic drinks/day (9)
- Women: No more than one standard alcoholic drink/day* (9)
- There is consensus that distributing total weekly alcohol intake evenly to most days is the healthiest drinking pattern.
Heavy alcohol consumption (8)
- Men: More than 14 standard alcoholic drinks/week or more than 4 standard alcoholic drinks in a day
- Women: More than 7 standard alcoholic drinks/week or more than 3 standard alcoholic drinks in a day*
*In addition to weighing less, on average, women absorb and metabolize alcohol differently than men. In general, women have less body water than men of similar body weight, so women achieve higher blood alcohol concentrations after drinking equivalent amounts of alcohol (10). Women also appear to be more vulnerable to adverse health effects of heavy drinking than men. Thus, most definitions of “moderate” or “heavy” drinking offer a lower threshold for women.
Potential Health Benefits of Moderate Alcohol Consumption
Mortality
Data from observational studies have shown that light-to-moderate alcohol consumption (≤1 drink/day for women and ≤2 drinks/day for men) is protective against all-cause mortality (4, 11-15). As mentioned above, a J-shaped relationship is apparent when all-cause mortality is plotted against alcohol consumption (alcohol abstinence on the left and heavy drinking on the right of the x-axis) (4, 16). In other words, those who drink moderately have the lowest risk of total mortality when compared to nondrinkers and heavy drinkers, and heavy drinkers have the highest risk of mortality.
The association of reduced mortality with moderate alcohol consumption is largely attributed to a decrease in cardiovascular mortality (14, 16-18), especially from coronary heart disease (see Cardiovascular disease below). However, concern has been raised that some earlier observational studies have misclassified former drinkers in the lifetime abstention group (i.e., the referent group), but most recent studies have not supported such a ‘misclassification hypothesis’ (15, 16, 19).
Cardiovascular disease
Coronary heart disease
Over the past four decades, the most consistent evidence of a health benefit associated with moderate alcohol consumption has been a significant reduction in the risk of coronary heart disease (CHD) — a finding confirmed by a large number of epidemiological studies. When the results of 28 prospective cohort studies were combined in a meta-analysis, adults who consumed an average of 25 grams/day of alcohol (the amount in two standard alcoholic drinks) had a risk of CHD that was 20% lower than adults who did not consume alcohol (20). More recent data from two large prospective cohort studies conducted in the US suggest that the magnitude of CHD risk reduction associated with moderate alcohol consumption may be closer to 30%. In a 12-year study of more than 38,000 male health professionals, those who consumed alcohol at least 3-4 times weekly had a risk of myocardial infarction (heart attack) that was 32% lower than men who drank alcohol less than once weekly (21). Similarly, in a 20-year study of more than 120,000 men and women, those who reported consuming 1-2 alcoholic drinks daily had a risk of death from CHD that was 30% lower than those who did not drink alcohol (22). A 2011 systematic review and meta-analysis of 29 studies found that alcohol consumption was associated with a 29% reduced risk of CHD compared to abstention; intakes of 2.5 to 60.0 grams/day of alcohol were associated with a lower risk of CHD (16).
How does alcohol consumption reduce CHD risk? The development of CHD is characterized by the formation of cholesterol-laden plaque in the arteries (atherosclerosis), vascular inflammation, and clot formation (23). Numerous small, randomized trials have examined the effect of daily alcohol consumption on markers of CHD risk, consistently finding that moderate alcohol consumption significantly increases concentrations of high-density lipoprotein (HDL)-cholesterol — the ‘good cholesterol’ (24, 25). HDLs transport cholesterol from tissues, including arterial walls, back to the liver for elimination or recycling. In addition to increasing HDL levels, moderate alcohol consumption has been shown to increase apolipoprotein A1, a major component of circulating HDL (25). Higher levels of high-density lipoprotein (HDL)-cholesterol have been associated with reductions in CHD risk (26).
Alcohol may also have anti-thrombotic properties. Clot formation is the result of complex interactions between factors that promote coagulation and factors that inhibit coagulation or promote the dissolution of clots. Several randomized trials have found that moderate alcohol consumption decreases serum levels of fibrinogen, a protein that promotes clot formation (25) and increases serum levels of an enzyme that helps dissolve clots (tissue type plasminogen activator) (24).
Further, moderate alcohol consumption may have an anti-inflammatory effect since serum levels of C-reactive protein (CRP), a marker of systemic inflammation and sensitive predictor of CHD risk, are lower in people who drink moderately than those who abstain from alcohol (27-32). Moderate alcohol consumption has also been associated with improvements in adiponectin levels (25), insulin sensitivity (see Type 2 diabetes mellitus below), abdominal obesity (33), and endothelial function (34).
Does the type of alcohol consumed (wine, beer, or liquor) affect CHD risk? Significant reductions in CHD risk have been associated with moderate consumption of wine, beer, and liquor. However, the “French Paradox” — the observation that mortality from CHD is relatively low in France despite relatively high levels of dietary saturated fat and cigarette smoking — led to the idea that regular consumption of red wine might provide additional protection from CHD (35, 36). Red wine contains the phenolic compound resveratrol — although usually at variable and low concentrations (see the article on Resveratrol) — as well as flavonoids like procyanidins; these compounds could provide additional cardiovascular benefits beyond those associated with ethanol. Beer also contains polyphenolic compounds that might confer some cardioprotection (37).
Some large prospective cohort studies have found wine drinkers to be at lower risk of CHD than beer or liquor drinkers (22, 38-40), but others have found no difference (21, 41, 42). Moreover, some studies have observed a decreased risk of myocardial infarction or CHD in predominantly beer-drinking populations in the Czech Republic (43), in Germany (44), and in Japanese men residing in Hawaii (45). A 2011 meta-analysis of prospective cohort and case-control studies found that moderate consumption of wine or beer was associated with a decreased risk of non-fatal vascular events (46). This analysis did not associate drinking liquor with cardiovascular benefit, although the authors noted that binge drinking — which is known to increase CHD risk — was apparent in several of the included studies (46).
Socioeconomic status and lifestyle characteristics (e.g., tobacco use, exercise habits) may differ among people who prefer wine, beer, or liquor, and this may in part explain any additional benefit of one beverage type observed in some studies. For example, several early studies found that people who prefer wine tend to have higher incomes, have more formal education, smoke less, and eat more fruit and vegetables and less saturated fat than people who prefer other alcoholic beverages (47-49). These potential confounders should be controlled or adjusted for in the analysis of observational data.
Thus, although moderate alcohol consumption has been consistently associated with 20%-30% reductions in CHD risk, it is not yet clear whether drinking a specific type of alcoholic beverage might confer additional cardiovascular benefit.
Stroke
Ischemic strokes, which represent 87% of all strokes, are the result of insufficient blood flow to an area of the brain, which may occur when an artery supplying the brain becomes blocked by a blood clot (50). Hemorrhagic strokes occur when a blood vessel ruptures and bleeds into the brain. Although they are less prevalent than ischemic strokes, hemorrhagic strokes are generally more severe and contribute disproportionately to overall stroke mortality (51). Light or moderate alcohol consumption has been associated with a reduced risk of ischemic stroke, but not hemorrhagic stroke, in a number of observational studies (52-58). When the results of 19 prospective cohort and 16 case-control studies of alcohol consumption and the risk of stroke were combined in a meta-analysis, moderate alcohol consumption was associated with a significant reduction in the risk of ischemic stroke (59). Overall, those who consumed one or two drinks daily had a 28% lower risk of ischemic stroke than those who did not consume alcohol. Another meta-analysis of more recent studies (1980-2009) confirmed that moderate alcohol consumption was protective against only ischemic stroke in both men and women (60). A more recent meta-analysis of 27 prospective cohort studies found that light-to-moderate alcohol consumption (<15 grams/day) in women was associated with a reduced risk of ischemic (RR, 0.72) but not hemorrhagic stroke; moderate alcohol consumption (15-30 grams/day) was not linked to either type of stroke in men in this analysis (61).
Thus, light-to-moderate alcohol consumption appears to decrease risk of ischemic stroke, but not hemorrhagic stroke, likely due to the anti-thrombotic effect of alcohol.
Peripheral arterial disease
Just as atherosclerosis of the arteries supplying the heart muscle leads to coronary heart disease, atherosclerosis of the arteries of the extremities leads to peripheral arterial disease (PAD). When atherosclerosis is severe enough to diminish blood flow to the legs, even walking may result in leg or hip pain known as intermittent claudication (62). Impaired vascular endothelial function is also characteristic of the disease and may contribute to the clinical symptoms (63).
Although much less consistent than the evidence for heart disease and stroke, there is limited evidence that moderate alcohol consumption is associated with decreased risk of PAD. Four prospective cohort studies have found moderate alcohol consumption to be associated with significant decreases in several different indicators of PAD (64-67). One of these studies found that the inverse association between alcohol intake and PAD risk was significant in nonsmokers but not smokers, suggesting that the adverse effects of cigarette smoking on PAD risk may outweigh any protective effects of alcohol consumption (64).
Heart failure
Coronary heart disease is a major cause of heart failure. A prospective study in a cohort of 21,601 men and another in a cohort of 126,236 men and women found that moderate alcohol intake was inversely associated with heart failure, especially heart disease related to CHD (68, 69). More recently, in a cohort of 4,490 older adults (65 years or older at baseline) followed for more than 20 years (1,380 cases of heart failure), drinking one or more alcoholic drink per week was associated with a 26% lower risk of heart failure compared to abstainers (70).
Sudden cardiac death
While several studies have found that heavy alcohol consumption increases risk of sudden cardiac death (SCD; see below), the association of light-to-moderate alcohol consumption and SCD is less clear. Studies on this association have reported mixed results, but the two largest prospective cohort studies to date have found a lower risk of SCD with light-to-moderate alcohol consumption (71, 72).
Type 2 diabetes mellitus
Three meta-analyses have found a U-shaped relationship between alcohol consumption and incidence of type 2 diabetes mellitus, with greater protection being observed for women (73-75). The most recent meta-analysis included 20 prospective cohort studies and associated moderate alcohol consumption (22-25 grams of alcohol daily or 1.6-1.8 drinks/day) with a 40% risk reduction for women and a 13% risk reduction for men compared to lifetime alcohol abstainers (74). Heavy alcohol consumption (62 grams/day or 4.4. drinks/day for men and 51 grams/day or 3.6 drinks/day for women) was associated with an increased risk for type 2 diabetes (74).
Increased insulin secretion by the pancreas and decreased insulin sensitivity are important factors leading to the development of type 2 diabetes. Research suggests that moderate alcohol intake may decrease serum insulin levels, increase adiponectin (an adipocyte hormone inversely associated with type 2 diabetes) levels (25), and improve insulin sensitivity (76-79). On the other hand, heavy alcohol consumption may increase the risk of type 2 diabetes by contributing to obesity, especially abdominal obesity, disturbing carbohydrate metabolism, and/or impairing pancreatic or liver function (80).
Osteoporosis
Osteoporosis, a condition common among the elderly, results from progressive loss of bone mineral density (BMD). Several observational studies have associated light or moderate alcohol consumption with higher BMD in older adults compared to abstainers (81-91). Some studies have found stronger protective relationships among wine (89) or beer drinkers (89, 90) in comparison to those who consume liquor, suggesting that non-alcohol components (e.g., silicon in beer) might help explain the association. The effects of alcohol on bone health may also be dependent on age, gender, and hormonal status (reviewed in 92).
It is important to note that the available data come from observational studies, and the observed associations may be confounded, e.g., individuals who consume alcohol in moderation may have an overall healthier lifestyle than those who drink heavily or abstain. However, a recent study in perimenopausal women found that moderate alcohol intake was associated with improved BMD independent of various lifestyle factors, including smoking status, fruit and vegetable intake, and physical activity level (90).
Cognitive decline, dementia, and Alzheimer’s disease
Although alcoholism and heavy alcohol consumption (>3-4 drinks/day) is known to increase the risk of cognitive impairment and dementia (93-95), recent meta-analyses and reviews have reported that light-to-moderate alcohol consumption in older adults is associated with a decreased risk of dementia and Alzheimer’s disease when compared to abstention (93, 96, 97). Some meta-analyses have not found a significant reduced risk for vascular type dementia (96, 98) or for cognitive decline (93, 96, 98, 99). A few studies have suggested that consumption of wine may be especially protective against dementia, although inconsistent findings have been observed, and many studies have not distinguished among the various types of alcohol.
At least three epidemiological studies have used magnetic resonance imaging (MRI) to examine relationships between alcohol intake and subclinical abnormalities in the brains of healthy middle-aged or older adults. Two studies found that infarctions (areas of dead tissue) were less frequent in the brains of those reporting light or moderate alcohol intake compared to those who abstained from alcohol (100, 101). However, another study found no relationship between alcohol intake and the presence of infarction (102). Two of the studies measuring brain atrophy, a characteristic of Alzheimer’s disease and alcoholic dementia, found brain atrophy to be lower in those who abstained from alcohol compared to alcohol consumers (100, 102). The other study found less brain atrophy with light-to-moderate alcohol consumption but only in carriers of the apolipoprotein E (APOE) ε4 allele, who are at increased risk for Alzheimer’s disease (101). Because of the complex nature of alcohol’s effects on the brain, further research is needed to determine the risks and benefits of alcohol consumption with respect to cognitive function and dementia.
Gallstones
The majority of prospective cohort studies (103-107) and case-control studies (108, 109) have found that men and women with moderate alcohol intakes have lower risks of gallstones or gallbladder surgery (cholecystectomy) than those who do not consume alcohol. Although the reasons for the consistent inverse association between moderate alcohol consumption and gallstone incidence are not entirely clear, regular alcohol intake may result in bile that is less likely to crystallize into gallstones or stimulate gallbladder emptying (106).
Health Risks of Moderate Alcohol Consumption
Pregnancy
Fetal alcohol spectrum disorders (FASD) is a continuum of developmental abnormalities resulting from gestational alcohol exposure; FASD may affect as many as 1%-2% of US children (110, 111). Fetal alcohol syndrome (FAS) — a severe FASD — is a cluster of physical and mental birth defects associated with heavy alcohol consumption during pregnancy. Some characteristics of FAS include facial abnormalities, mental retardation, and growth impairment. More moderate alcohol consumption during pregnancy (7-14 drinks/week) has been associated with more subtle effects on cognitive and behavioral development (112, 113). Children of mothers who drank moderately during pregnancy have been found to have problems with memory, attention and learning, and behavior (114). Overall, studies on the association of low-to-moderate drinking during pregnancy and mental health of offspring have reported mixed results (reviewed in 115). However, it is important to note that these studies are observational in nature and may have not adequately controlled for potential confounding factors (e.g., lifestyle differences [in women who drank alcohol during pregnancy versus those who abstained] that influence mental development) (115).
Since no safe level of alcohol consumption has been established at any stage of pregnancy, pregnant women and women who are planning a pregnancy should abstain from alcohol (116, 117).
Breast cancer
More than 100 observational studies have been completed on the association between alcohol consumption and female breast cancer, with most finding an increased risk (118-121). Even though the available data come from observational studies, many consider the association to be causal. Regular alcohol consumption as low as one or two drinks per day has been associated with modest but significant increases in breast cancer risk. A threshold for harm, however, is difficult to define due to potential underreporting of alcohol intake by heavy drinkers, which could result in heavy drinkers being misclassified as ‘moderate alcohol consumers’ (122).
A linear dose-dependent relationship between alcohol consumption and breast cancer risk has been observed for premenopausal and postmenopausal breast cancer regardless of the type of alcoholic beverage consumed. Pooled and meta-analyses have found that each 10-gram increase in daily alcohol consumption (slightly less than one drink) is associated with a 7%-10% increased risk of breast cancer in women (123-125). Studies of alcohol consumption and breast cancer-specific mortality have reported mixed results, with a recent meta-analysis of 25 prospective cohort studies finding an increased risk only with alcohol consumption in excess of 20 grams (1.4 drinks)/day (126). Moderate alcohol consumption has been consistently associated with reduced risk of all-cause mortality (see Mortality above).
Although the mechanisms for the consistent association between alcohol intake and breast cancer incidence have not been clearly identified, proposed mechanisms include acetaldehyde formation, induction of CYP2E1 metabolism and increased oxidative stress, increased circulating estrogen or androgen levels, and enhanced invasiveness of breast cancer cells (119, 127). Current estimates are that about one in eight women (12.4%) in the US will develop breast cancer at some point in her lifetime (128). Although there are many risk factors for breast cancer, alcohol consumption is one of only a few modifiable risk factors.
Folate and breast cancer
Alcohol interferes with the absorption, transport, and metabolism of folate, which is required for DNA methylation and DNA repair (see the article on Folate). Alterations in these processes may result in mutations or altered gene expression, which increase the risk of cancer (118). Several (129-134), but not all (135-139), studies have found that sufficient folate intake may modify the association between alcohol intake and breast cancer risk. Although the interactions between folate, alcohol, and breast cancer risk remain to be clarified, it makes sense for women who drink alcohol to take a daily multivitamin containing 400 μg of folic acid.
Progression to heavy or hazardous drinking
Some people, such as recovering alcoholics and those with family histories of alcohol abuse or alcoholism, may not be able to maintain moderate drinking habits. Susceptibility to alcoholism is affected by genetic, psychosocial, and environmental factors. Children of an alcoholic parent have been found to be at significantly higher risk of developing alcoholism than those without an alcoholic parent (140). This increase in risk is likely related to interactions between genetic factors and factors related to the family environment. The National Institute on Alcohol Abuse and Alcoholism recommends that people with a family history of alcoholism, especially in a parent, approach moderate drinking carefully (141).
Medication interactions
In the liver, alcohol is metabolized by the same enzymes as many medications. Therefore, alcohol consumption can affect the activation or breakdown of a number of medications. The consumption of alcohol may also increase sedation, drowsiness, and hypotensive effects caused by numerous prescription and over-the-counter medications. Although serious interactions between alcohol and medications are more common in the presence of heavy alcohol consumption, even moderate alcohol consumption may hypothetically increase the risk of some adverse reactions in susceptible people (142). Women and older adults are particularly at risk for interactions between alcohol and medications (143, 144).
Many different classes of prescription medication may interact adversely with alcohol, including antibiotics, anticonvulsants, anticoagulants (e.g., Coumadin), antidepressants, antidiabetic agents, antihypertensive agents, vasodilators (e.g., nitrates and calcium channel blockers), barbiturates, benzodiazepines (sedatives), histamine H2-receptor blockers, muscle relaxants, and narcotic and non-narcotic pain relievers. Over-the-counter medications and herbal preparations may also interact with alcohol, including pain medications like aspirin, acetaminophen (Tylenol), ibuprofen (Advil, Motrin), and naproxen sodium (Aleve); cold and allergy medications like diphenhydramine (Benadryl) and chlorpheniramine; heartburn medications like cimetidine (Tagamet) and ranitidine (Zantac); and herbal preparations like chamomile, valerian, and kava.
To help avoid potentially serious interactions between alcohol and medications, make sure your health care provider is aware of your alcohol intake. Before taking prescription or over-the-counter medications, read the product warning labels or consult a pharmacist or health care provider to determine whether alcohol consumption increases the risk of adverse effects. It may, in general, be advisable to separate taking any medication and drinking alcohol by two to three hours. For more information on potentially serious interactions between alcohol and medications, see the National Institute on Alcohol Abuse and Alcoholism website.
Health Benefits of Heavy Alcohol Consumption
None
Health Risks of Heavy Alcohol Consumption
Pregnancy
Heavy consumption of alcohol during pregnancy causes fetal alcohol syndrome (FAS). See above.
Cardiovascular disease
Hypertension
Heavy alcohol consumption has been consistently associated with an increased risk of high blood pressure (hypertension) in prospective cohort and case-control studies (145-147). A 2009 systematic review and meta-analysis of 12 prospective cohort studies found consuming 50 grams (3.6 drinks)/day of alcohol was associated with a 1.6-fold and 1.8-fold higher risk of hypertension in men and women, respectively; alcohol intake at twice that level (100 grams (~7 drinks)/day) was associated with a relative risk of 2.5 for men and 2.8 for women (148).
The results of numerous clinical trials indicate that reducing alcohol intake lowers blood pressure in hypertensive and normotensive individuals. A meta-analysis that combined the results of 15 randomized controlled trials found that reducing alcohol consumption resulted in significant decreases in both systolic and diastolic blood pressure (149).
Stroke
Ischemic strokes are the result of insufficient blood flow to an area of the brain, which may occur when an artery supplying the brain becomes blocked by a blood clot. Hemorrhagic strokes occur when a blood vessel ruptures and bleeds into the brain. Although regular, moderate alcohol consumption has been associated with decreased risk of ischemic stroke in some studies, heavy alcohol consumption has been associated with increased risk of both ischemic stroke and hemorrhagic stroke. A meta-analysis that combined the results of 19 prospective cohort and 16 case-control studies found that heavy drinking more than doubled the risk of hemorrhagic stroke and increased the risk of ischemic stroke by 70% (59). A meta-analysis of recent studies (1980-2009) confirmed that heavy drinking is associated with increased risks of ischemic and hemorrhagic stroke in both men and women (60). Heavy alcohol consumption may increase the risk of stroke by contributing to hypertension, cardiomyopathy (heart muscle damage), cardiac rhythm disturbances, and coagulation (clotting) disorders and impaired hemostasis.
Cardiac arrhythmias and sudden cardiac death
The long-recognized association between bouts of heavy alcohol consumption and cardiac rhythm disturbances (arrhythmias) was called “holiday heart syndrome” because it was first described in people who were admitted to hospitals after holidays or weekends (150). Atrial fibrillation is the cardiac arrhythmia most commonly associated with heavy alcohol use (151, 152). A 2010 systematic review and meta-analysis found a dose-dependent association between daily alcohol consumption and risk of this type of cardiac arrhythmia, with an increased risk being found with consumption greater than 24 grams/day (1.7 drinks/day) for women and 36 grams/day (2.6 drinks/day) for men (153). A 2014 meta-analysis of seven prospective studies found that consumption of more than two drinks per day was associated with increased risk of atrial fibrillation in men and women, and the risk increased by 8% with each additional daily drink (154, 155). Additionally, several studies have found that heavy alcohol consumption (>5 drinks per day) increases risk of sudden cardiac death (SCD) (156, 157).
The ways by which alcohol may trigger arrhythmias and SCD are not fully known. Alcohol may interfere with the contractility of heart muscle cells, change the shape and structure of heart muscle cells, contribute to electrolyte imbalance, and/or induce oxidative stress (158).
Alcoholic cardiomyopathy
Alcoholic cardiomyopathy is a heart muscle disease caused by long-term, heavy alcohol consumption (159); this disease likely occurs in only a small proportion (<10%) of heavy drinkers (160). Alcoholic cardiomyopathy occurs in two stages: (1) an early asymptomatic stage, when the damage to the heart muscle has no obvious symptoms; and (2) a symptomatic stage, when the heart muscle is too weak to pump effectively. Although the level of alcohol consumption resulting in alcoholic cardiomyopathy has not been clearly established, people consuming at least seven alcoholic drinks daily for more than five years are thought to be at risk of developing asymptomatic alcoholic cardiomyopathy. Those who continue to drink heavily ultimately develop heart failure. Research suggests that women may be more susceptible to alcohol’s toxic effects on the heart muscle than men (161, 162).
Alcoholic liver disease
Chronic excessive alcohol use is a major cause of illness and death from liver disease (163). Alcoholic liver disease is characterized by a spectrum of liver injury, including steatosis (fatty liver), hepatitis (a potentially fatal inflammation of the liver), fibrosis, and cirrhosis — the most advanced form of alcoholic liver disease. In cirrhosis, the formation of fibrotic scar tissue results in progressive deterioration of liver function. Complications of advanced liver disease include severe bleeding from distended veins in the esophagus (esophageal varices), brain damage (hepatic encephalopathy), fluid accumulation in the abdomen (ascites), and kidney failure.
A 2004 meta-analysis of nine studies found a dose-responsive increase in risk for liver cirrhosis with increasing amounts of alcohol consumed: relative risks (RR) of 2.9 for 25 grams (1.8 drinks)/day, 7.1 for 50 grams (3.6 drinks)/day, 26.5 for 100 grams (7.1 drinks)/day (164). Another meta-analysis found a higher RR for liver cirrhosis with increasing doses but also suggested a threshold response for morbidity from liver cirrhosis (higher risk in women with consumption >24 g (1.7 drinks)/day of alcohol, and higher risk in men with consumption >36 g (2.6 drinks)/day of alcohol) (165). Risk of mortality from liver cirrhosis was increased with any alcohol consumption in women and with consumption of >12 g (0.9 drinks)/day in men; a stronger relationship between alcohol consumption and mortality from liver cirrhosis versus morbidity might be expected because alcohol consumption is known to exacerbate any existing liver disease (165).
Serious liver disease has been found to develop in approximately 10% of those who consume more than 60 grams per day of alcohol (4.3 drinks/day). Women are more susceptible to serious alcoholic liver disease than men (165, 166), and individuals with hepatitis C infection have an increased risk of alcoholic liver disease (167).
Cancer
Heavy alcohol consumption has been found to increase the risk of cancer at a number of sites (168). Heavy alcohol consumption is consistently and dose-dependently associated with increases in risk of cancers of the mouth, pharynx, larynx, esophagus, liver, colon, rectum, and breast (165). Moreover, the combination of smoking and alcohol results in even more dramatic increases in cancer risks (169). Increased risk of liver cancer with long-term heavy alcohol consumption may be related to alcoholic cirrhosis of the liver or increased susceptibility to cancer caused by viral hepatitis.
Alcohol-related brain disorders
Chronic heavy alcohol use and alcohol dependence are associated with detrimental effects on the brain and its function, especially memory and executive functions (170). Alcoholics have been observed to suffer from cerebral atrophy (shrinkage of brain tissue), which likely contributes to alcohol-associated dementia and cognitive impairment (94). In contrast to the progressive cerebral atrophy observed in Alzheimer’s disease, alcohol-related cerebral atrophy may decrease after a period of abstinence. Alcohol-related brain disorders may be associated with nutritional deficiencies like thiamin (171) or niacin (172).
Pancreatitis
Pancreatitis is a painful inflammation of the pancreas. Acute pancreatitis is characterized by the sudden onset of severe upper abdominal pain, often accompanied by nausea and vomiting (173). Although most attacks of acute pancreatitis require only supportive care, a small percentage of people may experience serious or life-threatening complications. Studies estimate that 19%-32% of acute pancreatitis cases have an alcoholic etiology (reviewed in 174).
Chronic pancreatitis results in progressive destruction of the pancreas, leading to loss of pancreatic function (175). An estimated 60%-72% of chronic pancreatitis cases have an alcoholic etiology. The risk of developing chronic pancreatitis increases with the quantity and duration of alcohol consumed: an increased risk of chronic pancreatitis is observed with long-term consumption of five or more alcoholic drinks per day (174). Only a small percentage (<10%) of alcoholics develop clinical pancreatitis; thus, hereditary and environmental factors are also thought to play a role. The disease is more common in men than in women, in Blacks compared to Whites, and in smokers versus nonsmokers (176, 177).
Bone health
Chronic alcoholism has deleterious effects on bone health, including decreased bone mineral density and increased risk of fracture. Consumption of large quantities of alcohol (100-200 grams/day) directly impairs activity of osteoblasts — the bone-forming cells. Negative effects on bone health are also indirectly caused by the malnutrition experienced by alcoholics (92).
Accidents, injury, and violence
Alcohol use is associated with an increased risk of injury in a number of circumstances, including motor vehicle accidents, falls, and fires (178). Data from hospital emergency departments indicate that consuming as little as one or two alcoholic drinks in the previous six hours significantly increases the risk of injury (179). Thirty-one percent of all traffic fatalities in the US are alcohol-related (180). Although the legal blood alcohol concentration (BAC) limit for drivers is 0.08 (grams of alcohol/deciliter of blood) in the US, most scientific studies have found significant impairment of driving-related skills at a BAC of 0.05 (181). For reference, a BAC of 0.05 might be achieved by a 175-pound male consuming three standard alcoholic drinks in one hour or a 120-pound female consuming two drinks in one hour (182).
Excessive alcohol use is associated with all forms of violence, including suicide, homicide, domestic violence, sexual assault, and gang violence. Although the reasons for alcohol-associated violence are complex, alcohol use appears to increase the risk of violent behavior in some populations (183).
Mortality
Heavy alcohol consumption increases the risk of mortality (4, 16). As mentioned above, the relationship between alcohol consumption and mortality is often described as J-shaped, meaning those with high intakes of alcohol have a higher risk of mortality than nondrinkers. A 2011 meta-analysis of eight prospective cohort studies found that consumption of >60 grams/day of alcohol was associated with a 30% increase in mortality from all causes (16).
Authors and Reviewers
Originally written in 2004 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in December 2007 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in August 2015 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in August 2015 by:
Arthur L. Klatsky, M.D.
Senior Consultant in Cardiology
Adjunct Investigator, Division of Research
Kaiser Permanente Medical Care Program
Oakland, CA
Copyright 2004-2024 Linus Pauling Institute
References
1. Ecker RR, Klatsky AL. Doctor, should I have a drink? An algorithm for health professionals. Ann N Y Acad Sci. 2002;957:317-320. (PubMed)
2. US Department of Health and Human Services, US Department of Agriculture. Nutrition and your health: Dietary Guidelines for Americans [Website]. Available at: http://www.health.gov/dietaryguidelines/dga2000/document/choose.htm#alcohol. Accessed 11/4/03.
3. Klatsky AL. Drink to your health? Sci Am. 2003;288(2):74-81. (PubMed)
4. Di Castelnuovo A, Costanzo S, Bagnardi V, Donati MB, Iacoviello L, de Gaetano G. Alcohol dosing and total mortality in men and women: an updated meta-analysis of 34 prospective studies. Arch Intern Med. 2006;166(22):2437-2445. (PubMed)
5. Krenz M, Korthuis RJ. Moderate ethanol ingestion and cardiovascular protection: from epidemiologic associations to cellular mechanisms. J Mol Cell Cardiol. 2012;52(1):93-104. (PubMed)
6. Naimi TS, Brown DW, Brewer RD, et al. Cardiovascular risk factors and confounders among nondrinking and moderate-drinking U.S. adults. Am J Prev Med. 2005;28(4):369-373. (PubMed)
7. National Institute on Alcohol Abuse and Alcoholism. Helping patients who drink too much: a clinician's guide [Web page]. January 2007. Available at: http://pubs.niaaa.nih.gov/publications/Practitioner/CliniciansGuide2005/clinicians_guide.htm. Accessed 10/3/07.
8. National Institute on Alcohol Abuse and Alcoholism. Helping patients who drink too much: a clinician's guide: NIH Publication No. 07-3769; 2005.
9. US Department of Agriculture and US Department of Health and Human Services. Dietary Guidelines for Americans, 2010. Washington, D.C.: US Government Printing Office, December 2010.
10. National Institute on Alcohol Abuse and Alcoholism. Women and alcohol. Available at: http://pubs.niaaa.nih.gov/publications/womensfact/womensfact.htm. Accessed 7/29/15.
11. Thun MJ, Peto R, Lopez AD, et al. Alcohol consumption and mortality among middle-aged and elderly U.S. adults. N Engl J Med. 1997;337(24):1705-1714. (PubMed)
12. Poikolainen K. Alcohol and mortality: a review. J Clin Epidemiol. 1995;48(4):455-465. (PubMed)
13. Lee SJ, Sudore RL, Williams BA, Lindquist K, Chen HL, Covinsky KE. Functional limitations, socioeconomic status, and all-cause mortality in moderate alcohol drinkers. J Am Geriatr Soc. 2009;57(6):955-962. (PubMed)
14. Djousse L, Lee IM, Buring JE, Gaziano JM. Alcohol consumption and risk of cardiovascular disease and death in women: potential mediating mechanisms. Circulation. 2009;120(3):237-244. (PubMed)
15. Fuller TD. Moderate alcohol consumption and the risk of mortality. Demography. 2011;48(3):1105-1125. (PubMed)
16. Ronksley PE, Brien SE, Turner BJ, Mukamal KJ, Ghali WA. Association of alcohol consumption with selected cardiovascular disease outcomes: a systematic review and meta-analysis. BMJ. 2011;342:d671. (PubMed)
17. Mukamal KJ, Chen CM, Rao SR, Breslow RA. Alcohol consumption and cardiovascular mortality among U.S. adults, 1987 to 2002. J Am Coll Cardiol. 2010;55(13):1328-1335. (PubMed)
18. Fuchs CS, Stampfer MJ, Colditz GA, et al. Alcohol consumption and mortality among women. N Engl J Med. 1995;332(19):1245-1250. (PubMed)
19. Roerecke M, Rehm J. The cardioprotective association of average alcohol consumption and ischaemic heart disease: a systematic review and meta-analysis. Addiction. 2012;107(7):1246-1260. (PubMed)
20. Corrao G, Rubbiati L, Bagnardi V, Zambon A, Poikolainen K. Alcohol and coronary heart disease: a meta-analysis. Addiction. 2000;95(10):1505-1523. (PubMed)
21. Mukamal KJ, Conigrave KM, Mittleman MA, et al. Roles of drinking pattern and type of alcohol consumed in coronary heart disease in men. N Engl J Med. 2003;348(2):109-118. (PubMed)
22. Klatsky AL, Friedman GD, Armstrong MA, Kipp H. Wine, liquor, beer, and mortality. Am J Epidemiol. 2003;158(6):585-595. (PubMed)
23. Ross R. Atherosclerosis--an inflammatory disease. N Engl J Med. 1999;340(2):115-126. (PubMed)
24. Rimm EB, Williams P, Fosher K, Criqui M, Stampfer MJ. Moderate alcohol intake and lower risk of coronary heart disease: meta-analysis of effects on lipids and haemostatic factors. BMJ. 1999;319(7224):1523-1528. (PubMed)
25. Brien SE, Ronksley PE, Turner BJ, Mukamal KJ, Ghali WA. Effect of alcohol consumption on biological markers associated with risk of coronary heart disease: systematic review and meta-analysis of interventional studies. BMJ. 2011;342:d636. (PubMed)
26. Rader DJ. Regulation of reverse cholesterol transport and clinical implications. Am J Cardiol. 2003;92(4A):42J-49J. (PubMed)
27. Albert MA, Glynn RJ, Ridker PM. Alcohol consumption and plasma concentration of C-reactive protein. Circulation. 2003;107(3):443-447. (PubMed)
28. Stewart SH, Mainous AG, 3rd, Gilbert G. Relation between alcohol consumption and C-reactive protein levels in the adult US population. J Am Board Fam Pract. 2002;15(6):437-442. (PubMed)
29. Imhof A, Froehlich M, Brenner H, Boeing H, Pepys MB, Koenig W. Effect of alcohol consumption on systemic markers of inflammation. Lancet. 2001;357(9258):763-767. (PubMed)
30. Sierksma A, van der Gaag MS, Kluft C, Hendriks HF. Moderate alcohol consumption reduces plasma C-reactive protein and fibrinogen levels; a randomized, diet-controlled intervention study. Eur J Clin Nutr. 2002;56(11):1130-1136. (PubMed)
31. Whitfield JB, Heath AC, Madden PA, Pergadia ML, Montgomery GW, Martin NG. Metabolic and biochemical effects of low-to-moderate alcohol consumption. Alcohol Clin Exp Res. 2013;37(4):575-586. (PubMed)
32. Wang JJ, Tung TH, Yin WH, et al. Effects of moderate alcohol consumption on inflammatory biomarkers. Acta Cardiol. 2008;63(1):65-72. (PubMed)
33. O'Keefe JH, Bybee KA, Lavie CJ. Alcohol and cardiovascular health: the razor-sharp double-edged sword. J Am Coll Cardiol. 2007;50(11):1009-1014. (PubMed)
34. Suzuki K, Elkind MS, Boden-Albala B, et al. Moderate alcohol consumption is associated with better endothelial function: a cross sectional study. BMC Cardiovasc Disord. 2009;9:8. (PubMed)
35. St Leger AS, Cochrane AL, Moore F. Factors associated with cardiac mortality in developed countries with particular reference to the consumption of wine. Lancet. 1979;1(8124):1017-1020. (PubMed)
36. Criqui MH, Ringel BL. Does diet or alcohol explain the French paradox? Lancet. 1994;344(8939-8940):1719-1723. (PubMed)
37. Arranz S, Chiva-Blanch G, Valderas-Martinez P, Medina-Remon A, Lamuela-Raventos RM, Estruch R. Wine, beer, alcohol and polyphenols on cardiovascular disease and cancer. Nutrients. 2012;4(7):759-781. (PubMed)
38. Renaud SC, Gueguen R, Siest G, Salamon R. Wine, beer, and mortality in middle-aged men from eastern France. Arch Intern Med. 1999;159(16):1865-1870. (PubMed)
39. Gronbaek M, Becker U, Johansen D, et al. Type of alcohol consumed and mortality from all causes, coronary heart disease, and cancer. Ann Intern Med. 2000;133(6):411-419. (PubMed)
40. Streppel MT, Ocke MC, Boshuizen HC, Kok FJ, Kromhout D. Long-term wine consumption is related to cardiovascular mortality and life expectancy independently of moderate alcohol intake: the Zutphen Study. J Epidemiol Community Health. 2009;63(7):534-540. (PubMed)
41. Rimm EB, Klatsky A, Grobbee D, Stampfer MJ. Review of moderate alcohol consumption and reduced risk of coronary heart disease: is the effect due to beer, wine, or spirits. BMJ. 1996;312(7033):731-736. (PubMed)
42. Wannamethee SG, Shaper AG. Type of alcoholic drink and risk of major coronary heart disease events and all-cause mortality. Am J Public Health. 1999;89(5):685-690. (PubMed)
43. Bobak M, Skodova Z, Marmot M. Effect of beer drinking on risk of myocardial infarction: population based case-control study. BMJ. 2000;320(7246):1378-1379. (PubMed)
44. Keil U, Chambless LE, Doring A, Filipiak B, Stieber J. The relation of alcohol intake to coronary heart disease and all-cause mortality in a beer-drinking population. Epidemiology. 1997;8(2):150-156. (PubMed)
45. Yano K, Rhoads GG, Kagan A. Coffee, alcohol and risk of coronary heart disease among Japanese men living in Hawaii. N Engl J Med. 1977;297(8):405-409. (PubMed)
46. Costanzo S, Di Castelnuovo A, Donati MB, Iacoviello L, de Gaetano G. Wine, beer or spirit drinking in relation to fatal and non-fatal cardiovascular events: a meta-analysis. Eur J Epidemiol. 2011;26(11):833-850. (PubMed)
47. Mortensen EL, Jensen HH, Sanders SA, Reinisch JM. Better psychological functioning and higher social status may largely explain the apparent health benefits of wine: a study of wine and beer drinking in young Danish adults. Arch Intern Med. 2001;161(15):1844-1848. (PubMed)
48. Barefoot JC, Gronbaek M, Feaganes JR, McPherson RS, Williams RB, Siegler IC. Alcoholic beverage preference, diet, and health habits in the UNC Alumni Heart Study. Am J Clin Nutr. 2002;76(2):466-472. (PubMed)
49. McCann SE, Sempos C, Freudenheim JL, et al. Alcoholic beverage preference and characteristics of drinkers and nondrinkers in western New York (United States). Nutr Metab Cardiovasc Dis. 2003;13(1):2-11. (PubMed)
50. American Stroke Association. Ischemic strokes (clots) [Web page]. Available at: http://www.strokeassociation.org/STROKEORG/AboutStroke/TypesofStroke/IschemicClots/Ischemic-Strokes-Clots_UCM_310939_Article.jsp. Accessed 9/24/14.
51. Grysiewicz RA, Thomas K, Pandey DK. Epidemiology of ischemic and hemorrhagic stroke: incidence, prevalence, mortality, and risk factors. Neurol Clin. 2008;26(4):871-895, vii. (PubMed)
52. Berger K, Ajani UA, Kase CS, et al. Light-to-moderate alcohol consumption and risk of stroke among U.S. male physicians. N Engl J Med. 1999;341(21):1557-1564. (PubMed)
53. Sacco RL, Elkind M, Boden-Albala B, et al. The protective effect of moderate alcohol consumption on ischemic stroke. JAMA. 1999;281(1):53-60. (PubMed)
54. Malarcher AM, Giles WH, Croft JB, et al. Alcohol intake, type of beverage, and the risk of cerebral infarction in young women. Stroke. 2001;32(1):77-83. (PubMed)
55. Mukamal KJ, Chung H, Jenny NS, et al. Alcohol use and risk of ischemic stroke among older adults: the cardiovascular health study. Stroke. 2005;36(9):1830-1834. (PubMed)
56. Jimenez M, Chiuve SE, Glynn RJ, et al. Alcohol consumption and risk of stroke in women. Stroke. 2012;43(4):939-945. (PubMed)
57. Zhang Y, Tuomilehto J, Jousilahti P, Wang Y, Antikainen R, Hu G. Lifestyle factors on the risks of ischemic and hemorrhagic stroke. Arch Intern Med. 2011;171(20):1811-1818. (PubMed)
58. Iso H, Baba S, Mannami T, et al. Alcohol consumption and risk of stroke among middle-aged men: the JPHC Study Cohort I. Stroke. 2004;35(5):1124-1129. (PubMed)
59. Reynolds K, Lewis B, Nolen JD, Kinney GL, Sathya B, He J. Alcohol consumption and risk of stroke: a meta-analysis. JAMA. 2003;289(5):579-588. (PubMed)
60. Patra J, Taylor B, Irving H, et al. Alcohol consumption and the risk of morbidity and mortality for different stroke types--a systematic review and meta-analysis. BMC Public Health. 2010;10:258. (PubMed)
61. Zhang C, Qin YY, Chen Q, et al. Alcohol intake and risk of stroke: a dose-response meta-analysis of prospective studies. Int J Cardiol. 2014;174(3):669-677. (PubMed)
62. Mills JL. Peripheral arterial disease. In: Rakel RE, Bope ET, eds. Rakel: Conn's Current Therapy 2002. New York: W.B. Saunders Company; 2002:340-343.
63. Kiani S, Aasen JG, Holbrook M, et al. Peripheral artery disease is associated with severe impairment of vascular function. Vasc Med. 2013;18(2):72-78. (PubMed)
64. Vliegenthart R, Geleijnse JM, Hofman A, et al. Alcohol consumption and risk of peripheral arterial disease: the Rotterdam study. Am J Epidemiol. 2002;155(4):332-338. (PubMed)
65. Camargo CA, Jr., Hennekens CH, Gaziano JM, Glynn RJ, Manson JE, Stampfer MJ. Prospective study of moderate alcohol consumption and mortality in US male physicians. Arch Intern Med. 1997;157(1):79-85. (PubMed)
66. Djousse L, Levy D, Murabito JM, Cupples LA, Ellison RC. Alcohol consumption and risk of intermittent claudication in the Framingham Heart Study. Circulation. 2000;102(25):3092-3097. (PubMed)
67. Mukamal KJ, Kennedy M, Cushman M, et al. Alcohol consumption and lower extremity arterial disease among older adults: the cardiovascular health study. Am J Epidemiol. 2008;167(1):34-41. (PubMed)
68. Klatsky AL, Chartier D, Udaltsova N, et al. Alcohol drinking and risk of hospitalization for heart failure with and without associated coronary artery disease. Am J Cardiol. 2005;96(3):346-351. (PubMed)
69. Djousse L, Gaziano JM. Alcohol consumption and risk of heart failure in the Physicians' Health Study I. Circulation. 2007;115(1):34-39. (PubMed)
70. Del Gobbo LC, Kalantarian S, Imamura F, et al. Contribution of major lifestyle risk Factors for incident heart failure in older adults: the Cardiovascular Health Study. JACC Heart Fail. 2015;3(7):520-528. (PubMed)
71. Albert CM, Manson JE, Cook NR, Ajani UA, Gaziano JM, Hennekens CH. Moderate alcohol consumption and the risk of sudden cardiac death among US male physicians. Circulation. 1999;100(9):944-950. (PubMed)
72. Chiuve SE, Rimm EB, Mukamal KJ, et al. Light-to-moderate alcohol consumption and risk of sudden cardiac death in women. Heart Rhythm. 2010;7(10):1374-1380. (PubMed)
73. Koppes LL, Dekker JM, Hendriks HF, Bouter LM, Heine RJ. Moderate alcohol consumption lowers the risk of type 2 diabetes: a meta-analysis of prospective observational studies. Diabetes Care. 2005;28(3):719-725. (PubMed)
74. Baliunas DO, Taylor BJ, Irving H, et al. Alcohol as a risk factor for type 2 diabetes: A systematic review and meta-analysis. Diabetes Care. 2009;32(11):2123-2132. (PubMed)
75. Carlsson S, Hammar N, Grill V. Alcohol consumption and type 2 diabetes Meta-analysis of epidemiological studies indicates a U-shaped relationship. Diabetologia. 2005;48(6):1051-1054. (PubMed)
76. Meyer KA, Conigrave KM, Chu NF, et al. Alcohol consumption patterns and HbA1c, C-peptide and insulin concentrations in men. J Am Coll Nutr. 2003;22(3):185-194. (PubMed)
77. Kenkre PV, Lindeman RD, Lillian Yau C, Baumgartner RN, Garry PJ. Serum insulin concentrations in daily drinkers compared with abstainers in the New Mexico elder health survey. J Gerontol A Biol Sci Med Sci. 2003;58(10):M960-963. (PubMed)
78. Greenfield JR, Samaras K, Jenkins AB, Kelly PJ, Spector TD, Campbell LV. Moderate alcohol consumption, estrogen replacement therapy, and physical activity are associated with increased insulin sensitivity: is abdominal adiposity the mediator? Diabetes Care. 2003;26(10):2734-2740. (PubMed)
79. Bantle AE, Thomas W, Bantle JP. Metabolic effects of alcohol in the form of wine in persons with type 2 diabetes mellitus. Metabolism. 2008;57(2):241-245. (PubMed)
80. Kao WH, Puddey IB, Boland LL, Watson RL, Brancati FL. Alcohol consumption and the risk of type 2 diabetes mellitus: atherosclerosis risk in communities study. Am J Epidemiol. 2001;154(8):748-757. (PubMed)
81. Laitinen K, Valimaki M, Keto P. Bone mineral density measured by dual-energy X-ray absorptiometry in healthy Finnish women. Calcif Tissue Int. 1991;48(4):224-231. (PubMed)
82. Holbrook TL, Barrett-Connor E. A prospective study of alcohol consumption and bone mineral density. BMJ. 1993;306(6891):1506-1509. (PubMed)
83. Felson DT, Zhang Y, Hannan MT, Kannel WB, Kiel DP. Alcohol intake and bone mineral density in elderly men and women. The Framingham Study. Am J Epidemiol. 1995;142(5):485-492. (PubMed)
84. New SA, Bolton-Smith C, Grubb DA, Reid DM. Nutritional influences on bone mineral density: a cross-sectional study in premenopausal women. Am J Clin Nutr. 1997;65(6):1831-1839. (PubMed)
85. Kroger H, Tuppurainen M, Honkanen R, Alhava E, Saarikoski S. Bone mineral density and risk factors for osteoporosis--a population-based study of 1600 perimenopausal women. Calcif Tissue Int. 1994;55(1):1-7. (PubMed)
86. Hansen MA, Overgaard K, Riis BJ, Christiansen C. Potential risk factors for development of postmenopausal osteoporosis--examined over a 12-year period. Osteoporos Int. 1991;1(2):95-102. (PubMed)
87. Rapuri PB, Gallagher JC, Balhorn KE, Ryschon KL. Alcohol intake and bone metabolism in elderly women. Am J Clin Nutr. 2000;72(5):1206-1213. (PubMed)
88. Ganry O, Baudoin C, Fardellone P. Effect of alcohol intake on bone mineral density in elderly women: The EPIDOS Study. Epidemiologie de l'Osteoporose. Am J Epidemiol. 2000;151(8):773-780. (PubMed)
89. Tucker KL, Jugdaohsingh R, Powell JJ, et al. Effects of beer, wine, and liquor intakes on bone mineral density in older men and women. Am J Clin Nutr. 2009;89(4):1188-1196. (PubMed)
90. McLernon DJ, Powell JJ, Jugdaohsingh R, Macdonald HM. Do lifestyle choices explain the effect of alcohol on bone mineral density in women around menopause? Am J Clin Nutr. 2012;95(5):1261-1269. (PubMed)
91. Wosje KS, Kalkwarf HJ. Bone density in relation to alcohol intake among men and women in the United States. Osteoporos Int. 2007;18(3):391-400. (PubMed)
92. Maurel DB, Boisseau N, Benhamou CL, Jaffre C. Alcohol and bone: review of dose effects and mechanisms. Osteoporos Int. 2012;23(1):1-16. (PubMed)
93. Neafsey EJ, Collins MA. Moderate alcohol consumption and cognitive risk. Neuropsychiatr Dis Treat. 2011;7:465-484. (PubMed)
94. Tyas SL. Alcohol use and the risk of developing Alzheimer's disease. Alcohol Res Health. 2001;25(4):299-306. (PubMed)
95. Stavro K, Pelletier J, Potvin S. Widespread and sustained cognitive deficits in alcoholism: a meta-analysis. Addict Biol. 2013;18(2):203-213. (PubMed)
96. Peters R, Peters J, Warner J, Beckett N, Bulpitt C. Alcohol, dementia and cognitive decline in the elderly: a systematic review. Age Ageing. 2008;37(5):505-512. (PubMed)
97. Panza F, Frisardi V, Seripa D, et al. Alcohol consumption in mild cognitive impairment and dementia: harmful or neuroprotective? Int J Geriatr Psychiatry. 2012;27(12):1218-1238. (PubMed)
98. Panza F, Capurso C, D'Introno A, et al. Alcohol drinking, cognitive functions in older age, predementia, and dementia syndromes. J Alzheimers Dis. 2009;17(1):7-31. (PubMed)
99. Anstey KJ, Mack HA, Cherbuin N. Alcohol consumption as a risk factor for dementia and cognitive decline: meta-analysis of prospective studies. Am J Geriatr Psychiatry. 2009;17(7):542-555. (PubMed)
100. Mukamal KJ, Longstreth WT, Jr., Mittleman MA, Crum RM, Siscovick DS. Alcohol consumption and subclinical findings on magnetic resonance imaging of the brain in older adults: the cardiovascular health study. Stroke. 2001;32(9):1939-1946. (PubMed)
101. den Heijer T, Vermeer SE, van Dijk EJ, et al. Alcohol intake in relation to brain magnetic resonance imaging findings in older persons without dementia. Am J Clin Nutr. 2004;80(4):992-997. (PubMed)
102. Ding J, Eigenbrodt ML, Mosley TH, Jr., et al. Alcohol intake and cerebral abnormalities on magnetic resonance imaging in a community-based population of middle-aged adults: the Atherosclerosis Risk in Communities (ARIC) study. Stroke. 2004;35(1):16-21. (PubMed)
103. Kato I, Nomura A, Stemmermann GN, Chyou PH. Prospective study of clinical gallbladder disease and its association with obesity, physical activity, and other factors. Dig Dis Sci. 1992;37(5):784-790. (PubMed)
104. Misciagna G, Leoci C, Guerra V, et al. Epidemiology of cholelithiasis in southern Italy. Part II: Risk factors. Eur J Gastroenterol Hepatol. 1996;8(6):585-593. (PubMed)
105. Leitzmann MF, Giovannucci EL, Stampfer MJ, et al. Prospective study of alcohol consumption patterns in relation to symptomatic gallstone disease in men. Alcohol Clin Exp Res. 1999;23(5):835-841. (PubMed)
106. Leitzmann MF, Tsai CJ, Stampfer MJ, et al. Alcohol consumption in relation to risk of cholecystectomy in women. Am J Clin Nutr. 2003;78(2):339-347. (PubMed)
107. Maclure KM, Hayes KC, Colditz GA, Stampfer MJ, Speizer FE, Willett WC. Weight, diet, and the risk of symptomatic gallstones in middle-aged women. N Engl J Med. 1989;321(9):563-569. (PubMed)
108. Tseng M, Everhart JE, Sandler RS. Dietary intake and gallbladder disease: a review. Public Health Nutr. 1999;2(2):161-172. (PubMed)
109. Scragg RK, McMichael AJ, Baghurst PA. Diet, alcohol, and relative weight in gall stone disease: a case-control study. Br Med J (Clin Res Ed). 1984;288(6424):1113-1119. (PubMed)
110. Riley EP, Infante MA, Warren KR. Fetal alcohol spectrum disorders: an overview. Neuropsychol Rev. 2011;21(2):73-80. (PubMed)
111. Waterman EH, Pruett D, Caughey AB. Reducing fetal alcohol exposure in the United States. Obstet Gynecol Surv. 2013;68(5):367-378. (PubMed)
112. Jacobson JL, Jacobson SW. Drinking moderately and pregnancy. Effects on child development. Alcohol Res Health. 1999;23(1):25-30. (PubMed)
113. Jacobson JL, Dodge NC, Burden MJ, Klorman R, Jacobson SW. Number processing in adolescents with prenatal alcohol exposure and ADHD: differences in the neurobehavioral phenotype. Alcohol Clin Exp Res. 2011;35(3):431-442. (PubMed)
114. Jacobson JL, Jacobson SW. Effects of prenatal alcohol exposure on child development. Alcohol Res Health. 2002;26(4):282-286. (PubMed)
115. Niclasen J. Drinking or not drinking in pregnancy: the multiplicity of confounding influences. Alcohol Alcohol. 2014;49(3):349-355. (PubMed)
116. Centers for Disease Control and Prevention. Fetal Alcohol Spectrum Disorders (FASDs). Alcohol use in pregnancy. April 2014. Available at: http://www.cdc.gov/ncbddd/fasd/alcohol-use.html. Accessed 8/6/15.
117. American Academy of Pediatrics. Committee on Substance Abuse and Committee on Children With Disabilities. Fetal alcohol syndrome and alcohol-related neurodevelopmental disorders. Pediatrics. 2000;106(2 Pt 1):358-361. (PubMed)
118. Singletary KW, Gapstur SM. Alcohol and breast cancer: review of epidemiologic and experimental evidence and potential mechanisms. JAMA. 2001;286(17):2143-2151. (PubMed)
119. Seitz HK, Pelucchi C, Bagnardi V, La Vecchia C. Epidemiology and pathophysiology of alcohol and breast cancer: Update 2012. Alcohol Alcohol. 2012;47(3):204-212. (PubMed)
120. International Agency for Research on Cancer WHO. IARC monographs on the evaluation of carcingenic risks to humans. Vol 96; 2010. Available at: http://monographs.iarc.fr/ENG/Monographs/vol96/index.php. Accessed 8/24/15.
121. Scoccianti C, Lauby-Secretan B, Bello PY, Chajes V, Romieu I. Female breast cancer and alcohol consumption: a review of the literature. Am J Prev Med. 2014;46(3 Suppl 1):S16-25. (PubMed)
122. Klatsky AL, Udaltsova N, Li Y, Baer D, Nicole Tran H, Friedman GD. Moderate alcohol intake and cancer: the role of underreporting. Cancer Causes Control. 2014;25(6):693-699. (PubMed)
123. Smith-Warner SA, Spiegelman D, Yaun SS, et al. Alcohol and breast cancer in women: a pooled analysis of cohort studies. JAMA. 1998;279(7):535-540. (PubMed)
124. Hamajima N, Hirose K, Tajima K, et al. Alcohol, tobacco and breast cancer--collaborative reanalysis of individual data from 53 epidemiological studies, including 58,515 women with breast cancer and 95,067 women without the disease. Br J Cancer. 2002;87(11):1234-1245. (PubMed)
125. Key J, Hodgson S, Omar RZ, et al. Meta-analysis of studies of alcohol and breast cancer with consideration of the methodological issues. Cancer Causes Control. 2006;17(6):759-770. (PubMed)
126. Gou YJ, Xie DX, Yang KH, et al. Alcohol consumption and breast cancer survival: a meta-analysis of cohort studies. Asian Pac J Cancer Prev. 2013;14(8):4785-4790. (PubMed)
127. Brooks PJ, Zakhari S. Moderate alcohol consumption and breast cancer in women: from epidemiology to mechanisms and interventions. Alcohol Clin Exp Res. 2013;37(1):23-30. (PubMed)
128. National Cancer Institute. Breast Cancer Risk in American Women. [Web page]. Available at: http://www.cancer.gov/types/breast/risk-fact-sheet. Accessed 12/15/15.
129. Baglietto L, English DR, Gertig DM, Hopper JL, Giles GG. Does dietary folate intake modify effect of alcohol consumption on breast cancer risk? Prospective cohort study. BMJ. 2005;331(7520):807. (PubMed)
130. Rohan TE, Jain MG, Howe GR, Miller AB. Dietary folate consumption and breast cancer risk. J Natl Cancer Inst. 2000;92(3):266-269. (PubMed)
131. Sellers TA, Kushi LH, Cerhan JR, et al. Dietary folate intake, alcohol, and risk of breast cancer in a prospective study of postmenopausal women. Epidemiology. 2001;12(4):420-428. (PubMed)
132. Zhang S, Hunter DJ, Hankinson SE, et al. A prospective study of folate intake and the risk of breast cancer. JAMA. 1999;281(17):1632-1637. (PubMed)
133. Zhang SM, Willett WC, Selhub J, et al. Plasma folate, vitamin B6, vitamin B12, homocysteine, and risk of breast cancer. J Natl Cancer Inst. 2003;95(5):373-380. (PubMed)
134. Sellers TA, Vierkant RA, Cerhan JR, et al. Interaction of dietary folate intake, alcohol, and risk of hormone receptor-defined breast cancer in a prospective study of postmenopausal women. Cancer Epidemiol Biomarkers Prev. 2002;11(10 Pt 1):1104-1107. (PubMed)
135. Feigelson HS, Jonas CR, Robertson AS, McCullough ML, Thun MJ, Calle EE. Alcohol, folate, methionine, and risk of incident breast cancer in the American Cancer Society Cancer Prevention Study II Nutrition Cohort. Cancer Epidemiol Biomarkers Prev. 2003;12(2):161-164. (PubMed)
136. Tjonneland A, Christensen J, Olsen A, et al. Folate intake, alcohol and risk of breast cancer among postmenopausal women in Denmark. Eur J Clin Nutr. 2006;60(2):280-286. (PubMed)
137. Duffy CM, Assaf A, Cyr M, et al. Alcohol and folate intake and breast cancer risk in the WHI Observational Study. Breast Cancer Res Treat. 2009;116(3):551-562. (PubMed)
138. Suzuki R, Iwasaki M, Inoue M, et al. Alcohol consumption-associated breast cancer incidence and potential effect modifiers: the Japan Public Health Center-based Prospective Study. Int J Cancer. 2010;127(3):685-695. (PubMed)
139. Larsson SC, Bergkvist L, Wolk A. Folate intake and risk of breast cancer by estrogen and progesterone receptor status in a Swedish cohort. Cancer Epidemiol Biomarkers Prev. 2008;17(12):3444-3449. (PubMed)
140. Lieberman DZ. Children of alcoholics: an update. Curr Opin Pediatr. 2000;12(4):336-340. (PubMed)
141. National Institute on Alcohol Abuse and Alcoholism. A family history of alcoholism: are you at risk? [Web page]. June 2012. Available at: http://pubs.niaaa.nih.gov/publications/FamilyHistory/famhist.htm. Accessed 9/9/14.
142. Weathermon R, Crabb DW. Alcohol and medication interactions. Alcohol Res Health. 1999;23(1):40-54. (PubMed)
143. National Institute on Alcohol Abuse and Alcoholism. Older adults and alcohol. Available at: http://pubs.niaaa.nih.gov/publications/olderAdults/olderAdults.htm#toc03. Accessed 9/9/14.
144. National Institute on Alcohol Abuse and Alcoholism. Alcohol: a women's health issue. 2008. Available at: http://pubs.niaaa.nih.gov/publications/brochurewomen/women.htm. Accessed 8/24/15.
145. Klatsky AL. Alcohol and cardiovascular disease--more than one paradox to consider. Alcohol and hypertension: does it matter? Yes. J Cardiovasc Risk. 2003;10(1):21-24. (PubMed)
146. Cushman WC. Alcohol consumption and hypertension. J Clin Hypertens (Greenwich). 2001;3(3):166-170. (PubMed)
147. Briasoulis A, Agarwal V, Messerli FH. Alcohol consumption and the risk of hypertension in men and women: a systematic review and meta-analysis. J Clin Hypertens (Greenwich). 2012;14(11):792-798. (PubMed)
148. Taylor B, Irving HM, Baliunas D, et al. Alcohol and hypertension: gender differences in dose-response relationships determined through systematic review and meta-analysis. Addiction. 2009;104(12):1981-1990. (PubMed)
149. Xin X, He J, Frontini MG, Ogden LG, Motsamai OI, Whelton PK. Effects of alcohol reduction on blood pressure: a meta-analysis of randomized controlled trials. Hypertension. 2001;38(5):1112-1117. (PubMed)
150. Klatsky AL. Alcohol and cardiovascular diseases: a historical overview. Novartis Found Symp. 1998;216:2-12; discussion 12-18, 152-158. (PubMed)
151. Koskinen P, Kupari M, Leinonen H. Role of alcohol in recurrences of atrial fibrillation in persons less than 65 years of age. Am J Cardiol. 1990;66(12):954-958. (PubMed)
152. Ruigomez A, Johansson S, Wallander MA, Rodriguez LA. Incidence of chronic atrial fibrillation in general practice and its treatment pattern. J Clin Epidemiol. 2002;55(4):358-363. (PubMed)
153. Samokhvalov AV, Irving HM, Rehm J. Alcohol consumption as a risk factor for atrial fibrillation: a systematic review and meta-analysis. Eur J Cardiovasc Prev Rehabil. 2010;17(6):706-712. (PubMed)
154. Larsson SC, Drca N, Wolk A. Alcohol consumption and risk of atrial fibrillation: a prospective study and dose-response meta-analysis. J Am Coll Cardiol. 2014;64(3):281-289. (PubMed)
155. Conen D, Albert CM. Alcohol consumption and risk of atrial fibrillation: how much is too much? J Am Coll Cardiol. 2014;64(3):290-292. (PubMed)
156. Wannamethee G, Shaper AG. Alcohol and sudden cardiac death. Br Heart J. 1992;68(5):443-448. (PubMed)
157. Dyer AR, Stamler J, Paul O, et al. Alcohol consumption, cardiovascular risk factors, and mortality in two Chicago epidemiologic studies. Circulation. 1977;56(6):1067-1074. (PubMed)
158. Balbao CE, de Paola AA, Fenelon G. Effects of alcohol on atrial fibrillation: myths and truths. Ther Adv Cardiovasc Dis. 2009;3(1):53-63. (PubMed)
159. Piano MR, Phillips SA. Alcoholic cardiomyopathy: pathophysiologic insights. Cardiovasc Toxicol. 2014;14(4):291-308. (PubMed)
160. Klatsky AL. Alcohol and cardiovascular diseases: where do we stand today? J Intern Med. 2015;278(3):238-250. (PubMed)
161. Fernandez-Sola J, Nicolas-Arfelis JM. Gender differences in alcoholic cardiomyopathy. J Gend Specif Med. 2002;5(1):41-47. (PubMed)
162. Urbano-Marquez A, Estruch R, Fernandez-Sola J, Nicolas JM, Pare JC, Rubin E. The greater risk of alcoholic cardiomyopathy and myopathy in women compared with men. JAMA. 1995;274(2):149-154. (PubMed)
163. Louvet A, Mathurin P. Alcoholic liver disease: mechanisms of injury and targeted treatment. Nat Rev Gastroenterol Hepatol. 2015;12(4):231-242. (PubMed)
164. Corrao G, Bagnardi V, Zambon A, La Vecchia C. A meta-analysis of alcohol consumption and the risk of 15 diseases. Prev Med. 2004;38(5):613-619. (PubMed)
165. Rehm J, Baliunas D, Borges GL, et al. The relation between different dimensions of alcohol consumption and burden of disease: an overview. Addiction. 2010;105(5):817-843. (PubMed)
166. Maher JJ. Alcoholic liver disease. In: Feldman M, Friedman LS, Sleisenger LH, eds. Sleisenger & Fordtran's Gastrointestinal and Liver Disease. 7th ed. St. Louis: W.B. Saunders; 2002:1375-1387.
167. Lieber CS. Alcohol and hepatitis C. Alcohol Res Health. 2001;25(4):245-254. (PubMed)
168. Bagnardi V, Blangiardo M, La Vecchia C, Corrao G. Alcohol consumption and the risk of cancer: a meta-analysis. Alcohol Res Health. 2001;25(4):263-270. (PubMed)
169. Doll R, Forman D, La Vecchia C, Woutersen R. Alcoholic beverages and cancers of the digestive tract and larynx. In: MacDonald I, ed. Health Issues Related to Alcohol Consumption. Oxford: Blackwell Science Ltd; 1999:351-394.
170. Bernardin F, Maheut-Bosser A, Paille F. Cognitive impairments in alcohol-dependent subjects. Front Psychiatry. 2014;5:78. (PubMed)
171. Thomson AD. Mechanisms of vitamin deficiency in chronic alcohol misusers and the development of the Wernicke-Korsakoff syndrome. Alcohol Alcohol Suppl. 2000;35(1):2-7. (PubMed)
172. Greenberg DM, Lee JW. Psychotic manifestations of alcoholism. Curr Psychiatry Rep. 2001;3(4):314-318. (PubMed)
173. DiMagno EP, Chari S. Acute Pancreatitis. In: Feldman M, Friedman LS, Sleisenger LH, eds. Sleisenger & Fordtran's Gastrointestinal and Liver Disease. St. Louis: W.B. Saunders; 2002:913-942.
174. Yadav D, Whitcomb DC. The role of alcohol and smoking in pancreatitis. Nat Rev Gastroenterol Hepatol. 2010;7(3):131-145. (PubMed)
175. Forsmark CE. Chronic Pancreatitis. In: Feldman M, Friedman LS, Sleisenger LH, eds. Sleisenger & Fordtran's Gastrointestinal and Liver Disease. St. Louis: W.B. Saunders; 2002.
176. Yadav D, Papachristou GI, Whitcomb DC. Alcohol-associated pancreatitis. Gastroenterol Clin North Am. 2007;36(2):219-238, vii. (PubMed)
177. Pandol SJ, Lugea A, Mareninova OA, et al. Investigating the pathobiology of alcoholic pancreatitis. Alcohol Clin Exp Res. 2011;35(5):830-837. (PubMed)
178. Health risks and benefits of alcohol consumption. Alcohol Res Health. 2000;24(1):5-11. (PubMed)
179. Vinson DC, Maclure M, Reidinger C, Smith GS. A population-based case-crossover and case-control study of alcohol and the risk of injury. J Stud Alcohol. 2003;64(3):358-366. (PubMed)
180. US National Highway Traffic Safety Admininstration's National Center for Statistics and Analysis. Traffic safety facts. 2013 data: Alcohol-impaired driving. December 2014. Available at: http://www-nrd.nhtsa.dot.gov/Pubs/812102.pdf. Accessed 8/6/15.
181. Moskowitz H, Fiorentino DA. Review of the literature on the effects of low doses of alcohol on driving-related skills. Washington D.C.: National Highway Traffic Safety Administration; 2000.
182. Fisher HR, Simpson RI, Kapur BM. Calculation of blood alcohol concentration (BAC) by sex, weight, number of drinks and time. Can J Public Health. 1987;78(5):300-304. (PubMed)
183. National Institute on Alcohol Abuse and Alcoholism. Alcohol alert: alcohol, violence, and aggression [Web page]. October 2000. Available at: http://pubs.niaaa.nih.gov/publications/aa38.htm. Accessed 10/3/07.