Contents
Nutritional status plays an important role in the maintenance of healthy skin (1-4). Macronutrients (carbohydrates, proteins, and lipids) and micronutrients (vitamins and nutritionally essential minerals) work together to maintain the barrier functions of skin in the face of everyday challenges. Changes in nutritional status that alter skin structure and function can also directly affect skin appearance. Unlike many organs, skin nutrition may be enhanced directly through topical applications. Topical application of micronutrients can complement dietary consumption, leading to a stronger, healthier protective barrier for the body. This article discusses the roles of select micronutrients and other dietary compounds in the maintenance of skin health. Topical use of these compounds is discussed, and the effects of micronutrient deficiencies in the skin are also covered.
Skin provides a physical and chemical barrier between the outside environment and the inside tissues of the body (5, 6). This ‘barrier function’ is critical to protect underlying tissues from pathogens, chemicals, and environmental exposures (5, 7). Structurally, skin is comprised of two main layers, the epidermis and the dermis (Figure 1) (6, 7). The epidermis, the upper layer, is responsible for many of the barrier functions of skin. The dermis is the structural and nutritive support network underneath the epidermis. Since each layer has its own unique structure and function, the nutritive requirements of each must be considered separately.
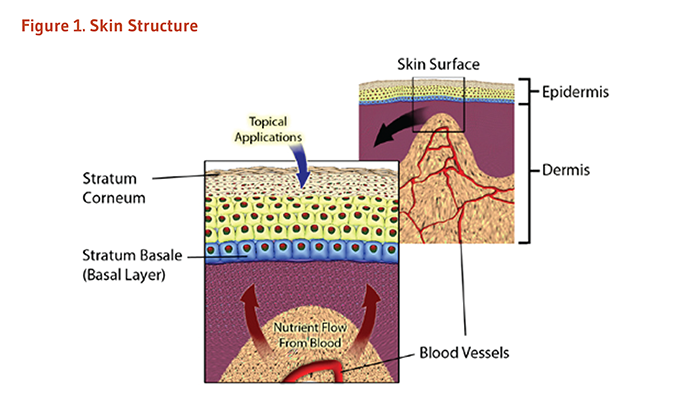
Human epidermis comprises the outermost layers of skin cells, ranging from 0.1 mm to 0.6 mm thick, depending on the location on the body (7). Keratinocytes compose the vast majority (90-95%) of cells within the epidermis. These cells are found in layers of varying size and thickness (6). The bottom or basal layer of the epidermis consists of a layer of round, undifferentiated keratinocytes that is supported by contact to the underlying dermis. Cells in this layer are constantly dividing in order to produce new cells that will make up the remainder of the epidermal layers (6, 7). Thus, it is in this fashion that skin is replenished, with new cells from the basal layer replacing the outer layer of skin cells that is shed over time.
Once a keratinocyte leaves the basal layer, it begins the process of cell differentiation known as keratinization (6). During keratinization, keratinocytes undergo several morphological changes that result in the synthesis of unique structural proteins (especially keratin) and the secretion of a variety of specialized lipids that will comprise key components of the epidermal barrier. These cells also undergo a process known as cornification, in which the synthesized proteins are assembled in an envelope just inside the cell membrane (5-7). At the end of this process, the large protein-laden cells, now known as corneocytes, lose their nuclei and other cellular organelles. Thus, there is little metabolic activity present in the outer layers of the skin (often referred to as ‘dead’ cells).
The outermost layer of skin, the stratum corneum, interacts with the outside environment. Lipids secreted by cells during the keratinization process are now assembled with extracellular proteins into a protective layer. This barrier is often likened to a ‘brick and mortar’ system: large, flattened, metabolically inactive corneocytes (the protein-rich ‘bricks’) are sealed together with a variety of extracellular lipids and proteins (the ‘mortar’) (5, 6). The chemical properties and structural design of this layer slow absorption and limit penetration of the skin, as well as limit the loss of vital nutrients and water from the underlying tissue. As new layers of cells are produced, the outer cells of the stratum corneum are enzymatically detached from this layer and shed in a process called desquamation (5).
Further, other cells contribute to the function of the epidermis. Melanocytes are cells that produce melanin, a compound involved in skin pigmentation produced in response to ultraviolet (UV) light exposure (6). Melanin can absorb energy from UV light to shield underlying tissues from damage. Langerhans cells are antigen-presenting cells involved in epidermal immunity. Merkel cells have various neuronal and endocrine roles; these cells function as mechanical sensors and produce growth factors that regulate sweat glands, hair follicles, nail growth, and nerve function in skin (6).
The dermis is the inner layer of skin situated between the epidermis and other tissues of the body, such as connective tissue, subcutaneous fat, muscle, and bone. The dermis can vary in depth from 0.3 mm to 4 mm depending on body location and is generally at least ten times thicker than the epidermis (7, 8). However, nearly 75% of the weight of the dermis is a matrix of collagen, an extracellular protein that allows for both structural support and elasticity of the skin. Thus, the primary role of the dermis is a mechanical support network for the epidermis, providing integrity and flexibility to skin. Blood vessels that supply nutrients for all skin layers are found in the dermis (6).
The dermis, which is less cell dense than the epidermis, is primarily comprised of a matrix of extracellular proteins. A majority of cells found in the dermis are fibroblasts that synthesize structural proteins like collagen. Immune cells, such as monocytes, macrophages, and dermal dendrocytes, also reside in the dermis, where they contribute to the defense of skin and modulate the response to injury (6). Subcutaneous fat cells provide structural support and energy reserves, as well assistance in thermoregulation. Hair follicles, sensory nerve endings, and sweat and sebaceous glands are also found in the dermis; each is made of specialized cells that support the various functions of the skin.
Although difficult to define, healthy skin has the ability to respond to challenges that would otherwise undermine its structure and function. Balanced nutrition complements the host of endogenous factors that preserve skin health. Moreover, skin that functions properly has aesthetically pleasing properties, giving skin a healthful appearance and feel. Therefore, conditions that adversely affect the biological functions of skin often correspond to a less attractive appearance of skin.
This section focuses on the everyday challenges to skin health and the prevention of chronic conditions (where applicable) rather than treatment of skin diseases.
Primary exposures to ultraviolet (UV) light are through sunlight and tanning beds. Although UV penetration of skin aids in vitamin D synthesis (see the article on Vitamin D and Skin Health), it has the potential to damage the cells and extracellular components of the skin (9-11). Free radicals are produced when light energy is absorbed by cellular components (10, 11). While the skin has endogenous antioxidant systems to combat free radicals and repair proteins, excessive exposures may overwhelm these defenses and lead to permanent damage (11, 12). This is often referred to as photodamage (11-13).
Sunburn is the most common form of acute photodamage, where the damage caused by excessive exposure of UV light leads to a large inflammatory response (erythema) (13). Prolonged or repeated exposures to UV light may cause permanent damage. This is often referred to as ‘photoaging’ due to the amount of time needed for these changes to appear and their similarities to intrinsic aging effects (see Skin aging) (12, 13). While skin laxity, wrinkling, and thickening are the most apparent signs of photodamage, changes in skin texture, abnormal skin growths, and impaired wound healing are also possible (13-15). Skin discoloration may also occur with chronic UV exposure, especially in the form of solar lentigines, also known as liver spots.
Prevention of photodamage begins with limiting exposure to UV light. Because avoidance of sunlight is not always practical, photoprotective agents are often used to limit exposure (9, 12). Sunscreens are the most commonly used photoprotective agents. However, use of sunscreens may only provide limited sun protection, expose the skin to chemicals that might otherwise damage or disrupt barrier function, or induce inflammatory reactions in the skin. Therefore, care must be taken in their use (12).
Since UV light exposure depletes antioxidant levels in the skin, including ascorbic acid (vitamin C) and α-tocopherol (vitamin E) (16-19), increasing antioxidant defenses in skin cells and/or nutritional support of cellular repair enzymes are also important routes to limit photodamage.
Although the lower layers of the epidermis are moist, there is a sharp decline in extracellular water content as the cells migrate outward toward the skin surface (20, 21). This is partially by design: the hydrophobic environment found in the stratum corneum slows the passage of water from the body out into the atmosphere, a phenomenon known as Transepidermal Water Loss (TEWL). Since water loss is directly related to the skin’s ability to maintain its barrier functions, measurements of TEWL are often used to determine skin dysfunction. However, small amounts of water are needed for the stratum corneum to maintain its structure. A mixture of stratum corneum components form a water-binding barrier, together known as natural moisturizing factor (NMF), to retain moisture content even in dry environments (20).
Dry skin can be caused by many factors, but it is usually accompanied by changes in the epidermal barrier and increased TEWL (more water lost to the environment). Intrinsic changes in the lipid barrier or NMF of the stratum corneum can disrupt the barrier and cause water loss. This can stem from simple chemical exposures, such as washing with detergents (20) or from more complex nutritional deficiencies, such as a lack of essential fatty acids (22). However, dry skin can also be an effect of atmospheric conditions or exposures. Changes in temperature, air flow, and humidity can pull water away from the skin and reduce barrier integrity. If left untreated, dry skin is often predisposed to insults from other sources, leading to cycles of cell damage and inflammation that perpetuate the condition.
An important strategy to treat dry skin is to maintain the lipid barrier and NMF components of the stratum corneum (20). This can be achieved either through nutritional support of the underlying epidermis or by the use of a variety of topical applications (23).
Little is known about the origin of wrinkles (24). However, skin laxity is associated with poor support of the epidermis by the underlying dermis. Loss of collagen, damage to collagen and elastin fibers in the dermis, or structural changes in the junction between the dermis and epidermis are thought to contribute to wrinkle formation (25-27).
Skin laxity may be accentuated by loss of blood vessels in the dermis (28). Additionally, smoking and photodamage increase skin wrinkling (27, 29). Thus, avoiding these sources of damage would benefit skin tone. Nutritional factors are thought to influence wrinkling (1), but their roles are not clear.
The process of wound healing involves a coordinated effort by a number of different cell types. The priorities of the skin during wound healing are to clear the damaged area of infection, re-establish the epidermal barrier, and reconstruct the damaged underlying dermis. Overall, healing of an injury to the skin requires interplay of the epidermis, dermis, and inflammatory mediators (30, 31).
After skin has been wounded, inflammatory cells are recruited to the area. The first responders are neutrophils, which clear the wounded area of damaged tissue and infectious particles and signal for the recruitment of macrophages to the wound (30). Macrophages, stimulated by growth factors at the wound site, take on a variety of roles in the wound healing process. Some macrophages assist in the removal of debris and damaged tissue, while others coordinate the remodeling of tissue at the wound site (31). Macrophages are essential to produce the signals for reepithelialization and dermal repair.
Reepithelialization is the process of restoring the skin’s barrier, thereby providing a safe environment for tissue remodeling underneath. Upon stimulation by the inflammatory response and loss of neighboring cells at wound edges, epidermal keratinocytes flow into a damaged area in an attempt to contact other keratinocytes and connect with the matrix of the dermis. This migration is coordinated with a rapid proliferation of the epidermis, which provides additional cells to fill in the wound site (31).
Fibroblasts of the surrounding dermis move into the wound during various stages of healing. Following a wound, a fibrin clot is often established if blood vessels were ruptured. Fibroblasts stimulate the orderly removal of fibrin, replacing the clot with a more stable collagen matrix (31). Fibroblasts also participate in wound contraction, which involves drawing together edges of the wound and undamaged tissue in order to restore skin integrity (30). During later stages of wound healing, fibroblasts are involved in reordering these collagen fibers into a more stable structure, integrated with elastin. Growth factors produced by macrophages, keratinocytes, and fibroblasts stimulate proliferation of blood vessels into the newly remodeled dermis to restore nutrient and oxygen flow (31).
Wound repair is never perfect. Skin tensile strength at a site of a healed injury is always lower than uninjured skin (30, 31). Scarring of the skin is a consequence of the repair process resulting from disorganized collagen deposits by fibroblasts and cytokine signals from inflammatory cells. Currently, no intervention has proven effective at preventing scar tissue formation.
While treatment of a wound involves preventing infection, support of the wound healing process can occur at different stages of repair. Efficient immune responses and support of reepithelialization are key factors in initial healing (31), removing damaged tissue, re-establishing the barrier from the environment, and remodeling the surrounding tissue. Nutritional support of the regenerating epidermis and dermis during the latter stages of wound healing is not only important in long-term wound resolution but is thought to promote the restoration of strong, healthy skin (32).
Skin is often the first visible manifestation of the aging process. However, the effects of age on skin appearance are often similar to the effects associated with photodamage and environmental exposures. This makes the changes induced by chronological age on skin — often referred to as intrinsic aging — difficult to distinguish from other effects (27).
Instrinsic skin aging is characterized by decreasing support from the dermis to the epidermis. Ridges on the interface between the two layers are diminished, preventing the dermis from providing adequate mechanical support to the epidermis (27, 33). Collagen levels are lower and extracellular proteins in the dermis are more disorganized in skin of older individuals compared to younger adults (27, 33, 34). These changes result in increased skin fragility and laxity, as well as decreased size of the dermis and reduced vascularization, which reduces nutrient availability to the skin (27, 29). Aged skin keratinocytes are relatively slow to differentiate and shed, which alters their ability to maintain the stratum corneum. These changes may lead to an overall dull skin appearance and loss of protective ability of skin (27).
Antioxidant protection is often thought to diminish in the skin of older individuals, but the extent of these changes is unclear. The results of one human study suggest that catalase (an antioxidant enzyme) levels in the epidermis of elderly subjects were increased compared to young subjects independent of sun exposure (16). The capacity of aging skin to maintain levels of other antioxidant enzymes was unchanged. However, elderly subjects had lower levels of α-tocopherol in the epidermis and lower levels of ascorbic acid in both skin layers (16).
Most of intrinsic skin aging cannot be avoided. The cumulative effects of decades of skin wear cannot be reversed, although the effects on skin appearance may be temporarily masked. Aging skin has a diminished ability to respond to stress; therefore, skin damage from other sources can enhance the signs of skin aging. This is particularly true for ultraviolet (UV) exposures, because antioxidant capacity, immune function, and melanin production may all be impaired in aged skin (27). Therefore, treatments designed to attenuate damage from other sources are also important factors in limiting age-associated skin damage.
The primary fuel for skin cells is glucose; glucose oxidation rates in skin are similar to those observed in resting skeletal muscle. Glucose also provides carbohydrate backbones for modification of proteins (glycoproteins) and lipids (glycoplipids) that comprise the extracellular environment of the epidermis (35). Aberrant glucose handling drastically affects skin structure and appearance (36).
Specialized lipids required for the development of the stratum corneum, such as sterols and ceramides, are synthesized in the epidermis from amino acids, carbohydrates, and phospholipids. However, differentiating keratinocytes also utilize fatty acids from circulating stores or dermal fat layers for energy. The extruded fatty acids that make up the mortar of the stratum corneum can absorb lipid-soluble materials placed on the external surface of this outermost skin layer. This is especially the case for sebum, a waxy substance secreted from the sebaceous glands that are attached to hair follicles, but it is also true for topically applied materials (35).
Amino acids are essential for the production of both dermal and epidermal structures, producing the extracellular matrix proteins and enzymes needed for the synthesis of the epidermal barrier. The amino acid requirements of skin are poorly understood (37).
Much of the role of nutrition in skin health focuses on the effects of deficiency, since the structural components of the skin are supported by a variety of nutritive factors, such as small peptides, minerals, and vitamins, which serve as enzyme cofactors, activators, or inhibitors (4). The skin is also constantly exposed to high concentrations of oxygen, UV light, and oxidizing chemicals, highlighting a role for antioxidant vitamins in skin function (38, 39). Further, nutritional support of the skin is important for inflammatory response during wound healing (32).
The architecture of skin deserves special consideration in regard to nutrition. The epidermal layers of the skin do not contain blood vessels that supply the cells with nutrients; blood vessels are found only in the dermis. Additionally, as the epidermis develops, its unique protein and lipid structure (the aforementioned “bricks and mortar” model) prevents the circulation of extracellular fluids (7). Therefore, the outer layers of the epidermis are provided with less nutritional support than the underlying cells. Dietary interventions to affect skin health may be limited in effect and slow to occur.
Unique among organs, the skin may be directly affected by the use of topical applications. The stratum corneum prevents the passage of many different types of molecules, but some compounds pass through to the underlying layers. In general, uncharged or lipid-soluble molecules pass through the epidermis and may also penetrate the dermis. Concentrations of nutrients in the skin may be comparable to that achieved through oral ingestion. Yet, topical application may be a more efficient, targeted method for supplying nutrients to the skin, especially to the epidermis (12, 39).
Changes in the appearance of skin were associated with poor nutrition long before any biochemical relationships were discovered. Among the first to make these associations was the Scottish physician James Lind, who described the bleeding gums, skin discoloration, abnormal hair growth, and poor wound healing associated with the vitamin C deficiency disease known as scurvy (see the article on Vitamin C and Skin Health). Other investigators would soon find skin abnormalities in vitamin deficiency diseases (pellagra, ariboflavinosis, etc.) that could be corrected with an appropriate diet. Thus, many micronutrients were initially identified for their impact on skin health.
The effect of micronutrient supplementation on skin health is a relatively new field of study, as associations between diet and skin conditions are only now starting to emerge (1-3, 40). In addition, topical applications of micronutrients have only recently been considered to be reasonable alternatives to oral supplementation (9, 12, 39).
The remainder of this article focuses on select micronutrients and other natural factors that are important for skin health. Please note that this site is under development, and new articles will be added once they are completed and reviewed. Commercially available products occasionally refer to these compounds as “bionutrients” due to their abundance in natural sources. The role of these factors in the unique environment of the skin will be explained, and the effects of micronutrient deficiencies on the skin will be discussed. Further, the effects of dietary intake and topical application of these natural products on skin health will be explored.
Essential Fatty Acids and Skin Health
Written in September 2011 by:
Alexander J. Michels, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in September 2011 by:
Zoe Diana Draelos, M.D.
Consulting Professor
Department of Dermatology
Duke University School of Medicine
Durham, NC
This article was underwritten, in part, by a grant from Neutrogena Corporation, Los Angeles, California.
Copyright 2011-2024 Linus Pauling Institute
1. Purba MB, Kouris-Blazos A, Wattanapenpaiboon N, et al. Skin wrinkling: can food make a difference? J Am Coll Nutr 2001;20:71-80. (PubMed)
2. Cosgrove MC, Franco OH, Granger SP, Murray PG, Mayes AE. Dietary nutrient intakes and skin-aging appearance among middle-aged American women. Am J Clin Nutr 2007;86:1225-1231. (PubMed)
3. Boelsma E, van de Vijver LP, Goldbohm RA, Klopping-Ketelaars IA, Hendriks HF, Roza L. Human skin condition and its associations with nutrient concentrations in serum and diet. Am J Clin Nutr 2003;77:348-355. (PubMed)
4. Roe DA. Nutrition and the skin. New York: Liss; 1986.
5. Madison KC. Barrier function of the skin: "la raison d'etre" of the epidermis. J Invest Dermatol 2003;121:231-241. (PubMed)
6. Freinkel RK, Woodley D. The biology of the skin. New York: Parthenon Pub. Group; 2001.
7. Odland GF. Structure of the skin. In: Goldsmith LA, ed. Physiology, biochemistry, and molecular biology of the skin. 2nd ed. New York: Oxford University Press; 1991:3-62.
8. Goldsmith LA. Physiology, biochemistry, and molecular biology of the skin. 2nd ed. New York: Oxford University Press; 1991.
9. DeBuys HV, Levy SB, Murray JC, Madey DL, Pinnell SR. Modern approaches to photoprotection. Dermatol Clin 2000;18:577-590. (PubMed)
10. Sander CS, Chang H, Salzmann S, et al. Photoaging is associated with protein oxidation in human skin in vivo. J Invest Dermatol 2002;118:618-625. (PubMed)
11. Darr D, Fridovich I. Free radicals in cutaneous biology. J Invest Dermatol 1994;102:671-675. (PubMed)
12. Pinnell SR. Cutaneous photodamage, oxidative stress, and topical antioxidant protection. J Am Acad Dermatol 2003;48:1-19; quiz 20-2. (PubMed)
13. Kligman LH, Kligman AM. The nature of photoaging: its prevention and repair. Photodermatol 1986;3:215-227. (PubMed)
14. Berneburg M, Plettenberg H, Krutmann J. Photoaging of human skin. Photodermatol Photoimmunol Photomed 2000;16:239-244. (PubMed)
15. Kang S, Fisher GJ, Voorhees JJ. Photoaging: pathogenesis, prevention, and treatment. Clin Geriatr Med 2001;17:643-659, v-vi. (PubMed)
16. Rhie G, Shin MH, Seo JY, et al. Aging- and photoaging-dependent changes of enzymic and nonenzymic antioxidants in the epidermis and dermis of human skin in vivo. J Invest Dermatol 2001;117:1212-1217. (PubMed)
17. Shindo Y, Witt E, Han D, Epstein W, Packer L. Enzymic and non-enzymic antioxidants in epidermis and dermis of human skin. J Invest Dermatol 1994;102:122-124. (PubMed)
18. Shindo Y, Witt E, Han D, Packer L. Dose-response effects of acute ultraviolet irradiation on antioxidants and molecular markers of oxidation in murine epidermis and dermis. J Invest Dermatol 1994;102:470-475. (PubMed)
19. Shindo Y, Witt E, Packer L. Antioxidant defense mechanisms in murine epidermis and dermis and their responses to ultraviolet light. J Invest Dermatol 1993;100:260-265. (PubMed)
20. Rawlings AV, Matts PJ. Stratum corneum moisturization at the molecular level: an update in relation to the dry skin cycle. J Invest Dermatol 2005;124:1099-1110. (PubMed)
21. Rawlings AV, Scott IR, Harding CR, Bowser PA. Stratum corneum moisturization at the molecular level. J Invest Dermatol 1994;103:731-741. (PubMed)
22. Proksch E, Feingold KR, Elias PM. Epidermal HMG CoA reductase activity in essential fatty acid deficiency: barrier requirements rather than eicosanoid generation regulate cholesterol synthesis. J Invest Dermatol 1992;99:216-220. (PubMed)
23. Rawlings AV. Trends in stratum corneum research and the management of dry skin conditions. Int J Cosmet Sci 2003;25:63-95. (PubMed)
24. Kligman AM, Zheng P, Lavker RM. The anatomy and pathogenesis of wrinkles. Br J Dermatol 1985;113:37-42. (PubMed)
25. Contet-Audonneau JL, Jeanmaire C, Pauly G. A histological study of human wrinkle structures: comparison between sun-exposed areas of the face, with or without wrinkles, and sun-protected areas. Br J Dermatol 1999;140:1038-1047. (PubMed)
26. Craven NM, Watson RE, Jones CJ, Shuttleworth CA, Kielty CM, Griffiths CE. Clinical features of photodamaged human skin are associated with a reduction in collagen VII. Br J Dermatol 1997;137:344-350. (PubMed)
27. Baumann L. Skin ageing and its treatment. J Pathol 2007;211:241-251. (PubMed)
28. Gilchrest BA, Stoff JS, Soter NA. Chronologic aging alters the response to ultraviolet-induced inflammation in human skin. J Invest Dermatol 1982;79:11-15. (PubMed)
29. Farage MA, Miller KW, Elsner P, Maibach HI. Intrinsic and extrinsic factors in skin ageing: a review. Int J Cosmet Sci 2008;30:87-95. (PubMed)
30. Martin P. Wound healing--aiming for perfect skin regeneration. Science 1997;276:75-81. (PubMed)
31. Singer AJ, Clark RA. Cutaneous wound healing. N Engl J Med 1999;341:738-746. (PubMed)
32. Thompson C, Fuhrman MP. Nutrients and wound healing: still searching for the magic bullet. Nutr Clin Pract 2005;20:331-347. (PubMed)
33. El-Domyati M, Attia S, Saleh F, et al. Intrinsic aging vs. photoaging: a comparative histopathological, immunohistochemical, and ultrastructural study of skin. Exp Dermatol 2002;11:398-405. (PubMed)
34. Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol 1986;15:571-585. (PubMed)
35. Freinkel RK. Metabolism of skin. In: Freinkel RK, Woodley D, eds. The biology of the skin. New York: Parthenon Pub. Group; 2001:191-199.
36. Van Hattem S, Bootsma AH, Thio HB. Skin manifestations of diabetes. Cleve Clin J Med 2008;75:772, 4, 6-7 passim. (PubMed)
37. Obled C, Arnal M. Contribution of skin to whole-body protein synthesis in rats at different stages of maturity. J Nutr 1992;122:2167-2173. (PubMed)
38. Steenvoorden DP, van Henegouwen GM. The use of endogenous antioxidants to improve photoprotection. J Photochem Photobiol B 1997;41:1-10. (PubMed)
39. Zussman J, Ahdout J, Kim J. Vitamins and photoaging: do scientific data support their use? J Am Acad Dermatol 2010;63:507-525. (PubMed)
40. Nagata C, Nakamura K, Wada K, et al. Association of dietary fat, vegetables and antioxidant micronutrients with skin ageing in Japanese women. Br J Nutr 2010;103:1493-1498. (PubMed)