Potassium
Contents
Summary
- Potassium is considered to be a "nutrient of public health concern" according to the 2015-2020 Dietary Guidelines for Americans since its underconsumption in the US population is associated with adverse health effects (hypertension and cardiovascular disease). (More information)
- Normal body function depends on tight regulation of potassium concentrations both inside and outside of cells. (More information)
- Low potassium concentration in blood (hypokalemia) can result in muscular paralysis or abnormal heart rhythms and can be fatal. Hypokalemia is usually due to excessive loss of potassium as with prolonged vomiting or diarrhea, use of diuretics, or with kidney disease. (More information)
- Chronic hypertension damages the heart, blood vessels, and kidneys, thereby increasing the risk of cardiovascular disease. Increasing dietary potassium intake may help lower blood pressure in normotensive and hypertensive individuals. (More information)
- Results from observational studies reported higher dietary potassium intakes to be associated with lower risks of stroke and kidney stone formation. Evidence of a role for potassium intakes in promoting bone health remains weak. (More information)
- The adequate intake (AI) for potassium is 2,600 mg/day for women and 3,400 mg/day for men. The AI for each age/life stage group was set based on the level of intake reported in apparently healthy populations. (More information)
- Good dietary sources of potassium include fruit and vegetables, some nuts and seeds, and dairy products. (More information)
- Safety concerns with consuming potassium are limited in healthy people because the kidneys adjust urinary potassium excretion to potassium intake. Concomitant use of potassium supplements with certain drugs can increase the risk of potassium toxicity. (More information)
Potassium is an essential dietary mineral and electrolyte. The term electrolyte refers to a substance that dissociates into ions (charged particles) in solution, making it capable of conducting electricity. Normal body function depends on tight regulation of potassium concentrations both inside and outside of cells (1).
Function
Maintenance of membrane potential
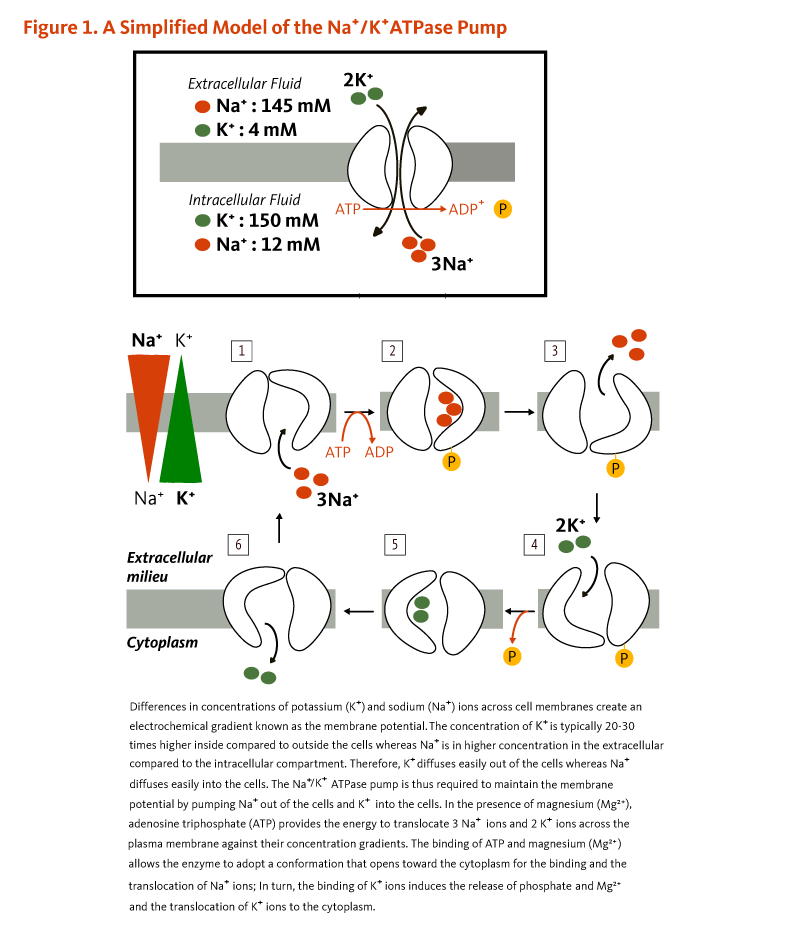
Potassium (K+) is the principal positively charged ion (cation) in the fluid inside of cells, while sodium (Na+) is the principal cation in the extracellular fluid. Potassium concentrations are about 30 times higher inside than outside cells, while sodium concentrations are more than 10 times lower inside than outside cells. The concentration differences between potassium and sodium across cell membranes create an electrochemical gradient known as the membrane potential. A cell's membrane potential is maintained by ion pumps in the cell membrane, especially the Na+/K+-ATPase pumps. These pumps use ATP (energy) to pump sodium out of the cell in exchange for potassium (Figure 1). Their activity has been estimated to account for 20%-40% of the resting energy expenditure in a typical adult. The large proportion of energy dedicated to maintaining sodium/potassium concentration gradients emphasizes the importance of this function in sustaining life. Tight control of cell membrane potential is critical for nerve impulse transmission, muscle contraction, and heart function (2-4).
Cofactor for enzymes
A limited number of enzymes require the presence of potassium for their activity. The activation of Na+/K+-ATPase requires the presence of sodium and potassium. The presence of potassium is also required for the activity of pyruvate kinase, an important enzyme in carbohydrate metabolism (5).
Deficiency
An abnormally low plasma potassium concentration is referred to as hypokalemia. Hypokalemia is most commonly a result of excessive loss of potassium, e.g., from prolonged vomiting or diarrhea, use of some diuretics and other medications (see Drug interactions), some forms of kidney disease, or metabolic disturbances. The symptoms of hypokalemia are related to alterations in membrane potential and cellular metabolism (1). They include fatigue, muscle weakness and cramps, and intestinal paralysis, which may lead to bloating, constipation, and abdominal pain. Chronic hypokalemia is associated with hypertension and kidney stone formation (see Disease Prevention and Disease Treatment). Severe hypokalemia may result in muscular paralysis or abnormal heart rhythms (cardiac arrhythmias) that can be fatal (1, 6).
Conditions that increase the risk of hypokalemia (see also Drug interactions; 1):
- The use of potassium-wasting diuretics (e.g., thiazide diuretics or furosemide)
- Prolonged vomiting or diarrhea
- Overuse or abuse of laxatives
- Anorexia nervosa or bulimia
- Excessive sweating
- Nephropathies
- Polyuria
- Abnormally high production of aldosterone (hyperaldosteronism)
- Magnesium depletion
- Recovery from prolonged undernutrition
Low dietary potassium intake alone does not generally result in hypokalemia. However, insufficient dietary potassium in patients at risk of hypokalemia can precipitate hypokalemia (1).
In rare cases, habitual consumption of large amounts of black licorice has resulted in hypokalemia (7, 8). Licorice contains a compound (i.e., glycyrrhizic acid) with similar physiologic effects to those of aldosterone, a hormone that increases urinary excretion of potassium.
The Adequate Intake (AI)
The Dietary Reference Intakes (DRIs) for potassium have been recently revised by the Food and Nutrition Board (FNB) of the National Academy of Medicine. The FNB did not find sufficient evidence to determine an Estimated Average Requirement (EAR) and derive a Recommended Dietary Allowance (RDA); instead, they established an adequate intake (AI) based on median intakes in generally healthy people (Table 1) (9). The FNB found insufficient evidence from human studies that examined potassium intakes in relation to chronic disease and mortality (reviewed recently by the Agency for Healthcare Research and Quality; 10) to inform the DRIs for potassium (11).
Disease Prevention
The diets of people residing in Western industrialized countries are quite different from those that were consumed before the agricultural revolution and the shift towards the consumption of highly refined, processed food (12). Among other differences, the daily intake of sodium chloride (salt) in modern diets is about three times higher than the daily intake of potassium on a molar basis, whereas salt intake in primitive cultures is about seven times lower than potassium intake (13). The relative deficiency of dietary potassium in the modern diet and a higher sodium-to-potassium ratio may contribute to the development of some chronic diseases.
Stroke
Observational studies have consistently reported an increased risk of cardiovascular disease with elevated dietary sodium intakes (14, 15). Several prospective cohort studies have also found an inverse association between potassium intake and risk of stroke. A meta-analysis of nine prospective cohort studies showed that daily potassium intakes ranging between 3,510 mg and 4,680 mg were associated with a 30% reduced risk of stroke (16). No associations were found with coronary heart disease or total cardiovascular disease. In a more recent meta-analysis of 16 studies, the highest versus lowest dietary potassium intake was found to be associated with a 13% lower risk of stroke after multiple adjustments (including for blood pressure) (17). The lowest risk of stroke corresponded to daily potassium intakes around 3,500 mg. Subgroup analyses showed a reduced risk of ischemic stroke, but not hemorrhagic stroke. Finally, in a recent meta-analysis of 16 observational studies, each 1-unit increase in the dietary sodium-to-potassium ratio was found to be associated with a 22% higher risk of stroke (12).
Kidney stones
Abnormally high urinary calcium (hypercalciuria) increases the risk of developing kidney stones. In individuals with a history of developing calcium-containing kidney stones, increased dietary acid load has been significantly associated with increased urinary calcium excretion (18). Increasing dietary potassium (and alkali) intake by increasing fruit and vegetable intake or by taking potassium bicarbonate (KHCO3) supplements has been found to decrease urinary calcium excretion. Conversely, potassium deprivation has been found to increase urinary calcium excretion (19, 20).
Three large US prospective cohort studies — the Health Professionals Follow-up Study and the Nurses’ Health Studies I and II — which included 193,676 participants, have examined dietary potassium intake and animal protein-to-potassium ratio (a marker of dietary acid load) in the diet in relation to the risk of developing kidney stones (21). In all three cohorts, dietary potassium intake was derived almost entirely from potassium-rich foods, such as fruit and vegetables. Across the three cohorts, individuals in the highest quintile of potassium intake were found to be 33%-56% less likely to develop symptomatic kidney stones than those in the lowest quintile of intake. Additionally, a pooled analysis of the data from all three cohorts showed that those with the highest versus lowest animal protein-to-potassium ratio were 41% more likely to develop kidney stones (21).
Urinary alkalinization with supplemental potassium citrate is used in stone formers to reduce the risk of recurrent stone formation (reviewed in 22). However, potassium citrate therapy should only be initiated under the supervision of a medical provider.
Osteoporosis
In a 2015 case-cohort study nested within the European Prospective Investigation into Cancer and Nutrition (EPIC)-Norfolk study, which included 5,319 individuals, dietary intakes of potassium (alone or combined with intakes of magnesium) were found to be inversely associated with heel bone (calcaneus) broadband ultrasound attenuation (BUA) measurements (a predictor of the risk of incidental fracture) and risk of hip fracture in women but not in men (23). More recently, a cross-sectional study in older Korean adults reported higher total hip and femur neck bone mineral density (BMD) in those in the top versus bottom tertile of potassium intakes (24). Although these observational studies suggest a link between potassium intakes and bone health, they cannot establish whether there is a cause-and-effect relationship.
The mechanisms by which potassium might influence bone health are poorly understood. Modern (Western) diets tend to be relatively low in sources of alkali (fruit and vegetables) and high in sources of acid (fish, meat, and cheese) (25). When the quantity of bicarbonate ions is insufficient to maintain normal pH, the body is capable of mobilizing alkaline calcium salts from bone in order to neutralize acids consumed in the diet or generated by metabolism (26). Because fruit and vegetables are rich in both potassium and precursors to bicarbonate ions, increasing their consumption might help reduce the net acid content of the diet and preserve calcium in bones, which might otherwise be mobilized to maintain normal pH (see the article on Fruit and Vegetables).
Alternatively, potassium bicarbonate supplementation might decrease urinary acid and calcium excretion and influence bone turnover — a small trial in postmenopausal women found that potassium bicarbonate supplementation increases biomarkers of bone formation and a decreased biomarkers of bone resorption (27). A two-year randomized, double-blind, controlled trial in 201 older adults without osteoporosis (mean age, 69 years) found evidence of increased lumbar spine, hip, femoral neck, and total-body BMD, as well as trabecular BMD of the radius and tibia, with supplemental potassium citrate (2,340 mg/day) compared to placebo (28). Potassium citrate was also found to increase the serum concentration of N-terminal propeptide of type I procollagen (PINP) — a marker of bone formation — and reduce the urine concentration of N-telopeptide of collagen type I (NTX) — a marker of bone resorption (28). Another three-month, randomized, placebo-controlled trial in 244 adults (≥50 years) examined the effect of oral potassium bicarbonate, at either 39 mg/kg/day or 58.5 mg/kg/day, on markers of bone turnover (29). Both dosage regimens led to reductions in serum PINP concentration and urine NTX concentration, yet evidence of an effect was stronger with the lowest dose (median dose administered, 3,160 mg/day) rather than with the highest dose (median dose administered, 4,760 mg/day). In contrast, a two-year randomized controlled trial found that neither supplementation with potassium citrate (721 mg/day or 2,165 mg/day) nor an increase in fruit and vegetable intake (300 mg/day) had an impact on markers of bone turnover or increased BMD in postmenopausal women (30). A 2015 meta-analysis of intervention studies found that supplemental potassium citrate or potassium bicarbonate could reduce urinary net acid and calcium excretion, but evidence to support an effect on markers of bone turnover and bone density was weak (31). The most recent randomized, double-blind, controlled trial in 40 postmenopausal women with osteopenia found no difference in markers of bone turnover over a six-month period between those supplemented with potassium citrate and those taking a placebo (32). The authors highlighted the possibility that an effect of supplemental potassium might have an impact on bone health in a subset of subjects with low potassium intakes and/or signs of low-grade acidosis. In this study, all participants received daily supplements of calcium carbonate (500 mg/day) and vitamin D (10 µg/day).
Overall, whether consuming potassium-rich fruit and vegetables can influence bone health and help lower the risk of osteoporosis remains uncertain (see also the article on Fruit and Vegetables).
Disease Treatment
Hypertension
Forty-five percent of US adults have hypertension (blood pressure levels ≥130/80 mm Hg) (33). Chronic hypertension damages the heart, blood vessels, and kidneys, thereby increasing the risk of heart disease and stroke, as well as hypertensive kidney disease (34, 35). Modern diets, which are high in sodium and low in potassium, are recognized as largely contributing to the high prevalence of hypertension (see the article on Sodium). Unlike 24-hour dietary recalls, 24-hour urine collections provide accurate estimates of dietary intakes of sodium and potassium (36). An analysis of the 2014 US National Health and Nutrition Examination Survey (NHANES) showed an increase in systolic blood pressure with increasing sodium excretion and increasing sodium-to-potassium ratio in the urine (37). In this study, the highest versus lowest quartile of urinary potassium excretion (mid-values, 3,043 mg/day versus 1,484 mg/day) was associated with a 62% lower risk of hypertension (37).
The Dietary Approaches to Stop Hypertension (DASH) trial provided evidence of the blood pressure-lowering effect of a diet higher in potassium and calcium, modestly higher in protein, and lower in total fat, saturated fat, cholesterol, red meat, sweets, and sugar-containing beverages compared to the typical US diet (38). Indeed, compared to the control diet providing only 3.5 servings/day of fruit and vegetables and 1,700 mg/day of potassium, adherence to the DASH diet that included 8.5 servings/day of fruit and vegetables and 4,100 mg/day of potassium lowered systolic/diastolic blood pressures by an average 11.4/5.5 mm Hg in people with hypertension and 3.5/2.1 mm Hg in those without hypertension (38). A 2014 meta-analysis of 17 randomized controlled trials that examined the effect of the DASH diet compared to a control diet in a total of 2,561 adults found overall reductions in systolic and diastolic blood pressure by 6.7 mm Hg and 3.5 mm Hg, respectively (39). However effective the DASH diet is, the blood pressure-lowering effects can hardly be solely attributed to potassium intakes (40).
A 2015 meta-analysis of 15 randomized controlled trials, including 917 individuals, assessed the effects of increased potassium intake, mostly in the form of potassium chloride (KCl) supplements, on blood pressure (41). Thirteen studies included hypertensive participants who were not taking anti-hypertensive medication and two studies included normotensive or at-risk subjects. Most studies used supplemental potassium doses between 2,340 and 2,535 mg/day (60-65 mmol/L). Increased potassium intake resulted in overall reductions of systolic blood pressure by 4.7 mm Hg and diastolic blood pressure by 3.5 mm Hg. The blood pressure-lowering effect of supplemental potassium was more pronounced when the analysis was restricted to individuals with hypertension: systolic and diastolic blood pressure were found to be reduced by 6.8 mm Hg and 4.6 mm Hg, respectively (41). Two additional meta-analyses published in 2017 also confirmed a blood pressure-lowering effect of supplemental potassium. Findings suggested some evidence of a greater effect when baseline potassium intake was less than 3,510 mg/day (vs. ≥3,510 mg/day [90 mmol]) (42). Meta-analyses have also reported a dose-response relationship between the intake of potassium and the lowering of blood pressure (42, 43).
Supplemental potassium can help lower blood pressure, but potassium supplements should only be used in consultation with a medical provider (see Supplements). Increasing potassium intake to recommended levels (see Adequate Intake) by consuming a diet rich in fruit and vegetables can help lower blood pressure and may have additional benefits to health (see the article on Fruit and Vegetables). Blood pressure is a reliable cardiovascular risk marker (44). Yet, although reducing sodium consumption while increasing potassium intake helps with lowering blood pressure (45), current evidence suggests that dietary advice and support interventions may not be sufficient to deliver long-term cardiovascular benefits in individuals with hypertension (46).
Sources
Food sources
The richest sources of potassium are fruit and vegetables. Nuts, seeds, and dairy products are also good sources of potassium. A dietary survey in the US indicated that the average dietary potassium intake was 2,408 mg/day for adult women and 3,172 mg/day for adult men (47). Because many individuals in the population consume potassium in amounts that are well below the AI and because underconsumption of potassium is linked with adverse health effects, potassium has been recognized as a "nutrient of public health concern" in the 2015-2020 Dietary Guidelines for Americans. In 2016, the US Food and Drug Administration (FDA) required manufacturers to display potassium content of foods on the Nutrition Facts food label (48).
Some relatively good dietary sources of potassium are listed in the Table 2, along with their potassium content in milligrams (mg). For more information on the nutrient content of foods, search USDA's FoodData Central (49).
Supplements
Multivitamin-mineral supplements in the US do not contain more than 99 mg of potassium per serving (50). One milliequivalent (mEq) or one millimole (mmol) corresponds to about 39 mg of potassium. Higher doses of supplemental potassium are generally prescribed to prevent and treat potassium depletion and hypokalemia. The use of more potent potassium supplements in potassium deficiency requires close monitoring of serum potassium concentrations. Potassium is available in different supplemental forms, including potassium chloride, potassium citrate, potassium gluconate, potassium bicarbonate, potassium aspartate, and potassium orotate (50). Because of the potential for serious side effects, one should seek medical advice before deciding to use a potassium supplement (see Safety). The best way to increase one’s potassium intake is by increasing the consumption of potassium-rich food and beverages (50).
Finally, many salt substitutes contain potassium chloride, and acesulfame potassium (Ace-K) is an FDA-approved general purpose sweetener.
Safety
Toxicity
Abnormally elevated serum potassium concentrations are referred to as hyperkalemia. Hyperkalemia occurs when potassium intake exceeds the capacity of the kidneys to eliminate it. Acute or chronic kidney failure, the use of potassium-sparing diuretics, and insufficient aldosterone secretion (hypoaldosteronism) may result in the accumulation of potassium due to a decreased urinary potassium excretion. Oral doses of potassium >18 g taken at one time in individuals not accustomed to high intakes may lead to severe hyperkalemia, even in those with normal kidney function (6, 50). Hyperkalemia may also result from a shift of intracellular potassium into the circulation, which may occur with the rupture of red blood cells (hemolysis) or tissue damage (e.g., trauma or severe burns). Symptoms of hyperkalemia may include tingling of the hands and feet, muscular weakness, and temporary paralysis. The most serious complication of hyperkalemia is the development of an abnormal heart rhythm (cardiac arrhythmia), which can lead to cardiac arrest (51). A meta-analysis of randomized controlled studies showed that heart rate in healthy adults was unlikely to be affected by the chronic use of supplemental potassium doses of 2 to 3 g/day (52).
See the section on Drug interactions for a discussion of the medications that increase the risk of hyperkalemia.
Adverse reactions to potassium supplements
Gastrointestinal symptoms are the most common side effects of potassium supplements, including nausea, vomiting, abdominal discomfort, and diarrhea. Intestinal ulceration has been reported after the use of enteric-coated potassium chloride tablets. Taking potassium with meals or taking a microencapsulated form of potassium may reduce gastrointestinal side effects (50). Rashes may occasionally occur. The most serious adverse reaction to potassium supplementation is hyperkalemia, yet is rare in subjects with normal kidney function (see Toxicity). Individuals with abnormal kidney function and those on potassium-sparing medications (see Drug interactions) should be monitored closely to prevent hyperkalemia (50, 53).
Drug interactions
Table 3 lists the classes of medications known to increase the risk of hyperkalemia (elevated serum potassium) in patients who also use potassium supplements (50, 51, 54).
Several classes of medications are known to induce hypokalemia (low serum potassium; Table 4; 55) . In the absence of treatment, hypokalemia can have serious complications and even be fatal (see Deficiency). Various mechanisms explain how certain medications can lead to potassium depletion. For example, both loop and thiazide diuretics increase the urinary excretion of potassium. Corticoids cause sodium retention that leads to a compensatory increase in urinary potassium excretion. Penicillins formulated as sodium salts also stimulate potassium excretion. Several medications, including aminoglycosides, anti-fungal agents (amphotericin-B, fluconazole), and cisplatin, can damage the renal tubular epithelium and lead to severe potassium loss. Outdated tetracycline antibiotics have been linked to electrolyte disturbances.
Linus Pauling Institute Recommendation
There is substantial evidence suggesting that a diet high in potassium-rich food and beverages may be associated with lower risks of stroke, hypertension, kidney stones, and possibly osteoporosis. However, currently there is insufficient evidence to establish a causal relationship between potassium intakes and the risk of these chronic conditions (10). As a consequence, median potassium intakes observed in apparently healthy people were used to set adequate intakes (AI) by age/life stage in the recent revision of the Dietary Reference Intakes (DRIs) for potassium. The revised AI values are 2.6 g/day for women and 3.4 g/day for men (see The Adequate Intake).
Fruit and vegetables are among the richest sources of dietary potassium, and a large body of evidence supports the association of increased fruit and vegetable intakes with reduced risk of cardiovascular disease (see the article on Fruit and Vegetables). The Linus Pauling Institute recommends the consumption of a diet high in potassium-rich foods (see Sources), especially fruit, vegetables, nuts, and dairy products to ensure adequate potassium intakes.
Older adults (>50 years)
A diet rich in fruit and vegetables that supplies 2.6-3.4 g/day of potassium (see AI) should contribute to maintaining a low risk of chronic disease in generally healthy older adults. This recommendation does not apply to individuals who have been advised to limit potassium consumption by a health care professional (see Safety).
Authors and Reviewers
Originally written in 2001 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in February 2004 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in December 2010 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in April 2019 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in April 2019 by
Connie Weaver, Ph.D.
Distinguished Professor and Department Head
Department of Nutrition Science
Purdue University
Copyright 2001-2024 Linus Pauling Institute
References
1. Bailey JL, Sands JM, Franch HA. Water, electrolytes, and acid — Base Metabolism In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease: Lippincott Williams & Wilkins; 2014:102-132.
2. Clausen T. Quantification of Na+,K+ pumps and their transport rate in skeletal muscle: functional significance. J Gen Physiol. 2013;142(4):327-345. (PubMed)
3. Larsen BR, Stoica A, MacAulay N. Managing brain extracellular K(+) during neuronal activity: the physiological role of the Na(+)/K(+)-ATPase subunit isoforms. Front Physiol. 2016;7:141. (PubMed)
4. Shattock MJ, Ottolia M, Bers DM, et al. Na+/Ca2+ exchange and Na+/K+-ATPase in the heart. J Physiol. 2015;593(6):1361-1382. (PubMed)
5. Sheng H-W. Sodium, chloride and potassium. In: Stipanuk M, ed. Biochemical and Physiological Aspects of Human Nutrition. Philadelphia: W.B. Saunders Company; 2000:686-710.
6. Food and Nutrition Board, Institute of Medicine. Potassium. Dietary Reference Intakes for Water, Potassium, Sodium, Chloride, and Sulfate. Washington, D.C.: National Academies Press; 2005:186-268. (The National Academies Press)
7. Mumoli N, Cei M. Licorice-induced hypokalemia. Int J Cardiol. 2008;124(3):e42-44. (PubMed)
8. Walker BR, Edwards CR. Licorice-induced hypertension and syndromes of apparent mineralocorticoid excess. Endocrinol Metab Clin North Am. 1994;23(2):359-377. (PubMed)
9. Food and Nutrition Board, National Academy of Medicine. Dietary Reference Intakes for Sodium and Potassium - uncorrected proofs. The National Academies of Sciences, Engineering, and Medicine. Washington, D.C.: The National Academies Press; 2019. (The National Academies Press)
10. Newberry SJ, Chung M, Anderson CAM, et al. AHRQ Comparative Effectiveness Reviews. Sodium and potassium intake: effects on chronic disease outcomes and risks. Rockville (MD): Agency for Healthcare Research and Quality (US); 2018. (PubMed)
11. Food and Nutrition Board, National Academy of Medicine. Potassium: Dietary Reference Intakes based on chronic disease. Dietary Reference Intakes for Sodium and Potassium - uncorrected proofs. The National Academies of Sciences, Engineering, and Medicine. Washington, D.C.: National Academy Press; 2019:121-154. (The National Academies Press)
12. Weaver CM. Potassium and health. Adv Nutr. 2013;4(3):368s-377s. (PubMed)
13. Young DB, Lin H, McCabe RD. Potassium's cardiovascular protective mechanisms. Am J Physiol. 1995;268(4 Pt 2):R825-837. (PubMed)
14. Aburto NJ, Ziolkovska A, Hooper L, Elliott P, Cappuccio FP, Meerpohl JJ. Effect of lower sodium intake on health: systematic review and meta-analyses. BMJ. 2013;346:f1326. (PubMed)
15. Jayedi A, Ghomashi F, Zargar MS, Shab-Bidar S. Dietary sodium, sodium-to-potassium ratio, and risk of stroke: A systematic review and nonlinear dose-response meta-analysis. Clin Nutr. 2018; doi: 10.1016/j.clnu.2018.05.017. [Epub ahead of print]. (PubMed)
16. Aburto NJ, Hanson S, Gutierrez H, Hooper L, Elliott P, Cappuccio FP. Effect of increased potassium intake on cardiovascular risk factors and disease: systematic review and meta-analyses. BMJ. 2013;346:f1378. (PubMed)
17. Vinceti M, Filippini T, Crippa A, de Sesmaisons A, Wise LA, Orsini N. Meta-analysis of potassium intake and the risk of stroke. J Am Heart Assoc. 2016;5(10). (PubMed)
18. Trinchieri A, Zanetti G, Curro A, Lizzano R. Effect of potential renal acid load of foods on calcium metabolism of renal calcium stone formers. Eur Urol. 2001;39 Suppl 2:33-36; discussion 36-37. (PubMed)
19. Lemann J, Jr., Pleuss JA, Gray RW. Potassium causes calcium retention in healthy adults. J Nutr. 1993;123(9):1623-1626. (PubMed)
20. Morris RC, Jr., Schmidlin O, Tanaka M, Forman A, Frassetto L, Sebastian A. Differing effects of supplemental KCl and KHCO3: pathophysiological and clinical implications. Semin Nephrol. 1999;19(5):487-493. (PubMed)
21. Ferraro PM, Mandel EI, Curhan GC, Gambaro G, Taylor EN. Dietary protein and potassium, diet-dependent net acid load, and risk of incident kidney stones. Clin J Am Soc Nephrol. 2016;11(10):1834-1844. (PubMed)
22. Suarez M, Youssef RF. Potassium citrate: treatment and prevention of recurrent calcium nephrolithiasis. J Clin Nephrol Res. 2015;2(1).
23. Hayhoe RP, Lentjes MA, Luben RN, Khaw KT, Welch AA. Dietary magnesium and potassium intakes and circulating magnesium are associated with heel bone ultrasound attenuation and osteoporotic fracture risk in the EPIC-Norfolk cohort study. Am J Clin Nutr. 2015;102(2):376-384. (PubMed)
24. Kong SH, Kim JH, Hong AR, Lee JH, Kim SW, Shin CS. Dietary potassium intake is beneficial to bone health in a low calcium intake population: the Korean National Health and Nutrition Examination Survey (KNHANES) (2008-2011). Osteoporos Int. 2017;28(5):1577-1585. (PubMed)
25. Fenton TR, Eliasziw M, Lyon AW, Tough SC, Hanley DA. Meta-analysis of the quantity of calcium excretion associated with the net acid excretion of the modern diet under the acid-ash diet hypothesis. Am J Clin Nutr. 2008;88(4):1159-1166. (PubMed)
26. Morris RC, Jr., Frassetto LA, Schmidlin O, Forman A, Sabastian A. Expression of osteoporosis as determined by diet-disordered electrolyte and acid-base metabolism. In: Burckhardt P, Dawson-Hughes B, Heaney R, eds. Nutritional Aspects of Osteoporosis. San Diego: Academic Press; 2001:357-378.
27. Sebastian A, Harris ST, Ottaway JH, Todd KM, Morris RC, Jr. Improved mineral balance and skeletal metabolism in postmenopausal women treated with potassium bicarbonate. N Engl J Med. 1994;330(25):1776-1781. (PubMed)
28. Jehle S, Hulter HN, Krapf R. Effect of potassium citrate on bone density, microarchitecture, and fracture risk in healthy older adults without osteoporosis: a randomized controlled trial. J Clin Endocrinol Metab. 2013;98(1):207-217. (PubMed)
29. Dawson-Hughes B, Harris SS, Palermo NJ, et al. Potassium bicarbonate supplementation lowers bone turnover and calcium excretion in older men and women: a randomized dose-finding trial. J Bone Miner Res. 2015;30(11):2103-2111. (PubMed)
30. Macdonald HM, Black AJ, Aucott L, et al. Effect of potassium citrate supplementation or increased fruit and vegetable intake on bone metabolism in healthy postmenopausal women: a randomized controlled trial. Am J Clin Nutr. 2008;88(2):465-474. (PubMed)
31. Lambert H, Frassetto L, Moore JB, et al. The effect of supplementation with alkaline potassium salts on bone metabolism: a meta-analysis. Osteoporos Int. 2015;26(4):1311-1318. (PubMed)
32. Granchi D, Caudarella R, Ripamonti C, et al. Potassium citrate supplementation decreases the biochemical markers of bone loss in a group of osteopenic women: the results of a randomized, double-blind, placebo-controlled pilot study. Nutrients. 2018;10(9). (PubMed)
33. Centers for Disease Control and Prevention. High Blood Pressure Facts. November 2016. Available at: https://www.cdc.gov/bloodpressure/facts.htm. Accessed 11/30/18.
34. Mente A, O'Donnell M, Rangarajan S, et al. Associations of urinary sodium excretion with cardiovascular events in individuals with and without hypertension: a pooled analysis of data from four studies. Lancet. 2016;388(10043):465-475. (PubMed)
35. Sanghavi S, Vassalotti JA. Dietary sodium: a therapeutic target in the treatment of hypertension and CKD. J Ren Nutr. 2013;23(3):223-227. (PubMed)
36. Cogswell ME, Loria CM, Terry AL, et al. Estimated 24-hour urinary sodium and potassium excretion in US adults. JAMA. 2018;319(12):1209-1220. (PubMed)
37. Jackson SL, Cogswell ME, Zhao L, et al. Association between urinary sodium and potassium excretion and blood pressure among adults in the United States: National Health and Nutrition Examination Survey, 2014. Circulation. 2018;137(3):237-246. (PubMed)
38. Appel LJ, Moore TJ, Obarzanek E, et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N Engl J Med. 1997;336(16):1117-1124. (PubMed)
39. Saneei P, Salehi-Abargouei A, Esmaillzadeh A, Azadbakht L. Influence of Dietary Approaches to Stop Hypertension (DASH) diet on blood pressure: a systematic review and meta-analysis on randomized controlled trials. Nutr Metab Cardiovasc Dis. 2014;24(12):1253-1261. (PubMed)
40. Weaver CM, Stone MS, Lobene AJ, Cladis DP, Hodges JK. What is the evidence base for a potassium requirement? Nutr Today. 2018;53(5):184-195. (PubMed)
41. Binia A, Jaeger J, Hu Y, Singh A, Zimmermann D. Daily potassium intake and sodium-to-potassium ratio in the reduction of blood pressure: a meta-analysis of randomized controlled trials. J Hypertens. 2015;33(8):1509-1520. (PubMed)
42. Filippini T, Violi F, D'Amico R, Vinceti M. The effect of potassium supplementation on blood pressure in hypertensive subjects: A systematic review and meta-analysis. Int J Cardiol. 2017;230:127-135. (PubMed)
43. Poorolajal J, Zeraati F, Soltanian AR, Sheikh V, Hooshmand E, Maleki A. Oral potassium supplementation for management of essential hypertension: A meta-analysis of randomized controlled trials. PLoS One. 2017;12(4):e0174967. (PubMed)
44. Viera AJ. Screening for hypertension and lowering blood pressure for prevention of cardiovascular disease events. Med Clin North Am. 2017;101(4):701-712. (PubMed)
45. Appel LJ, Giles TD, Black HR, et al. ASH position paper: dietary approaches to lower blood pressure. J Am Soc Hypertens. 2010;4(2):79-89. (PubMed)
46. Adler AJ, Taylor F, Martin N, Gottlieb S, Taylor RS, Ebrahim S. Reduced dietary salt for the prevention of cardiovascular disease. Cochrane Database Syst Rev. 2014(12):Cd009217. (PubMed)
47. Hoy MK, Goldman JD. Potassium Intake of the US Population: What We Eat In America, NHANES 2009-2010. 2012.
48. Food and Drug Administration. Food Labeling: Revision of the Nutrition and Supplement Facts Labels. Available at: https://www.regulations.gov/document?D=FDA-2012-N-1210-0875. Accessed 12/12/18.
49. US Department of Agriculture, Agricultural Research Service. FoodData Central, 2019. fdc.nal.usda.gov.
50. Hendler SS, Rorvik DR. PDR for Nutrititional Supplements. Montvale: Thomson Reuters; 2008.
51. Mandal AK. Hypokalemia and hyperkalemia. Med Clin North Am. 1997;81(3):611-639. (PubMed)
52. Gijsbers L, Molenberg FJ, Bakker SJ, Geleijnse JM. Potassium supplementation and heart rate: A meta-analysis of randomized controlled trials. Nutr Metab Cardiovasc Dis. 2016;26(8):674-682. (PubMed)
53. Gennari FJ. Hypokalemia. N Engl J Med. 1998;339(7):451-458. (PubMed)
54. Natural Medicines. Potassium/Professional Monograph/Interactions with Drugs. June 26, 2018. Available at: https://naturalmedicines.therapeuticresearch.com. Accessed 11/19/18.
55. Natural Medicines. Potassium/Professional Monograph/Nutrient Depletion. June 26, 2018. Available at: https://naturalmedicines.therapeuticresearch.com. Accessed 11/19/18.