Indole-3-Carbinol
Contents
Summary
- Indole-3-carbinol (I3C) is derived from the breakdown of glucobrassicin, a compound found in cruciferous vegetables. (More information)
- In the stomach, I3C molecules undergo acid-catalyzed condensation that generates a number of biologically active I3C oligomers, such as 3,3'-diindolylmethane (DIM) and 5,11-dihydroindolo-[3,2-b]carbazole (ICZ). (More information)
- I3C and DIM have been found to modulate the expression and activity of biotransformation enzymes that are involved in the metabolism and elimination of many biologically active compounds, including steroid hormones, drugs, carcinogens, and toxins. (More information)
- Preclinical studies suggested that anti-estrogenic activities of I3C and DIM might help reduce the risk of hormone-dependent cancers. Although supplementation with I3C and DIM could alter urinary estrogen metabolite profiles in women, the effects of I3C and DIM on breast cancer risk are not known. (More information)
- Preclinical studies showed that I3C and I3C oligomers could affect multiple signaling pathways that are dysregulated in cancer cells, such as those controlling cell proliferation, apoptosis, migration, invasion, and angiogenesis. (More information)
- Limited evidence from preliminary trials suggested that I3C supplementation may help treat conditions related to human papilloma virus (HPV) infection, such as cervical/vulvar intraepithelial neoplasias and recurrent respiratory papillomatosis. However, randomized controlled trials are needed to determine whether I3C supplementation is beneficial. (More information)
- The timing of I3C exposure in animal models of chemically-induced cancers seems to determine whether I3C inhibits or promotes the development of tumors. Some experts have cautioned against the widespread use of I3C and DIM supplements for cancer prevention in humans until their potential risks and benefits are better understood. (More information)
Introduction
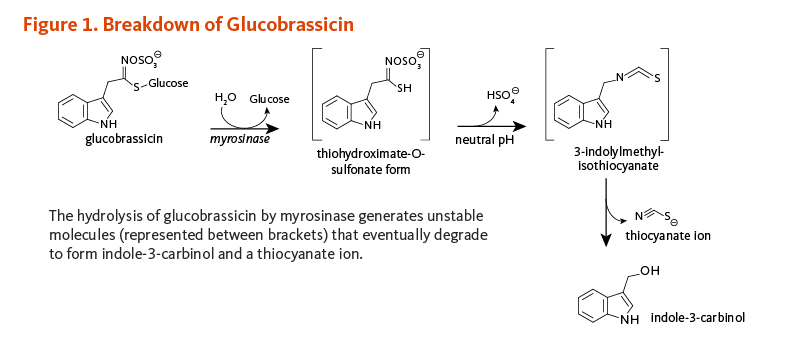
Some observational studies have reported significant associations between high intakes of cruciferous vegetables and lower risk of several types of cancer (1). Cruciferous vegetables differ from other classes of vegetables in that they are rich sources of sulfur-containing compounds known as glucosinolates (for detailed information, see the article on Cruciferous Vegetables) (2). The potential health benefits of consuming cruciferous vegetables are attributed to compounds derived from the enzymatic hydrolysis (breakdown) of glucosinolates. Among these compounds is indole-3-carbinol (I3C), a compound derived from the degradation of an indole glucosinolate commonly known as glucobrassicin (Figure 1).
Metabolism and Bioavailability
A number of commonly consumed cruciferous vegetables, including broccoli, Brussels sprouts, and cabbage, are good sources of glucobrassicin — the glucosinolate precursor of I3C (see Food sources).
Myrosinase (β-thioglucosidase), an enzyme that catalyzes the hydrolysis of glucosinolates, is physically separated from glucosinolates in intact plant cells (3). When raw cruciferous vegetables are chopped or chewed, plant cells are damaged such that glucobrassicin is exposed to myrosinase. The hydrolysis of glucobrassicin initially produces a glucose molecule and the unstable aglycone, thiohydroximate-O-sulfonate. The spontaneous release of a sulfate ion results in the formation of another unstable intermediate form, 3-indolylmethylisothiocyanate (4). This compound easily splits into thiocyanate ion and I3C (Figure 1). In the acidic environment of the stomach, I3C molecules can combine with each other to form a complex mixture of polycyclic aromatic compounds, known collectively as acid condensation products (Figure 2) (5). Some of the most prominent acid condensation products include 3,3'-diindolylmethane (DIM), 5,11-dihydroindolo-[3,2-b]carbazole (ICZ), and a cyclic triindole (CT) (Figure 2). The biological activities of individual acid condensation products may differ from those of I3C (see Biological Activities).
When cruciferous vegetables are cooked, plant myrosinase is inactivated thus the hydrolysis of glucosinolates is prevented. Intact glucosinolates then transit to the colon and are metabolized by human intestinal bacteria. The generation of I3C from glucobrassicin may still occur to a lesser degree in the large intestine, due to the myrosinase activity of colonic bacteria (4). Thus, when cruciferous vegetables are cooked, I3C can still form in the colon, but I3C-derived acid condensation products are less likely to form in the more alkaline environment of the intestine.
No I3C could be detected in plasma following oral administration of single doses of I3C, ranging from 200 to 1,200 mg, to healthy women at high risk for breast cancer (6). However, DIM was detected and peaked in plasma around two hours after I3C ingestion, at concentrations from <100 nanograms per milliliter (ng/mL) with oral doses of 400 to 600 mg of I3C up to 500 ng/mL-600 ng/mL with oral doses of 1,000 to 1,200 mg of I3C. All DIM disappeared from the blood within 24 hours (6).
Formulation strategies, such as the encapsulation of I3C and DIM into nanoparticles or liposomes (7-9), are being developed with the aim of increasing the bioavailability and evaluating the safety and efficacy of these compounds in humans.
Biological Activities
Effects on biotransformation enzymes
Biotransformation enzymes play major roles in the metabolism and elimination of many biologically active compounds, including physiologic regulators (e.g., estrogens), drugs, and environmental chemicals (xenobiotics; e.g., carcinogens, toxins). In general, phase I metabolizing enzymes, including the cytochrome P450 (CYP) family, catalyze reactions that increase the reactivity of hydrophobic (fat-soluble) compounds, which prepares them for reactions catalyzed by phase II detoxifying enzymes. Reactions catalyzed by phase II enzymes usually increase water solubility and promote the elimination of these compounds (10).
Aryl hydrocarbon receptor (AhR) pathway
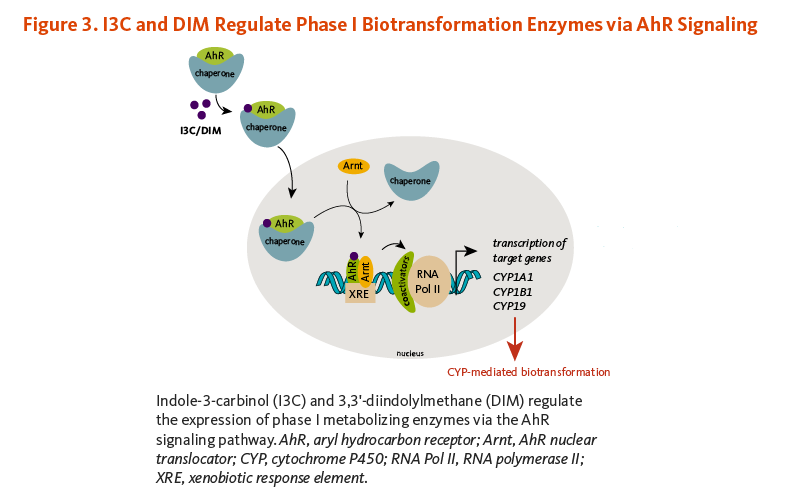
I3C and some I3C condensation products can bind to a protein in the cytoplasm of cells called the aryl hydrocarbon receptor (AhR) (Figure 3) (11-13). In fact, ICZ is one of the most potent ligands for the AhR known with an affinity approaching that of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). I3C acid condensation products, as well as indoles and their acid condensation products formed from tryptophan metabolism, appear to be important endogenous ligands for the AhR (13). Binding allows AhR to enter the nucleus where it forms a complex with the AhR nuclear translocator (Arnt) protein. This AhR/Arnt complex binds to specific DNA sequences, known as xenobiotic response elements (XRE), in the regulatory regions (promoters) of target genes, especially those involved in xenobiotic metabolism (14). The promoters of genes coding for a number of CYP enzymes and several phase II enzymes contain XREs. Microarray gene expression profiling of I3C- or DIM-treated human prostate cancer cells showed that both compounds upregulated the phase I enzyme, CYP1A1, and the phase II enzymes, glutathione S-transferase theta-1 (GST q1) and aldo-keto reductase (15). Another study in human prostate cancer cells demonstrated that the removal of AhR abolished I3C- or DIM-induced CYP1A1 mRNA expression (16). The expression of CYP1A1 and CYP1A2 was also upregulated in human primary liver cells challenged with DIM (17). Further, I3C and DIM have been found to interfere with CYP activities involved in estrogen metabolism (see Anti-estrogenic activities).
Increasing the activity of biotransformation enzymes is generally considered a beneficial effect because the elimination of potential carcinogens or toxins is enhanced. However, there is a potential for adverse effects because some procarcinogens require biotransformation by phase I enzymes to become active carcinogens (18).
Nuclear factor E2-related factor 2-dependent pathway
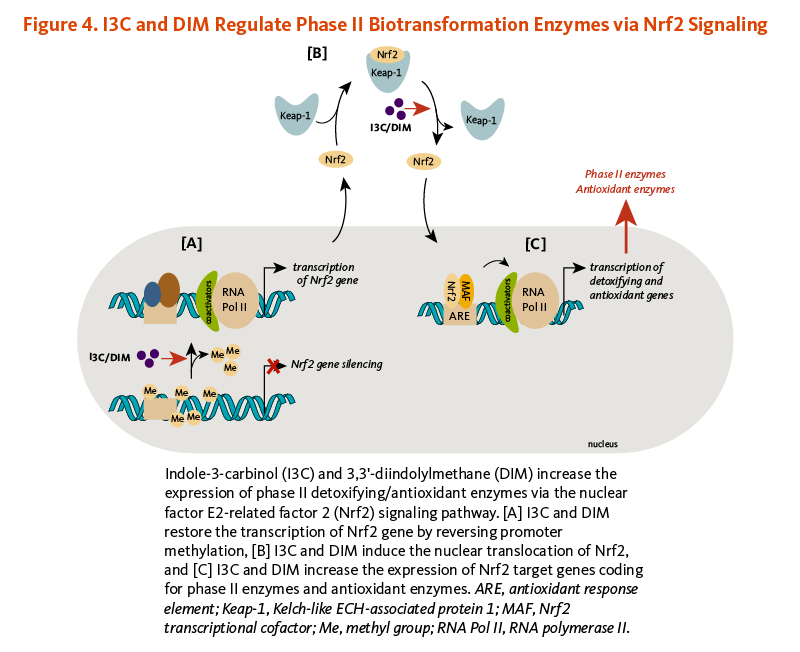
I3C and DIM have been shown to induce the expression of phase II detoxifying and antioxidant enzymes via the activation of the nuclear factor E2-related factor 2 (Nrf2)-dependent pathway.
Briefly, Nrf2 is a transcription factor that is bound to the protein Kelch-like ECH-associated protein 1 (Keap1) in the cytosol (Figure 4). Keap1 responds to oxidative stress signals or chemical inducers by freeing Nrf2. Upon release, Nrf2 translocates to the nucleus and binds to the antioxidant response element (ARE) located in the promoters of genes coding for antioxidant/detoxifying enzymes and scavengers. Nrf2/ARE-dependent genes code for numerous mediators of the antioxidant response, including glutathione S-transferases (GSTs), thioredoxin, NAD(P)H quinone oxidoreductase 1 (NQO-1), and heme oxygenase 1 (HO-1) (19). DIM induced Nrf2/ARE-dependent upregulation of HO-1, and I3C stimulated NQO-1 and GST (µ2 isoform) expression in liver cancer cells (20). In addition, the transcription of Nrf2 coding gene, which was abnormally repressed through promoter DNA hypermethylation, was enhanced in mouse prostate cancer cells treated with DIM. DIM subsequently restored the expression of the Nrf2-target genes, NQO1 and GSTµ1 (21). DIM also reversed Nrf2 gene silencing in transgenic mouse prostate cancer tissues, inducing Nrf2 expression, and subsequently, NQO1 expression (Figure 4). This was accompanied by the suppression of proliferation and the induction of apoptosis in prostate cancer tissues (21). Similar observations have been reported with I3C (22).
Anti-estrogenic activities
Endogenous estrogens are steroid hormones synthesized by humans and other mammals.
Inhibition of estrogen synthesis
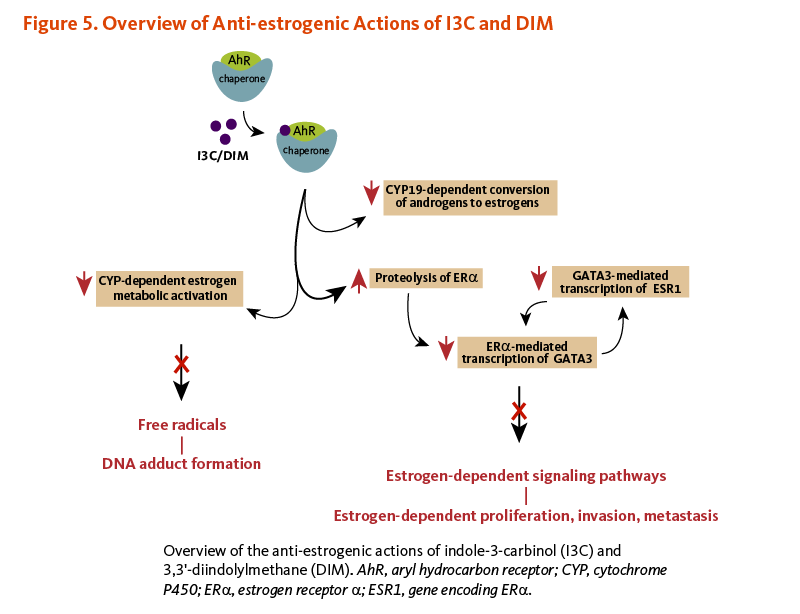
In breast tissue, CYP19 (aromatase) catalyzes the final steps in the conversion of androgens (testosterone or androstenedione) to estrogens (17β-estradiol or estrone, respectively). Both I3C and DIM have been found to downregulate the expression of CYP19 in non-tumorigenic and tumorigenic estrogen-responsive (ER+) breast cells, whereas CYP19 expression was increased in I3C/DIM-treated tumorigenic estrogen-independent (ER-) breast cells (23).
Inhibition of estrogen metabolic activation
Prolonged exposure to estrogens is thought to play a role in cancer development through CYP-mediated generation of estrogen reactive metabolites that can damage DNA (24, 25).
Phase I metabolizing enzymes, CYP1A1, CYP1A2, and CYP1B1, have been involved in the oxidative metabolism of estrogens. 17β-estradiol can be converted to 2-hydroxyestradiol (2HE2) and 4-hydroxyestradiol (4HE2) by CYP1A1/2 and CYP1B1, respectively. 2HE2 and 4HE2 are further metabolized to 2- and 4-metoxymetabolites by the phase II enzyme, catechol-O-methyltransferase (COMT) (25). 2HE2 is a noncarcinogenic agent with weaker estrogenic potential than 17β-estradiol, while 4-HE2 can be converted to free radicals that can form DNA adducts and promote carcinogenesis (26, 27). In different breast cancer cell lines, I3C and DIM have been shown to upregulate the expression of CYP1A1, CYP1A2, and CYP1B1 at the transcript (mRNA) level but not at the protein level (28).
Additionally, the endogenous estrogens 17β-estradiol and estrone can be irreversibly metabolized to 16a-hydroxyestrone (16HE1) (29). In contrast to 2-hydroxyestrone (2HE1), 16HE1 is highly estrogenic and has been found to stimulate the proliferation of several estrogen-sensitive cancer cell lines (30, 31). It has been hypothesized that shifting the metabolism of 17β-estradiol toward 2HE1, and away from 16HE1, could decrease the risk of estrogen-sensitive cancers, such as breast cancer (32). In controlled clinical trials, oral supplementation with I3C or DIM has consistently increased urinary 2HE1 concentrations or urinary 2HE1:16HE1 ratios in women (33-39). However, large case-control and prospective cohort studies have failed to find significant associations between urinary 2HE1:16HE1 ratios and risk of breast and endometrial cancer (40-43).
Inhibition of estrogen signaling
Endogenous estrogens, including 17β-estradiol, exert their estrogenic effects by binding to specific nuclear receptors called estrogen receptors (ERs). Within the nucleus, estrogen-activated ERs can bind to specific DNA sequences, known as estrogen response elements (EREs), in the promoters of estrogen-responsive genes. ERE-bound estrogen-ER complexes act as transcription factors by recruiting coactivator proteins and chromatin remodeling factors to promoters, thereby triggering the transcription of target genes (44). There are two major ER subtypes, ERα and ERβ, coded by two separate genes ESR1 and ESR2, respectively. ERα is the main driver of the proliferative effect of estrogens, while the expression of ERβ has been inversely associated with mammary gland tumorigenesis (45). Elevated ERα levels promote cellular proliferation in the breast and uterus, possibly increasing the risk of developing estrogen-sensitive cancers (46).
Inhibition of estrogen-dependent cell proliferation
In estrogen-sensitive human breast cancer cells challenged with 17β-estradiol, I3C has been found to inhibit the transcription of estrogen-responsive genes without binding to either ERβ or ERα (47, 48). In fact, the binding of I3C to AhR was shown to trigger the proteasome-dependent degradation of ERα (49). I3C-induced loss of ERα resulted in the downregulation of ERα-responsive gene products like the transcription factor GATA3. Since GATA3 regulates the transcription of the ERα coding gene ESR1, I3C prevented the synthesis of new ERα transcripts and proteins, eventually abolishing the ERα signaling pathway. The disruption of the GATA3/ERα cross-regulatory loop by I3C ultimately halted ERα-dependent cell proliferation (49). Acid condensation products of I3C that bind and activate AhR may also inhibit the transcription of estrogen-responsive genes by competing for co-activators or increasing ERα degradation (14, 50). I3C treatment also affected the expression of other ERα-responsive genes, including those coding for insulin-like growth factor-1 receptor (IGFR1) and insulin receptor substrate-1 (IRS-1), involved in cell proliferation and deregulated in breast cancer (Figure 5) (51).
Modulation of cell-signaling pathways
I3C and condensation derivatives have been found to affect multiple signaling pathways that are often deregulated in cancer cells. Below are some examples illustrating how I3C, DIM, or ICZ may influence cell proliferation, apoptosis, migration, invasion, angiogenesis, and immunity by targeting specific signaling pathways (23).
Induction of cell cycle arrest and apoptosis
Once a cell divides, it passes through a sequence of stages — collectively known as the cell cycle — before it divides again. Following DNA damage, the cell cycle can be transiently arrested at damage checkpoints, which allows for DNA repair or activation of pathways leading to cell death (apoptosis) if the damage is irreparable (52). Defective cell cycle regulation may result in the propagation of mutations that contribute to the development of cancer. In addition, unlike normal cells, cancerous cells lose their ability to respond to death signals that initiate apoptosis.
I3C-induced downregulation of the phosphatidylinositol 3-kinase (PI3K)/serine-threonine kinase (Akt) cell survival signaling pathway in mice with nasopharyngeal carcinoma resulted in inhibition of cell proliferation and induction of apoptosis (53). Inactivation of the Wnt/β-catenin signaling pathway in DIM-treated colon cancer cells decreased the expression of downstream targets, c-myc and cyclin D1, that promote cell proliferation and survival (54). In prostate cancer cells, I3C opposed the anti-apoptotic effect of epidermal growth factor (EGF) by limiting EGF receptor autophosphorylation (activation) and reducing EGF-induced activation of the PI3K/Akt signaling pathway and expression of the pro-survival target molecules, Bcl-x(L) and BAD (55). In another study, DIM caused apoptosis of prostate cancer cells by stimulating p38 mitogen-activated protein kinase (p38 MAPK)-induced upregulation of tumor suppressor p75NTR (56). The anti-proliferative effect of DIM in cancer cells has also been linked to the inhibition of histone deacetylase (HDAC) activity. Specifically, DIM was found to reverse HDAC-mediated epigenetic silencing of genes coding for key regulators of the cell cycle (57, 58). A recent genome-wide analysis of DNA methylation also showed that DIM could reverse aberrant promoter methylation in prostate cancer cells, at least partly through downregulating the expression of DNA methyltransferases (DNMTs) (59).
Inhibition of cell migration and invasion
The epithelial-to-mesenchymal transition (EMT) describes a process of epithelial cell transformation whereby cells lose their polarity and adhesion properties while gaining migratory and invasive properties through the expression of mesenchymal genes. Inhibition of EMT by I3C and ICZ in breast cancer cells has been associated with upregulation of an epithelial marker, E-cadherin, and downregulation of vimentin, focal adhesion kinase (FAK), and matrix metalloproteins (MMPs) — proteins and enzymes known to promote migration (60). DIM also inhibited migration and invasion of liver cancer cells in vitro and in vivo through inactivating the FAK signaling pathway (61). Moreover, DIM has been shown to reverse methylation-associated dysregulation of genes involved in cell adhesion, chemotaxis, and inflammation that contributes to cancer progression (59). DIM was able to inhibit lung metastasis in mice with liver (61) or mammary tumors (62).
Inhibition of angiogenesis
To fuel their rapid growth, invasive tumors must also develop new capillaries from preexisting blood vessels by a process known as angiogenesis. I3C inhibited lipopolysaccharide (LPS)-induced macrophage activation and secretion of proangiogenic molecules, such as nitric oxide (NO), vascular endothelial growth factor (VEGF), interleukin-6 (IL-6), and MMP-9, and prevented the formation of capillary-like structures from co-cultured human umbilical endothelial cells (63). Similarly, I3C inhibited capillary-like tube formation from phorbol myristate acetate (PMA)-stimulated endothelial cells (64). DIM also blocked PMA-induced angiogenic activities in human umbilical endothelial cells (65).
Regulation of inflammation and cell-mediated immunity
Uncontrolled inflammation has been associated with several chronic diseases, including cancer. In the mouse ear edema model, 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced upregulation of pro-inflammatory mediators, such as cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS), has been found to be mitigated by DIM treatment (66). Nuclear factor-kappa B (NF-κB) is a major transcription factor regulating the expression of many pro-inflammatory genes like those coding for COX-2 and iNOS. Specifically, DIM inhibited TPA-induced activation of kinases (inhibitor of kappa B kinase [IκK] and extracellular signal-regulated kinase [ERK]) that control the transcriptional activity of NF-κB (66). In addition, recent animal studies showed that I3C and/or DIM could modulate cell-mediated immune response in experimental autoimmune encephalomyelitis (67), staphylococcal enterotoxin-induced lung inflammation (68), and delayed-type hypersensitivity (69). Specifically, I3C and/or DIM differentially regulated T-cell subpopulations via the activation or suppression of microRNA-dependent pathways controlling cell cycle progression and apoptosis.
Transplacental cancer prevention
The inclusion of I3C in the maternal diet was found to protect the offspring from lymphoma and lung tumors induced by dibenzo[a]pyrene, a polycyclic aromatic hydrocarbon (70, 71). Polycyclic aromatic hydrocarbons are chemical pollutants formed during incomplete combustion of organic substances, such as coal, oil, wood, and tobacco (72).
However, the physiological relevance of cell culture and animal studies to human health is unclear since little or no I3C is available to tissues after oral administration (see Metabolism and Bioavailability) (6).
Disease Prevention
Cancer
Some observational studies provide some support for the hypothesis that higher intakes of cruciferous vegetables are associated with lower risk for some types of cancer (see the article on Cruciferous Vegetables) (1). Cruciferous vegetables are relatively good sources of nutrients that may have protective effects against cancer, including vitamin C, folate, selenium, carotenoids, and fiber. In addition, glucosinolates can be hydrolyzed to a variety of potentially protective isothiocyanates, in addition to indole-3-carbinol (see the article on Isothiocyanates). Consequently, evidence for an inverse association between cruciferous vegetable intake and cancer risk provides relatively little information about the specific effects of indole-3-carbinol on cancer risk.
At present, the effects of I3C or DIM supplementation on cancer risk in humans are not known.
Disease Treatment
Human papilloma virus infection-related diseases
Cervical intraepithelial neoplasia
Infection with certain strains of human papilloma virus (HPV) is an important risk factor for cervical cancer (73). Transgenic mice that express cancer-promoting HPV genes develop cervical cancer with chronic 17β-estradiol administration. In this model, feeding I3C markedly reduced the number of mice that developed cervical cancer (74). A small placebo-controlled trial in women examined the effect of oral I3C supplementation on the progression of precancerous cervical lesions classified as cervical intraepithelial neoplasia (CIN) 2 or CIN 3 (75). After 12 weeks, four out of the eight women who took 200 mg/day had complete regression of CIN, and four out of the nine who took 400 mg/day had complete regression; none of the 10 women who took a placebo had complete regression. HPV was present in 7 out of the 10 women in the placebo group, seven out of eight women in the 200 mg I3C group, and eight out of nine women in the 400 mg I3C group (75). However compared to placebo, oral supplementation with DIM (2 mg/kg/day) for 12 weeks in 64 women with CIN 2 or CIN 3 lesions failed to improve clinical parameters during a one-year follow-up period (76). In another six-month randomized, double-blind controlled trial, DIM supplementation (150 mg/day) failed to promote HPV clearance and prevent CIN progression in 551 women with low-grade cell abnormalities in cervical smears (77).
Although oral supplementation of I3C or DIM appears relatively safe and well tolerated, results obtained with I3C are only preliminary, and interventions with DIM failed to show preventative or therapeutic efficacy in women with precancerous lesions of the cervix. The intravaginal administration of DIM in the form of suppositories may prove to be a more effective approach to reverse CIN in women (78).
Vulvar intraepithelial neoplasia
HPV infection has also been associated with vulvar intraepithelial neoplasia (VIN), a precancerous condition that may progress to vulval cancer (79). A small randomized trial in 12 women with VIN found that supplementation with 200 mg/day or 400 mg/day of I3C for six months improved overall symptoms, as well as lesion size and appearance (80). Additional trials are necessary to determine whether I3C might be an effective treatment for VIN.
Recurrent respiratory papillomatosis
Recurrent respiratory papillomatosis (RRP) is a rare disease of children and adults, characterized by generally benign growths (papillomas) in the respiratory tract, which are caused by HPV infection (81). These papillomas occur most commonly on or around the vocal cords in the larynx (voice box), but they may also affect the trachea, bronchi, and lungs. The most common treatment for RRP is surgical removal of the papillomas. Since papillomas often recur, adjunct treatments may be used to help prevent or reduce recurrences (82). In immune-compromised mice transplanted with HPV-infected laryngeal tissue, only 25% of the mice fed I3C developed laryngeal papillomas compared to 100% of the control mice (83). In a small observational study of RRP patients, increased ratios of urinary 2HE1:16HE1 resulting from increased cruciferous vegetable consumption were associated with less severe RRP (84). In an uncontrolled pilot study, the effect of daily I3C supplementation (400 mg/day for adults and 10 mg/kg daily for children) on papilloma recurrence has been examined in RRP patients (85). Over a five-year follow-up period, 11 of the original 49 patients experienced no recurrence, 10 experienced a reduction in the rate of recurrence, 12 experienced no improvement, and 12 were lost to follow-up (86). I3C given for 6 months to 3 years to five children with an aggressive form of the disease halted the growth of the papillomas in three children after two years of treatment (87). In some patients, I3C may be an effective adjunct treatment to reduce the growth or recurrence of respiratory papillomas.
Systemic lupus erythematosus
Systemic lupus erythematosus (SLE) is an autoimmune disorder characterized by chronic inflammation that may result in damage to the joints, skin, kidneys, heart, lungs, blood vessels, or brain (88). Estrogen is thought to play a role in the pathology of SLE because the disorder is much more common in women than men, and its onset is most common during the reproductive years when endogenous estrogen levels are highest (89). The potential for I3C supplementation to shift endogenous estrogen metabolism toward the less estrogenic metabolite 2HE1, and away from the highly estrogenic metabolite 16HE1 (see Anti-estrogenic activities), led to interest in its use in SLE (35). In an animal model of SLE, I3C feeding decreased the severity of kidney disease and prolonged survival (90). A small uncontrolled trial of I3C supplementation (375 mg/day) in female SLE patients found that I3C supplementation increased urinary 2HE1:16HE1 ratios, but the trial found no significant change in SLE symptoms after three months (90). Controlled clinical trials are needed to determine whether I3C supplementation could have beneficial effects in SLE patients.
Sources
Food sources
Glucobrassicin, the glucosinolate precursor of I3C, is found in a number of cruciferous vegetables, including broccoli, Brussels sprouts, cabbage, cauliflower, collard greens, kale, kohlrabi, mustard greens, radish, rutabaga, and turnip (91, 92). Although glucosinolates are present in relatively high concentrations in cruciferous vegetables, glucobrassicin makes up only about 8%-12% of the total glucosinolates (93). Total glucosinolate contents of selected cruciferous vegetables are presented in Table 1. However, the amount of total glucosinolates and the amount of indole-3-carbinol formed from glucobrassicin in food is variable and depends, in part, on the processing and preparation of foods (for more detailed information, see the article on Cruciferous Vegetables).
| Food (raw) | Serving | Total Glucosinolates (mg) |
|---|---|---|
| Brussels sprouts | ½ cup |
104
|
| Garden cress | ½ cup |
98
|
| Mustard greens | ½ cup, chopped |
79
|
| Kale | 1 cup, chopped |
67
|
| Turnip | ½ cup, cubes |
60
|
| Cabbage, savoy | ½ cup, chopped |
35
|
| Watercress | 1 cup, chopped |
32
|
| Kohlrabi | ½ cup, chopped |
31
|
| Cabbage, red | ½ cup, chopped |
29
|
| Broccoli | ½ cup, chopped |
27
|
| Horseradish | 1 tablespoon (15 g) |
24
|
| Cauliflower | ½ cup, chopped |
22
|
| Bok choi (pak choi) | ½ cup, chopped |
19
|
Supplements
Indole-3-Carbinol (I3C)
I3C is available without a prescription as a dietary supplement, alone or in combination products. Dosage ranges between 200 mg/day and 800 mg/day (95). I3C supplementation increased urinary 2HE1 concentrations in adults at doses of 300 to 400 mg/day (39). I3C doses of 200 mg/day or 400 mg/day improved the regression of cervical intraepithelial neoplasia (CIN) in a preliminary clinical trial (75). I3C in doses up to 400 mg/day has been used to treat recurrent respiratory papillomatosis (see Disease Treatment) (85, 86).
3,3'-Diindolylmethane (DIM)
DIM is available without a prescription as a dietary supplement, alone or in combination products. In a small clinical trial, DIM supplementation at a dose of 108 mg/day for 30 days increased urinary 2HE1 excretion in postmenopausal women with a history of breast cancer (34).
Safety
Adverse effects
Slight increases in the serum concentrations of the liver enzyme, alanine aminotransferase (ALT) were observed in two women who took unspecified doses of I3C supplements for four weeks (39). One person reported a skin rash while taking 375 mg/day of I3C (35). High doses of I3C (800 mg/day) have been associated with symptoms of disequilibrium and tremor, which resolved when the dose was decreased (85). In a phase I study in women at high risk for breast cancer, 5 out of 20 participants had gastrointestinal symptoms with single doses ≥600 mg, although others had no adverse effects with single doses up to 1,200 mg (6). No adverse effects were reported with daily consumption of 400 mg of I3C for four weeks (6).
In some animal models, I3C supplementation was found to enhance carcinogen-induced cancer development when given chronically after the carcinogen (96-99). When administered before or at the same time as the carcinogen, oral I3C inhibited tumorigenesis in animal models of cancers of the mammary gland (100, 101), uterus (102), stomach (103), colon (104, 105), lung (106), and liver (107, 108). Although the long-term effects of I3C supplementation on cancer risk in humans are not known, the contradictory results of animal studies have led several experts to caution against the widespread use of I3C and DIM supplements in humans until their potential risks and benefits are better understood (99, 109, 110).
Pregnancy and lactation
The safety of I3C or DIM supplements during pregnancy or lactation has not been established (95).
Drug interactions
No drug interactions with I3C or DIM supplementation in humans have been reported. However, preliminary evidence that I3C and DIM can increase the activity of CYP1A2 (111, 112) suggests the potential for I3C or DIM supplementation to decrease serum concentrations of medications metabolized by CYP1A2 (113). Both I3C and DIM modestly increase the activity of CYP3A4 in rats when administered chronically (114). This observation raises the potential for adverse drug interactions in humans since CYP3A4 is involved in the metabolism of approximately 60% of therapeutic drugs.
The acidic environment of the stomach allows I3C molecules to condense and generate a number of biologically active I3C oligomers (Figure 2). Drugs that block the production of stomach acids, like antacids, Histamine2 (H2) receptor antagonists, and proton-pump inhibitors, would likely prevent the generation of DIM and ICZ. However, it is not known whether these drugs limit the biological activities attributed to I3C and its derivatives (95).
Authors and Reviewers
Originally written in 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in December 2008 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in January 2017 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in July 2017 by:
David E. Williams, Ph.D.
Principal Investigator, and Helen P. Rumbel Professor for Cancer Prevention
Linus Pauling Institute
Professor, Department of Environmental and Molecular Toxicology
Oregon State University
Copyright 2005-2024 Linus Pauling Institute
References
1. Traka M. Chapter nine-health benefits of glucosinolates. Advances in Botanical Research. 2016;80:247-279.
2. Ishida M, Hara M, Fukino N, Kakizaki T, Morimitsu Y. Glucosinolate metabolism, functionality and breeding for the improvement of Brassicaceae vegetables. Breed Sci. 2014;64(1):48-59. (PubMed)
3. Holst B, Williamson G. A critical review of the bioavailability of glucosinolates and related compounds. Nat Prod Rep. 2004;21(3):425-447. (PubMed)
4. Barba FJ, Nikmaram N, Roohinejad S, Khelfa A, Zhu Z, Koubaa M. Bioavailability of glucosinolates and their breakdown products: impact of processing. Front Nutr. 2016;3:24. (PubMed)
5. Wang SQ, Cheng LS, Liu Y, Wang JY, Jiang W. Indole-3-carbinol (I3C) and its major derivatives: their pharmacokinetics and important roles in hepatic protection. Curr Drug Metab. 2016;17(4):401-409. (PubMed)
6. Reed GA, Arneson DW, Putnam WC, et al. Single-dose and multiple-dose administration of indole-3-carbinol to women: pharmacokinetics based on 3,3'-diindolylmethane. Cancer Epidemiol Biomarkers Prev. 2006;15(12):2477-2481. (PubMed)
7. Anderton MJ, Manson MM, Verschoyle R, et al. Physiological modeling of formulated and crystalline 3,3'-diindolylmethane pharmacokinetics following oral administration in mice. Drug Metab Dispos. 2004;32(6):632-638. (PubMed)
8. Luo Y, Wang TT, Teng Z, Chen P, Sun J, Wang Q. Encapsulation of indole-3-carbinol and 3,3'-diindolylmethane in zein/carboxymethyl chitosan nanoparticles with controlled release property and improved stability. Food Chem. 2013;139(1-4):224-230. (PubMed)
9. Song JM, Kirtane AR, Upadhyaya P, et al. Intranasal delivery of liposomal indole-3-carbinol improves its pulmonary bioavailability. Int J Pharm. 2014;477(1-2):96-101. (PubMed)
10. Lampe JW, Peterson S. Brassica, biotransformation and cancer risk: genetic polymorphisms alter the preventive effects of cruciferous vegetables. J Nutr. 2002;132(10):2991-2994. (PubMed)
11. Bjeldanes LF, Kim JY, Grose KR, Bartholomew JC, Bradfield CA. Aromatic hydrocarbon responsiveness-receptor agonists generated from indole-3-carbinol in vitro and in vivo: comparisons with 2,3,7,8-tetrachlorodibenzo-p-dioxin. Proc Natl Acad Sci U S A. 1991;88(21):9543-9547. (PubMed)
12. Bonnesen C, Eggleston IM, Hayes JD. Dietary indoles and isothiocyanates that are generated from cruciferous vegetables can both stimulate apoptosis and confer protection against DNA damage in human colon cell lines. Cancer Res. 2001;61(16):6120-6130. (PubMed)
13. Hubbard TD, Murray IA, Perdew GH. Indole and tryptophan metabolism: endogenous and dietary routes to Ah receptor activation. Drug Metab Dispos. 2015;43(10):1522-1535. (PubMed)
14. Safe S. Molecular biology of the Ah receptor and its role in carcinogenesis. Toxicol Lett. 2001;120(1-3):1-7. (PubMed)
15. Li Y, Li X, Sarkar FH. Gene expression profiles of I3C- and DIM-treated PC3 human prostate cancer cells determined by cDNA microarray analysis. J Nutr. 2003;133(4):1011-1019. (PubMed)
16. Wang TT, Schoene NW, Milner JA, Kim YS. Broccoli-derived phytochemicals indole-3-carbinol and 3,3'-diindolylmethane exerts concentration-dependent pleiotropic effects on prostate cancer cells: comparison with other cancer preventive phytochemicals. Mol Carcinog. 2012;51(3):244-256. (PubMed)
17. Gross-Steinmeyer K, Stapleton PL, Liu F, et al. Phytochemical-induced changes in gene expression of carcinogen-metabolizing enzymes in cultured human primary hepatocytes. Xenobiotica. 2004;34(7):619-632. (PubMed)
18. Baird WM, Hooven LA, Mahadevan B. Carcinogenic polycyclic aromatic hydrocarbon-DNA adducts and mechanism of action. Environ Mol Mutagen. 2005;45(2-3):106-114. (PubMed)
19. Watson GW, Beaver LM, Williams DE, Dashwood RH, Ho E. Phytochemicals from cruciferous vegetables, epigenetics, and prostate cancer prevention. AAPS J. 2013;15(4):951-961. (PubMed)
20. Saw CL, Cintron M, Wu TY, et al. Pharmacodynamics of dietary phytochemical indoles I3C and DIM: Induction of Nrf2-mediated phase II drug metabolizing and antioxidant genes and synergism with isothiocyanates. Biopharm Drug Dispos. 2011;32(5):289-300. (PubMed)
21. Wu TY, Khor TO, Su ZY, et al. Epigenetic modifications of Nrf2 by 3,3'-diindolylmethane in vitro in TRAMP C1 cell line and in vivo TRAMP prostate tumors. AAPS J. 2013;15(3):864-874. (PubMed)
22. Wu TY, Saw CL, Khor TO, Pung D, Boyanapalli SS, Kong AN. In vivo pharmacodynamics of indole-3-carbinol in the inhibition of prostate cancer in transgenic adenocarcinoma of mouse prostate (TRAMP) mice: involvement of Nrf2 and cell cycle/apoptosis signaling pathways. Mol Carcinog. 2012;51(10):761-770. (PubMed)
23. Licznerska BE, Szaefer H, Murias M, Bartoszek A, Baer-Dubowska W. Modulation of CYP19 expression by cabbage juices and their active components: indole-3-carbinol and 3,3'-diindolylmethene in human breast epithelial cell lines. Eur J Nutr. 2013;52(5):1483-1492. (PubMed)
24. Belous AR, Hachey DL, Dawling S, Roodi N, Parl FF. Cytochrome P450 1B1-mediated estrogen metabolism results in estrogen-deoxyribonucleoside adduct formation. Cancer Res. 2007;67(2):812-817. (PubMed)
25. Jefcoate CR, Liehr JG, Santen RJ, et al. Tissue-specific synthesis and oxidative metabolism of estrogens. J Natl Cancer Inst Monogr. 2000(27):95-112. (PubMed)
26. Kwon YJ, Baek HS, Ye DJ, Shin S, Kim D, Chun YJ. CYP1B1 enhances cell proliferation and metastasis through induction of EMT and activation of Wnt/beta-catenin signaling via Sp1 upregulation. PLoS One. 2016;11(3):e0151598. (PubMed)
27. Park SA, Lee MH, Na HK, Surh YJ. 4-Hydroxyestradiol induces mammary epithelial cell transformation through Nrf2-mediated heme oxygenase-1 overexpression. Oncotarget. 2016;8(1):164-178. (PubMed)
28. Szaefer H, Licznerska B, Krajka-Kuzniak V, Bartoszek A, Baer-Dubowska W. Modulation of CYP1A1, CYP1A2 and CYP1B1 expression by cabbage juices and indoles in human breast cell lines. Nutr Cancer. 2012;64(6):879-888. (PubMed)
29. Ziegler RG, Fuhrman BJ, Moore SC, Matthews CE. Epidemiologic studies of estrogen metabolism and breast cancer. Steroids. 2015;99(Pt A):67-75. (PubMed)
30. Telang NT, Suto A, Wong GY, Osborne MP, Bradlow HL. Induction by estrogen metabolite 16 alpha-hydroxyestrone of genotoxic damage and aberrant proliferation in mouse mammary epithelial cells. J Natl Cancer Inst. 1992;84(8):634-638. (PubMed)
31. Yuan F, Chen DZ, Liu K, Sepkovic DW, Bradlow HL, Auborn K. Anti-estrogenic activities of indole-3-carbinol in cervical cells: implication for prevention of cervical cancer. Anticancer Res. 1999;19(3A):1673-1680. (PubMed)
32. Bradlow HL, Telang NT, Sepkovic DW, Osborne MP. 2-Hydroxyestrone: the 'good' estrogen. J Endocrinol. 1996;150 Suppl:S259-265. (PubMed)
33. Bradlow HL, Michnovicz JJ, Halper M, Miller DG, Wong GY, Osborne MP. Long-term responses of women to indole-3-carbinol or a high fiber diet. Cancer Epidemiol Biomarkers Prev. 1994;3(7):591-595. (PubMed)
34. Dalessandri KM, Firestone GL, Fitch MD, Bradlow HL, Bjeldanes LF. Pilot study: effect of 3,3'-diindolylmethane supplements on urinary hormone metabolites in postmenopausal women with a history of early-stage breast cancer. Nutr Cancer. 2004;50(2):161-167. (PubMed)
35. McAlindon TE, Gulin J, Chen T, Klug T, Lahita R, Nuite M. Indole-3-carbinol in women with SLE: effect on estrogen metabolism and disease activity. Lupus. 2001;10(11):779-783. (PubMed)
36. Michnovicz JJ. Increased estrogen 2-hydroxylation in obese women using oral indole-3-carbinol. Int J Obes Relat Metab Disord. 1998;22(3):227-229. (PubMed)
37. Michnovicz JJ, Adlercreutz H, Bradlow HL. Changes in levels of urinary estrogen metabolites after oral indole-3-carbinol treatment in humans. J Natl Cancer Inst. 1997;89(10):718-723. (PubMed)
38. Reed GA, Peterson KS, Smith HJ, et al. A phase I study of indole-3-carbinol in women: tolerability and effects. Cancer Epidemiol Biomarkers Prev. 2005;14(8):1953-1960. (PubMed)
39. Wong GY, Bradlow L, Sepkovic D, Mehl S, Mailman J, Osborne MP. Dose-ranging study of indole-3-carbinol for breast cancer prevention. J Cell Biochem Suppl. 1997;28-29:111-116. (PubMed)
40. Arslan AA, Shore RE, Afanasyeva Y, Koenig KL, Toniolo P, Zeleniuch-Jacquotte A. Circulating estrogen metabolites and risk for breast cancer in premenopausal women. Cancer Epidemiol Biomarkers Prev. 2009;18(8):2273-2279. (PubMed)
41. Eliassen AH, Missmer SA, Tworoger SS, Hankinson SE. Circulating 2-hydroxy- and 16alpha-hydroxy estrone levels and risk of breast cancer among postmenopausal women. Cancer Epidemiol Biomarkers Prev. 2008;17(8):2029-2035. (PubMed)
42. Modugno F, Kip KE, Cochrane B, et al. Obesity, hormone therapy, estrogen metabolism and risk of postmenopausal breast cancer. Int J Cancer. 2006;118(5):1292-1301. (PubMed)
43. Zeleniuch-Jacquotte A, Shore RE, Afanasyeva Y, et al. Postmenopausal circulating levels of 2- and 16alpha-hydroxyestrone and risk of endometrial cancer. Br J Cancer. 2011;105(9):1458-1464. (PubMed)
44. Jordan VC, Gapstur S, Morrow M. Selective estrogen receptor modulation and reduction in risk of breast cancer, osteoporosis, and coronary heart disease. J Natl Cancer Inst. 2001;93(19):1449-1457. (PubMed)
45. Omoto Y, Iwase H. Clinical significance of estrogen receptor beta in breast and prostate cancer from biological aspects. Cancer Sci. 2015;106(4):337-343. (PubMed)
46. Liehr JG. Is estradiol a genotoxic mutagenic carcinogen? Endocr Rev. 2000;21(1):40-54. (PubMed)
47. Ashok BT, Chen Y, Liu X, Bradlow HL, Mittelman A, Tiwari RK. Abrogation of estrogen-mediated cellular and biochemical effects by indole-3-carbinol. Nutr Cancer. 2001;41(1-2):180-187. (PubMed)
48. Meng Q, Yuan F, Goldberg ID, Rosen EM, Auborn K, Fan S. Indole-3-carbinol is a negative regulator of estrogen receptor-alpha signaling in human tumor cells. J Nutr. 2000;130(12):2927-2931. (PubMed)
49. Marconett CN, Sundar SN, Poindexter KM, Stueve TR, Bjeldanes LF, Firestone GL. Indole-3-carbinol triggers aryl hydrocarbon receptor-dependent estrogen receptor (ER)alpha protein degradation in breast cancer cells disrupting an ERalpha-GATA3 transcriptional cross-regulatory loop. Mol Biol Cell. 2010;21(7):1166-1177. (PubMed)
50. Chen I, McDougal A, Wang F, Safe S. Aryl hydrocarbon receptor-mediated antiestrogenic and antitumorigenic activity of diindolylmethane. Carcinogenesis. 1998;19(9):1631-1639. (PubMed)
51. Marconett CN, Singhal AK, Sundar SN, Firestone GL. Indole-3-carbinol disrupts estrogen receptor-alpha dependent expression of insulin-like growth factor-1 receptor and insulin receptor substrate-1 and proliferation of human breast cancer cells. Mol Cell Endocrinol. 2012;363(1-2):74-84. (PubMed)
52. Stewart ZA, Westfall MD, Pietenpol JA. Cell-cycle dysregulation and anticancer therapy. Trends Pharmacol Sci. 2003;24(3):139-145. (PubMed)
53. Mao CG, Tao ZZ, Chen Z, Chen C, Chen SM, Wan LJ. Indole-3-carbinol inhibits nasopharyngeal carcinoma cell growth in vivo and in vitro through inhibition of the PI3K/Akt pathway. Exp Ther Med. 2014;8(1):207-212. (PubMed)
54. Leem SH, Li XJ, Park MH, Park BH, Kim SM. Genome-wide transcriptome analysis reveals inactivation of Wnt/beta-catenin by 3,3'-diindolylmethane inhibiting proliferation of colon cancer cells. Int J Oncol. 2015;47(3):918-926. (PubMed)
55. Chinni SR, Li Y, Upadhyay S, Koppolu PK, Sarkar FH. Indole-3-carbinol (I3C) induced cell growth inhibition, G1 cell cycle arrest and apoptosis in prostate cancer cells. Oncogene. 2001;20(23):2927-2936. (PubMed)
56. Khwaja FS, Wynne S, Posey I, Djakiew D. 3,3'-diindolylmethane induction of p75NTR-dependent cell death via the p38 mitogen-activated protein kinase pathway in prostate cancer cells. Cancer Prev Res. 2009;2(6):566-571. (PubMed)
57. Beaver LM, Yu TW, Sokolowski EI, Williams DE, Dashwood RH, Ho E. 3,3'-Diindolylmethane, but not indole-3-carbinol, inhibits histone deacetylase activity in prostate cancer cells. Toxicol Appl Pharmacol. 2012;263(3):345-351. (PubMed)
58. Li Y, Li X, Guo B. Chemopreventive agent 3,3'-diindolylmethane selectively induces proteasomal degradation of class I histone deacetylases. Cancer Res. 2010;70(2):646-654. (PubMed)
59. Wong CP, Hsu A, Buchanan A, et al. Effects of sulforaphane and 3,3'-diindolylmethane on genome-wide promoter methylation in normal prostate epithelial cells and prostate cancer cells. PLoS One. 2014;9(1):e86787. (PubMed)
60. Ho JN, Jun W, Choue R, Lee J. I3C and ICZ inhibit migration by suppressing the EMT process and FAK expression in breast cancer cells. Mol Med Rep. 2013;7(2):384-388. (PubMed)
61. Li WX, Chen LP, Sun MY, Li JT, Liu HZ, Zhu W. 3'3-Diindolylmethane inhibits migration, invasion and metastasis of hepatocellular carcinoma by suppressing FAK signaling. Oncotarget. 2015;6(27):23776-23792. (PubMed)
62. Kim EJ, Shin M, Park H, et al. Oral administration of 3,3'-diindolylmethane inhibits lung metastasis of 4T1 murine mammary carcinoma cells in BALB/c mice. J Nutr. 2009;139(12):2373-2379. (PubMed)
63. Wang ML, Shih CK, Chang HP, Chen YH. Antiangiogenic activity of indole-3-carbinol in endothelial cells stimulated with activated macrophages. Food Chem. 2012;134(2):811-820. (PubMed)
64. Wu HT, Lin SH, Chen YH. Inhibition of cell proliferation and in vitro markers of angiogenesis by indole-3-carbinol, a major indole metabolite present in cruciferous vegetables. J Agric Food Chem. 2005;53(13):5164-5169. (PubMed)
65. Kunimasa K, Kobayashi T, Kaji K, Ohta T. Antiangiogenic effects of indole-3-carbinol and 3,3'-diindolylmethane are associated with their differential regulation of ERK1/2 and Akt in tube-forming HUVEC. J Nutr. 2010;140(1):1-6. (PubMed)
66. Kim EJ, Park H, Kim J, Park JH. 3,3'-diindolylmethane suppresses 12-O-tetradecanoylphorbol-13-acetate-induced inflammation and tumor promotion in mouse skin via the downregulation of inflammatory mediators. Mol Carcinog. 2010;49(7):672-683. (PubMed)
67. Rouse M, Rao R, Nagarkatti M, Nagarkatti PS. 3,3'-diindolylmethane ameliorates experimental autoimmune encephalomyelitis by promoting cell cycle arrest and apoptosis in activated T cells through microRNA signaling pathways. J Pharmacol Exp Ther. 2014;350(2):341-352. (PubMed)
68. Elliott DM, Nagarkatti M, Nagarkatti PS. 3,3-Diindolylmethane ameliorates Staphylococcal enterotoxin B-induced acute lung injury through alterations in the expression of microRNA that target apoptosis and cell-cycle arrest in activated T cells. J Pharmacol Exp Ther. 2016;357(1):177-187. (PubMed)
69. Singh NP, Singh UP, Rouse M, et al. Dietary indoles suppress delayed-type hypersensitivity by inducing a switch from proinflammatory Th17 Cells to anti-inflammatory regulatory T cells through regulation of microRNA. J Immunol. 2016;196(3):1108-1122. (PubMed)
70. Shorey LE, Madeen EP, Atwell LL, et al. Differential modulation of dibenzo[def,p]chrysene transplacental carcinogenesis: maternal diets rich in indole-3-carbinol versus sulforaphane. Toxicol Appl Pharmacol. 2013;270(1):60-69. (PubMed)
71. Yu Z, Mahadevan B, Lohr CV, et al. Indole-3-carbinol in the maternal diet provides chemoprotection for the fetus against transplacental carcinogenesis by the polycyclic aromatic hydrocarbon dibenzo[a,l]pyrene. Carcinogenesis. 2006;27(10):2116-2123. (PubMed)
72. ATSDR. Toxicological profile for polycyclic aromatic hydrocarbons (PAHs). Agency for Toxic Substances and Disease Registry, U.S. Department of Health and Human Services. Atlanta, GA; August 1995.
73. Rambout L, Hopkins L, Hutton B, Fergusson D. Prophylactic vaccination against human papillomavirus infection and disease in women: a systematic review of randomized controlled trials. CMAJ. 2007;177(5):469-479. (PubMed)
74. Jin L, Qi M, Chen DZ, et al. Indole-3-carbinol prevents cervical cancer in human papilloma virus type 16 (HPV16) transgenic mice. Cancer Res. 1999;59(16):3991-3997. (PubMed)
75. Bell MC, Crowley-Nowick P, Bradlow HL, et al. Placebo-controlled trial of indole-3-carbinol in the treatment of CIN. Gynecol Oncol. 2000;78(2):123-129. (PubMed)
76. Del Priore G, Gudipudi DK, Montemarano N, Restivo AM, Malanowska-Stega J, Arslan AA. Oral diindolylmethane (DIM): pilot evaluation of a nonsurgical treatment for cervical dysplasia. Gynecol Oncol. 2010;116(3):464-467. (PubMed)
77. Castanon A, Tristram A, Mesher D, et al. Effect of diindolylmethane supplementation on low-grade cervical cytological abnormalities: double-blind, randomised, controlled trial. Br J Cancer. 2012;106(1):45-52. (PubMed)
78. Ashrafian L, Sukhikh G, Kiselev V, et al. Double-blind randomized placebo-controlled multicenter clinical trial (phase IIa) on diindolylmethane's efficacy and safety in the treatment of CIN: implications for cervical cancer prevention. EPMA J. 2015;6:25. (PubMed)
79. Pepas L, Kaushik S, Nordin A, Bryant A, Lawrie TA. Medical interventions for high-grade vulval intraepithelial neoplasia. Cochrane Database Syst Rev. 2015(8):Cd007924. (PubMed)
80. Naik R, Nixon S, Lopes A, Godfrey K, Hatem MH, Monaghan JM. A randomized phase II trial of indole-3-carbinol in the treatment of vulvar intraepithelial neoplasia. Int J Gynecol Cancer. 2006;16(2):786-790. (PubMed)
81. Recurrent Respiratory Papillomatosis Foundation. What is recurrent respiratory papillomatosis? Recurrent Respiratory Papillomatosis Foundation [Web page]. Available at: https://rrpf.org/what-is-rrp/. Accessed 1/4/22.
82. Auborn KJ. Therapy for recurrent respiratory papillomatosis. Antivir Ther. 2002;7(1):1-9. (PubMed)
83. Newfield L, Goldsmith A, Bradlow HL, Auborn K. Estrogen metabolism and human papillomavirus-induced tumors of the larynx: chemo-prophylaxis with indole-3-carbinol. Anticancer Res. 1993;13(2):337-341. (PubMed)
84. Auborn K, Abramson A, Bradlow HL, Sepkovic D, Mullooly V. Estrogen metabolism and laryngeal papillomatosis: a pilot study on dietary prevention. Anticancer Res. 1998;18(6B):4569-4573. (PubMed)
85. Rosen CA, Woodson GE, Thompson JW, Hengesteg AP, Bradlow HL. Preliminary results of the use of indole-3-carbinol for recurrent respiratory papillomatosis. Otolaryngol Head Neck Surg. 1998;118(6):810-815. (PubMed)
86. Rosen CA, Bryson PC. Indole-3-carbinol for recurrent respiratory papillomatosis: long-term results. J Voice. 2004;18(2):248-253. (PubMed)
87. Boltezar IH, Bahar MS, Zargi M, Gale N, Maticic M, Poljak M. Adjuvant therapy for laryngeal papillomatosis. Acta Dermatovenerol Alp Pannonica Adriat. 2011;20(3):175-180. (PubMed)
88. Handout on health: systemic lupus erythematosus. National Institute of Arthritis and Musculoskeletal and Skin Diseases [Web page]. June 2016. Available at: https://www.niams.nih.gov/health_info/lupus/. Accessed 1/21/17.
89. McMurray RW, May W. Sex hormones and systemic lupus erythematosus: review and meta-analysis. Arthritis Rheum. 2003;48(8):2100-2110. (PubMed)
90. Auborn KJ, Qi M, Yan XJ, et al. Lifespan is prolonged in autoimmune-prone (NZB/NZW) F1 mice fed a diet supplemented with indole-3-carbinol. J Nutr. 2003;133(11):3610-3613. (PubMed)
91. Carlson DG, Kwolek WF, Williams PH. Glucosinolates in crucifer vegetables: broccoli, Brussels sprouts, cauliflower, collards, kale, mustard greens, and kohlrabi. J Amer Soc Hort Sci. 1987;112(1):173-178.
92. Fenwick GR, Heaney RK, Mullin WJ. Glucosinolates and their breakdown products in food and food plants. Crit Rev Food Sci Nutr. 1983;18(2):123-201. (PubMed)
93. Kushad MM, Brown AF, Kurilich AC, et al. Variation of glucosinolates in vegetable crops of Brassica oleracea. J Agric Food Chem. 1999;47(4):1541-1548. (PubMed)
94. McNaughton SA, Marks GC. Development of a food composition database for the estimation of dietary intakes of glucosinolates, the biologically active constituents of cruciferous vegetables. Br J Nutr. 2003;90(3):687-697. (PubMed)
95. Hendler SS, Rorvik DM. PDR for Nutritional Supplements. 2nd ed: Thomson Reuters; 2008.
96. Kim DJ, Han BS, Ahn B, et al. Enhancement by indole-3-carbinol of liver and thyroid gland neoplastic development in a rat medium-term multiorgan carcinogenesis model. Carcinogenesis. 1997;18(2):377-381. (PubMed)
97. Yoshida M, Katashima S, Ando J, et al. Dietary indole-3-carbinol promotes endometrial adenocarcinoma development in rats initiated with N-ethyl-N'-nitro-N-nitrosoguanidine, with induction of cytochrome P450s in the liver and consequent modulation of estrogen metabolism. Carcinogenesis. 2004;25(11):2257-2264. (PubMed)
98. Pence BC, Buddingh F, Yang SP. Multiple dietary factors in the enhancement of dimethylhydrazine carcinogenesis: main effect of indole-3-carbinol. J Natl Cancer Inst. 1986;77(1):269-276. (PubMed)
99. Stoner G, Casto B, Ralston S, Roebuck B, Pereira C, Bailey G. Development of a multi-organ rat model for evaluating chemopreventive agents: efficacy of indole-3-carbinol. Carcinogenesis. 2002;23(2):265-272. (PubMed)
100. Grubbs CJ, Steele VE, Casebolt T, et al. Chemoprevention of chemically-induced mammary carcinogenesis by indole-3-carbinol. Anticancer Res. 1995;15(3):709-716. (PubMed)
101. Bradlow HL, Michnovicz J, Telang NT, Osborne MP. Effects of dietary indole-3-carbinol on estradiol metabolism and spontaneous mammary tumors in mice. Carcinogenesis. 1991;12(9):1571-1574. (PubMed)
102. Kojima T, Tanaka T, Mori H. Chemoprevention of spontaneous endometrial cancer in female Donryu rats by dietary indole-3-carbinol. Cancer Res. 1994;54(6):1446-1449. (PubMed)
103. Wattenberg LW, Loub WD. Inhibition of polycyclic aromatic hydrocarbon-induced neoplasia by naturally occurring indoles. Cancer Res. 1978;38(5):1410-1413. (PubMed)
104. Wargovich MJ, Chen CD, Jimenez A, et al. Aberrant crypts as a biomarker for colon cancer: evaluation of potential chemopreventive agents in the rat. Cancer Epidemiol Biomarkers Prev. 1996;5(5):355-360. (PubMed)
105. Guo D, Schut HA, Davis CD, Snyderwine EG, Bailey GS, Dashwood RH. Protection by chlorophyllin and indole-3-carbinol against 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP)-induced DNA adducts and colonic aberrant crypts in the F344 rat. Carcinogenesis. 1995;16(12):2931-2937. (PubMed)
106. Morse MA, LaGreca SD, Amin SG, Chung FL. Effects of indole-3-carbinol on lung tumorigenesis and DNA methylation induced by 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) and on the metabolism and disposition of NNK in A/J mice. Cancer Res. 1990;50(9):2613-2617. (PubMed)
107. Dashwood RH, Arbogast DN, Fong AT, Hendricks JD, Bailey GS. Mechanisms of anti-carcinogenesis by indole-3-carbinol: detailed in vivo DNA binding dose-response studies after dietary administration with aflatoxin B1. Carcinogenesis. 1988;9(3):427-432. (PubMed)
108. Oganesian A, Hendricks JD, Williams DE. Long term dietary indole-3-carbinol inhibits diethylnitrosamine-initiated hepatocarcinogenesis in the infant mouse model. Cancer Lett. 1997;118(1):87-94. (PubMed)
109. Dashwood RH. Indole-3-carbinol: anticarcinogen or tumor promoter in brassica vegetables? Chem Biol Interact. 1998;110(1-2):1-5. (PubMed)
110. Lee BM, Park KK. Beneficial and adverse effects of chemopreventive agents. Mutat Res. 2003;523-524:265-278. (PubMed)
111. He YH, Friesen MD, Ruch RJ, Schut HA. Indole-3-carbinol as a chemopreventive agent in 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP) carcinogenesis: inhibition of PhIP-DNA adduct formation, acceleration of PhIP metabolism, and induction of cytochrome P450 in female F344 rats. Food Chem Toxicol. 2000;38(1):15-23. (PubMed)
112. Lake BG, Tredger JM, Renwick AB, Barton PT, Price RJ. 3,3'-Diindolylmethane induces CYP1A2 in cultured precision-cut human liver slices. Xenobiotica. 1998;28(8):803-811. (PubMed)
113. Natural Medicines. Professional monograph: Indole-3-carbinol/Interactions with drugs; 2016.
114. Leibelt DA, Hedstrom OR, Fischer KA, Pereira CB, Williams DE. Evaluation of chronic dietary exposure to indole-3-carbinol and absorption-enhanced 3,3'-diindolylmethane in Sprague-Dawley rats. Toxicol Sci. 2003;74(1):10-21. (PubMed)
![Figure 2. Condensation Derivatives of Indole-3-Carbinol: 3,3'-diindolylmethane (DIM), 5,6,11,12,17,18-hexahydrocyclononal [1,2-b:4,5-b':7,8-b"]triindole (CT), and 5,11-dihydroindolo-[3,2-b]carbazole (ICZ)](/sites/lpi.oregonstate.edu/files/indole-3-carbinol-figure-2-800px.png)