Chlorophyll and Metallo-Chlorophyll Derivatives
Contents
Summary
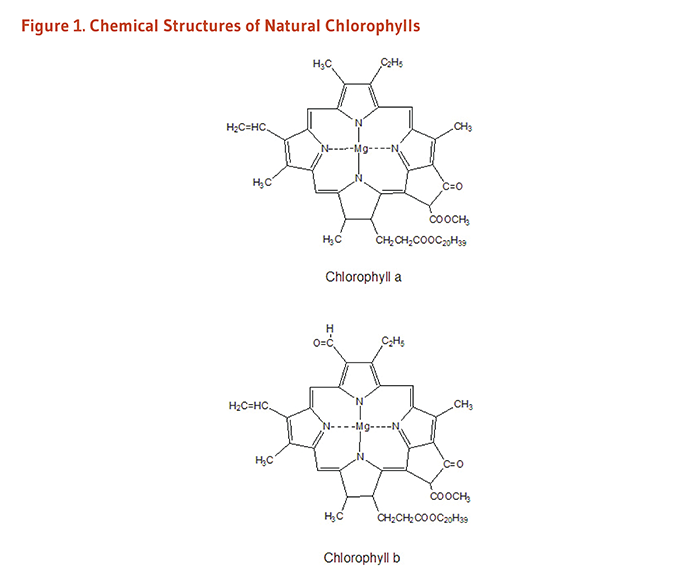
- Chlorophyll a and chlorophyll b are natural, fat-soluble chlorophylls found in plants. (More information)
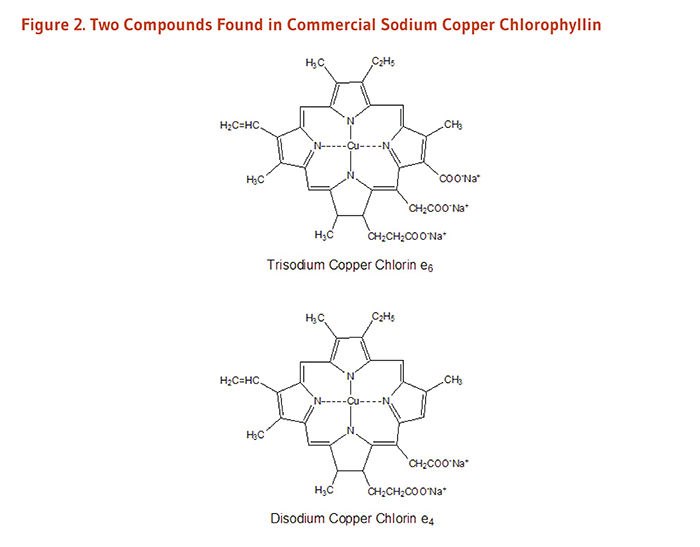
- Sodium copper chlorophyllin (SCC) is a semi-synthetic mixture of water-soluble sodium copper salts derived from chlorophyll. (More information)
- SCC has been used orally as an internal deodorant and topically in the treatment of slow-healing wounds for more than 50 years without any serious side effects. (More information)
- Chlorophylls and SCC form tight molecular complexes with some chemicals known or suspected to cause cancer, and in doing so, may block carcinogenic effects. Carefully controlled studies have not been undertaken to determine whether a similar mechanism might limit uptake of required nutrients. (More information)
- Supplementation with SCC before meals substantially decreased a urinary biomarker of aflatoxin-induced DNA damage in a Chinese population at high risk of liver cancer due to unavoidable, dietary aflatoxin exposure from moldy grains and legumes. (More information)
- Scientists are hopeful that SCC supplementation will be helpful in decreasing the risk of liver cancer in high-risk populations with unavoidable, dietary aflatoxin exposure. However, it is not yet known whether SCC or natural chlorophylls will be useful in the prevention of cancers in people who are not exposed to significant levels of dietary aflatoxin. (More information)
Introduction
Chlorophyll is the pigment that gives plants and algae their green color. Plants use chlorophyll to trap light needed for photosynthesis (1). The basic structure of chlorophyll is a porphyrin ring similar to that of heme in hemoglobin, although the central atom in chlorophyll is magnesium instead of iron. The long hydrocarbon (phytol) tail attached to the porphyrin ring makes chlorophyll fat-soluble and insoluble in water. Chlorophyll a and chlorophyll b represent about 99% of the chlorophyll species found in edible plants (Figure 1; 2), while some algae and microalgae contain minor quantities of chlorophyll c pigments (e.g., Laminaria ochroleuca, Undaria pinnatifida) (3). Chlorophyll a and b only have a small difference in one of the side chains but an intact phytol tail, while the common characteristic of chlorophyll c isoforms is the absence of a phytol tail. These structural differences cause each type of chlorophyll to absorb light at slightly different wavelengths.
Metallo-chlorophyll derivatives, including chlorophyllins, can be chemically synthesized or produced in industrial food processing; these compounds contain zinc, iron, or copper in place of the central magnesium atom (2). The most studied chlorophyllin, sodium copper chlorophyllin (SCC), is a semi-synthetic mixture of sodium copper salts derived from chlorophyll (4, 5). SCC is often simply called ‘chlorophyllin’ in the older scientific literature, with newer publications specifying whether iron, zinc, copper, or magnesium chlorophyllin were studied. During its synthesis, the magnesium atom at the center of the ring is replaced with copper (or other metals), and the phytol tail is lost. Unlike natural chlorophyll, chlorophyllins (regardless of the metal used) are water-soluble. Although the content of different SCC mixtures may vary, two compounds commonly found in commercial SCC are trisodium copper chlorin e6 and disodium copper chlorin e4 (Figure 2).
Metabolism and Bioavailability
Little is known about the bioavailability and metabolism of chlorophyll in humans, although it is known that chlorophyll undergoes extensive metabolism once consumed. Animal model studies show only about 1%-3% of chlorophyll is absorbed, while the rest is excreted in the feces, primarily as pheopytin and pyropheophytin metabolites, indicating that significant transformation and microbial metabolism occur in the gastrointestinal tract (reviewed in 2). A recent study in eight healthy adults found pheophytin and pheophorbide derivatives in the blood of most subjects following consumption of 1.2 kg boiled spinach, a concentrated source of chlorophyll (6).
Sodium copper chlorophyllin was originally thought to be poorly absorbed because of its lack of apparent toxicity. However, a placebo-controlled clinical trial found that significant amounts of copper chlorin e4 in the serum of people taking chlorophyllin tablets (300 mg/day) (7), indicating that it is indeed absorbed. In vitro studies have found chlorin e4 to have a higher stability than chlorin e6 (2).
More research, however, is needed to understand the bioavailability and metabolism of natural chlorophylls and chlorin compounds in synthetic chlorophyllin.
Biological Activities
Complex formation with other molecules
Chlorophyll and sodium copper chlorophyllin are able to form tight molecular complexes with certain chemicals known or suspected to cause cancer, including polyaromatic hydrocarbons found in tobacco smoke (8), some heterocyclic amines found in cooked meat (9), and aflatoxin-B1 (10). The binding of chlorophyll or SCC to these potential carcinogens may interfere with gastrointestinal absorption of potential carcinogens, reducing the amount that reaches susceptible tissues (11). This has been demonstrated in humans: a cross-over study in three volunteers that used accelerator mass spectrometry to study the pharmacokinetics of an ultra-low dose of aflatoxin-B1 found a 150-mg dose of either SCC or chlorophyll could decrease absorption of aflatoxin-B1 (12).
Antioxidant effects
SCC can neutralize several physically relevant oxidants in vitro (13-16), and limited data from animal studies suggest that SCC supplementation may decrease oxidative damage induced by chemical carcinogens and radiation (17, 18). While chlorophyll and its derivatives have demonstrated antioxidant activity in in vitro assays (15, 19), the relevance of these findings to humans is not clear.
Modification of the metabolism and detoxification of carcinogens
To initiate the development of cancer, some chemicals (procarcinogens) must first be metabolized to active carcinogens that are capable of damaging DNA or other critical molecules in susceptible tissues. Since enzymes in the cytochrome P450 family are required for the activation of some procarcinogens, inhibition of cytochrome P450 enzymes may decrease the risk of some types of chemically induced cancers. In vitro studies indicate that SCC may decrease the activity of cytochrome P450 enzymes (8, 20, 21). Phase II biotransformation enzymes promote the elimination of potentially harmful toxins and carcinogens from the body. Limited data from animal studies indicate that SCC may increase the activity of the phase II enzyme quinone reductase (22).
Therapeutic effects
One in vitro study showed that human colon cancer cells undergo cell cycle arrest after treatment with SCC (23). The mechanism involved inhibition of ribonucleotide reductase activity. Ribonucleotide reductase plays a pivotal role in DNA synthesis and repair and is a target of currently used cancer therapeutic agents, such as hydroxyurea (23). While this may provide a potential avenue for SCC in the clinical setting, sensitizing cancer cells to DNA damaging agents, in vivo studies are needed.
Metal absorption
The porphyrin structure of chlorophyll is analogous to the heme structure found in blood and muscle tissue. Because heme-bound iron has higher bioavailability than nonheme iron (the most common form of iron in plant-based sources, e.g., legumes, spinach), iron uptake from iron chlorophyll is of interest. Toxicity studies in rats suggest iron chlorophyllin is generally safe for mammalian consumption (24). In vitro studies demonstrate that iron chlorophyllin is as good as heme in delivering iron to intestinal cells, and significantly better than the most common supplemental form of iron (i.e., ferrous sulfate) when incorporated into most food matrices (25). However, work in this area is nascent and has not yet been validated in humans. It is also not known if metallo-chlorophyll derivatives of copper or zinc increase absorption of these essential divalent metals.
Disease Prevention
Aflatoxin-associated liver cancer
Aflatoxin-B1 (AFB1), a liver carcinogen produced by certain species of fungus, is found in moldy grains and legumes, including corn, peanuts, and soybeans (4, 11). In hot, humid regions of Africa and Asia with improper grain storage facilities, high levels of dietary AFB1 are associated with increased risk of hepatocellular carcinoma. Moreover, the combination of hepatitis B infection and high dietary AFB1 exposure increases the risk of hepatocellular carcinoma still further. In the liver, AFB1 is metabolized to a carcinogen capable of binding DNA and causing mutations. In animal models of AFB1-induced liver cancer, administration of SCC at the same time as dietary AFB1 exposure significantly reduces AFB1-induced DNA damage in the livers of rainbow trout and rats (26-28) and dose-dependently inhibits the development of liver cancer in trout (29). Likewise, natural chlorophyll has also been found to inhibit AFB1-induced liver cancer in the rat (28). Collectively, this evidence supports a role for SCC and/or chlorophyll itself in limiting cancer initiation. In contrast, data suggest a limited role for SCC in influencing cancer progression. For example, one rat study found that SCC did not protect against aflatoxin-induced liver damage when given after tumor initiation (30).
Because of the long time period between AFB1 exposure and the development of cancer in humans, an intervention trial might require as long as 20 years to determine whether SCC supplementation can reduce the incidence of hepatocellular carcinoma in people exposed to high levels of dietary AFB1. However, a biomarker of AFB1-induced DNA damage (AFB1-N7-guanine) can be measured in the urine, and high urinary levels of AFB1-N7-guanine have been associated with significantly increased risk of developing hepatocellular carcinoma (31). In order to determine whether chlorophyllin could decrease AFB1-induced DNA damage in humans, a randomized, placebo-controlled intervention trial was conducted in 180 adults residing in a region in China where the risk of hepatocellular carcinoma is very high due to unavoidable, dietary AFB1 exposure and a high prevalence of chronic hepatitis B infection (32). Participants took either 100 mg of SCC or a placebo before meals three times daily. After 16 weeks of treatment, urinary levels of AFB1-N7-guanine were 55% lower in those taking SCC than in those taking the placebo, suggesting that SCC supplementation before meals can substantially decrease AFB1-induced DNA damage. Although a reduction in hepatocellular carcinoma has not yet been demonstrated in humans taking SCC, scientists are hopeful that supplementation will provide some protection to high-risk populations with unavoidable, dietary AFB1 exposure (11).
It is not known whether SCC will be useful in the prevention of cancers in people who are not exposed to significant levels of dietary AFB1, as is the case for most people living in the US. Many questions remain to be answered regarding the exact mechanisms of cancer prevention by SCC, the implications for the prevention of other types of cancer, and the potential for natural chlorophylls in the diet to provide cancer protection.
Therapeutic Uses of Chlorophyllin
Internal deodorant
Observations in the 1940s and 1950s that topical SCC had deodorizing effects on foul-smelling wounds led clinicians to administer SCC orally to patients with colostomies and ileostomies in order to control fecal odor (33). While early case reports indicated that SCC doses of 100 to 200 mg/day were effective in reducing fecal odor in ostomy patients (34, 35), a placebo-controlled trial found that 75 mg of oral SCC three times daily was no more effective than placebo in decreasing fecal odor assessed by colostomy patients (36). Several case reports have been published indicating that oral SCC (100-300 mg/day) decreased subjective assessments of urinary and fecal odor in incontinent patients (33, 37).
Trimethylaminuria is a hereditary disorder characterized by the excretion of trimethylamine, a compound with a “fishy” or foul odor. One study in a small number of Japanese patients with trimethylaminuria found that oral SCC (60 mg three times daily) for three weeks significantly decreased urinary trimethylamine concentrations (38).
Wound healing
Research in the 1940s indicated that chlorophyllin slowed the growth of certain anaerobic bacteria in the test tube and accelerated the healing of experimental wounds in animals. These findings led to the use of topical SCC solutions and ointments in the treatment of persistent open wounds in humans (39). During the late 1940s and 1950s, a series of largely uncontrolled studies in patients with slow-healing wounds, such as vascular ulcers and pressure (decubitus) ulcers, reported that the application of topical SCC promoted healing more effectively than other commonly used treatments (40, 41). In the late 1950s, SCC was added to papain and urea-containing ointments used for the chemical debridement of wounds in order to reduce local inflammation, promote healing, and control odor (33). SCC-containing papain/urea ointments are still available in the US by prescription (42). Several studies have reported that such ointments are effective in wound healing (43). A spray formulation of the papain/urea/SCC therapy is also available (44).
Skin conditions
A few small studies have investigated SCC as a topical treatment for various skin conditions. In a pilot study of 10 adults (ages 18-30 years) who had mild-to-moderate acne vulgaris and enlarged facial pores, twice daily application of a 0.1% liposomal SCC gel for three weeks improved a number of clinical parameters of the Global Acne Assessment Scale (i.e., facial oiliness, facial blotchiness, presence and size of facial pores, and number of acne lesions) compared to baseline (45). Additionally, a pilot study in 10 women (ages 40 years or older) with noticeable photodamage and solar lentigines found that twice daily topical application of a gel containing 0.66% SCC complex salts for eight weeks improved various clinical measures, including tactile and visual roughness of facial skin, skin radiance, fine lines, pore size, and overall photodamage (46). A few case reports have also observed some improvement in facial redness and rosacea with application of topical SCC (47).
While the reports from these studies are interesting, placebo-controlled clinical trials are needed to determine whether SCC may have utility in treating various skin conditions.
Sources
Chlorophylls
Chlorophylls are the most abundant pigments in plants, with chlorophyll a being two to four times as prevalent as chlorophyll b (6, 48). Dark-green leafy vegetables like spinach are rich sources of natural chlorophylls. The chlorophyll content of selected vegetables are presented in Table 1 (49).
Food and supplements
Chlorophyll
Green algae like chlorella are often marketed as supplemental sources of chlorophyll. Because natural chlorophyll is not as stable as SCC and is much more expensive, most over-the-counter chlorophyll supplements actually contain sodium copper chlorophyllin.
Sodium copper chlorophyllin
Oral preparations of sodium copper chlorophyllin (also called chlorophyllin copper complex) are available as a dietary supplement and as an over-the-counter drug (Derifil) used to reduce odor from colostomies and ileostomies, or to reduce fecal odor due to incontinence (50). Oral doses of 100 to 300 mg/day in three divided doses have been used to control fecal and urinary odor (see Therapeutic Uses of Chlorophyllin).
In the US, SCC is found in minor quantities in some types of green table olives (51). It is also approved for use as a green color additive in a limited number of foods like chewing gum (52), as well as in drugs and cosmetics, as detailed in the Code of Federal Regulations 21 (53).
Zinc chlorophyll derivatives
In US supermarkets, canned green beans thermally processed in a zinc chloride solution to produce zinc chlorophyll derivatives within the green beans themselves are sold under the trademarked name "veri-green" (54). Because zinc chlorophyll derivatives are more robust to heat and acid treatment, they better retain a bright green color as compared to native magnesium-bound chlorophyll (48).
Safety
Natural chlorophylls are not known to be toxic, and no toxic effects have been attributed to chlorophyllin despite more than 50 years of clinical use in humans (11, 33, 39). When taken orally, supplemental chlorophyll or sodium copper chlorophyllin may cause green discoloration of urine or feces, or yellow or black discoloration of the tongue (55). There have also been occasional reports of diarrhea related to oral SCC use. When applied topically to wounds, SCC has been reported to cause mild burning or itching in some cases (56). Oral chlorophyllin may result in false positive results on guaiac card tests for occult blood (57). Since the safety of chlorophyll or chlorophyllin supplements has not been tested in pregnant or lactating women, they should be avoided during pregnancy and lactation.
Authors and Reviewers
Originally written in 2004 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in December 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in June 2009 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in September 2021 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in March 2022 by:
Rachel E. Kopec, Ph.D.
Assistant Professor of Human Nutrition
The Ohio State University
Copyright 2004-2024 Linus Pauling Institute
References
1. Matthews CK, van Holde KE. Biochemistry. 2nd ed. Menlo Park: The Benjamin/Cummings Publishing Company; 1996.
2. Zhong S, Bird A, Kopec RE. The metabolism and potential bioactivity of chlorophyll and metallo-chlorophyll derivatives in the gastrointestinal tract. Mol Nutr Food Res. 2021;65(7):e2000761. (PubMed)
3. Chen K, Rios JJ, Perez-Galvez A, Roca M. Comprehensive chlorophyll composition in the main edible seaweeds. Food Chem. 2017;228:625-633. (PubMed)
4. Sudakin DL. Dietary aflatoxin exposure and chemoprevention of cancer: a clinical review. J Toxicol Clin Toxicol. 2003;41(2):195-204. (PubMed)
5. Dashwood RH. The importance of using pure chemicals in (anti) mutagenicity studies: chlorophyllin as a case in point. Mutat Res. 1997;381(2):283-286. (PubMed)
6. Chao PY, Huang MY, Huang WD, Lin KH, Chen SY, Yang CM. Study of chlorophyll-related compounds from dietary spinach in human blood. Not Bot Horti Agrobo. 2018;46(2):309-316.
7. Egner PA, Stansbury KH, Snyder EP, Rogers ME, Hintz PA, Kensler TW. Identification and characterization of chlorin e(4) ethyl ester in sera of individuals participating in the chlorophyllin chemoprevention trial. Chem Res Toxicol. 2000;13(9):900-906. (PubMed)
8. Tachino N, Guo D, Dashwood WM, Yamane S, Larsen R, Dashwood R. Mechanisms of the in vitro antimutagenic action of chlorophyllin against benzo[a]pyrene: studies of enzyme inhibition, molecular complex formation and degradation of the ultimate carcinogen. Mutat Res. 1994;308(2):191-203. (PubMed)
9. Dashwood R, Yamane S, Larsen R. Study of the forces of stabilizing complexes between chlorophylls and heterocyclic amine mutagens. Environ Mol Mutagen. 1996;27(3):211-218. (PubMed)
10. Breinholt V, Schimerlik M, Dashwood R, Bailey G. Mechanisms of chlorophyllin anticarcinogenesis against aflatoxin B1: complex formation with the carcinogen. Chem Res Toxicol. 1995;8(4):506-514. (PubMed)
11. Egner PA, Munoz A, Kensler TW. Chemoprevention with chlorophyllin in individuals exposed to dietary aflatoxin. Mutat Res. 2003;523-524:209-216. (PubMed)
12. Jubert C, Mata J, Bench G, et al. Effects of chlorophyll and chlorophyllin on low-dose aflatoxin B(1) pharmacokinetics in human volunteers. Cancer Prev Res (Phila). 2009;2(12):1015-1022. (PubMed)
13. Kumar SS, Devasagayam TP, Bhushan B, Verma NC. Scavenging of reactive oxygen species by chlorophyllin: an ESR study. Free Radic Res. 2001;35(5):563-574. (PubMed)
14. Kamat JP, Boloor KK, Devasagayam TP. Chlorophyllin as an effective antioxidant against membrane damage in vitro and ex vivo. Biochim Biophys Acta. 2000;1487(2-3):113-127. (PubMed)
15. Vankova K, Markova I, Jasprova J, et al. Chlorophyll-mediated changes in the redox status of pancreatic cancer cells are associated with its anticancer effects. Oxid Med Cell Longev. 2018;2018:4069167. (PubMed)
16. Domijan AM, Gajski G, Novak Jovanovic I, Geric M, Garaj-Vrhovac V. In vitro genotoxicity of mycotoxins ochratoxin A and fumonisin B(1) could be prevented by sodium copper chlorophyllin--implication to their genotoxic mechanism. Food Chem. 2015;170:455-462. (PubMed)
17. Park KK, Park JH, Jung YJ, Chung WY. Inhibitory effects of chlorophyllin, hemin and tetrakis(4-benzoic acid)porphyrin on oxidative DNA damage and mouse skin inflammation induced by 12-O-tetradecanoylphorbol-13-acetate as a possible anti-tumor promoting mechanism. Mutat Res. 2003;542(1-2):89-97. (PubMed)
18. Kumar SS, Shankar B, Sainis KB. Effect of chlorophyllin against oxidative stress in splenic lymphocytes in vitro and in vivo. Biochim Biophys Acta. 2004;1672(2):100-111. (PubMed)
19. Perez-Galvez A, Viera I, Roca M. Carotenoids and chlorophylls as antioxidants. Antioxidants (Basel). 2020;9(6):505. (PubMed)
20. Yun CH, Jeong HG, Jhoun JW, Guengerich FP. Non-specific inhibition of cytochrome P450 activities by chlorophyllin in human and rat liver microsomes. Carcinogenesis. 1995;16(6):1437-1440. (PubMed)
21. John K, Divi RL, Keshava C, et al. CYP1A1 and CYP1B1 gene expression and DNA adduct formation in normal human mammary epithelial cells exposed to benzo[a]pyrene in the absence or presence of chlorophyllin. Cancer Lett. 2010;292(2):254-260. (PubMed)
22. Dingley KH, Ubick EA, Chiarappa-Zucca ML, et al. Effect of dietary constituents with chemopreventive potential on adduct formation of a low dose of the heterocyclic amines PhIP and IQ and phase II hepatic enzymes. Nutr Cancer. 2003;46(2):212-221. (PubMed)
23. Chimploy K, Diaz GD, Li Q, et al. E2F4 and ribonucleotide reductase mediate S-phase arrest in colon cancer cells treated with chlorophyllin. Int J Cancer. 2009;125(9):2086-2094. (PubMed)
24. Toyoda T, Cho YM, Mizuta Y, Akagi J, Nishikawa A, Ogawa K. A 13-week subchronic toxicity study of sodium iron chlorophyllin in F344 rats. J Toxicol Sci. 2014;39(1):109-119. (PubMed)
25. Miret S, Tascioglu S, van der Burg M, Frenken L, Klaffke W. In vitro bioavailability of iron from the heme analogue sodium iron chlorophyllin. J Agric Food Chem. 2010;58(2):1327-1332. (PubMed)
26. Dashwood RH, Breinholt V, Bailey GS. Chemopreventive properties of chlorophyllin: inhibition of aflatoxin B1 (AFB1)-DNA binding in vivo and anti-mutagenic activity against AFB1 and two heterocyclic amines in the Salmonella mutagenicity assay. Carcinogenesis. 1991;12(5):939-942. (PubMed)
27. Kensler TW, Groopman JD, Roebuck BD. Use of aflatoxin adducts as intermediate endpoints to assess the efficacy of chemopreventive interventions in animals and man. Mutat Res. 1998;402(1-2):165-172. (PubMed)
28. Simonich MT, Egner PA, Roebuck BD, et al. Natural chlorophyll inhibits aflatoxin B1-induced multi-organ carcinogenesis in the rat. Carcinogenesis. 2007;28(6):1294-1302. (PubMed)
29. Breinholt V, Hendricks J, Pereira C, Arbogast D, Bailey G. Dietary chlorophyllin is a potent inhibitor of aflatoxin B1 hepatocarcinogenesis in rainbow trout. Cancer Res. 1995;55(1):57-62. (PubMed)
30. Orner GA, Roebuck BD, Dashwood RH, Bailey GS. Post-initiation chlorophyllin exposure does not modulate aflatoxin-induced foci in the liver and colon of rats. J Carcinog. 2006;5:6. (PubMed)
31. Qian GS, Ross RK, Yu MC, et al. A follow-up study of urinary markers of aflatoxin exposure and liver cancer risk in Shanghai, People's Republic of China. Cancer Epidemiol Biomarkers Prev. 1994;3(1):3-10. (PubMed)
32. Egner PA, Wang JB, Zhu YR, et al. Chlorophyllin intervention reduces aflatoxin-DNA adducts in individuals at high risk for liver cancer. Proc Natl Acad Sci U S A. 2001;98(25):14601-14606. (PubMed)
33. Chernomorsky SA, Segelman AB. Biological activities of chlorophyll derivatives. N J Med. 1988;85(8):669-673. (PubMed)
34. Siegel LH. The control of ileostomy and colostomy odors. Gastroenterology. 1960;38:634-636. (PubMed)
35. Weingarten M, Payson B. Deodorization of colostomies with chlorophyll. Rev Gastroenterol. 1951;18(8):602-604. (PubMed)
36. Christiansen SB, Byel SR, Stromsted H, Stenderup JK, Eickhoff JH. [Can chlorophyll reduce fecal odor in colostomy patients?]. Ugeskr Laeger. 1989;151(27):1753-1754. (PubMed)
37. Young RW, Beregi JS, Jr. Use of chlorophyllin in the care of geriatric patients. J Am Geriatr Soc. 1980;28(1):46-47. (PubMed)
38. Yamazaki H, Fujieda M, Togashi M, et al. Effects of the dietary supplements, activated charcoal and copper chlorophyllin, on urinary excretion of trimethylamine in Japanese trimethylaminuria patients. Life Sci. 2004;74(22):2739-2747. (PubMed)
39. Kephart JC. Chlorophyll derivatives - their chemistry, commercial preparation and uses. Econ Bot. 1955;9:3-38.
40. Bowers WF. Chlorophyll in wound healing and suppurative disease. Am J Surg. 1947;73:37-50. (PubMed)
41. Carpenter EB. Clinical experiences with chlorophyll preparations. Am J Surg. 1949;77(2):167-171. (PubMed)
42. Physicians' Desk Reference. 58th ed. Stamford: Thomson Health Care, Inc.; 2003.
43. Smith RG. Enzymatic debriding agents: an evaluation of the medical literature. Ostomy Wound Manage. 2008;54(8):16-34. (PubMed)
44. Weir D, Farley KL. Relative delivery efficiency and convenience of spray and ointment formulations of papain/urea/chlorophyllin enzymatic wound therapies. J Wound Ostomy Continence Nurs. 2006;33(5):482-490. (PubMed)
45. Stephens TJ, McCook JP, Herndon JH, Jr. Pilot study of topical copper chlorophyllin complex in subjects with facial acne and large pores. J Drugs Dermatol. 2015;14(6):589-592. (PubMed)
46. Sigler ML, Stephens TJ. Assessment of the safety and efficacy of topical copper chlorophyllin in women with photodamaged facial skin. J Drugs Dermatol. 2015;14(4):401-404. (PubMed)
47. Vasily DB. Topical treatment with liposomal sodium copper chlorophyllin complex in subjects with facial redness and erythematotelangiectatic rosacea: case studies. J Drugs Dermatol. 2015;14(10):1157-1159. (PubMed)
48. Hayes M, Ferruzzi MG. Update on the bioavailability and chemopreventative mechanisms of dietary chlorophyll derivatives. Nutr Res. 2020;81:19-37. (PubMed)
49. Bohn T, Walczyk S, Leisibach S, Hurrell RF. Chlorophyll-bound magnesium in commonly consumed vegetables and fruits: relevance to magnesium nutrition. J Food Sci. 2004;69(9):S347-S350.
50. Food and Drug Administration. Code of Federal Regulations: Miscellaneous Internal Drug Products for Over the Counter Use [Web page]. April 1, 2002. Available at: http://www.fda.gov/cder/otcmonographs/Internal_Deodorant/internal_deodorant(357I).html. Accessed 6/9/04.
51. Aparicio-Ruiz R, Riedl KM, Schwartz SJ. Identification and quantification of metallo-chlorophyll complexes in bright green table olives by high-performance liquid chromatography-mass spectrometry quadrupole/time-of-flight. J Agric Food Chem. 2011;59(20):11100-11108. (PubMed)
52. Viera I, Perez-Galvez A, Roca M. Green natural colorants. Molecules. 2019;24(1):154. (PubMed)
53. US Food and Drug Administration. CFR - Code of Federal Regulations Title 21. Available at: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=73.3110. Accessed 9/29/21.
54. Von Elbe JH, Huang AS, Attoe EL, Nank WK. Pigment composition and color of conventional and Veri-Green canned beans. J Agric Food Chem. 1986;34(1):52-54.
55. Hendler SS, Rorvik DR, eds. PDR for Nutritional Supplements. 2nd ed. Montvale: Physicians' Desk Reference, Inc.; 2008.
56. Smith LW. The present status of topical chlorophyll therapy. N Y State J Med. 1955;55(14):2041-2050. (PubMed)
57. Gogel HK, Tandberg D, Strickland RG. Substances that interfere with guaiac card tests: implications for gastric aspirate testing. Am J Emerg Med. 1989;7(5):474-480. (PubMed)