Summary
- Coffee is a complex mixture of chemicals that provides significant amounts of chlorogenic acid and caffeine. (More information)
- Unfiltered coffee is a significant source of diterpenes (mainly cafestol and kahweol) that appear to raise serum total and LDL-cholesterol concentrations in humans. (More information)
- The results of observational studies suggest that coffee consumption is associated with lower risks of type 2 diabetes mellitus, Parkinson’s disease, liver disease, and mortality. However, it is premature to recommend coffee consumption for disease prevention based on this evidence. (More information)
- Coffee consumption has been associated with lower risks of cirrhosis, cirrhosis-related mortality, and liver cancer. Coffee consumption was also found to be inversely associated with the risk of oral/pharyngeal cancer, colon cancer, prostate cancer, endometrial cancer, and melanoma. Evidence also suggests that coffee consumption is not a risk factor for lung cancer. (More information)
- Despite evidence from clinical trials that caffeine in coffee can increase blood pressure, most prospective cohort studies have not found moderate coffee consumption to be associated with increased risk of cardiovascular disease. (More information)
- Caffeine intake comparable to the amount in 2-3 cups of coffee may raise blood pressure, especially in people with borderline or high blood pressure. However, regular coffee consumption in hypertensive subjects has not been associated with an increased risk of cardiovascular disease. (More information)
- Current evidence from dose-response meta-analyses of observational studies does not exclude that moderate maternal coffee consumption could adversely affect fetal growth during pregnancy. Limiting intakes of caffeinated coffee to ≤1 cup/day during pregnancy and 2-3 cups/day during breast-feeding is recommended. (More information)
- Ensuring adequate calcium and vitamin D intakes and limiting coffee consumption to 3 cups/day (~300 mg/day of caffeine) are unlikely to cause any adverse effects on calcium absorption and bone health. (More information)
- Overall, there is little evidence of health risks and some evidence of health benefits for adults consuming moderate amounts of filtered coffee (3-4 cups/day providing ~300-400 mg/day of caffeine). (More information)
Other Articles
Introduction
Coffee, an infusion of ground, roasted coffee beans, is among the most widely consumed beverages in the world. The main types of coffee consumed are (1) boiled unfiltered coffee, (2) filtered coffee, and (3) decaffeinated coffee (1). Although caffeine has received the most attention from scientists, coffee is a complex mixture of many chemicals, including carbohydrates, lipids (fats), amino acids, vitamins, minerals, alkaloids, and phenolic compounds (2). The composition of coffee varies with the source of coffee beans (Coffea arabica or Coffea canephora var. robusta) (3), as well as with the method of preparation (i.e., filtration methods, boiling, steeping, or brewing under pressure) (1, 4).
Some Bioactive Compounds in Coffee
Chlorogenic acids
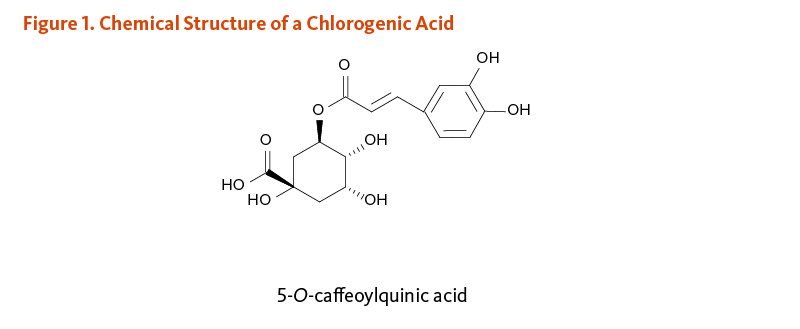
Chlorogenic acids are a family of esters formed between quinic acid and phenolic compounds known as cinnamic acids (mostly caffeic acid and ferulic acid) (1). The most abundant chlorogenic acid in coffee is 5-O-caffeoylquinic acid, an ester formed between quinic acid and caffeic acid (Figure 1). Coffee represents one of the richest dietary sources of chlorogenic acids. The chlorogenic acid content of a 200 mL (7-oz) cup of coffee has been reported to range from 70 to 350 mg, which would provide about 35 to 175 mg of caffeic acid. Although chlorogenic and caffeic acids have demonstrated antioxidant activities in vitro (5), it is unclear how much antioxidant activity they contribute in vivo because they are extensively metabolized, and the metabolites often have lower antioxidant activity than the parent compounds (6, 7). Additionally, the antioxidant capacity of coffee is attenuated by the decaffeination process, which decreases total polyphenol content (8). Other phenolic compounds in coffee, though less abundant than chlorogenic acids, include tannins, lignans, and anthocyanine (1).
Caffeine
Caffeine (1,3,7-trimethylxanthine) is a purine alkaloid that occurs naturally in coffee beans (Figure 2). At intake levels associated with coffee consumption, caffeine appears to exert most of its biological effects through antagonism of the A1 and A2A subtypes of the adenosine receptor (1). Adenosine is an endogenous compound that modulates the response of neurons to neurotransmitters. Adenosine has mostly inhibitory effects in the central nervous system, so the effects of adenosine antagonism by caffeine are generally stimulatory. Caffeine is rapidly and almost completely absorbed in the stomach and small intestine and then distributed to all tissues, including the brain. Caffeine concentrations in coffee beverages can be quite variable. A standard cup of coffee is often assumed to provide 100 mg of caffeine, but an analysis of 14 different specialty coffees purchased at coffee shops in the US found that the amount of caffeine in 8 oz (~240 mL) of brewed coffee ranged from 72 to 130 mg (9). Caffeine in espresso coffees ranged from 58 to 76 mg in a single shot (~35 to 50 mL). In countries other than the US, coffee is often stronger, but the volume per cup is smaller, making 100 mg of caffeine/cup a reasonable estimate.
Diterpenes
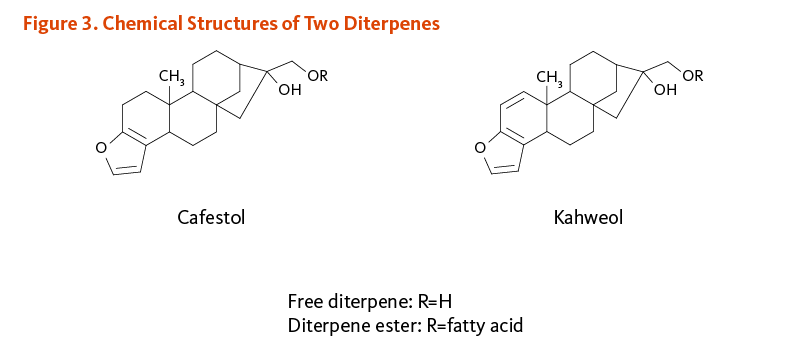
Cafestol and kahweol are fat-soluble compounds known as diterpenes (Figure 3), which have been found to raise serum total and LDL-cholesterol concentrations in humans (10). Some cafestol and kahweol are extracted from ground coffee during brewing, but are largely removed from coffee by paper filters. Scandinavian boiled coffee, Turkish coffee, and French press (cafetiere) coffee contain relatively high levels of cafestol and kahweol (6 to 12 mg/cup), while filtered coffee, percolated coffee, and instant coffee contain low levels of cafestol and kahweol (0.2 to 0.6 mg/cup) (11). Although diterpene concentrations are relatively high in espresso coffee, the small serving size makes it an intermediate source of cafestol and kahweol (4 mg/cup). Since coffee beans are high in cafestol and kahweol, ingestion of coffee beans or grounds on a regular basis may also raise serum total and LDL-cholesterol.
Trigonelline
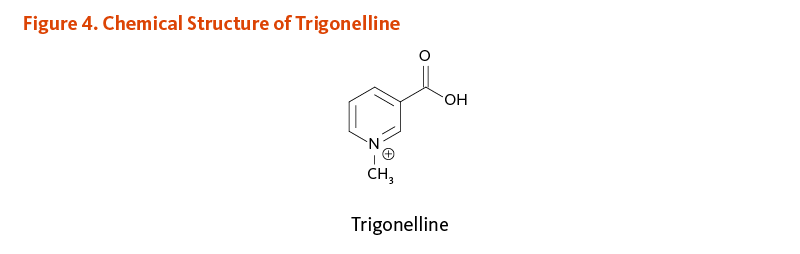
Trigonelline (N-methylnicotinic acid) is a plant alkaloid derived from niacin (vitamin B3) (Figure 4). Trigonelline is largely broken down to nicotinic acid during the roasting process, although some intact molecules remain in roasted beans. Trigonelline has been found to exert antioxidant, hypoglycemic, and hypolipidemic activities (reviewed in 1).
Disease Prevention
Type 2 diabetes mellitus
Observational studies
The three largest prospective cohort studies in the US to examine the relationship between caffeinated coffee consumption and type 2 diabetes mellitus were the Health Professionals Follow-up Study ([HPFS]; 41,934 men), the Nurses’ Health Study ([NHS I]; 84,276 women), and the NHS II (88,259 women). Men who drank at least six cups of coffee daily had a 54% lower risk of developing type 2 diabetes than men who did not drink coffee. In the NHS I cohort, women who drank at least six cups of coffee daily had a 29% lower risk of type 2 diabetes than women who did not drink coffee (12). In the NHS II cohort, women who consumed ≥4 cups of coffee daily had a 39% lower risk of developing type 2 diabetes; similar results were found in women who drank 2 to 3 cups/day of coffee (13). In a pooled analysis of all three cohorts, an increase in caffeinated coffee intake by >1 cup/day over a four-year period was associated with a 13% decreased risk of type 2 diabetes in the subsequent four years; those who decreased their intake of caffeinated coffee by >1 cup/day had a 20% increased risk of type 2 diabetes over the four-year period (14).
Several other cohort studies have found higher coffee intakes to be associated with significant reductions in the risk of type 2 diabetes. A systematic review and meta-analysis of 18 prospective cohort studies (published between 1966 and 2009), including more than 450,000 men and women, found that the risk of developing type 2 diabetes was 24% lower in those consuming 3 to 4 cups/day of coffee compared to those consuming ≤2 cups/day or none (15). Two additional meta-analyses that included data from more recent prospective cohort studies were published concomitantly and found similar results (16, 17). The data analysis of 28 prospective studies in over 1 million participants with 45,335 incident cases of diabetes reported a 30% decreased risk of type 2 diabetes with coffee intake of 5 cups/day versus 0 cups/day (16). In addition, a 9% reduction in the incidence of type 2 diabetes was estimated for every one cup per day increase in total coffee intake. Likewise, a dose-response analysis of 11 studies found a 6% reduction in type 2 diabetes incidence for every one cup per day increase in decaffeinated coffee intake. Every 140 mg/day (~1 cup/day of coffee) increment in caffeine intake was also associated with an 8% reduction in the risk of type 2 diabetes (16). Although decaffeinated coffee consumption is associated with a more modest decrease in the risk of type 2 diabetes, it is likely that compounds other than caffeine contribute to the reduction in diabetes risk.
Intervention studies
The mechanisms that might contribute to the association between coffee consumption and lower risk of type 2 diabetes in prospective cohort studies are unclear. Bioactive compounds other than caffeine appear to show temporary hypoglycemic effects. For example, the acute ingestion of chlorogenic acid (1 g) and trigonelline (0.5 g) transiently lowered blood glucose concentration shortly after the administration of 75 g of glucose in an oral glucose tolerance test (18). Conversely, short-term clinical trials have found that acute administration of caffeine (3 to 6 mg/kg) impaired glucose tolerance and decreased insulin sensitivity in healthy participants (19). In addition, incremental doses of decaffeinated coffee (1, 2, and 4 servings) failed to lower postprandial blood glucose in the presence of 100 mg of caffeine (20). However, despite the deleterious effect of caffeine on glucose homeostasis, caffeinated coffee consumption may favorably affect other metabolic pathways. In a single-blinded clinical trial, subjects at risk for type 2 diabetes abstained from drinking caffeinated coffee for one month, then consumed 4 cups/day of coffee for another month, and finally consumed 8 cups/day for a third month. Compared to one month of coffee abstinence, the consumption of 8 cups/day of coffee for one month appeared to increase antioxidant capacity and reduce subclinical inflammation, as indicated by changes in plasma markers of oxidative stress and inflammation (21).
Until the relationship between long-term coffee consumption and type 2 diabetes risk is better understood, it is premature to recommend coffee consumption as a means of preventing type 2 diabetes (12, 22).
Parkinson’s disease
Studies in animal models of Parkinson’s disease suggest that caffeine may protect dopaminergic neurons by acting as an adenosine A2A-receptor antagonist in the brain (23). Several large prospective cohort studies have examined coffee and/or caffeine intakes in association with Parkinson’s disease risk. A meta-analysis of nine prospective cohort studies found higher caffeine intake to be associated with significant reductions in Parkinson’s disease risk in both men (-39%) and women (-29%) (24). In another meta-analysis of six case-control studies and seven prospective studies, including 901,764 participants and 3,954 cases, an inverse association between coffee intake and Parkinson’s disease risk — only significant in men — was found to be nonlinear, with no further risk reduction beyond 3 to 4 cups/day of coffee. This meta-analysis also reported significant reductions in Parkinson’s disease risk in men (-43%) and women (-36%) with the highest (700 mg/day) versus lowest (100 mg/day) intake of caffeine (25).
However, while prospective studies have consistently found a lower risk of developing Parkinson’s disease with higher coffee and caffeine intakes in men, such an association has not always been observed in women (26-28). It is hypothesized that estrogen replacement therapy may modify the interaction between caffeine and risk of Parkinson’s disease in postmenopausal women. Indeed, because estrogen and caffeine are metabolized by hepatic cytochrome P450 (CYP) 1A2 in the body, estrogen might compete for CYP1A2 activity and hinder the metabolism of caffeine in estrogen users (29). An analysis of data from more than 77,000 female nurses, followed for 18 years in NHS I, revealed that coffee consumption was inversely associated with Parkinson’s disease risk in women who had never used postmenopausal estrogen (30). However, drinking ≥6 cups of coffee was associated with an increased risk or Parkinson’s disease in women who had used postmenopausal estrogen (30). In a prospective cohort study that included more than 238,000 women, a significant inverse association between coffee consumption and Parkinson’s disease mortality was also observed in women who had never used postmenopausal estrogen, but not in those who had used postmenopausal estrogen (31). Yet, in a recent analysis of the National Institutes of Health (NIH)-AARP Diet and Health Study, which included 303,880 participants and 1,100 cases, the highest versus lowest intake of caffeine was associated with a reduced risk of Parkinson’s disease in postmenopausal women who ever used hormones but not among never users (24).
At present, it remains unclear whether caffeine consumption can prevent Parkinson’s disease, particularly in women taking estrogen. Of note, whether caffeine could help reduce some common symptoms associated with Parkinson’s disease (e.g., sleepiness, freezing of gait) is under investigation (32, 33).
Cognitive decline and dementia
Results from observational studies regarding a possible link between coffee consumption and cognitive disorders are inconclusive. A recent meta-analysis of nine prospective cohort studies in 34,282 older adults reported an 18% reduced risk of cognitive disorders with 1 to 2 cups/day of coffee compared to <1 cup/day (34). Yet, there was no difference in risk of cognitive disorders between daily coffee intakes >3 cups and <1 cup (34). Two other meta-analyses of prospective studies failed to find an association between high versus low intakes of coffee and risk of cognitive disorders (35, 36). No dose-response relationship was reported between coffee intake and risk of cognitive disorders (36).
Whether moderate coffee intake may reduce the risk of cognitive decline and dementia later in life is still not known.
Cirrhosis and liver cancer
Chronic inflammation-inducing liver injury may result in cirrhosis. In cirrhosis, the formation of fibrotic scar tissue leads to the progressive deterioration of liver function and other complications, including liver cancer (primarily hepatocellular carcinoma [HCC]) (37). The most common causes of cirrhosis in developed countries are alcohol abuse and chronic infections with hepatitis B and C viruses. Often associated with metabolic disorders, nonalcoholic fatty liver disease (NAFLD) is a liver condition that can progress to nonalcoholic steatohepatitis (NASH) in about one-third of NAFLD patients, thereby increasing the risk of cirrhosis and HCC (38).
A cross-sectional study in 177 subjects with chronic liver disease (especially chronic hepatitis B or C and NASH) found an association between daily caffeine intake >308 mg — equivalent to >2 cups/day of coffee — and a lower risk of having advanced liver fibrosis (39). Of note, no association was reported with non-coffee sources of caffeine like caffeinated soda or green and black tea (39). Recent cross-sectional studies also suggested a protective association of coffee intake against fibrosis development in patients with hepatitis C (40, 41). Additional studies have suggested that consumption of coffee, but not of caffeine, was inversely associated with the risk of advanced liver fibrosis in patients with NAFLD or NASH (42-44).
A recent meta-analysis of four case-control and three prospective cohort studies reported an inverse association between coffee consumption and risk of cirrhosis (45). In addition, a few large prospective cohort studies found that coffee drinking was associated with reduced mortality from alcoholic cirrhosis (46-49). A 17-year study of more than 51,000 men and women in Norway found that those who consumed ≥2 cups/day of coffee had a risk of cirrhosis-related death that was 40% lower than those who never consumed coffee (49). A 22-year prospective cohort study in 125,580 US adults found that coffee drinking was protective against alcoholic cirrhosis but not nonalcoholic cirrhosis (48). Specifically, the risk of developing alcoholic cirrhosis was 40% lower in those who drank 1 to 3 cups/day of coffee and 80% lower in those who drank ≥4 cups/day (48). Recent data from 63,275 Chinese participants of the Singapore Chinese Health Study showed that consumption of ≥2 cups/day of coffee was associated with a 66% lower risk of death from non-viral hepatitis-related cirrhosis — no such association was found with mortality from cirrhosis due to viral hepatitis (46).
Several case-control and prospective cohort studies have found significant inverse associations between coffee consumption and the risk of HCC (reviewed in 50). In a recent 18-year prospective cohort study of 162,022 US adults — comprising Japanese Americans, Caucasians, Mexican Americans, African Americans, and Native Hawaiians — consumption of coffee, but not decaffeinated coffee, was inversely associated with risk of developing HCC (51). Specifically, drinking 2 to 3 cups/day of coffee was associated with a 38% reduced risk of HCC compared to no coffee drinking (51). In addition, the risk of chronic liver disease-related mortality was 71% lower in individuals consuming ≥4 cups/day of regular coffee and 46% lower in those consuming ≥2 cups/day of decaffeinated coffee compared to non-consumers (51). A pooled analysis of this study with 10 other prospective cohort studies found an overall 46% lower risk of liver cancer with coffee consumption (see also Cancer) (52).
Cancer
Numerous observational studies have examined the relationship between coffee consumption and cancer risk (53, 54). Results from recently published meta-analyses of observational studies are reported in Table 1. In addition, a recent meta-analysis of prospective cohort studies by Wang et al. (54) investigated the relationships between the highest versus lowest categories of coffee intake and the risk of most cancer types. Coffee consumption was found to be associated with reduced risks of oral/pharyngeal cancer (6 studies; -31%), colon cancer (10 studies; -13%), liver cancer (9 studies; -54%), prostate cancer (14 studies; -11%), endometrial cancer (12 studies; -27%), and melanoma (6 studies; -11%) (54). Overall, these results are in agreement with those from other pooled analyses of prospective studies presented in Table 1. However, unlike the results reported by Wang et al. (54), coffee consumption has been inversely associated with the risk of colon cancer in case-control studies, but not in prospective cohort studies (see Table 1). Also, in the case of prostate cancer, several meta-analyses suggested a reduced risk with increased coffee intake in prospective but not case-control studies (54-56). However, whether this association exists only in certain study populations or at specific cancer stages remains unclear (57, 58). There seems to be little evidence of associations between coffee consumption and breast cancer, esophageal cancer, glioma, laryngeal cancer, pancreatic cancer, rectal cancer, stomach cancer, and thyroid cancer (Table 1) (54).
| Type of Cancer | Type of Observational Studies | Relative Risk [RR] or Odds Ratio [OR]# (95% Confidence Interval) | Relative Risk [RR] or Odds Ratio [OR] in Subgroup Analyses (e.g., by study types) | References |
|---|---|---|---|---|
| Breast cancer | 10 case-control and 16 prospective cohort studies |
RR: 0.96 (0.93-1.00) |
RR: 0.93 (0.86-1.00) for case-control studies RR: 0.98 (0.93-1.02) for cohort studies RR: 0.81 (0.67-0.97) for ER-negative cancer RR: 1.01 (0.93-1.09) for ER-positive cancer |
Li et al. (2013; 59) |
| 20 case-control and 17 prospective cohort studies | RR: 0.97 (0.93-1.00) |
RR: 0.94 (0.89-1.00) for case-control studies |
Jiang et al. (2013; 60) | |
| Colorectal cancer |
25 case-control studies | OR: 0.85 (0.75-0.97) | OR: 0.68 (0.57-0.81) for colon cancer OR: 0.95 (0.79-1.15) for rectal cancer |
Li et al. (2013; 61) |
| 16 prospective cohort studies | RR: 0.94 (0.88-1.01) | RR: 0.93 (0.86-1.01) for colon cancer RR: 0.98 (0.88-1.09) for rectal cancer |
Li et al. (2013; 61) | |
| 7 case-control and 5 prospective cohort studies | - | RR: 0.78 (0.65-0.95) for case-control studies RR: 0.82 (0.65-1.02) for cohort studies |
Akter et al. (2016; 62) | |
| 19 prospective cohort studies | RR: 0.98 (0.90-1.06) | RR: 0.92 (0.83-1.02) for colon cancer RR: 1.06 (0.95-1.19) for rectal cancer |
Gan et al. (2017; 63) | |
| Endometrial cancer |
10 case-control and 6 prospective cohort studies | RR: 0.71 (0.62-0.81) | RR: 0.69 (0.55-0.87) for case-control studies RR: 0.70 (0.61-0.80) for cohort studies |
Je et al. (2012; 64) |
| 13 prospective cohort studies | RR: 0.80 (0.74-0.86) | RR: 0.66 (0.52-0.84) for caffeinated coffee RR: 0.77 (0.63-0.94) for decaffeinated coffee |
Zhou et al. (2015; 65) | |
| Esophageal cancer | 10 case-control and 4 prospective cohort studies | RR: 0.88 (0.76-1.01) | Zheng et al. (2013; 66) | |
| Glioma | 2 case-control and 4 prospective cohort studies | RR: 1.01 (0.83-1.22) | Malerba et al. (2013; 67) | |
| Laryngeal cancer | 5 case-control studies and 1 prospective cohort study | RR: 1.47 (1.03-2.11) | Chen et al. (2014; 68) | |
| 7 case-control studies and 1 prospective cohort study | RR: 1.22 (0.92-1.62) | Ouyang et al. (2014; 69) | ||
| Liver cancer | 10 prospective cohort studies | RR: 0.55 (0.44-0.67) | RR: 0.57 (0.42-0.79) for women RR: 0.58 (0.40-0.83) for men |
Yu et al. (2016; 52) |
| Lung cancer | 12 case-control and 5 prospective cohort studies | OR: 1.31 (1.11-1.55) | OR: 1.36 (1.10-1.69) for hospital-based case-control studies OR: 0.99 (0.77-1.28) for community-based case-control studies OR: 1.59 (1.26-2.00) for cohort studies OR: 1.41 (1.21-1.63) for men OR:1.16 (0.86-1.56) for women OR: 1.24 (1.00-1.54) for smokers OR: 0.85 (0.64-1.11) for non-smokers |
Xie et al. (2016; 70) |
| Melanoma | 4 case-control and 8 prospective cohort studies | RR: 0.80 (0.69-0.93) | RR: 0.85 (0.71-1.01) for caffeinated coffee RR: 0.92 (0.81-1.05) for decaffeinated coffee |
Wang et al. (2016; 71) |
| Oral cancer | 9 case-control and 3 prospective cohort studies | RR: 0.69 (0.54-0.89) | RR: 0.65 (0.46-0.92) for case-control studies RR: 0.81 (0.62-1.05) for cohort studies RR: 0.81 (0.58-1.13) for studies in the US RR: 0.57 (0.38-0.86) for studies in Europe |
Zhang et al. (2015; 72) |
| Pancreatic cancer |
22 case-control and 15 prospective cohort studies | RR: 1.08 (0.94-1.25) | RR: 1.10 (0.92-1.31) for case-control studies RR: 1.04 (0.80-1.36) for cohort studies |
Turati et al. (2012; 73) |
| 20 prospective cohort studies | RR: 0.88 (0.64-1.12) | Ran et al. (2016; 74) | ||
| 21 prospective cohort studies | RR: 0.99 (0.81-1.21) | Nie et al. (2016; 75) | ||
| Prostate cancer | 12 case-control and 12 prospective cohort studies | RR: 0.94 (0.85-1.05) | RR: 1.36 (1.06-1.75) for studies in the US RR: 1.08 (0.80-1.45) for studies in Europe RR: 0.92 (0.66-1.28) for studies in Asia RR: 0.61 (0.42-0.90) for fatal cancer RR: 0.70 (0.52-0.94) for high-grade tumors RR: 1.07 (0.89-1.29) for low-grade tumors |
Zhong et al. (2014; 58) |
| 12 case-control and 9 prospective cohort studies | RR: 0.91 (0.86-0.97) | RR: 0.91 (0.95-1.26) for case-control studies RR: 0.89 (0.84-0.95) for cohort studies |
Lu et al. (2014; 56) | |
| 13 prospective cohort studies | RR: 0.90 (0.85-0.95) | RR: 0.93 (0.87-1.00) for studies in the US RR: 0.83 (0.75-0.92) for studies in Europe RR: 0.82 (0.51-1.31) for studies in Asia RR: 0.76 (0.55-1.06) for fatal cancer RR: 0.82 (0.61-1.10) for advanced tumors RR: 0.89 (0.83-0.96) for non-advanced tumors |
Liu et al. (2015; 57) | |
| Stomach cancer |
9 prospective cohort studies | RR: 1.18 (0.90-1.55) | Zeng et al. (2015; 76) | |
| 13 prospective cohort studies | RR: 1.13 (0.94-1.35) | RR: 1.36 (1.06-1.75) for studies in the US RR: 1.08 (0.80-1.45) for studies in Europe RR: 0.92 (0.66-1.28) for studies in Asia |
Li et al. (2015; 77) | |
| 13 case-control and 9 prospective cohort studies | RR: 0.96 (0.82-1.12) | RR: 0.85 (0.77-0.95) for case-control studies RR: 1.12 (0.94-1.33) for cohort studies |
Xie et al. (2016; 78) | |
| Thyroid cancer | 5 case-control and 2 prospective cohort studies | OR: 0.88 (0.71-1.07) | Han et al. (2017; 79) | |
| #Odds ratio or relative risk of cancer for the highest vs. lowest categories of coffee intake. | ||||
Finally, there is some evidence suggesting a potential increase in the risk of lung cancer with the highest versus lowest levels of coffee intakes. Yet, cigarette smoking has a major confounding effect on this association (see Health risks associated with coffee consumption) (54, 70).
Mortality
Three large US prospective cohort studies, namely NHS I (74,890 women), NHS II (93,054 women), and HPFS (40,557 men), examined whether coffee drinking was associated with all-cause, cardiovascular disease-related, or cancer-related mortality (80). Compared to no coffee consumption, the consumption of coffee, whether caffeinated or decaffeinated, up to 5 cups/day, was inversely associated with all-cause mortality. There was no difference in risk of death between non-consumers and consumers of >5 cups/day of coffee. Coffee consumption was also found to be inversely associated with mortality related to cardiovascular disease, neurological disease, and suicide (80). Other large cohort studies have reported habitual consumption of any coffee being inversely associated with all-cause and cardiovascular disease-related mortality, but generally not with cancer-related mortality (81-86). Moreover, the associations have not always been consistent among women and men, especially regarding cancer-related mortality (85, 86). A dose-response meta-analysis of 21 prospective studies found a nonlinear inverse association between coffee consumption and all-cause and cardiovascular disease-related mortality (87). Consumption of only 1 cup/day of coffee was significantly associated with lower risk of all-cause (-8%) and cardiovascular disease-related mortality (-11%). The largest risk reductions for all-cause (-16%) and cardiovascular disease-related mortality (-21%) were found to be associated with the consumption of 4 cups/day and 3 cups/day of coffee, respectively (87).
Safety
Adverse effects
Most adverse effects attributed to coffee consumption are related to caffeine. In healthy adults, daily caffeine consumption ≤400 mg — corresponding to 6.5 mg/kg body weight/day for a 70-kg person — is usually not associated with adverse effects (88). Caffeine intakes of less than 300 mg/day in women of childbearing age (equivalent to 4.3 mg/kg body weight/day for a 70-kg woman) and less than 2.5 mg/kg body weight/day in children are unlikely to cause adverse effects (88).
Adverse reactions to caffeine may include tachycardia (rapid heart rate), palpitations, insomnia, restlessness, nervousness, tremor, headache, abdominal pain, nausea, vomiting, diarrhea, and diuresis (increased urination) (89). Very high caffeine intakes, not usually from coffee, may induce hypokalemia (abnormally low serum potassium) (90). Sudden cessation of caffeine consumption after long-term use may result in caffeine withdrawal symptoms (91). Gradual withdrawal from caffeine appears less likely to result in withdrawal symptoms than abrupt withdrawal (92). Commonly reported caffeine withdrawal symptoms include headache, fatigue, drowsiness, irritability, difficulty concentrating, and depressed mood.
Potential health risks associated with regular coffee consumption
Cardiovascular disease
Serum lipids
An early meta-analysis of nine randomized controlled trials found that the consumption of unfiltered, boiled coffee dose-dependently increased serum total and LDL-cholesterol concentrations, while the consumption of filtered coffee resulted in very little change (93). A more recent meta-analysis of 12 randomized controlled trials reported that the consumption of coffee increased serum total cholesterol by 8.1 mg/dL, LDL-cholesterol by 5.1 mg/dL, and triglycerides by 12.6 mg/dL (94). The consumption of filtered coffee raised total cholesterol by only 3.6 mg/dL compared to an increase of 12.9 mg/dL with unfiltered coffee consumption. Unlike filtered coffee, consumption of unfiltered coffee significantly increased LDL-cholesterol and triglycerides by 11.9 mg/dL and 18.8 mg/dL, respectively (94). The cholesterol-raising factors in unfiltered coffee have been identified as cafestol and kahweol, two diterpenes that are largely removed from coffee by paper filters (see Diterpenes) (10).
Homocysteine
An elevated plasma total homocysteine concentration has been associated with increased risk of cardiovascular disease, including coronary heart disease, stroke, and peripheral vascular disease, but the relationship may not be causal (95). Higher coffee intakes have been associated with increased plasma homocysteine concentrations in cross-sectional studies conducted in Europe, Scandinavia, and the US (96-100). Controlled clinical trials have confirmed the homocysteine-raising effect of coffee at intakes of about 4 cups/day (101-103).
Hypertension
Hypertension is a well-recognized risk factor for cardiovascular disease. Two meta-analyses of randomized controlled trials showed that high intakes of coffee (3 to 5 cups/day) for <85 days significantly increased systolic/diastolic blood pressure by 1.2/0.5 mm Hg (104) or 2.4/1.2 mm Hg (105). Although the increases in blood pressure seem modest by individual standards, it has been estimated that an average systolic blood pressure reduction of 2 mm Hg in a population may result in 10% lower mortality from stroke and 7% lower mortality from coronary heart disease (106). However, a more recent pooled analysis excluding the trials that used decaffeinated coffee in control groups — and thus assessed the effect of caffeine rather than that of coffee — found no significant changes in systolic blood pressure and diastolic blood pressure with high coffee intakes (107). Additionally, two meta-analyses combined the data from observational studies that examined prospectively the association between habitual coffee consumption and risk of hypertension (107, 108). One meta-analysis of six prospective cohort studies included 172,567 non-hypertensive participants (one study enrolled pre-hypertensive subjects) of which 37,135 cases of incident hypertension were reported over follow-up periods spanning from 6.4 years to 33 years (108). Compared to coffee intakes of <1 cup/day, intakes of 1 to 3 cups/day were found to be associated with a 9% increased risk of hypertension. However, no such association could be observed for coffee intakes >3 cups/day (108). Another meta-analysis of four prospective cohort studies found no association between coffee consumption and risk of hypertension (107). Also, a recent analysis of data from 29,985 postmenopausal women followed for nearly four years in the Women’s Health Initiative Observational Study found no increased risk of hypertension with intakes of caffeinated coffee, decaffeinated coffee, or caffeine (109).
While there is little evidence of an association between long-term coffee consumption and risk of hypertension, the available evidence from trials suggests that consumption of caffeine modestly raises systolic blood pressure. Whether this may result in increased risk of stroke and coronary heart disease in the population, particularly in those with hypertension, is still uncertain. Yet, to date, regular coffee consumption in hypertensive subjects has not been associated with an increased risk of cardiovascular disease (110).
Coronary heart disease
A meta-analysis of 20 prospective cohort studies, including 407,806 participants and 15,599 incident coronary heart disease (CHD) cases, found no significant association between coffee consumption and CHD risk (111). Yet, a more recent meta-analysis of 22 prospective cohort studies reported a modest reduction in CHD risk with moderate (3-5 cups/day) — but not high (≥6 cups/day) — coffee intakes compared to low intakes (<1 cup/day) (112).
In addition, cross-sectional studies have provided little evidence that the formation of atherosclerotic plaques, an early event in the development of CHD, is more prevalent in regular coffee drinkers than in non-drinkers (113-116). Coffee consumption has not been linked to the development of atherosclerosis in two prospective cohort studies. In the Coronary Artery Risk Development in Young Adults (CARDIA) study that followed approximately 4,000 young US adults for 15 to 20 years, there was no evidence of an association between regular coffee intake and progression of coronary artery calcification, a measure of subclinical atherosclerosis (117). Moreover, in the Multi-Ethnic Study of Atherosclerosis (MESA) that followed about 6,500 US adults over a median period of 11.1 years, occasional but not regular drinking of coffee was associated with a 28% increased risk of cardiovascular disease compared with no drinking (114). This study found no association between coffee intake and progression of coronary artery calcification (114).
Cardiac arrhythmias
Early clinical trials found coffee or caffeine intake equivalent to 5 to 6 cups/day did not increas the frequency or severity of cardiac arrhythmias in healthy people or in people with coronary heart disease (118, 119). A meta-analysis of six prospective cohort studies in 214,316 participants found no association between coffee consumption and risk of atrial fibrillation, the most common type of cardiac arrhythmias. In addition, a recent meta-analysis of 11 short-term intervention studies (single dose to two-week trials) found that caffeine consumption did not increase the occurrence of ventricular arrhythmias (120). Finally, two meta-analyses of observational studies found no evidence to suggest that caffeine consumption was associated with an increased risk of atrial fibrillation and even reported a modest reduction in risk with moderate intakes (121, 122).
Thus, consumption of coffee or caffeine at usual intakes does not appear to increase the risk of cardiac arrhythmias. The current evidence does not support clinical recommendations that discourage moderate consumption of coffee in patients at risk or with suspicions of cardiac arrhythmia (120, 123).
Stroke
A 2011 meta-analysis that included eight prospective cohort studies — all following participants who were free of cardiovascular disease and diabetes mellitus at baseline — found that consumption of 3 to 4 cups/day of coffee was associated with an 18% lower risk of stroke compared with no consumption and that higher intakes were not associated with an increased risk (124). Since this meta-analysis, a few large prospective studies have reported mixed results on the association between coffee consumption and stroke incidence or stroke-related mortality. Results from 42,659 participants in the German cohort of the European Prospective Investigation in Cancer and Nutrition (EPIC-Germany) reported no association between coffee consumption and stroke incidence over an 8.9 year-period (125). Compared to no intake, consumption of ≥3-6 cups/week of coffee was associated with a reduced risk of stroke in a prospective study of 82,369 Japanese participants (126). Also, the consumption of 4 to 5 cups/day of coffee was associated with a reduced risk of stroke-related mortality among men, but not women, followed in the large prospective NIH-AARP Diet and Health Study (81).
Lung cancer
Several observational studies have examined the relationship between coffee intake and lung cancer risk in humans. A recent meta-analysis of 12 case-control and 5 prospective cohort studies, including a total of 12,276 cases and 102,516 controls, found an overall 31% increased risk of lung cancer with the highest versus lowest levels of coffee intake (see Table 1). Subgroup analyses outlined a significant increase in lung cancer risk with coffee intake in hospital-based case-control (+36%) and prospective cohort studies (+59%), in studies conducted in American (+34%) and Asian (+49%) populations, in men (+41%), and in smokers (+24%); conversely, no significant association between coffee intake and lung cancer risk was found in community-based case-control studies, in studies conducted in European populations, in women, and in non-smokers (70). Another meta-analysis of 13 case-control and 8 prospective cohort studies, including 19,892 cases and 623,645 controls, found a 9% increased risk of lung cancer in coffee drinkers compared to non-drinkers (127). However, a pooled analysis restricted to the 16 (out of 21) studies that adjusted for smoking found no significant association between coffee intake and risk of lung cancer [RR: 1.03 (0.95-1.12)].
Cigarette smoking is a major confounding factor in the association between coffee consumption and lung cancer risk, and the evidence suggests that coffee intake is unlikely to be a risk factor for lung cancer. Of note, residual confounding by smoking remains a concern when a slight increase in lung cancer risk is still observed in studies even after adjustment for tobacco smoking (127).
Adverse pregnancy outcomes
It has been suggested that in utero exposure to caffeine through maternal coffee consumption might have adverse effects on the embryo/fetus during pregnancy and the offspring.
Miscarriage
The results of observational studies that have examined the relationship between maternal coffee or caffeine intake and the risk of miscarriage (spontaneous abortion) have been conflicting. While some prospective cohort studies have observed significant associations between high caffeine intakes, particularly from coffee, and the risk of spontaneous abortion (128-132), other studies have not (133-136). The most recent meta-analysis of 14 prospective cohort studies in 130,456 pregnant women and 3,429 cases of miscarriage found risk of miscarriage increased by 40% and 72% with maternal caffeine intakes of 350 to 699 mg/day and ≥700 mg/day during pregnancy, respectively (137). No significant associations were found for daily doses of caffeine less than 350 mg. A dose-response analysis found a 7% increase in the risk of miscarriage per 100 mg/day-increment in caffeine intake during pregnancy (137). Of note, one prospective cohort study that assessed caffeine intake by measuring serum concentrations of paraxanthine, a caffeine metabolite, found that the risk of spontaneous abortion was only elevated in women with paraxanthine concentrations that suggested caffeine intakes of ≥600 mg/day (138).
It has been proposed that an association between caffeine consumption and the risk of spontaneous abortion could be explained by the relationship between nausea and fetal viability (139). Nausea is more common in women with viable pregnancies than nonviable pregnancies, such that women with viable pregnancies may be more likely avoid or limit caffeine consumption due to nausea (140). However, at least one study found that the significant increase in risk of spontaneous abortion observed in women with caffeine intakes >300 mg/day was independent of nausea in pregnancy (141). Additionally, two other studies found that caffeine consumption was associated with an increased risk of spontaneous abortion in women who experienced nausea or aversion to coffee during pregnancy (131, 142). Nonetheless, this does not exclude the possibility of reverse causality, when the loss of fetal viability results in reduction of pregnancy symptoms, like nausea and aversion to coffee, and may be followed by an increase in coffee intake (137).
Of note, consumption of <400 mg/day of caffeine or <4 cups/day of caffeinated coffee by women prior to pregnancy has not been linked to the risk of miscarriage in an analysis of a large prospective study (NHS I) that followed 11,072 women and a total of 15,590 pregnancies (143).
Although the topic remains unsettled, the American College of Obstetricians and Gynecologists recommends that women limit their daily caffeine intake to <200 mg during pregnancy (144).
Intrauterine growth restriction and low birth weight
Observational studies examining the effects of maternal caffeine and coffee consumption on fetal growth have assessed intrauterine growth restriction (IUGR; also known as small-for-gestational age; defined as fetal weight <10th percentile for gestational age), and/or incidence of low birth weight (defined as weight at birth <2,500 g [5.5 pounds]).
A recent meta-analysis of eight prospective cohort and four case-control studies reported a 38% increased risk of low birth weight with the highest versus lowest intakes of caffeine during pregnancy (145). This risk appeared to increase linearly with incremental doses of caffeine (145, 146). A dose-response meta-analysis of six prospective cohort and five case-control studies found a 7% increased risk of low-birth-weight infants per 100 mg of caffeine consumed daily during pregnancy (146). Likewise, a 100 mg-increment in maternal caffeine intakes has been associated with a 10% increased risk of small-for-gestational age infants in the dose-response analysis of 10 prospective cohort and 5 case-control studies (146).
At present, only one study has examined the impact of limiting caffeine intake during pregnancy on birth weight. In a double-blind, intervention trial that randomized 1,197 regular coffee drinkers (≥3 cups/day of coffee) to drink decaffeinated (median caffeine intake of 117 mg/day) or caffeinated coffee (median caffeine intake of 317 mg/day) throughout the second half of their pregnancy, no differences in length of gestation or infant birth weight were found between the two groups (147).
Although the relationship between maternal caffeine consumption and fetal growth requires further clarification, it appears that even moderate caffeine intakes might adversely affect fetal growth in non-smoking women (145, 146, 148). Limiting caffeine intake to ≤100 mg/day (≤1 cup/day of coffee) during pregnancy may be recommended to avoid any adverse effect, assuming that the associations of caffeine intake with the risk of IUGR and low birth weight are causal (145, 146).
Birth defects
Potential relationships between coffee consumption during pregnancy and congenital birth defects have been investigated in the US population-based National Birth Defects Prevention Study (NBDPS), an ongoing multi-site case-control study. In an analysis that included mothers of 1,531 infants with cleft lips (with or without cleft palates), 813 infants with cleft palates only, and 5,711 control infants, no association was found between the highest versus lowest intakes of coffee and caffeine and the risk of orofacial clefts (149). Another analysis, including mothers of 3,346 cases and 6,642 control infants, suggested an increased risk of anotia/microtia and craniosynostosis with the consumption of coffee or caffeine. However, no dose-response could be detected (150). Further, an analysis of the NBDPS in mothers of 844 infants with limb deficiencies and 8,069 control infants found no increased risk associated with coffee and/or caffeine intake during pregnancy (151). There was no association between maternal coffee or caffeine intake during pregnancy and risk of congenital talipes equinovarus (known as "clubfoot") in another US population-based case-control study of mothers of 646 infants with isolated clubfoot and 2,037 control infants (152). Finally, a recent meta-analysis that combined data from one prospective cohort study and six case-control studies found no association between maternal coffee consumption during pregnancy and risk of neural tube defects (153).
At present, there is no convincing evidence that maternal consumption of 3 cups/day of coffee or 300 mg/day of caffeine during pregnancy increases the risk of congenital malformations in humans.
Childhood acute leukemia
The etiology of acute lymphoblastic leukemia (ALL) and acute myeloblastic leukemia (AML), which primarily affect children, is unclear. It has been suggested that exposure to caffeine during pregnancy might have long-lasting adverse effects on the health of the offspring. A meta-analysis of seven case-control studies that examined maternal coffee consumption during pregnancy in relation to the incidence of childhood acute leukemia found coffee consumption was associated with increased risks of overall acute leukemia (+72%), ALL (+65%), and AML (+58%) (154). Another meta-analysis of eight case-control studies found an increased risk of ALL (+43%), but not AML, with the highest versus lowest intakes of coffee during pregnancy (155). High versus low consumption of other sources of caffeine during pregnancy (tea, cola beverages) and childhood (cola beverages) were not found to be associated with childhood acute leukemia (155).
The evidence of a positive association between maternal coffee intake and childhood acute leukemia is currently limited to case-control studies. Case-control studies usually include more cancer cases than prospective cohort studies, but they are subject to recall bias with respect to coffee consumption and selection bias with respect to the control group (156). Further studies with a prospective design are needed to confirm the possible link between coffee intake during pregnancy and childhood acute leukemia.
Lactation
The American Academy of Pediatrics categorizes caffeine as a maternal medication that is usually compatible with breastfeeding (157). Although high maternal caffeine intakes have been reported to cause irritability and poor sleeping patterns in infants, no adverse effects have been reported with moderate maternal intake of caffeinated beverages equivalent to 2 to 3 cups of coffee daily.
Nutrient interactions
Calcium, osteoporosis, and risk of fracture
Osteoporosis is a multifactorial bone disorder that compromises bone mass and strength and increases the risk of fracture. The results of early controlled studies in humans indicated that coffee and caffeine consumption decreased the efficiency of calcium absorption resulting in a loss of about 4 to 6 mg of calcium per cup of coffee (158, 159). However, there is little evidence to suggest detrimental effects of coffee on bone health in populations with adequate calcium intakes (160). To date, results from observational studies that examined associations between coffee intakes and measures of bone mineral density (BMD) loss — generally used to diagnose osteoporosis — have been mixed (161-164). Further, two meta-analyses of observational studies reported no significant associations between coffee intake and risk of hip fracture (165, 166). A third meta-analysis of six case-control and nine prospective cohort studies found no overall association between coffee intake and total fracture (167). Yet, a gender subgroup analysis of eight studies showed a 14% increased risk of fracture in women — but not in men — with the highest versus lowest intakes of coffee. Another subgroup analysis of six studies found a 35% increased risk of osteoporotic fracture in participants with the highest versus lowest intakes of coffee (167).
Current evidence is scarce to suggest that coffee consumption could increase the risk of bone loss and fracture. Limiting coffee consumption to ≤3 cups/day while ensuring adequate calcium and vitamin D intakes should prevent any potential adverse effects on calcium absorption and bone health.
Nonheme iron
Phenolic compounds in coffee can bind nonheme iron and inhibit its intestinal absorption (168). Drinking 150 to 250 mL of coffee with a test meal has been found to inhibit iron absorption by 24%-73% (169, 170). To maximize iron absorption from a meal or supplements, people with poor iron status should not consume coffee at the same time.
Drug interactions
Habitual caffeine consumption increases hepatic cytochrome P450 (CYP) 1A2 activity, which has implications for the metabolism for a number of medications (171). Conversely, drugs that inhibit the activity of CYP1A2 interfere with the metabolism and elimination of caffeine, thereby increasing the risk of adverse effects (172).
Drugs that alter caffeine metabolism
The following medications may impair the hepatic metabolism of caffeine, delaying its excretion and potentially increasing the risk of caffeine-related side effects: cimetidine (Tagamet), disulfiram (Antabuse), estrogens, fluconazole (Diflucan), fluvoxamine (Luvox), mexiletine (Mexitil), quinolone class antibiotics (Cipro, Avelox), riluzole (Rilutek), terbinafine (Lamisil), and Verapamil (Calan) (173). Concomitant use of ephedrine and caffeine can lead to life-threatening adverse effects, including heart attack, stroke, seizures, and death (173). Use of the drug phenytoin (Dilantin) or cigarette smoking increases the hepatic metabolism of caffeine, resulting in increased elimination and decreased plasma caffeine concentrations (89).
Caffeine effects on other drugs
Caffeine and other methylxanthines may enhance the effects and side effects of β-adrenergic stimulating agents, such as epinephrine and albuterol (89, 171). Caffeine doses of 400 to 1,000 mg may inhibit the hepatic metabolism of the antipsychotic medication, clozapine (Clozaril), potentially elevating serum clozapine concentration and increasing the risk of toxicity. Those taking levothyroxine are advised to avoid drinking coffee at the same time they take their medication because coffee may reduce the absorption of levothyroxine in some patients. Caffeine consumption can decrease the elimination of theophylline, potentially increasing serum theophylline levels. Caffeine has been also found to decrease the systemic elimination of acetaminophen (i.e., paracetamol) and to increase the bioavailability of aspirin, which may partially explain its efficacy in enhancing their analgesic effects. This is important because many pain-relievers on the market today combine caffeine with aspirin and/or acetaminophen. Further, caffeine may decrease the bioavailability of lithium and alendronate (Fosamax) by enhancing their elimination (173).
Authors and Reviewers
Originally written in 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in September 2008 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in April 2017 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in June 2017 by:
Rob van Dam, Ph.D.
Adjunct Associate Professor of Nutrition and Epidemiology
Harvard T.H. Chan School of Public Health
Copyright 2005-2026 Linus Pauling Institute
Figure Alternative Text
Figure 1. Chemical structure of a chlorogenic acid
The figure shows the chemical structure of 5-O-caffeoylquinic acid, a chlorogenic acid.
Figure 2. Chemical structures of caffeine and adenosine
The figure shows the chemical structures of two compounds: caffeine and adenosine.
Figure 3. Chemical structures of two diterpenes
The figure shows the chemical structures of the diterpenes, cafestol and kahweol. Each structure has a multiring diterpene backbone with a terminal ‘OR’ group, where ‘R’ represents hydrogen in free diterpenes or a fatty acid in diterpene esters.
Figure 4. Chemical structure of trigonelline
The figure shows the chemical structure of trigonelline.
References
- Godos J, Pluchinotta FR, Marventano S, et al. Coffee components and cardiovascular risk: beneficial and detrimental effects. Int J Food Sci Nutr. 2014;65(8):925-936. (PubMed)
- Spiller MA. The chemical components of coffee. In: Spiller GA, ed. Caffeine. Boca Raton: CRC Press; 1998:97-161.
- Cagliani LR, Pellegrino G, Giugno G, Consonni R. Quantification of Coffea arabica and Coffea canephora var. robusta in roasted and ground coffee blends. Talanta. 2013;106:169-173. (PubMed)
- Caprioli G, Cortese M, Sagratini G, Vittori S. The influence of different types of preparation (espresso and brew) on coffee aroma and main bioactive constituents. Int J Food Sci Nutr. 2015;66(5):505-513. (PubMed)
- Ozgen M, Reese RN, Tulio AZ, Jr., Scheerens JC, Miller AR. Modified 2,2-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (abts) method to measure antioxidant capacity of Selected small fruits and comparison to ferric reducing antioxidant power (FRAP) and 2,2'-diphenyl-1-picrylhydrazyl (DPPH) methods. J Agric Food Chem. 2006;54(4):1151-1157. (PubMed)
- Olthof MR, Hollman PC, Buijsman MN, van Amelsvoort JM, Katan MB. Chlorogenic acid, quercetin-3-rutinoside and black tea phenols are extensively metabolized in humans. J Nutr. 2003;133(6):1806-1814. (PubMed)
- Piazzon A, Vrhovsek U, Masuero D, Mattivi F, Mandoj F, Nardini M. Antioxidant activity of phenolic acids and their metabolites: synthesis and antioxidant properties of the sulfate derivatives of ferulic and caffeic acids and of the acyl glucuronide of ferulic acid. J Agric Food Chem. 2012;60(50):12312-12323. (PubMed)
- Niseteo T, Komes D, Belscak-Cvitanovic A, Horzic D, Budec M. Bioactive composition and antioxidant potential of different commonly consumed coffee brews affected by their preparation technique and milk addition. Food Chem. 2012;134(4):1870-1877. (PubMed)
- McCusker RR, Goldberger BA, Cone EJ. Caffeine content of specialty coffees. J Anal Toxicol. 2003;27(7):520-522. (PubMed)
- Urgert R, Katan MB. The cholesterol-raising factor from coffee beans. Annu Rev Nutr. 1997;17:305-324. (PubMed)
- Naidoo N, Chen C, Rebello SA, et al. Cholesterol-raising diterpenes in types of coffee commonly consumed in Singapore, Indonesia and India and associations with blood lipids: a survey and cross sectional study. Nutr J. 2011;10:48. (PubMed)
- Salazar-Martinez E, Willett WC, Ascherio A, et al. Coffee consumption and risk for type 2 diabetes mellitus. Ann Intern Med. 2004;140(1):1-8. (PubMed)
- van Dam RM, Willett WC, Manson JE, Hu FB. Coffee, caffeine, and risk of type 2 diabetes: a prospective cohort study in younger and middle-aged U.S. women. Diabetes Care. 2006;29(2):398-403. (PubMed)
- Bhupathiraju SN, Pan A, Manson JE, Willett WC, van Dam RM, Hu FB. Changes in coffee intake and subsequent risk of type 2 diabetes: three large cohorts of US men and women. Diabetologia. 2014;57(7):1346-1354. (PubMed)
- Huxley R, Lee CM, Barzi F, et al. Coffee, decaffeinated coffee, and tea consumption in relation to incident type 2 diabetes mellitus: a systematic review with meta-analysis. Arch Intern Med. 2009;169(22):2053-2063. (PubMed)
- Ding M, Bhupathiraju SN, Chen M, van Dam RM, Hu FB. Caffeinated and decaffeinated coffee consumption and risk of type 2 diabetes: a systematic review and a dose-response meta-analysis. Diabetes Care. 2014;37(2):569-586. (PubMed)
- Jiang X, Zhang D, Jiang W. Coffee and caffeine intake and incidence of type 2 diabetes mellitus: a meta-analysis of prospective studies. Eur J Nutr. 2014;53(1):25-38. (PubMed)
- van Dijk AE, Olthof MR, Meeuse JC, Seebus E, Heine RJ, van Dam RM. Acute effects of decaffeinated coffee and the major coffee components chlorogenic acid and trigonelline on glucose tolerance. Diabetes Care. 2009;32(6):1023-1025. (PubMed)
- Shi X, Xue W, Liang S, Zhao J, Zhang X. Acute caffeine ingestion reduces insulin sensitivity in healthy subjects: a systematic review and meta-analysis. Nutr J. 2016;15(1):103. (PubMed)
- Robertson TM, Clifford MN, Penson S, Chope G, Robertson MD. A single serving of caffeinated coffee impairs postprandial glucose metabolism in overweight men. Br J Nutr. 2015;114(8):1218-1225. (PubMed)
- Kempf K, Herder C, Erlund I, et al. Effects of coffee consumption on subclinical inflammation and other risk factors for type 2 diabetes: a clinical trial. Am J Clin Nutr. 2010;91(4):950-957. (PubMed)
- van Dam RM, Hu FB. Coffee consumption and risk of type 2 diabetes: a systematic review. JAMA. 2005;294(1):97-104. (PubMed)
- Kolahdouzan M, Hamadeh MJ. The neuroprotective effects of caffeine in neurodegenerative diseases. CNS Neurosci Ther. 2017;23(4):272-290. (PubMed)
- Liu R, Guo X, Park Y, et al. Caffeine intake, smoking, and risk of Parkinson disease in men and women. Am J Epidemiol. 2012;175(11):1200-1207. (PubMed)
- Qi H, Li S. Dose-response meta-analysis on coffee, tea and caffeine consumption with risk of Parkinson's disease. Geriatr Gerontol Int. 2014;14(2):430-439. (PubMed)
- Ascherio A, Zhang SM, Hernan MA, et al. Prospective study of caffeine consumption and risk of Parkinson's disease in men and women. Ann Neurol. 2001;50(1):56-63. (PubMed)
- Hu G, Bidel S, Jousilahti P, Antikainen R, Tuomilehto J. Coffee and tea consumption and the risk of Parkinson's disease. Mov Disord. 2007;22(15):2242-2248. (PubMed)
- Palacios N, Gao X, McCullough ML, et al. Caffeine and risk of Parkinson's disease in a large cohort of men and women. Mov Disord. 2012;27(10):1276-1282. (PubMed)
- Pollock BG, Wylie M, Stack JA, et al. Inhibition of caffeine metabolism by estrogen replacement therapy in postmenopausal women. J Clin Pharmacol. 1999;39(9):936-940. (PubMed)
- Ascherio A, Chen H, Schwarzschild MA, Zhang SM, Colditz GA, Speizer FE. Caffeine, postmenopausal estrogen, and risk of Parkinson's disease. Neurology. 2003;60(5):790-795. (PubMed)
- Ascherio A, Weisskopf MG, O'Reilly EJ, et al. Coffee consumption, gender, and Parkinson's disease mortality in the cancer prevention study II cohort: the modifying effects of estrogen. Am J Epidemiol. 2004;160(10):977-984. (PubMed)
- Kitagawa M, Houzen H, Tashiro K. Effects of caffeine on the freezing of gait in Parkinson's disease. Mov Disord. 2007;22(5):710-712. (PubMed)
- Postuma RB, Lang AE, Munhoz RP, et al. Caffeine for treatment of Parkinson disease: a randomized controlled trial. Neurology. 2012;79(7):651-658. (PubMed)
- Wu L, Sun D, He Y. Coffee intake and the incident risk of cognitive disorders: A dose-response meta-analysis of nine prospective cohort studies. Clin Nutr. 2016;36(3):730-736. (PubMed)
- Kim YS, Kwak SM, Myung SK. Caffeine intake from coffee or tea and cognitive disorders: a meta-analysis of observational studies. Neuroepidemiology. 2015;44(1):51-63. (PubMed)
- Liu QP, Wu YF, Cheng HY, et al. Habitual coffee consumption and risk of cognitive decline/dementia: A systematic review and meta-analysis of prospective cohort studies. Nutrition. 2016;32(6):628-636. (PubMed)
- Friedman SL, Schiano TD. Cirrhosis and its sequelae. In: Goldman L, Ausiello D, eds. Cecil Textbook of Medicine. 22nd ed. St. Louis: W. B. Saunders; 2004:940-944.
- Michelotti GA, Machado MV, Diehl AM. NAFLD, NASH and liver cancer. Nat Rev Gastroenterol Hepatol. 2013;10(11):656-665. (PubMed)
- Modi AA, Feld JJ, Park Y, et al. Increased caffeine consumption is associated with reduced hepatic fibrosis. Hepatology. 2010;51(1):201-209. (PubMed)
- Khalaf N, White D, Kanwal F, et al. Coffee and caffeine are associated with decreased risk of advanced hepatic fibrosis among patients with hepatitis C. Clin Gastroenterol Hepatol. 2015;13(8):1521-1531.e1523. (PubMed)
- Machado SR, Parise ER, Carvalho L. Coffee has hepatoprotective benefits in Brazilian patients with chronic hepatitis C even in lower daily consumption than in American and European populations. Braz J Infect Dis. 2014;18(2):170-176. (PubMed)
- Anty R, Marjoux S, Iannelli A, et al. Regular coffee but not espresso drinking is protective against fibrosis in a cohort mainly composed of morbidly obese European women with NAFLD undergoing bariatric surgery. J Hepatol. 2012;57(5):1090-1096. (PubMed)
- Molloy JW, Calcagno CJ, Williams CD, Jones FJ, Torres DM, Harrison SA. Association of coffee and caffeine consumption with fatty liver disease, nonalcoholic steatohepatitis, and degree of hepatic fibrosis. Hepatology. 2012;55(2):429-436. (PubMed)
- Shen H, Rodriguez AC, Shiani A, et al. Association between caffeine consumption and nonalcoholic fatty liver disease: a systemic review and meta-analysis. Therap Adv Gastroenterol. 2016;9(1):113-120. (PubMed)
- Liu F, Wang X, Wu G, et al. Coffee consumption decreases risks for hepatic fibrosis and cirrhosis: a meta-analysis. PLoS One. 2015;10(11):e0142457. (PubMed)
- Goh GB, Chow WC, Wang R, Yuan JM, Koh WP. Coffee, alcohol and other beverages in relation to cirrhosis mortality: the Singapore Chinese Health Study. Hepatology. 2014;60(2):661-669. (PubMed)
- Klatsky AL, Armstrong MA, Friedman GD. Coffee, tea, and mortality. Ann Epidemiol. 1993;3(4):375-381. (PubMed)
- Klatsky AL, Morton C, Udaltsova N, Friedman GD. Coffee, cirrhosis, and transaminase enzymes. Arch Intern Med. 2006;166(11):1190-1195. (PubMed)
- Tverdal A, Skurtveit S. Coffee intake and mortality from liver cirrhosis. Ann Epidemiol. 2003;13(6):419-423. (PubMed)
- Saab S, Mallam D, Cox GA, 2nd, Tong MJ. Impact of coffee on liver diseases: a systematic review. Liver Int. 2014;34(4):495-504. (PubMed)
- Setiawan VW, Wilkens LR, Lu SC, Hernandez BY, Le Marchand L, Henderson BE. Association of coffee intake with reduced incidence of liver cancer and death from chronic liver disease in the US multiethnic cohort. Gastroenterology. 2015;148(1):118-125; quiz e115. (PubMed)
- Yu C, Cao Q, Chen P, et al. An updated dose-response meta-analysis of coffee consumption and liver cancer risk. Sci Rep. 2016;6:37488. (PubMed)
- Alicandro G, Tavani A, La Vecchia C. Coffee and cancer risk: a summary overview. Eur J Cancer Prev. 2017. (PubMed)
- Wang A, Wang S, Zhu C, et al. Coffee and cancer risk: A meta-analysis of prospective observational studies. Sci Rep. 2016;6:33711. (PubMed)
- Cao S, Liu L, Yin X, Wang Y, Liu J, Lu Z. Coffee consumption and risk of prostate cancer: a meta-analysis of prospective cohort studies. Carcinogenesis. 2014;35(2):256-261. (PubMed)
- Lu Y, Zhai L, Zeng J, et al. Coffee consumption and prostate cancer risk: an updated meta-analysis. Cancer Causes Control. 2014;25(5):591-604. (PubMed)
- Liu H, Hu GH, Wang XC, et al. Coffee consumption and prostate cancer risk: a meta-analysis of cohort studies. Nutr Cancer. 2015;67(3):392-400. (PubMed)
- Zhong S, Chen W, Yu X, Chen Z, Hu Q, Zhao J. Coffee consumption and risk of prostate cancer: an up-to-date meta-analysis. Eur J Clin Nutr. 2014;68(3):330-337. (PubMed)
- Li XJ, Ren ZJ, Qin JW, et al. Coffee consumption and risk of breast cancer: an up-to-date meta-analysis. PLoS One. 2013;8(1):e52681. (PubMed)
- Jiang W, Wu Y, Jiang X. Coffee and caffeine intake and breast cancer risk: an updated dose-response meta-analysis of 37 published studies. Gynecol Oncol. 2013;129(3):620-629. (PubMed)
- Li G, Ma D, Zhang Y, Zheng W, Wang P. Coffee consumption and risk of colorectal cancer: a meta-analysis of observational studies. Public Health Nutr. 2013;16(2):346-357. (PubMed)
- Akter S, Kashino I, Mizoue T, et al. Coffee drinking and colorectal cancer risk: an evaluation based on a systematic review and meta-analysis among the Japanese population. Jpn J Clin Oncol. 2016;46(8):781-787. (PubMed)
- Gan Y, Wu J, Zhang S, et al. Association of coffee consumption with risk of colorectal cancer: a meta-analysis of prospective cohort studies. Oncotarget. 2016; 8(12):18699-18711. (PubMed)
- Je Y, Giovannucci E. Coffee consumption and risk of endometrial cancer: findings from a large up-to-date meta-analysis. Int J Cancer. 2012;131(7):1700-1710. (PubMed)
- Zhou Q, Luo ML, Li H, Li M, Zhou JG. Coffee consumption and risk of endometrial cancer: a dose-response meta-analysis of prospective cohort studies. Sci Rep. 2015;5:13410. (PubMed)
- Zheng JS, Yang J, Fu YQ, Huang T, Huang YJ, Li D. Effects of green tea, black tea, and coffee consumption on the risk of esophageal cancer: a systematic review and meta-analysis of observational studies. Nutr Cancer. 2013;65(1):1-16. (PubMed)
- Malerba S, Galeone C, Pelucchi C, et al. A meta-analysis of coffee and tea consumption and the risk of glioma in adults. Cancer Causes Control. 2013;24(2):267-276. (PubMed)
- Chen J, Long S. Tea and coffee consumption and risk of laryngeal cancer: a systematic review meta-analysis. PLoS One. 2014;9(12):e112006. (PubMed)
- Ouyang Z, Wang Z, Jin J. Association between tea and coffee consumption and risk of laryngeal cancer: a meta-analysis. Int J Clin Exp Med. 2014;7(12):5192-5200. (PubMed)
- Xie Y, Qin J, Nan G, Huang S, Wang Z, Su Y. Coffee consumption and the risk of lung cancer: an updated meta-analysis of epidemiological studies. Eur J Clin Nutr. 2016;70(2):199-206. (PubMed)
- Wang J, Li X, Zhang D. Coffee consumption and the risk of cutaneous melanoma: a meta-analysis. Eur J Nutr. 2016;55(4):1317-1329. (PubMed)
- Zhang Y, Wang X, Cui D. Association between coffee consumption and the risk of oral cancer: a meta-analysis of observational studies. Int J Clin Exp Med. 2015;8(7):11657-11665. (PubMed)
- Turati F, Galeone C, Edefonti V, et al. A meta-analysis of coffee consumption and pancreatic cancer. Ann Oncol. 2012;23(2):311-318. (PubMed)
- Ran HQ, Wang JZ, Sun CQ. Coffee consumption and pancreatic cancer risk: an update meta-analysis of cohort studies. Pak J Med Sci. 2016;32(1):253-259. (PubMed)
- Nie K, Xing Z, Huang W, Wang W, Liu W. Coffee intake and risk of pancreatic cancer: an updated meta-analysis of prospective studies. Minerva Med. 2016;107(4):270-278. (PubMed)
- Zeng SB, Weng H, Zhou M, Duan XL, Shen XF, Zeng XT. Long-term coffee consumption and risk of gastric cancer: a PRISMA-compliant dose-response meta-analysis of prospective cohort studies. Medicine (Baltimore). 2015;94(38):e1640. (PubMed)
- Li L, Gan Y, Wu C, Qu X, Sun G, Lu Z. Coffee consumption and the risk of gastric cancer: a meta-analysis of prospective cohort studies. BMC Cancer. 2015;15:733. (PubMed)
- Xie Y, Huang S, He T, Su Y. Coffee consumption and risk of gastric cancer: an updated meta-analysis. Asia Pac J Clin Nutr. 2016;25(3):578-588. (PubMed)
- Han MA, Kim JH. Coffee consumption and the risk of thyroid cancer: a systematic review and meta-analysis. Int J Environ Res Public Health. 2017;14(2). (PubMed)
- Ding M, Satija A, Bhupathiraju SN, et al. Association of coffee consumption with total and cause-specific mortality in 3 large prospective cohorts. Circulation. 2015;132(24):2305-2315. (PubMed)
- Freedman ND, Park Y, Abnet CC, Hollenbeck AR, Sinha R. Association of coffee drinking with total and cause-specific mortality. N Engl J Med. 2012;366(20):1891-1904. (PubMed)
- Lof M, Sandin S, Yin L, Adami HO, Weiderpass E. Prospective study of coffee consumption and all-cause, cancer, and cardiovascular mortality in Swedish women. Eur J Epidemiol. 2015;30(9):1027-1034. (PubMed)
- Loftfield E, Freedman ND, Graubard BI, et al. Association of coffee consumption with overall and cause-specific mortality in a large US prospective cohort study. Am J Epidemiol. 2015;182(12):1010-1022. (PubMed)
- Saito E, Inoue M, Sawada N, et al. Association of coffee intake with total and cause-specific mortality in a Japanese population: the Japan Public Health Center-based Prospective Study. Am J Clin Nutr. 2015;101(5):1029-1037. (PubMed)
- Sugiyama K, Kuriyama S, Akhter M, et al. Coffee consumption and mortality due to all causes, cardiovascular disease, and cancer in Japanese women. J Nutr. 2010;140(5):1007-1013. (PubMed)
- Tamakoshi A, Lin Y, Kawado M, Yagyu K, Kikuchi S, Iso H. Effect of coffee consumption on all-cause and total cancer mortality: findings from the JACC study. Eur J Epidemiol. 2011;26(4):285-293. (PubMed)
- Crippa A, Discacciati A, Larsson SC, Wolk A, Orsini N. Coffee consumption and mortality from all causes, cardiovascular disease, and cancer: a dose-response meta-analysis. Am J Epidemiol. 2014;180(8):763-775. (PubMed)
- Nawrot P, Jordan S, Eastwood J, Rotstein J, Hugenholtz A, Feeley M. Effects of caffeine on human health. Food Addit Contam. 2003;20(1):1-30. (PubMed)
- CNS Stimulants: Caffeine. In: Novak K, ed. Drug Facts and Comparisons. St. Louis: Wolters Kluwer Health; 2005:917-919.
- Engebretsen KM, Harris CR. Caffeine and Related Nonprescription Sympathomimetics. In: Ford MD, Delaney KA, Ling LJ, Erickson T, eds. Clinical Toxicology. Philadelphia: W. B. Saunders; 2001:310-315.
- Juliano LM, Griffiths RR. A critical review of caffeine withdrawal: empirical validation of symptoms and signs, incidence, severity, and associated features. Psychopharmacology (Berl). 2004;176(1):1-29. (PubMed)
- Dews PB, Curtis GL, Hanford KJ, O'Brien CP. The frequency of caffeine withdrawal in a population-based survey and in a controlled, blinded pilot experiment. J Clin Pharmacol. 1999;39(12):1221-1232. (PubMed)
- Jee SH, He J, Appel LJ, Whelton PK, Suh I, Klag MJ. Coffee consumption and serum lipids: a meta-analysis of randomized controlled clinical trials. Am J Epidemiol. 2001;153(4):353-362. (PubMed)
- Cai L, Ma D, Zhang Y, Liu Z, Wang P. The effect of coffee consumption on serum lipids: a meta-analysis of randomized controlled trials. Eur J Clin Nutr. 2012;66(8):872-877. (PubMed)
- Splaver A, Lamas GA, Hennekens CH. Homocysteine and cardiovascular disease: biological mechanisms, observational epidemiology, and the need for randomized trials. Am Heart J. 2004;148(1):34-40. (PubMed)
- Husemoen LL, Thomsen TF, Fenger M, Jorgensen T. Effect of lifestyle factors on plasma total homocysteine concentrations in relation to MTHFR(C677T) genotype. Inter99 (7). Eur J Clin Nutr. 2004;58(8):1142-1150. (PubMed)
- Mennen LI, de Courcy GP, Guilland JC, et al. Homocysteine, cardiovascular disease risk factors, and habitual diet in the French Supplementation with Antioxidant Vitamins and Minerals Study. Am J Clin Nutr. 2002;76(6):1279-1289. (PubMed)
- de Bree A, Verschuren WM, Blom HJ, Kromhout D. Lifestyle factors and plasma homocysteine concentrations in a general population sample. Am J Epidemiol. 2001;154(2):150-154. (PubMed)
- Stolzenberg-Solomon RZ, Miller ER, 3rd, Maguire MG, Selhub J, Appel LJ. Association of dietary protein intake and coffee consumption with serum homocysteine concentrations in an older population. Am J Clin Nutr. 1999;69(3):467-475. (PubMed)
- Nygard O, Refsum H, Ueland PM, et al. Coffee consumption and plasma total homocysteine: The Hordaland Homocysteine Study. Am J Clin Nutr. 1997;65(1):136-143. (PubMed)
- Christensen B, Mosdol A, Retterstol L, Landaas S, Thelle DS. Abstention from filtered coffee reduces the concentrations of plasma homocysteine and serum cholesterol--a randomized controlled trial. Am J Clin Nutr. 2001;74(3):302-307. (PubMed)
- Urgert R, van Vliet T, Zock PL, Katan MB. Heavy coffee consumption and plasma homocysteine: a randomized controlled trial in healthy volunteers. Am J Clin Nutr. 2000;72(5):1107-1110. (PubMed)
- Grubben MJ, Boers GH, Blom HJ, et al. Unfiltered coffee increases plasma homocysteine concentrations in healthy volunteers: a randomized trial. Am J Clin Nutr. 2000;71(2):480-484. (PubMed)
- Noordzij M, Uiterwaal CS, Arends LR, Kok FJ, Grobbee DE, Geleijnse JM. Blood pressure response to chronic intake of coffee and caffeine: a meta-analysis of randomized controlled trials. J Hypertens. 2005;23(5):921-928. (PubMed)
- Jee SH, He J, Whelton PK, Suh I, Klag MJ. The effect of chronic coffee drinking on blood pressure: a meta-analysis of controlled clinical trials. Hypertension. 1999;33(2):647-652. (PubMed)
- Lewington S, Clarke R, Qizilbash N, Peto R, Collins R. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet. 2002;360(9349):1903-1913. (PubMed)
- Steffen M, Kuhle C, Hensrud D, Erwin PJ, Murad MH. The effect of coffee consumption on blood pressure and the development of hypertension: a systematic review and meta-analysis. J Hypertens. 2012;30(12):2245-2254. (PubMed)
- Zhang Z, Hu G, Caballero B, Appel L, Chen L. Habitual coffee consumption and risk of hypertension: a systematic review and meta-analysis of prospective observational studies. Am J Clin Nutr. 2011;93(6):1212-1219. (PubMed)
- Rhee JJ, Qin F, Hedlin HK, et al. Coffee and caffeine consumption and the risk of hypertension in postmenopausal women. Am J Clin Nutr. 2016;103(1):210-217. (PubMed)
- Mesas AE, Leon-Munoz LM, Rodriguez-Artalejo F, Lopez-Garcia E. The effect of coffee on blood pressure and cardiovascular disease in hypertensive individuals: a systematic review and meta-analysis. Am J Clin Nutr. 2011;94(4):1113-1126. (PubMed)
- Wu JN, Ho SC, Zhou C, et al. Coffee consumption and risk of coronary heart diseases: a meta-analysis of 21 prospective cohort studies. Int J Cardiol. 2009;137(3):216-225. (PubMed)
- Ding M, Bhupathiraju SN, Satija A, van Dam RM, Hu FB. Long-term coffee consumption and risk of cardiovascular disease: a systematic review and a dose-response meta-analysis of prospective cohort studies. Circulation. 2014;129(6):643-659. (PubMed)
- Choi Y, Chang Y, Ryu S, et al. Coffee consumption and coronary artery calcium in young and middle-aged asymptomatic adults. Heart. 2015;101(9):686-691. (PubMed)
- Miller PE, Zhao D, Frazier-Wood AC, et al. Associations of Coffee, Tea, and Caffeine Intake with Coronary Artery Calcification and Cardiovascular Events. Am J Med. 2017;130(2):188-197.e185. (PubMed)
- Patel YR, Gadiraju TV, Ellison RC, et al. Coffee consumption and calcified atherosclerotic plaques in the coronary arteries: The NHLBI Family Heart Study. Clin Nutr ESPEN. 2017;17:18-21. (PubMed)
- van Woudenbergh GJ, Vliegenthart R, van Rooij FJ, et al. Coffee consumption and coronary calcification: the Rotterdam Coronary Calcification Study. Arterioscler Thromb Vasc Biol. 2008;28(5):1018-1023. (PubMed)
- Reis JP, Loria CM, Steffen LM, et al. Coffee, decaffeinated coffee, caffeine, and tea consumption in young adulthood and atherosclerosis later in life: the CARDIA study. Arterioscler Thromb Vasc Biol. 2010;30(10):2059-2066. (PubMed)
- Chelsky LB, Cutler JE, Griffith K, Kron J, McClelland JH, McAnulty JH. Caffeine and ventricular arrhythmias. An electrophysiological approach. JAMA. 1990;264(17):2236-2240. (PubMed)
- Myers MG. Caffeine and cardiac arrhythmias. Ann Intern Med. 1991;114(2):147-150. (PubMed)
- Zuchinali P, Ribeiro PA, Pimentel M, da Rosa PR, Zimerman LI, Rohde LE. Effect of caffeine on ventricular arrhythmia: a systematic review and meta-analysis of experimental and clinical studies. Europace. 2016;18(2):257-266. (PubMed)
- Caldeira D, Martins C, Alves LB, Pereira H, Ferreira JJ, Costa J. Caffeine does not increase the risk of atrial fibrillation: a systematic review and meta-analysis of observational studies. Heart. 2013;99(19):1383-1389. (PubMed)
- Cheng M, Hu Z, Lu X, Huang J, Gu D. Caffeine intake and atrial fibrillation incidence: dose response meta-analysis of prospective cohort studies. Can J Cardiol. 2014;30(4):448-454. (PubMed)
- Glatter KA, Myers R, Chiamvimonvat N. Recommendations regarding dietary intake and caffeine and alcohol consumption in patients with cardiac arrhythmias: what do you tell your patients to do or not to do? Curr Treat Options Cardiovasc Med. 2012;14(5):529-535. (PubMed)
- Larsson SC, Orsini N. Coffee consumption and risk of stroke: a dose-response meta-analysis of prospective studies. Am J Epidemiol. 2011;174(9):993-1001. (PubMed)
- Floegel A, Pischon T, Bergmann MM, Teucher B, Kaaks R, Boeing H. Coffee consumption and risk of chronic disease in the European Prospective Investigation into Cancer and Nutrition (EPIC)-Germany study. Am J Clin Nutr. 2012;95(4):901-908. (PubMed)
- Kokubo Y, Iso H, Saito I, et al. The impact of green tea and coffee consumption on the reduced risk of stroke incidence in Japanese population: the Japan public health center-based study cohort. Stroke. 2013;44(5):1369-1374. (PubMed)
- Galarraga V, Boffetta P. Coffee Drinking and Risk of Lung Cancer-A Meta-Analysis. Cancer Epidemiol Biomarkers Prev. 2016;25(6):951-957. (PubMed)
- Bech BH, Nohr EA, Vaeth M, Henriksen TB, Olsen J. Coffee and fetal death: a cohort study with prospective data. Am J Epidemiol. 2005;162(10):983-990. (PubMed)
- Greenwood DC, Alwan N, Boylan S, et al. Caffeine intake during pregnancy, late miscarriage and stillbirth. Eur J Epidemiol. 2010;25(4):275-280. (PubMed)
- Tolstrup JS, Kjaer SK, Munk C, et al. Does caffeine and alcohol intake before pregnancy predict the occurrence of spontaneous abortion? Hum Reprod. 2003;18(12):2704-2710. (PubMed)
- Wen W, Shu XO, Jacobs DR, Jr., Brown JE. The associations of maternal caffeine consumption and nausea with spontaneous abortion. Epidemiology. 2001;12(1):38-42. (PubMed)
- Weng X, Odouli R, Li DK. Maternal caffeine consumption during pregnancy and the risk of miscarriage: a prospective cohort study. Am J Obstet Gynecol. 2008;198(3):279 e271-278. (PubMed)
- Fenster L, Hubbard AE, Swan SH, et al. Caffeinated beverages, decaffeinated coffee, and spontaneous abortion. Epidemiology. 1997;8(5):515-523. (PubMed)
- Mills JL, Holmes LB, Aarons JH, et al. Moderate caffeine use and the risk of spontaneous abortion and intrauterine growth retardation. JAMA. 1993;269(5):593-597. (PubMed)
- Pollack AZ, Buck Louis GM, Sundaram R, Lum KJ. Caffeine consumption and miscarriage: a prospective cohort study. Fertil Steril. 2010;93(1):304-306. (PubMed)
- Savitz DA, Chan RL, Herring AH, Howards PP, Hartmann KE. Caffeine and miscarriage risk. Epidemiology. 2008;19(1):55-62. (PubMed)
- Chen LW, Wu Y, Neelakantan N, Chong MF, Pan A, van Dam RM. Maternal caffeine intake during pregnancy and risk of pregnancy loss: a categorical and dose-response meta-analysis of prospective studies. Public Health Nutr. 2016;19(7):1233-1244. (PubMed)
- Klebanoff MA, Levine RJ, DerSimonian R, Clemens JD, Wilkins DG. Maternal serum paraxanthine, a caffeine metabolite, and the risk of spontaneous abortion. N Engl J Med. 1999;341(22):1639-1644. (PubMed)
- Leviton A, Cowan L. A review of the literature relating caffeine consumption by women to their risk of reproductive hazards. Food Chem Toxicol. 2002;40(9):1271-1310. (PubMed)
- Peck JD, Leviton A, Cowan LD. A review of the epidemiologic evidence concerning the reproductive health effects of caffeine consumption: a 2000-2009 update. Food Chem Toxicol. 2010;48(10):2549-2576. (PubMed)
- Giannelli M, Doyle P, Roman E, Pelerin M, Hermon C. The effect of caffeine consumption and nausea on the risk of miscarriage. Paediatr Perinat Epidemiol. 2003;17(4):316-323. (PubMed)
- Cnattingius S, Signorello LB, Anneren G, et al. Caffeine intake and the risk of first-trimester spontaneous abortion. N Engl J Med. 2000;343(25):1839-1845. (PubMed)
- Gaskins AJ, Rich-Edwards JW, Williams PL, Toth TL, Missmer SA, Chavarro JE. Pre-pregnancy caffeine and caffeinated beverage intake and risk of spontaneous abortion. Eur J Nutr. 2016. [Epub ahead of print]. (PubMed)
- American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 462: Moderate caffeine consumption during pregnancy. Obstet Gynecol. 2010 (Reaffirmed 2016);116(2 Pt 1):467-468.
- Rhee J, Kim R, Kim Y, et al. Maternal caffeine consumption during pregnancy and risk of low birth weight: a dose-response meta-analysis of observational studies. PLoS One. 2015;10(7):e0132334. (PubMed)
- Greenwood DC, Thatcher NJ, Ye J, et al. Caffeine intake during pregnancy and adverse birth outcomes: a systematic review and dose-response meta-analysis. Eur J Epidemiol. 2014;29(10):725-734. (PubMed)
- Bech BH, Obel C, Henriksen TB, Olsen J. Effect of reducing caffeine intake on birth weight and length of gestation: randomised controlled trial. BMJ. 2007;334(7590):409. (PubMed)
- Chen LW, Wu Y, Neelakantan N, Chong MF, Pan A, van Dam RM. Maternal caffeine intake during pregnancy is associated with risk of low birth weight: a systematic review and dose-response meta-analysis. BMC Med. 2014;12:174. (PubMed)
- Collier SA, Browne ML, Rasmussen SA, Honein MA. Maternal caffeine intake during pregnancy and orofacial clefts. Birth Defects Res A Clin Mol Teratol. 2009;85(10):842-849. (PubMed)
- Browne ML, Hoyt AT, Feldkamp ML, et al. Maternal caffeine intake and risk of selected birth defects in the National Birth Defects Prevention Study. Birth Defects Res A Clin Mol Teratol. 2011;91(2):93-101. (PubMed)
- Chen L, Bell EM, Browne ML, et al. Maternal caffeine consumption and risk of congenital limb deficiencies. Birth Defects Res A Clin Mol Teratol. 2012;94(12):1033-1043. (PubMed)
- Werler MM, Yazdy MM, Kasser JR, et al. Maternal cigarette, alcohol, and coffee consumption in relation to risk of clubfoot. Paediatr Perinat Epidemiol. 2015;29(1):3-10. (PubMed)
- Li ZX, Gao ZL, Wang JN, Guo QH. Maternal coffee consumption during pregnancy and neural tube defects in offspring: a meta-analysis. Fetal Pediatr Pathol. 2016;35(1):1-9. (PubMed)
- Cheng J, Su H, Zhu R, et al. Maternal coffee consumption during pregnancy and risk of childhood acute leukemia: a metaanalysis. Am J Obstet Gynecol. 2014;210(2):151.e151-151.e110. (PubMed)
- homopoulos TP, Ntouvelis E, Diamantaras AA, et al. Maternal and childhood consumption of coffee, tea and cola beverages in association with childhood leukemia: a meta-analysis. Cancer Epidemiol. 2015;39(6):1047-1059. (PubMed)
- Song JW, Chung KC. Observational studies: cohort and case-control studies. Plast Reconstr Surg. 2010;126(6):2234-2242. (PubMed)
- The American Academy of Pediatrics. Things to Avoid When Breastfeeding. 21 November 2015 Available at: https://www.healthychildren.org/English/ages-stages/baby/breastfeeding/Pages/Things-to-Avoid-When-Breastfeeding.aspx. Accessed 4/11/17.
- Barger-Lux MJ, Heaney RP. Caffeine and the calcium economy revisited. Osteoporos Int. 1995;5(2):97-102. (PubMed)
- Hasling C, Sondergaard K, Charles P, Mosekilde L. Calcium metabolism in postmenopausal osteoporotic women is determined by dietary calcium and coffee intake. J Nutr. 1992;122(5):1119-1126. (PubMed)
- Heaney RP. Effects of caffeine on bone and the calcium economy. Food Chem Toxicol. 2002;40(9):1263-1270. (PubMed)
- Choi E, Choi KH, Park SM, Shin D, Joh HK, Cho E. The benefit of bone health by drinking coffee among Korean postmenopausal women: a cross-sectional analysis of the fourth & fifth Korea National Health and Nutrition Examination Surveys. PLoS One. 2016;11(1):e0147762. (PubMed)
- Hallstrom H, Byberg L, Glynn A, Lemming EW, Wolk A, Michaelsson K. Long-term coffee consumption in relation to fracture risk and bone mineral density in women. Am J Epidemiol. 2013;178(6):898-909. (PubMed)
- Harris SS, Dawson-Hughes B. Caffeine and bone loss in healthy postmenopausal women. Am J Clin Nutr. 1994;60(4):573-578. (PubMed)
- Rapuri PB, Gallagher JC, Kinyamu HK, Ryschon KL. Caffeine intake increases the rate of bone loss in elderly women and interacts with vitamin D receptor genotypes. Am J Clin Nutr. 2001;74(5):694-700. (PubMed)
- Li S, Dai Z, Wu Q. Effect of coffee intake on hip fracture: a meta-analysis of prospective cohort studies. Nutr J. 2015;14:38. (PubMed)
- Sheng J, Qu X, Zhang X, et al. Coffee, tea, and the risk of hip fracture: a meta-analysis. Osteoporos Int. 2014;25(1):141-150. (PubMed)
- Lee DR, Lee J, Rota M, et al. Coffee consumption and risk of fractures: a systematic review and dose-response meta-analysis. Bone. 2014;63:20-28. (PubMed)
- Fairweather-Tait SJ. Iron nutrition in the UK: getting the balance right. Proc Nutr Soc. 2004;63(4):519-528. (PubMed)
- Morck TA, Lynch SR, Cook JD. Inhibition of food iron absorption by coffee. Am J Clin Nutr. 1983;37(3):416-420. (PubMed)
- Hallberg L, Rossander L. Effect of different drinks on the absorption of non-heme iron from composite meals. Hum Nutr Appl Nutr. 1982;36(2):116-123. (PubMed)
- Carrillo JA, Benitez J. Clinically significant pharmacokinetic interactions between dietary caffeine and medications. Clin Pharmacokinet. 2000;39(2):127-153. (PubMed)
- Faber MS, Fuhr U. Time response of cytochrome P450 1A2 activity on cessation of heavy smoking. Clin Pharmacol Ther. 2004;76(2):178-184. (PubMed)
- Natural Medicines. Coffee - professional handout: drug interactions. © 2017 Therapeutic Research Center 27 May 2015. Available at: http://naturaldatabase.therapeuticresearch.com/. Accessed 4/11/17.
Disclaimer
The Linus Pauling Institute's Micronutrient Information Center provides scientific information on the health aspects of dietary factors and supplements, food, and beverages for the general public. The information is made available with the understanding that the author and publisher are not providing medical, psychological, or nutritional counseling services on this site. The information should not be used in place of a consultation with a competent health care or nutrition professional.
The information on dietary factors and supplements, food, and beverages contained on this website does not cover all possible uses, actions, precautions, side effects, and interactions. It is not intended as nutritional or medical advice for individual problems. Liability for individual actions or omissions based upon the contents of this site is expressly disclaimed.
You may not copy, modify, distribute, display, transmit, perform, publish or sell any of the copyrightable material on this website. You may hyperlink to this website but must include the following statement:
"This link leads to a website provided by the Linus Pauling Institute at Oregon State University. [Your name] is not affiliated or endorsed by the Linus Pauling Institute or Oregon State University."