Summary
- Resveratrol is a polyphenolic compound naturally found in peanuts, grapes, red wine, and some berries. (More information)
- When taken orally, resveratrol is well absorbed by humans, but its bioavailability is relatively low because it is rapidly metabolized and eliminated. (More information)
- In preclinical studies, resveratrol has been shown to possess numerous biological activities, which could possibly be applied to the prevention and/or treatment of cancer, cardiovascular disease, and neurodegenerative diseases. (More information)
- Although resveratrol can inhibit the growth of cancer cells in culture and in some animal models, it is not known whether resveratrol can prevent and/or help treat cancer in humans. (More information)
- The presence of resveratrol in red wine was initially thought to be responsible for red wine’s beneficial cardiovascular effects. Two randomized, placebo-controlled trials reported that one-year consumption of a grape supplement containing 8 mg/day of resveratrol improved inflammatory and atherogenic status in subjects at risk for cardiovascular disease, as well as in patients with established coronary heart disease. Yet, there is currently no evidence that the content of resveratrol in red wine confers any additional risk reduction beyond that attributed to the alcohol content and to other wine polyphenols. (More information)
- Resveratrol administration has increased the lifespans of yeast, worms, fruit flies, fish, and mice fed a high-calorie diet, but it is not known whether resveratrol will have similar effects in humans. (More information)
- Experimental animal studies have suggested that resveratrol might be neuroprotective and be beneficial in the prevention and/or treatment of neurodegenerative diseases; however, clinical trials in healthy or cognitively impaired older people are currently very limited. (More information)
- In randomized controlled trials, short-term supplementation with resveratrol significantly improved glucose and lipid metabolic disorders in patients with type 2 diabetes. (More information)
- Long-term, high resveratrol intake might affect the pharmacokinetics of certain drugs (i.e., those metabolized by cytochrome P450 enzymes), potentially reducing their efficacy or increasing their toxicity. (More information)
Other Articles
Introduction
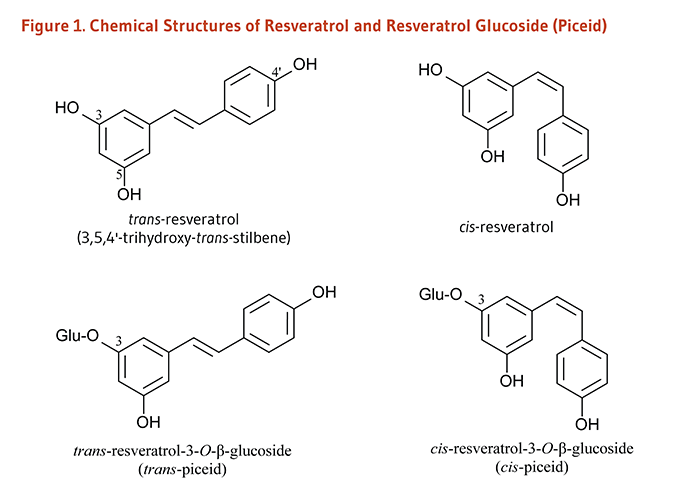
Resveratrol (3,4',5-trihydroxystilbene) belongs to a class of polyphenolic compounds called stilbenes (1). Certain plants produce resveratrol and other stilbenoids in response to stress, injury, fungal infection, or ultraviolet (UV) radiation (2). Resveratrol is a fat-soluble compound that occurs in both trans and cis molecular configurations (Figure 1). Both cis- and trans-resveratrol also occur as glucosides, i.e., bound to a glucose molecule. One major resveratrol derivative is resveratrol-3-O-β-glucoside, also called piceid (Figure 1) (3).
Since the early 1990s, when the presence of resveratrol in red wine was established (4), the scientific community has been exploring the effects of resveratrol on health. Specifically, it was postulated that resveratrol intake via moderate red wine consumption might help explain the fact that French people have a relatively low incidence of coronary heart disease (CHD) in spite of consuming foods high in saturated fat, a phenomenon dubbed the “French Paradox” (see Cardiovascular disease) (5). Since then, reports on the potential for resveratrol to prevent cancer, delay the development of cardiovascular and neurodegenerative diseases, improve glycemic control in type 2 diabetes, and extend lifespan in experimental models have continued to generate scientific interest (see Disease Prevention).
Metabolism and Bioavailability
Initial studies of the pharmacokinetics of trans-resveratrol in humans found only traces of the unmetabolized resveratrol in the plasma upon oral exposure of single trans-resveratrol doses of 5 to 25 mg. Indeed, trans-resveratrol appears to be well absorbed by humans when taken orally, but its bioavailability is relatively low due to its rapid metabolism and elimination (6). Once absorbed, resveratrol is rapidly metabolized by conjugation to glucuronic acid and/or sulfate, forming resveratrol glucuronides, sulfates, and/or sulfoglucuronides. Sulfate conjugates are the major forms of resveratrol metabolites found in plasma and urine in humans (7).
Preliminary studies found that the administration of single oral doses of 25 mg of trans-resveratrol to healthy volunteers resulted in peak blood concentrations of total resveratrol (i.e., trans-resveratrol plus its metabolites) around 60 minutes later, at about 1.8-2 μmoles/liter (μM), depending on whether resveratrol was administered in wine, vegetable juice, or grape juice (8, 9). A study in 40 healthy subjects who received single ascending doses of oral trans-resveratrol (i.e., 0.5 g, 1 g, 2.5 g, and 5 g) showed that plasma concentrations of unmetabolized resveratrol peaked between 0.8 and 1.5 hours after resveratrol administration at levels ranging from 0.3 μM to 2.3 μM (10). Of note, these values were markedly below those used to elicit chemopreventive effects of resveratrol in in vitro experiments (>5 μM). In contrast, following a single oral dose of 5 g of trans-resveratrol, the peak plasma concentrations of certain resveratrol conjugates were found to be about two to eight times higher than those of unmetabolized resveratrol (10). Also, compared to a single dose administration, the repeated intake of 5 g/day of trans-resveratrol for 29 days was found to result in significantly greater peak plasma concentrations of trans-resveratrol and two resveratrol glucuronide conjugates (11). Repeated doses of 1 g/day of trans-resveratrol (a dose less likely to cause side effects; see Safety) could yield maximum plasma concentrations of about 22 μM for resveratrol-3-O-sulfate (the most abundant sulfate conjugate in humans) and about 7-8 μM for typical monoglucuronide conjugates (12).
A few studies have examined the influence of food matrix on resveratrol absorption and/or bioavailability (reviewed in 13). One study has reported that bioavailability of trans-resveratrol from red wine did not differ when the wine was consumed with a meal (low- or high-fat) versus on an empty stomach (14). Yet, in another study, the absorption of supplemental resveratrol was found to be delayed, but not reduced, by the presence of food in the stomach (15). A third study found that the bioavailability of supplemental resveratrol was reduced by the amount of fat in the diet, but not by the co-administration of quercetin (another polyphenol) or alcohol (16).
Information about the bioavailability of resveratrol in humans is important because most of the experimental research conducted to date has been ‘preclinical,’ i.e., in vitro, exposing cells to resveratrol concentrations up to 100 times greater than peak plasma concentrations observed in humans, and in animal models given very high (non dietary) doses of resveratrol (13). While cells that line the digestive tract are exposed to unmetabolized resveratrol, other tissues are likely exposed to resveratrol metabolites. At present, little is known about the biological activity of resveratrol metabolites. Yet, if some tissues are capable of converting resveratrol metabolites back to resveratrol, stable resveratrol conjugates in tissues could serve as a pool in the body from which resveratrol might be regenerated (6, 12).
Biological Activities
The biological significance of resveratrol has been primarily investigated in test tubes and cultured cells, and to a lesser extent, in animal models. Of note, a recent publication by Tomé-Carneiro et al. (13) thoroughly reviewed the most relevant preclinical studies published in the most recent decades. It is important to keep in mind that many of the biological activities discussed below were observed in cells cultured in the presence of resveratrol at higher concentrations than those likely to be achieved in humans consuming resveratrol orally (see Metabolism and Bioavailability).
Direct antioxidant activity
In the test tube, resveratrol effectively scavenges (neutralizes) free radicals and other oxidants (17, 18) and inhibits low-density lipoprotein (LDL) oxidation (19, 20). Resveratrol was found to induce antioxidant enzymes, including superoxide dismutase (SOD), thioredoxin, glutathione peroxidase-1, heme oxygenase-1, and catalase, and/or inhibit reactive oxygen species (ROS) production by nicotinamide adenine dinucleotide phosphate (NADPH) oxidases (NOX) (21). However, there is little evidence that resveratrol is an important antioxidant in vivo. Upon oral consumption of resveratrol, circulating and intracellular levels of resveratrol in humans are likely to be much lower than that of other important antioxidants, such as vitamin C, uric acid, vitamin E, and glutathione. Moreover, the antioxidant activity of resveratrol metabolites, which comprise most of the circulating resveratrol, may be lower than that of resveratrol (22).
Estrogenic and anti-estrogenic activities
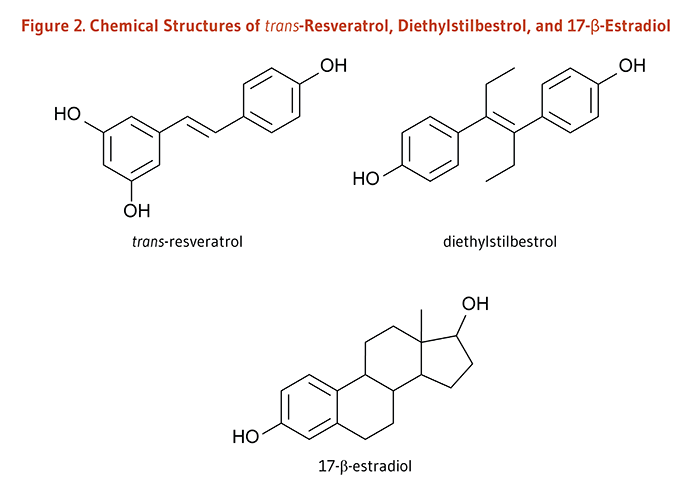
Endogenous estrogens are steroid hormones synthesized by humans and other mammals; these hormones bind to estrogen receptors within cells. The estrogen-receptor complex interacts with unique sequences in DNA (estrogen response elements; EREs) to modulate the expression of estrogen-responsive genes (23). The chemical structure of resveratrol is very similar to that of the synthetic estrogen agonist, diethylstilbestrol (Figure 2), suggesting that resveratrol might also function as an estrogen agonist, i.e., might bind to estrogen receptors and elicit similar responses to endogenous estrogens. However, in cell culture experiments, resveratrol was found to act either as an estrogen agonist or as an estrogen antagonist depending on such factors as cell type, estrogen receptor isoform (ERα or ERβ), and the presence of endogenous estrogens (23). Most recently, resveratrol was shown to improve endothelial wound healing through an ERα-dependent pathway in an animal model of arterial injury (24).
Biological activities related to cancer prevention
Effects on biotransformation enzymes
Some compounds are not carcinogenic until they have been metabolized in the body by phase I biotransformation enzymes, especially cytochrome P450 enzymes (2). By inhibiting the expression and activity of certain cytochrome P450 enzymes (25, 26), resveratrol might help prevent cancer by limiting the activation of procarcinogens. In contrast, increasing the activity of phase II detoxification enzymes generally promotes the excretion of potentially toxic or carcinogenic chemicals. Resveratrol has been found to increase the expression and activity of NAD(P)H:quinone oxidoreductase-1 (NQO1) in cultured cells (27) and may be a weak inducer of other phase II enzymes (28).
Inhibition of proliferation and induction of apoptosis
Following DNA damage, the cell cycle can be transiently arrested to allow for DNA repair or activation of pathways leading to cell death (apoptosis) if the damage is irreparable (29). Defective cell cycle regulation may result in the propagation of mutations that contribute to the development of cancer. Moreover, unlike normal cells, cancer cells proliferate rapidly and are unable to respond to cell death signals that initiate apoptosis. Resveratrol has been found to induce cell cycle arrest and/or apoptosis (programmed cell death) in a number of cancer cell lines (reviewed in 13).
Inhibition of tumor invasion and angiogenesis
Cancerous cells invade normal tissue aided by enzymes called matrix metalloproteinases. Resveratrol has been found to inhibit the activity of at least one type of matrix metalloproteinase (30, 31). To fuel their rapid growth, invasive tumors must also develop new blood vessels by a process known as angiogenesis. Resveratrol has been found to inhibit angiogenesis in vitro (32-34) and in vivo (35).
Anti-inflammatory effects
Inflammation promotes cellular proliferation and angiogenesis and inhibits apoptosis (36). Resveratrol has been found to inhibit the activity of several inflammatory enzymes in vitro, including cyclooxygenases and lipoxygenases (37, 38). Resveratrol may also inhibit pro-inflammatory transcription factors, such as NFκB or AP-1 (39, 40).
Biological activities related to cardiovascular disease prevention
Inhibition of vascular cell adhesion molecule (VCAM) expression
Atherosclerosis is an inflammatory process in which lipids deposit in plaques (known as atheromas) within arterial walls and increase the risk of myocardial infarction (41). One of the earliest events in the development of atherosclerosis is the recruitment of inflammatory white blood cells from the blood to the arterial wall by vascular cell adhesion molecules (42). Resveratrol has been found to inhibit the expression of adhesion molecules in cultured endothelial cells (43, 44).
Inhibition of vascular smooth muscle cell (VSMC) proliferation
The proliferation of vascular smooth muscle cells (VSMCs) plays an important role in the progression of hypertension, atherosclerosis, and restenosis (when treated arteries become blocked again). Resveratrol has been found to inhibit the proliferation of VSMCs in culture (45-47), as well as in vivo (48). Resveratrol appeared to reduce VSMC proliferation via an ERα-dependent mechanism, hence preventing the narrowing of vessels in a mouse model of arterial injury (48).
Stimulation of endolethelial nitric oxide synthase (eNOS) activity
Endothelial nitric oxide synthase (eNOS) is an enzyme that catalyzes the formation of nitric oxide (NO) by vascular endothelial cells. NO is needed to maintain arterial relaxation (vasodilation), and impaired NO-dependent vasodilation is associated with an increased risk of cardiovascular disease (49). Because physiological concentrations of resveratrol were found to stimulate eNOS activity in cultured endothelial cells (50-52), resveratrol might help maintain or improve endothelial function in vivo (see Cardiovascular disease).
Inhibition of platelet activiation and aggregation
Platelet aggregation is one of the first steps in the formation of a blood clot that can occlude a coronary or cerebral artery, resulting in myocardial infarction or stroke, respectively. Resveratrol has been found to inhibit platelet activation and aggregation in vitro (53-55).
Biological activities related to neurodegenerative disease prevention and treatment
Stimulation of neurogenesis and microvessel formation
Age-related mood alterations and memory deficits result from a decrease in the function of the hippocampus in the elderly. Resveratrol was shown to stimulate neurogenesis and blood vessel formation in the hippocampus of healthy old rats. These structural changes were associated with significant improvements in spatial learning, memory formation, and mood function (56).
Stimulation of β-amyloid peptide clearance
One feature of Alzheimer’s disease (AD) is the accumulation of β-amyloid peptide into senile (amyloid) plaques outside neurons in the hippocampus and cortex of AD patients (57). Senile plaques are toxic to cells, resulting in progressive neuronal dysfunction and death. Resveratrol was found to facilitate the clearance of β-amyloid peptide and promote cell survival in primary neurons in culture and neuronal cell lines (58-60). Resveratrol also reduced senile plaque counts in various brain regions of a transgenic AD mouse model (61).
Inhibition of neuroinflammation
Abnormally activated microglia and hypertrophic astrocytes around the senile plaques in AD brains release cytotoxic molecules, such as proinflammatory mediators and ROS, which enhance the formation and deposition of β-amyloid peptides and further damage neurons (57). Resveratrol was found able to inhibit the inflammatory response triggered by β-amyloid peptide-induced microglial activation in microglial cell lines and in a mouse model of cerebral amyloid deposition (62). A decreased occurrence of microglial activation and astrocyte hypertrophy was also reported in healthy aged rats treated with resveratrol (56).
Reduction of oxidative stress
Mitochondrial dysfunction and oxidative stress are thought to be involved in the etiology and/or progression of several neurodegenerative disorders (63). Resveratrol counteracted oxidative stress and β-amyloid peptide-induced toxicity in cultured neuroblastoma (64). Resistance against oxidative stress-related damage in primary neuronal cells treated with resveratrol has been associated with the induction of heme oxygenase-1 (HO-1), an enzyme that degrades pro-oxidant heme (65). In an experimental model of stroke, resveratrol limited infarct size during ischemia-reperfusion in wild-type mice but not in mice lacking the HO-1 gene (66). Also, resveratrol was able to correct experimentally induced oxidative stress and the associated cognitive dysfunction in rats (67).
Disease Prevention
Cancer
Resveratrol has been found to inhibit the proliferation of a variety of human cancer cell lines, including those from breast, prostate, stomach, colon, pancreatic, and thyroid cancers (2). In animal models, oral administration, topical application, and/or injection of resveratrol inhibited the development of chemically-induced cancer at many sites, including gastrointestinal tract, liver, skin, breast, prostate, and lung (reviewed in 68, 69). The anti-cancer effects of resveratrol in rodent models involved the reduction of cell proliferation, the induction of apoptosis, and the inhibition of angiogenesis, tumor growth, and metastasis (reviewed in 13). Yet, a few animal studies have reported a lack of an effect of oral resveratrol in inhibiting the development of lung cancer induced by carcinogens in cigarette smoke (70, 71), and the study of resveratrol administration on colon cancer has given mixed results (72-74).
At present, it is not known whether resveratrol might be beneficial in the prevention and/or the treatment of cancer in humans. The low bioavailability of resveratrol reported in human studies limits the clinical evaluation of possible systemic health effects of resveratrol in humans (see Metabolism and Bioavailability). Yet, in a pilot study, unmetabolized resveratrol and conjugates have been detected in colorectal tumor tissues from 20 cancer patients following daily oral supplementation with either 4 g or 8 g of resveratrol for 29 days. Resveratrol appeared to be well tolerated and significantly, though modestly, reduced cell proliferation compared to baseline (75). A micronized formulation of resveratrol (named SRT501), which was meant to increase resveratrol delivery to target tissues, was given for 14 days to 6 patients with colorectal cancer and liver metastasis in a small randomized, double-blind, placebo-controlled trial (76). Unmetabolized resveratrol was measurable in the liver of five out of six patients who consumed 5 g of SRT501, and SRT501 administration resulted in an increased detection of the apoptotic marker, cleaved caspase-3, in hepatic tumor tissues. Yet, in an unrandomized and unblinded trial in patients with multiple myeloma, the administration of SRT501 was associated with a number of serious adverse effects, including kidney failure, such that the trial was halted (77). Since kidney failure is a frequent complication in myeloma patients, it is unclear whether kidney failure cases should be solely attributed to the use of SRT501. Nevertheless, there is a need to find safe ways to increase resveratrol bioavailability in humans before exploring its putative benefits in clinical settings (6, 78).
Cardiovascular disease
Red wine polyphenols
Significant reductions in cardiovascular disease risk have been associated with moderate consumption of alcoholic beverages (79). The “French Paradox” — the observation that incidence of coronary heart disease was relatively low in France despite high levels of dietary saturated fat and cigarette smoking — led to the idea that regular consumption of red wine might provide additional protection from cardiovascular disease (80). Red wine contains variable and usually low concentrations of resveratrol (see Sources) and higher concentrations of flavonoids like procyanidins. These polyphenolic compounds have displayed antioxidant, anti-inflammatory, and other potentially anti-atherogenic effects in the test tube and in some animal models of atherosclerosis (81). The results of epidemiological studies addressing this question have been inconsistent. While some large prospective cohort studies found that wine drinkers were at lower risk of cardiovascular disease than beer or liquor drinkers (82-84), others found no difference (85-87). Socioeconomic and lifestyle differences between people who prefer wine and those who prefer beer or liquor may explain part of the additional benefit observed in some studies: people who prefer wine tend to have higher incomes, more education, smoke less, and eat more fruit and vegetables and less saturated fat than those who prefer other alcoholic beverages (87-92).
Although moderate alcohol consumption has been consistently associated with reductions in coronary heart disease risk, it is not yet clear whether red wine polyphenols confer any additional risk reduction. Interestingly, studies that administered alcohol-free red wine to rodents noted improvements in various parameters related to cardiovascular disease (93, 94), and a placebo-controlled human study found that heart disease patients administered red grape polyphenol extract experienced acute improvements in endothelial function (95). Whether increased consumption of polyphenols from red wine provides any additional cardiovascular benefits beyond those associated with red wine’s alcohol content needs to be further examined (see the article on Alcoholic Beverages) (96).
Resveratrol and endothelial function
Endothelial dysfunction is usually associated with the presence of cardiovascular risk factors (e.g., insulin resistance, hypertension, and hypercholesterolemia) and is thought to precede the clinical manifestation of cardiovascular and metabolic disorders. Endothelial dysfunction is characterized by abnormal vasoconstriction, leukocyte adherence to vascular endothelial cells, platelet activation and aggregation, smooth muscle cell proliferation, vascular inflammation, thrombosis (clot formation), impaired coagulation, and atherosclerosis (97).
Experimental studies
Resveratrol has been found to exert a number of protective effects on the cardiovascular system in vitro, including inhibition of both platelet activation and aggregation (53, 98, 99), promotion of vasodilation by enhancing the production of nitric oxide (NO) (52), and control of the production of inflammatory lipid mediators (38, 100, 101). However, the concentrations of resveratrol required to produce these effects are often higher than those measured in human plasma after oral consumption of resveratrol (9). Some animal studies also suggested that high oral doses of resveratrol could decrease the risk of thrombosis and atherosclerosis (102, 103), although one study found increased atherosclerosis in animals fed resveratrol (104). Other protective effects of resveratrol in vivo include the reduction of cardiac hypertrophy and the lowering of blood pressure in various models, as well as the limitation of infarct size in post myocardial infarction rats (reviewed in 13).
Randomized controlled studies
In a six-month, cross-over study, 34 patients with metabolic syndrome were randomized to receive resveratrol (100 mg/day) for three months either immediately at the beginning of the study or three months later. Resveratrol supplementation resulted in improved values of flow-mediated dilation (FMD) of the brachial artery, a surrogate marker of vascular health. Yet, FMD returned to baseline values within three months after discontinuing resveratrol (105). One study limitation was that the resveratrol formulation contained additional compounds (i.e., vitamin D3, quercetin, and rice bran phytate), which may also affect endothelial function. One randomized, placebo-controlled study in healthy overweight or obese volunteers (BMI, 25-34 kg/m2) found that a single dose of trans-resveratrol (30 mg, 90 mg, or 270 mg) improved brachial FMD around 60 minutes after administration (106). In a second study, the same investigators found that FMD improvements were similar whether participants had received a single dose of resveratrol (75 mg) or a daily dose (75 mg/day of resveratrol) for six months (107).
In a few additional studies, resveratrol was shown to improve endothelial function by reducing vascular inflammation and endothelial activation. A randomized, double-blind, placebo-controlled study in 41 healthy subjects found that daily supplementation with resveratrol (400 mg), grapeseed extract (400 mg), and quercetin (100 mg), for one month significantly reduced the expression of interleukin-8 (IL-8) and cell adhesion molecules (ICAM-1 and VCAM-1) in endothelial cells, suggestive of a protective effect against endothelial dysfunction (108). The daily intake of a resveratrol-rich grape supplement was compared to resveratrol-free grape supplement in a year-long, randomized, double blind, placebo-controlled study in 75 individuals at high risk for cardiovascular disease (CVD). Administration of the resveratrol-rich supplement (resveratrol: 8 mg/day for 6 months, then 16 mg/day for another 6 months) significantly improved the profile of circulating inflammatory markers, reducing levels of C-reactive protein (CRP) and Tumor Necrosis Factor-α (TNF-α), as well as the level of thrombogenic factor, Plasminogen Activator Inhibitor-1 (PAI-1) (109). The decreased concentrations of two CVD risk markers, oxidized low-density lipoprotein (oxLDL) and apolipoprotein B (ApoB) after six months further suggested a cardioprotective effect of resveratrol (110). Supplementation of patients with stable coronary heart disease with the same regimen also improved the profile of circulating inflammatory markers and reduced the expression of proinflammatory genes in peripheral blood mononuclear cells (PBMCs) (111). The expression of microRNAs and cytokines specifically involved in atherogenic and pro-inflammatory signals were also found to be downregulated in the PBMCs of supplemented patients (112). Finally, although it is not clear whether hypertension is a cause or an effect of endothelial dysfunction, a recent meta-analysis of randomized controlled trials suggested that high doses of resveratrol (≥150 mg/for at least one month) might help lower systolic blood pressure in individuals at risk for CVD (113).
While preliminary human studies suggest that resveratrol may have beneficial effects on cardiovascular health, there is currently no convincing evidence that these effects can be achieved in the amounts present in one to two glasses of red wine (see Sources). For more information regarding resveratrol and cardiovascular disease, see (114).
Longevity
Caloric restriction is known to extend the lifespan of a number of species, including yeast, worms, flies, fish, rats, and mice (115). In yeast (Saccharomyces cerevisiae), caloric restriction stimulates the activity of an enzyme known as Silent information regulator 2 protein (Sir2) or sirtuin (116). Yeast Sir2 is a nicotinamide adenine dinucleotide (NAD)-dependent deacetylase enzyme that removes the acetyl group from acetylated lysine residues in target proteins (see the article on Niacin).
Providing resveratrol to yeast increased Sir2 activity in the absence of caloric restriction and extended the replicative (but not the chronological) lifespan of yeast by 70% (117). Resveratrol feeding also extended the lifespan of worms (Caenorhabditis elegans) and fruit flies (Drosophila melanogaster) by a similar mechanism (118). Additionally, resveratrol dose-dependently increased the lifespan of a vertebrate fish (Nothobranchius furzeri) (119). Resveratrol was also found to extend the lifespan of mice on a high-calorie diet such that their lifespan was similar to that of mice fed a standard diet (120). Although resveratrol increased the activity of the Sir2 homologous human sirtuin 1 (SIRT1) in the test tube (117), there are no epidemiological data to link resveratrol, SIRT1 activation, and extended human lifespan. Moreover, the supraphysiological concentrations of resveratrol required to increase human SIRT1 activity were considerably higher than concentrations that have been measured in human plasma after oral consumption.
The results of a nine-year prospective cohort study in over 700 older adults (≥65 years) indicated that participants who were alive at the end of the study had baseline concentrations of total urinary resveratrol metabolites (used as a biomarker of resveratrol intake) similar to those who died during the study period (121). Based on a lack of correlation with baseline inflammatory markers, cardiovascular disease and cancer incidence, and all-cause mortality, the authors concluded that higher versus lower quartiles of urinary resveratrol metabolite concentrations did not predict risk of chronic disease or mortality. However, key experts identified several limitations regarding the quality of the research (122, 123). Specifically, the use of single measures of total urinary resveratrol metabolites at baseline has been highlighted as being unlikely to reflect lifetime consumption of wine or exposure to dietary resveratrol (122).
Cognitive decline
In a mouse model of Alzheimer’s disease (AD), caloric restriction has been shown to limit the production and deposition of neurotoxic β-amyloid peptide in the brain (124). Similar to the effect of caloric restriction, resveratrol was found to improve obesity and diabetes-related metabolic deregulations via the activation of metabolic sensors, including SIRT and the AMP-activated protein kinase (AMPK) (125), as well as to promote the AMPK-dependent clearance of β-amyloid peptide in the brain of an AD mouse model (60). Resveratrol has also exhibited additional neuroprotective properties in cultured cells and animal models (see Biological Activities).
Although resveratrol bioavailability to the brain is uncertain (78), a randomized, double-blind, placebo-controlled study has reported an increase in cerebral blood flow in the prefrontal cortex of healthy young subjects (ages, 18-29 years) following a single oral dose of 500 mg of resveratrol. However, resveratrol intake did not improve performance in cognitively demanding tasks undertaken during the post-administration period (126). More recently, the co-administration of resveratrol (200 mg/day) and quercetin (320 mg/day) for 26 weeks in a double-blind, placebo-controlled study significantly improved measures of memory function and enhanced blood glucose control in 46 healthy, overweight older adults (ages, 50-80 years; BMI, 25-30 kg/m2) (127). Additional evidence of the potential of resveratrol to mimic the metabolic benefits of caloric restriction on cognitive health may come from ongoing clinical trials in both healthy older individuals and AD patients (128).
Disease Treatment
Impaired glucose tolerance and type 2 diabetes mellitus
More than one out of three American adults has impaired glucose tolerance (also known as prediabetes), which places them at increased risk of developing type 2 diabetes (129). Impaired glucose tolerance is associated with insulin resistance in skeletal muscle — the major peripheral tissue for insulin-mediated glucose uptake — as well as defective insulin secretion by pancreatic β-cells. Muscle insulin resistance, which is thought to be the earliest stage in the development of type 2 diabetes, is characterized by excess lipid exposure, impaired insulin receptor signaling, impaired glucose uptake, mitochondrial dysfunction, reduced fatty acid oxidation, and increased expression of pro-inflammatory cytokines.
In animal studies, resveratrol has been shown to improve insulin sensitivity, glucose tolerance, and lipid profiles in obese and/or metabolically abnormal animals (reviewed in 130).
In humans, short-term supplementation with resveratrol has been associated with beneficial effects on glucose and lipid metabolism in individuals with type 2 diabetes. In a randomized, double-blind, placebo-controlled study, the effect of oral resveratrol supplementation (1,000 mg/day for 45 days) on the control of glucose metabolism was assessed in 70 subjects with type 2 diabetes (131). Comparison of changes between baseline and end-of-study measures between placebo and intervention groups showed that resveratrol significantly lowered both fasting glucose and fasting insulin concentrations and improved measures of glycemic control (HbA1c level) and insulin sensitivity (HOMA-IR). In addition, the level of HDL-cholesterol was increased while the level of LDL-cholesterol and systolic blood pressure were significantly reduced. No changes were found in measures of diastolic blood pressure, total cholesterol, triglycerides, and markers of liver function (131). Additionally, in a randomized, open-label, and controlled study, the effect of oral resveratrol (250 mg/day) on glycemic control and lipid metabolism was assessed in 62 type 2 diabetics (132). During the three-month study period, changes in biochemical and clinical parameters, including fasting glucose concentration, HbA1c level, systolic and diastolic blood pressure, total cholesterol, and LDL-cholesterol, were significantly improved with resveratrol compared to control (i.e., no resveratrol). Doses as low as 10 mg/day of resveratrol also resulted in lower insulin resistance in a four-week, randomized, placebo-controlled study in 19 male subjects with type 2 diabetes (133).
Obesity (defined as a body mass index [BMI] ≥ 30 kg/m2) is a well-known risk factor for the development of type 2 diabetes. A few clinical studies have evaluated the effects of resveratrol on key metabolic variables in overweight or obese subjects with no overt metabolic dysfunction and found little or no metabolic benefits following resveratrol treatment (134-136). Yet, at present, there is no available evidence to suggest whether overweight or obese individuals with impaired glucose tolerance could benefit from resveratrol supplements and reduce their risk of developing type 2 diabetes (137).
Current data suggest that resveratrol could improve specific metabolic variables in individuals with type 2 diabetes (138, 139), but more research is needed to assess its effect in individuals at risk for diabetes, including obese subjects with impaired glucose tolerance.
Sources
Food sources
Resveratrol is found in grapes, wine, grape juice, peanuts, cocoa, and berries of Vaccinium species, including blueberries, bilberries, and cranberries (140-143). In grapes, resveratrol is found only in the skins (144). The amount of resveratrol in grape skins varies with the grape cultivar, its geographic origin, and exposure to fungal infection (145). The amount of fermentation time a wine spends in contact with grape skins is also an important determinant of its resveratrol content. Because grape skins are removed early during the production process of white and rosé wines, these wines generally contain less resveratrol than red wines (4). Therefore, because of variations between types of wine, vintages, and regions, it is very difficult to provide accurate estimates of resveratrol content in the thousands of wines from worldwide wineries. Yet, it appears that resveratrol content in wine is usually low, highly variable and unpredictable, and resveratrol is only a minor compound in the complete set of grape and wine polyphenols (13).
The predominant form of resveratrol in grapes and grape juice is trans-resveratrol-3-O-β-glucoside (trans-piceid), and wines contain significant amounts of resveratrol aglycones, thought to be the result of sugar cleavage during fermentation (3, 140). Many wines also contain significant amounts of cis-resveratrol (see Figure 1 above), which may be produced during fermentation or released from viniferins (resveratrol polymers) (146). Red wine is a relatively rich source of resveratrol, but other polyphenols are present in red wine at considerably higher concentrations than resveratrol (see the article on Flavonoids) (147). Estimates of resveratrol content of some beverages and foods are listed in Table 1 and Table 2. These values should be considered approximate since the resveratrol content of foods and beverages can vary considerably.
| Variety | Lowest (mg/L) | Highest (mg/L) | Mean (mg/L) | 5-oz Glass (mg) |
|---|---|---|---|---|
| Pinot Noir | 0.2 | 11.9 | 3.6 ± 2.9 | 0.5 |
| Merlot | 0.3 | 14.3 | 2.8 ± 2.6 | 0.4 |
| Zweigelt | 0.6 | 4.7 | 1.9 ± 1.2 | 0.3 |
| Shiraz | 0.2 | 3.2 | 1.8 ± 0.9 | 0.3 |
| Cabernet Sauvignon | - | 9.3 | 1.7 ± 1.7 | 0.2 |
| Red wines (global) | - | 14.3 | 1.9 ± 1.7 | 0.3 |
| Food | Serving | Total Resveratrol (mg) |
|---|---|---|
| Peanuts (raw) | 1 cup (146 g) | 0.01-0.26 |
| Peanuts (boiled) | 1 cup (180 g) | 0.32-1.28 |
| Peanut butter | 1 cup (258 g) | 0.04-0.13 |
| Red grapes | 1 cup (160 g) | 0.24-1.25 |
Supplements
Most resveratrol supplements available in the US contain extracts of the root of Polygonum cuspidatum, also known as Fallopia japonica, Japanese knotweed, or Hu Zhang (150). Red wine extracts and grape extracts (from Vitis vinifera) containing resveratrol and other polyphenols are also available as dietary supplements. Resveratrol supplements may contain anywhere from less than 1 milligram (mg) to 500 mg of resveratrol per tablet or capsule, but it is not known whether there is a safe and effective dosage for chronic disease prevention in humans (also see the section on Safety).
Safety
Adverse effects
In rats, daily oral administration of trans-resveratrol at doses up to 700 mg/kg of body weight for 90 days resulted in no apparent adverse effects (151). Other toxicity studies conducted in animal models estimated that the no-observed-adverse-effect-level (NOAEL) for resveratrol was 200 mg/kg/days and 600 mg/kg/day in rats and dogs, respectively (152). Resveratrol is not known to be toxic or cause significant adverse effects in humans, but there have been only a few controlled clinical trials to date (reviewed in 153). A trial evaluating the safety of oral trans-resveratrol in 10 subjects found that a single dose of 5,000 mg resulted in no serious adverse effects (10). In a follow-up study, mild-to-moderate gastrointestinal side effects, including nausea, abdominal pain, flatulence, and diarrhea, have been reported in participants who consume more than 1,000 mg/day of resveratrol for up to 29 consecutive days (11). Mild diarrhea was also reported in six out of eight individuals who consumed 2,000 mg of resveratrol twice daily for two periods of eight days in an open-label and within subject-control study (16).
Pregnancy and lactation
The safety of resveratrol-containing supplements during pregnancy and lactation has not been established (150). Because there is no known safe amount of alcohol consumption at any stage of pregnancy (154), pregnant women should avoid consuming wine as a source of resveratrol.
Estrogen-sensitive conditions
Until more is known about the estrogenic activity of resveratrol in humans, women with a history of estrogen-sensitive cancers, such as breast, ovarian, and uterine cancers, should avoid resveratrol supplements (see Estrogenic and anti-estrogenic activities) (150).
Drug interactions
Anticoagulant and antiplatelet drugs
Resveratrol has been found to inhibit human platelet aggregation in vitro (53, 155). Theoretically, high intakes of resveratrol (i.e., from supplements) could increase the risk of bruising and bleeding when taken with anticoagulant drugs, such as warfarin (Coumadin) and heparin; antiplatelet drugs, such as clopidogrel (Plavix) and dipyridamole (Persantine); and non-steroidal anti-inflammatory drugs (NSAIDs), including aspirin, ibuprofen, diclofenac, naproxen, and others.
Drugs metabolized by cytochrome P450
Cytochrome P450 (CYP) enzymes are phase I biotransformation enzymes involved in the metabolism of a broad range of compounds, from endogenous molecules to therapeutic agents. The most abundant CYP isoform in the human liver and intestines is cytochrome P450 3A4 (CYP3A4), which catalyzes the metabolism of about half of all marketed drugs in the US (156). Resveratrol has been reported to inhibit CYP3A4 activity in vitro (157, 158) and in healthy volunteers (28). Therefore, high intakes of resveratrol (i.e., from supplements) could potentially reduce the metabolic clearance of drugs that undergo extensive first-pass metabolism by CYP3A4, hence increasing the bioavailability and risk of toxicity of these drugs. Some of the many drugs metabolized by CYP3A4 include HMG-CoA reductase inhibitors (statins), calcium channel antagonists (felodipine, nicardipine, nifedipine, nisoldipine, nitrendipine, nimodipine, and verapamil), anti-arrhythmic agents (amiodarone), HIV protease inhibitors (saquinavir), immunosuppressants (cyclosporine and tacrolimus), antihistamines (terfenadine), benzodiazepines (midazolam and triazolam), and drugs used to treat erectile dysfunction (sildenafil). Of note, a recently completed clinical trial (NCT01173640) examined the potential for single and multiple doses of resveratrol (1,000 mg) to interfere with the metabolism of midazolam in healthy volunteers, and results are soon to be published (153). Other CYP enzymes (e.g., CYP2D6 and CYP2C9) may also be inhibited by resveratrol (reviewed in 159).
Finally, resveratrol was found to be a weak inducer of the expression and activity of CYP1A2, which catalyzes the metabolism of several drugs, including acetaminophen (paracetamol) and the antidepressant drugs, clomipramine and imipramine (28, 156). This suggests that resveratrol may interfere with CYP1A2-mediated drug metabolism by increasing drug clearance, possibly lowering circulating drug concentrations below therapeutic levels.
Authors and Reviewers
Originally written in 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in May 2008 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in May 2015 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in May 2015 by:
Juan Carlos Espín, Ph.D.
Research Professor
Consejo Superior de Investigaciones Científicas (CSIC)
Department of Food Science & Technology
Murcia, Spain
Last updated 6/11/15 Copyright 2005-2026 Linus Pauling Institute
Figure Alternative Text
Figure 1. Chemical structures of resveratrol and resveratrol glucoside (piceid)
The figure shows the chemical structures of trans-resveratrol (3,5,4’-trihydroxy-trans-stilbene), cis-resveratrol, trans-resveratrol-3-O-β-glucoside (trans-piceid), and cis-resveratrol-3-O-β-glucoside (cis-piceid).
Figure 2. Chemical structures of trans-resveratrol, diethylstilbestrol, and 17-β-estradiol
The figure shows the chemical structures of trans-resveratrol, diethylstilbestrol, and 17-β-estradiol.
References
- Soleas GJ, Diamandis EP, Goldberg DM. Resveratrol: a molecule whose time has come? And gone? Clin Biochem. 1997;30(2):91-113. (PubMed)
- Aggarwal BB, Bhardwaj A, Aggarwal RS, Seeram NP, Shishodia S, Takada Y. Role of resveratrol in prevention and therapy of cancer: preclinical and clinical studies. Anticancer Res. 2004;24(5A):2783-2840. (PubMed)
- Romero-Perez AI, Ibern-Gomez M, Lamuela-Raventos RM, de La Torre-Boronat MC. Piceid, the major resveratrol derivative in grape juices. J Agric Food Chem. 1999;47(4):1533-1536. (PubMed)
- Siemann EH, Creasey LL. Concentration of the phytoalexin resveratrol in wine. Am J Enol Vitic. 1992;43(1):49-52.
- Renaud S, de Lorgeril M. Wine, alcohol, platelets, and the French paradox for coronary heart disease. Lancet. 1992;339(8808):1523-1526. (PubMed)
- Walle T. Bioavailability of resveratrol. Ann N Y Acad Sci. 2011;1215:9-15. (PubMed)
- Burkon A, Somoza V. Quantification of free and protein-bound trans-resveratrol metabolites and identification of trans-resveratrol-C/O-conjugated diglucuronides - two novel resveratrol metabolites in human plasma. Mol Nutr Food Res. 2008;52(5):549-557. (PubMed)
- Goldberg DM, Yan J, Soleas GJ. Absorption of three wine-related polyphenols in three different matrices by healthy subjects. Clin Biochem. 2003;36(1):79-87. (PubMed)
- Walle T, Hsieh F, Delegge MH, Oatis JE, Jr., Walle UK. High absorption but very low bioavailability of oral resveratrol in humans. Drug Metab Dispos. 2004;32(12):1377-1382. (PubMed)
- Boocock DJ, Faust GE, Patel KR, et al. Phase I dose escalation pharmacokinetic study in healthy volunteers of resveratrol, a potential cancer chemopreventive agent. Cancer Epidemiol Biomarkers Prev. 2007;16(6):1246-1252. (PubMed)
- Brown VA, Patel KR, Viskaduraki M, et al. Repeat dose study of the cancer chemopreventive agent resveratrol in healthy volunteers: safety, pharmacokinetics, and effect on the insulin-like growth factor axis. Cancer Res. 2010;70(22):9003-9011. (PubMed)
- Patel KR, Andreadi C, Britton RG, et al. Sulfate metabolites provide an intracellular pool for resveratrol generation and induce autophagy with senescence. Sci Transl Med. 2013;5(205):205ra133. (PubMed)
- Tome-Carneiro J, Larrosa M, Gonzalez-Sarrias A, Tomas-Barberan FA, Garcia-Conesa MT, Espin JC. Resveratrol and clinical trials: the crossroad from in vitro studies to human evidence. Curr Pharm Des. 2013;19(34):6064-6093. (PubMed)
- Vitaglione P, Sforza S, Galaverna G, et al. Bioavailability of trans-resveratrol from red wine in humans. Mol Nutr Food Res. 2005;49(5):495-504. (PubMed)
- Vaz-da-Silva M, Loureiro AI, Falcao A, et al. Effect of food on the pharmacokinetic profile of trans-resveratrol. Int J Clin Pharmacol Ther. 2008;46(11):564-570. (PubMed)
- la Porte C, Voduc N, Zhang G, et al. Steady-State pharmacokinetics and tolerability of trans-resveratrol 2000 mg twice daily with food, quercetin and alcohol (ethanol) in healthy human subjects. Clin Pharmacokinet. 2010;49(7):449-454. (PubMed)
- Leonard SS, Xia C, Jiang BH, et al. Resveratrol scavenges reactive oxygen species and effects radical-induced cellular responses. Biochem Biophys Res Commun. 2003;309(4):1017-1026. (PubMed)
- Vlachogianni IC, Fragopoulou E, Kostakis IK, Antonopoulou S. In vitro assessment of antioxidant activity of tyrosol, resveratrol and their acetylated derivatives. Food Chem. 2015;177:165-173. (PubMed)
- Brito P, Almeida LM, Dinis TC. The interaction of resveratrol with ferrylmyoglobin and peroxynitrite; protection against LDL oxidation. Free Radic Res. 2002;36(6):621-631. (PubMed)
- Frankel EN, Waterhouse AL, Kinsella JE. Inhibition of human LDL oxidation by resveratrol. Lancet. 1993;341(8852):1103-1104. (PubMed)
- Wang H, Yang YJ, Qian HY, Zhang Q, Xu H, Li JJ. Resveratrol in cardiovascular disease: what is known from current research? Heart Fail Rev. 2012;17(3):437-448. (PubMed)
- Bradamante S, Barenghi L, Villa A. Cardiovascular protective effects of resveratrol. Cardiovasc Drug Rev. 2004;22(3):169-188. (PubMed)
- Tangkeangsirisin W, Serrero G. Resveratrol in the chemoprevention and chemotherapy of breast cancer. In: Bagchi D, Preuss HG, eds. Phytopharmaceuticals in Cancer Chemoprevention. Boca Raton: CRC Press; 2005:449-463.
- Yurdagul A, Jr., Kleinedler JJ, McInnis MC, et al. Resveratrol promotes endothelial cell wound healing under laminar shear stress through an estrogen receptor-alpha-dependent pathway. Am J Physiol Heart Circ Physiol. 2014;306(6):H797-806. (PubMed)
- Chen ZH, Hurh YJ, Na HK, et al. Resveratrol inhibits TCDD-induced expression of CYP1A1 and CYP1B1 and catechol estrogen-mediated oxidative DNA damage in cultured human mammary epithelial cells. Carcinogenesis. 2004;25(10):2005-2013. (PubMed)
- Ciolino HP, Yeh GC. Inhibition of aryl hydrocarbon-induced cytochrome P-450 1A1 enzyme activity and CYP1A1 expression by resveratrol. Mol Pharmacol. 1999;56(4):760-767. (PubMed)
- Hsieh TC, Lu X, Wang Z, Wu JM. Induction of quinone reductase NQO1 by resveratrol in human K562 cells involves the antioxidant response element ARE and is accompanied by nuclear translocation of transcription factor Nrf2. Med Chem. 2006;2(3):275-285. (PubMed)
- Chow HH, Garland LL, Hsu CH, et al. Resveratrol modulates drug- and carcinogen-metabolizing enzymes in a healthy volunteer study. Cancer Prev Res (Phila). 2010;3(9):1168-1175. (PubMed)
- Stewart ZA, Westfall MD, Pietenpol JA. Cell-cycle dysregulation and anticancer therapy. Trends Pharmacol Sci. 2003;24(3):139-145. (PubMed)
- Woo JH, Lim JH, Kim YH, et al. Resveratrol inhibits phorbol myristate acetate-induced matrix metalloproteinase-9 expression by inhibiting JNK and PKC delta signal transduction. Oncogene. 2004;23(10):1845-1853. (PubMed)
- Yu H, Pan C, Zhao S, Wang Z, Zhang H, Wu W. Resveratrol inhibits tumor necrosis factor-alpha-mediated matrix metalloproteinase-9 expression and invasion of human hepatocellular carcinoma cells. Biomed Pharmacother. 2008;62(6):366-372. (PubMed)
- Igura K, Ohta T, Kuroda Y, Kaji K. Resveratrol and quercetin inhibit angiogenesis in vitro. Cancer Lett. 2001;171(1):11-16. (PubMed)
- Lin MT, Yen ML, Lin CY, Kuo ML. Inhibition of vascular endothelial growth factor-induced angiogenesis by resveratrol through interruption of Src-dependent vascular endothelial cadherin tyrosine phosphorylation. Mol Pharmacol. 2003;64(5):1029-1036. (PubMed)
- Chen Y, Tseng SH. Review. Pro- and anti-angiogenesis effects of resveratrol. In Vivo. 2007;21(2):365-370. (PubMed)
- Kanavi MR, Darjatmoko S, Wang S, et al. The sustained delivery of resveratrol or a defined grape powder inhibits new blood vessel formation in a mouse model of choroidal neovascularization. Molecules. 2014;19(11):17578-17603. (PubMed)
- Steele VE, Hawk ET, Viner JL, Lubet RA. Mechanisms and applications of non-steroidal anti-inflammatory drugs in the chemoprevention of cancer. Mutat Res. 2003;523-524:137-144. (PubMed)
- Donnelly LE, Newton R, Kennedy GE, et al. Anti-inflammatory effects of resveratrol in lung epithelial cells: molecular mechanisms. Am J Physiol Lung Cell Mol Physiol. 2004;287(4):L774-783. (PubMed)
- Pinto MC, Garcia-Barrado JA, Macias P. Resveratrol is a potent inhibitor of the dioxygenase activity of lipoxygenase. J Agric Food Chem. 1999;47(12):4842-4846. (PubMed)
- Shankar S, Singh G, Srivastava RK. Chemoprevention by resveratrol: molecular mechanisms and therapeutic potential. Front Biosci. 2007;12:4839-4854. (PubMed)
- de la Lastra CA, Villegas I. Resveratrol as an anti-inflammatory and anti-aging agent: mechanisms and clinical implications. Mol Nutr Food Res. 2005;49(5):405-430. (PubMed)
- Hartman J, Frishman WH. Inflammation and atherosclerosis: a review of the role of interleukin-6 in the development of atherosclerosis and the potential for targeted drug therapy. Cardiol Rev. 2014;22(3):147-151. (PubMed)
- Stocker R, Keaney JF, Jr. Role of oxidative modifications in atherosclerosis. Physiol Rev. 2004;84(4):1381-1478. (PubMed)
- Carluccio MA, Siculella L, Ancora MA, et al. Olive oil and red wine antioxidant polyphenols inhibit endothelial activation: antiatherogenic properties of Mediterranean diet phytochemicals. Arterioscler Thromb Vasc Biol. 2003;23(4):622-629. (PubMed)
- Ferrero ME, Bertelli AE, Fulgenzi A, et al. Activity in vitro of resveratrol on granulocyte and monocyte adhesion to endothelium. Am J Clin Nutr. 1998;68(6):1208-1214. (PubMed)
- Ekshyyan VP, Hebert VY, Khandelwal A, Dugas TR. Resveratrol inhibits rat aortic vascular smooth muscle cell proliferation via estrogen receptor dependent nitric oxide production. J Cardiovasc Pharmacol. 2007;50(1):83-93. (PubMed)
- Haider UG, Sorescu D, Griendling KK, Vollmar AM, Dirsch VM. Resveratrol increases serine15-phosphorylated but transcriptionally impaired p53 and induces a reversible DNA replication block in serum-activated vascular smooth muscle cells. Mol Pharmacol. 2003;63(4):925-932. (PubMed)
- Mnjoyan ZH, Fujise K. Profound negative regulatory effects by resveratrol on vascular smooth muscle cells: a role of p53-p21(WAF1/CIP1) pathway. Biochem Biophys Res Commun. 2003;311(2):546-552. (PubMed)
- Khandelwal AR, Hebert VY, Dugas TR. Essential role of ER-alpha-dependent NO production in resveratrol-mediated inhibition of restenosis. Am J Physiol Heart Circ Physiol. 2010;299(5):H1451-1458. (PubMed)
- Duffy SJ, Vita JA. Effects of phenolics on vascular endothelial function. Curr Opin Lipidol. 2003;14(1):21-27. (PubMed)
- Klinge CM, Blankenship KA, Risinger KE, et al. Resveratrol and estradiol rapidly activate MAPK signaling through estrogen receptors alpha and beta in endothelial cells. J Biol Chem. 2005;280(9):7460-7468. (PubMed)
- Klinge CM, Wickramasinghe NS, Ivanova MM, Dougherty SM. Resveratrol stimulates nitric oxide production by increasing estrogen receptor alpha-Src-caveolin-1 interaction and phosphorylation in human umbilical vein endothelial cells. FASEB J. 2008;22(7):2185-2197. (PubMed)
- Takahashi S, Nakashima Y. Repeated and long-term treatment with physiological concentrations of resveratrol promotes NO production in vascular endothelial cells. Br J Nutr. 2012;107(6):774-780. (PubMed)
- Pace-Asciak CR, Hahn S, Diamandis EP, Soleas G, Goldberg DM. The red wine phenolics trans-resveratrol and quercetin block human platelet aggregation and eicosanoid synthesis: implications for protection against coronary heart disease. Clin Chim Acta. 1995;235(2):207-219. (PubMed)
- Shen MY, Hsiao G, Liu CL, et al. Inhibitory mechanisms of resveratrol in platelet activation: pivotal roles of p38 MAPK and NO/cyclic GMP. Br J Haematol. 2007;139(3):475-485. (PubMed)
- Yang YM, Chen JZ, Wang XX, Wang SJ, Hu H, Wang HQ. Resveratrol attenuates thromboxane A2 receptor agonist-induced platelet activation by reducing phospholipase C activity. Eur J Pharmacol. 2008;583(1):148-155. (PubMed)
- Kodali M, Parihar VK, Hattiangady B, Mishra V, Shuai B, Shetty AK. Resveratrol prevents age-related memory and mood dysfunction with increased hippocampal neurogenesis and microvasculature, and reduced glial activation. Sci Rep. 2015;5:8075. (PubMed)
- Ma T, Tan MS, Yu JT, Tan L. Resveratrol as a therapeutic agent for Alzheimer's disease. Biomed Res Int. 2014;2014:350516. (PubMed)
- Chen J, Zhou Y, Mueller-Steiner S, et al. SIRT1 protects against microglia-dependent amyloid-beta toxicity through inhibiting NF-kappaB signaling. J Biol Chem. 2005;280(48):40364-40374. (PubMed)
- Marambaud P, Zhao H, Davies P. Resveratrol promotes clearance of Alzheimer's disease amyloid-beta peptides. J Biol Chem. 2005;280(45):37377-37382. (PubMed)
- Vingtdeux V, Giliberto L, Zhao H, et al. AMP-activated protein kinase signaling activation by resveratrol modulates amyloid-beta peptide metabolism. J Biol Chem. 2010;285(12):9100-9113. (PubMed)
- Karuppagounder SS, Pinto JT, Xu H, Chen HL, Beal MF, Gibson GE. Dietary supplementation with resveratrol reduces plaque pathology in a transgenic model of Alzheimer's disease. Neurochem Int. 2009;54(2):111-118. (PubMed)
- Capiralla H, Vingtdeux V, Zhao H, et al. Resveratrol mitigates lipopolysaccharide- and Abeta-mediated microglial inflammation by inhibiting the TLR4/NF-kappaB/STAT signaling cascade. J Neurochem. 2012;120(3):461-472. (PubMed)
- Ruszkiewicz J, Albrecht J. Changes in the mitochondrial antioxidant systems in neurodegenerative diseases and acute brain disorders. Neurochem Int. 2015; doi: 10.1016/j.neuint.2014.12.012. [Epub ahead of print]. (PubMed)
- Albani D, Polito L, Batelli S, et al. The SIRT1 activator resveratrol protects SK-N-BE cells from oxidative stress and against toxicity caused by alpha-synuclein or amyloid-beta (1-42) peptide. J Neurochem. 2009;110(5):1445-1456. (PubMed)
- Zhuang H, Kim YS, Koehler RC, Dore S. Potential mechanism by which resveratrol, a red wine constituent, protects neurons. Ann N Y Acad Sci. 2003;993:276-286; discussion 287-278. (PubMed)
- Sakata Y, Zhuang H, Kwansa H, Koehler RC, Dore S. Resveratrol protects against experimental stroke: putative neuroprotective role of heme oxygenase 1. Exp Neurol. 2010;224(1):325-329. (PubMed)
- Kumar A, Naidu PS, Seghal N, Padi SS. Neuroprotective effects of resveratrol against intracerebroventricular colchicine-induced cognitive impairment and oxidative stress in rats. Pharmacology. 2007;79(1):17-26. (PubMed)
- Bishayee A. Cancer prevention and treatment with resveratrol: from rodent studies to clinical trials. Cancer Prev Res (Phila). 2009;2(5):409-418. (PubMed)
- Bishayee A, Darvesh AS, Politis T, McGory R. Resveratrol and liver disease: from bench to bedside and community. Liver Int. 2010;30(8):1103-1114. (PubMed)
- Hecht SS, Kenney PM, Wang M, et al. Evaluation of butylated hydroxyanisole, myo-inositol, curcumin, esculetin, resveratrol and lycopene as inhibitors of benzo[a]pyrene plus 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone-induced lung tumorigenesis in A/J mice. Cancer Lett. 1999;137(2):123-130. (PubMed)
- Berge G, Ovrebo S, Eilertsen E, Haugen A, Mollerup S. Analysis of resveratrol as a lung cancer chemopreventive agent in A/J mice exposed to benzo[a]pyrene. Br J Cancer. 2004;91(7):1380-1383. (PubMed)
- Schneider Y, Duranton B, Gosse F, Schleiffer R, Seiler N, Raul F. Resveratrol inhibits intestinal tumorigenesis and modulates host-defense-related gene expression in an animal model of human familial adenomatous polyposis. Nutr Cancer. 2001;39(1):102-107. (PubMed)
- Sengottuvelan M, Nalini N. Dietary supplementation of resveratrol suppresses colonic tumour incidence in 1,2-dimethylhydrazine-treated rats by modulating biotransforming enzymes and aberrant crypt foci development. Br J Nutr. 2006;96(1):145-153. (PubMed)
- Ziegler CC, Rainwater L, Whelan J, McEntee MF. Dietary resveratrol does not affect intestinal tumorigenesis in Apc(Min/+) mice. J Nutr. 2004;134(1):5-10. (PubMed)
- Patel KR, Brown VA, Jones DJ, et al. Clinical pharmacology of resveratrol and its metabolites in colorectal cancer patients. Cancer Res. 2010;70(19):7392-7399. (PubMed)
- Howells LM, Berry DP, Elliott PJ, et al. Phase I randomized, double-blind pilot study of micronized resveratrol (SRT501) in patients with hepatic metastases--safety, pharmacokinetics, and pharmacodynamics. Cancer Prev Res (Phila). 2011;4(9):1419-1425. (PubMed)
- Popat R, Plesner T, Davies F, et al. A phase 2 study of SRT501 (resveratrol) with bortezomib for patients with relapsed and or refractory multiple myeloma. Br J Haematol. 2013;160(5):714-717. (PubMed)
- Smoliga JM, Blanchard O. Enhancing the delivery of resveratrol in humans: if low bioavailability is the problem, what is the solution? Molecules. 2014;19(11):17154-17172. (PubMed)
- Ronksley PE, Brien SE, Turner BJ, Mukamal KJ, Ghali WA. Association of alcohol consumption with selected cardiovascular disease outcomes: a systematic review and meta-analysis. BMJ. 2011;342:d671. (PubMed)
- Lippi G, Franchini M, Favaloro EJ, Targher G. Moderate red wine consumption and cardiovascular disease risk: beyond the "French paradox". Semin Thromb Hemost. 2010;36(1):59-70. (PubMed)
- Salvamani S, Gunasekaran B, Shaharuddin NA, Ahmad SA, Shukor MY. Antiartherosclerotic effects of plant flavonoids. Biomed Res Int. 2014;2014:480258. (PubMed)
- Gronbaek M, Becker U, Johansen D, et al. Type of alcohol consumed and mortality from all causes, coronary heart disease, and cancer. Ann Intern Med. 2000;133(6):411-419. (PubMed)
- Klatsky AL, Friedman GD, Armstrong MA, Kipp H. Wine, liquor, beer, and mortality. Am J Epidemiol. 2003;158(6):585-595. (PubMed)
- Renaud SC, Gueguen R, Siest G, Salamon R. Wine, beer, and mortality in middle-aged men from eastern France. Arch Intern Med. 1999;159(16):1865-1870. (PubMed)
- Mukamal KJ, Conigrave KM, Mittleman MA, et al. Roles of drinking pattern and type of alcohol consumed in coronary heart disease in men. N Engl J Med. 2003;348(2):109-118. (PubMed)
- Rimm EB, Klatsky A, Grobbee D, Stampfer MJ. Review of moderate alcohol consumption and reduced risk of coronary heart disease: is the effect due to beer, wine, or spirits. Bmj. 1996;312(7033):731-736. (PubMed)
- Wannamethee SG, Shaper AG. Type of alcoholic drink and risk of major coronary heart disease events and all-cause mortality. Am J Public Health. 1999;89(5):685-690. (PubMed)
- Barefoot JC, Gronbaek M, Feaganes JR, McPherson RS, Williams RB, Siegler IC. Alcoholic beverage preference, diet, and health habits in the UNC Alumni Heart Study. Am J Clin Nutr. 2002;76(2):466-472. (PubMed)
- McCann SE, Sempos C, Freudenheim JL, et al. Alcoholic beverage preference and characteristics of drinkers and nondrinkers in western New York (United States). Nutr Metab Cardiovasc Dis. 2003;13(1):2-11. (PubMed)
- Mortensen EL, Jensen HH, Sanders SA, Reinisch JM. Better psychological functioning and higher social status may largely explain the apparent health benefits of wine: a study of wine and beer drinking in young Danish adults. Arch Intern Med. 2001;161(15):1844-1848. (PubMed)
- Johansen D, Friis K, Skovenborg E, Gronbaek M. Food buying habits of people who buy wine or beer: cross sectional study. Bmj. 2006;332(7540):519-522. (PubMed)
- Ruidavets JB, Bataille V, Dallongeville J, et al. Alcohol intake and diet in France, the prominent role of lifestyle. Eur Heart J. 2004;25(13):1153-1162. (PubMed)
- Stocker R, O'Halloran RA. Dealcoholized red wine decreases atherosclerosis in apolipoprotein E gene-deficient mice independently of inhibition of lipid peroxidation in the artery wall. Am J Clin Nutr. 2004;79(1):123-130. (PubMed)
- De Curtis A, Murzilli S, Di Castelnuovo A, et al. Alcohol-free red wine prevents arterial thrombosis in dietary-induced hypercholesterolemic rats: experimental support for the 'French paradox'. J Thromb Haemost. 2005;3(2):346-350. (PubMed)
- Lekakis J, Rallidis LS, Andreadou I, et al. Polyphenolic compounds from red grapes acutely improve endothelial function in patients with coronary heart disease. Eur J Cardiovasc Prev Rehabil. 2005;12(6):596-600. (PubMed)
- Karatzi K, Karatzis E, Papamichael C, Lekakis J, Zampelas A. Effects of red wine on endothelial function: postprandial studies vs clinical trials. Nutr Metab Cardiovasc Dis. 2009;19(10):744-750. (PubMed)
- Grover-Paez F, Zavalza-Gomez AB. Endothelial dysfunction and cardiovascular risk factors. Diabetes Res Clin Pract. 2009;84(1):1-10. (PubMed)
- Wang Z, Huang Y, Zou J, Cao K, Xu Y, Wu JM. Effects of red wine and wine polyphenol resveratrol on platelet aggregation in vivo and in vitro. Int J Mol Med. 2002;9(1):77-79. (PubMed)
- Kirk RI, Deitch JA, Wu JM, Lerea KM. Resveratrol decreases early signaling events in washed platelets but has little effect on platalet in whole blood. Blood Cells Mol Dis. 2000;26(2):144-150. (PubMed)
- Szewczuk LM, Forti L, Stivala LA, Penning TM. Resveratrol is a peroxidase-mediated inactivator of COX-1 but not COX-2: a mechanistic approach to the design of COX-1 selective agents. J Biol Chem. 2004;279(21):22727-22737. (PubMed)
- Tsai SH, Lin-Shiau SY, Lin JK. Suppression of nitric oxide synthase and the down-regulation of the activation of NFkappaB in macrophages by resveratrol. Br J Pharmacol. 1999;126(3):673-680. (PubMed)
- Fukao H, Ijiri Y, Miura M, et al. Effect of trans-resveratrol on the thrombogenicity and atherogenicity in apolipoprotein E-deficient and low-density lipoprotein receptor-deficient mice. Blood Coagul Fibrinolysis. 2004;15(6):441-446. (PubMed)
- Wang Z, Zou J, Huang Y, Cao K, Xu Y, Wu JM. Effect of resveratrol on platelet aggregation in vivo and in vitro. Chin Med J (Engl). 2002;115(3):378-380. (PubMed)
- Wilson T, Knight TJ, Beitz DC, Lewis DS, Engen RL. Resveratrol promotes atherosclerosis in hypercholesterolemic rabbits. Life Sci. 1996;59(1):PL15-21. (PubMed)
- Fujitaka K, Otani H, Jo F, et al. Modified resveratrol Longevinex improves endothelial function in adults with metabolic syndrome receiving standard treatment. Nutr Res. 2011;31(11):842-847. (PubMed)
- Wong RH, Howe PR, Buckley JD, Coates AM, Kunz I, Berry NM. Acute resveratrol supplementation improves flow-mediated dilatation in overweight/obese individuals with mildly elevated blood pressure. Nutr Metab Cardiovasc Dis. 2011;21(11):851-856. (PubMed)
- Wong RH, Berry NM, Coates AM, et al. Chronic resveratrol consumption improves brachial flow-mediated dilatation in healthy obese adults. J Hypertens. 2013;31(9):1819-1827. (PubMed)
- Agarwal B, Campen MJ, Channell MM, et al. Resveratrol for primary prevention of atherosclerosis: clinical trial evidence for improved gene expression in vascular endothelium. Int J Cardiol. 2013;166(1):246-248. (PubMed)
- Tome-Carneiro J, Gonzalvez M, Larrosa M, et al. One-year consumption of a grape nutraceutical containing resveratrol improves the inflammatory and fibrinolytic status of patients in primary prevention of cardiovascular disease. Am J Cardiol. 2012;110(3):356-363. (PubMed)
- Tome-Carneiro J, Gonzalvez M, Larrosa M, et al. Consumption of a grape extract supplement containing resveratrol decreases oxidized LDL and ApoB in patients undergoing primary prevention of cardiovascular disease: a triple-blind, 6-month follow-up, placebo-controlled, randomized trial. Mol Nutr Food Res. 2012;56(5):810-821. (PubMed)
- Tome-Carneiro J, Gonzalvez M, Larrosa M, et al. Grape resveratrol increases serum adiponectin and downregulates inflammatory genes in peripheral blood mononuclear cells: a triple-blind, placebo-controlled, one-year clinical trial in patients with stable coronary artery disease. Cardiovasc Drugs Ther. 2013;27(1):37-48. (PubMed)
- Tome-Carneiro J, Larrosa M, Yanez-Gascon MJ, et al. One-year supplementation with a grape extract containing resveratrol modulates inflammatory-related microRNAs and cytokines expression in peripheral blood mononuclear cells of type 2 diabetes and hypertensive patients with coronary artery disease. Pharmacol Res. 2013;72:69-82. (PubMed)
- Liu Y, Ma W, Zhang P, He S, Huang D. Effect of resveratrol on blood pressure: A meta-analysis of randomized controlled trials. Clin Nutr. 2015;34(1):27-34. (PubMed)
- Tome-Carneiro J, Gonzalvez M, Larrosa M, et al. Resveratrol in primary and secondary prevention of cardiovascular disease: a dietary and clinical perspective. Ann N Y Acad Sci. 2013;1290:37-51. (PubMed)
- Heilbronn LK, Ravussin E. Calorie restriction and aging: review of the literature and implications for studies in humans. Am J Clin Nutr. 2003;78(3):361-369. (PubMed)
- Lin SJ, Defossez PA, Guarente L. Requirement of NAD and SIR2 for life-span extension by calorie restriction in Saccharomyces cerevisiae. Science. 2000;289(5487):2126-2128. (PubMed)
- Howitz KT, Bitterman KJ, Cohen HY, et al. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature. 2003;425(6954):191-196. (PubMed)
- Wood JG, Rogina B, Lavu S, et al. Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature. 2004;430(7000):686-689. (PubMed)
- Valenzano DR, Terzibasi E, Genade T, Cattaneo A, Domenici L, Cellerino A. Resveratrol prolongs lifespan and retards the onset of age-related markers in a short-lived vertebrate. Curr Biol. 2006;16(3):296-300. (PubMed)
- Baur JA, Pearson KJ, Price NL, et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature. 2006;444(7117):337-342. (PubMed)
- Semba RD, Ferrucci L, Bartali B, et al. Resveratrol levels and all-cause mortality in older community-dwelling adults. JAMA Intern Med. 2014;174(7):1077-1084. (PubMed)
- Brown K, Rufini A, Gescher A. Do not throw out the resveratrol with the bath water. JAMA Intern Med. 2015;175(1):140-141. (PubMed)
- Glaser JH. Effect of wine consumption on mortality. JAMA Intern Med. 2015;175(4):650. (PubMed)
- Wang J, Ho L, Qin W, et al. Caloric restriction attenuates beta-amyloid neuropathology in a mouse model of Alzheimer's disease. FASEB J. 2005;19(6):659-661. (PubMed)
- Aguirre L, Fernandez-Quintela A, Arias N, Portillo MP. Resveratrol: anti-obesity mechanisms of action. Molecules. 2014;19(11):18632-18655. (PubMed)
- Kennedy DO, Wightman EL, Reay JL, et al. Effects of resveratrol on cerebral blood flow variables and cognitive performance in humans: a double-blind, placebo-controlled, crossover investigation. Am J Clin Nutr. 2010;91(6):1590-1597. (PubMed)
- Witte AV, Kerti L, Margulies DS, Floel A. Effects of resveratrol on memory performance, hippocampal functional connectivity, and glucose metabolism in healthy older adults. J Neurosci. 2014;34(23):7862-7870. (PubMed)
- Pasinetti GM, Wang J, Ho L, Zhao W, Dubner L. Roles of resveratrol and other grape-derived polyphenols in Alzheimer's disease prevention and treatment. Biochim Biophys Acta. 2015;1852(6):1202-1208. (PubMed)
- Centers for Disease Control and Prevention. National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States, 2014. Atlanta, GA: US Department of Health and Human Services. http://www.cdc.gov/diabetes/data/statistics/2014StatisticsReport.html.
- Szkudelski T, Szkudelska K. Resveratrol and diabetes: from animal to human studies. Biochim Biophys Acta. 2015; 1852(6):1145-1154. (PubMed)
- Movahed A, Nabipour I, Lieben Louis X, et al. Antihyperglycemic effects of short term resveratrol supplementation in type 2 diabetic patients. Evid Based Complement Alternat Med. 2013;2013:851267. (PubMed)
- Bhatt JK, Thomas S, Nanjan MJ. Resveratrol supplementation improves glycemic control in type 2 diabetes mellitus. Nutr Res. 2012;32(7):537-541. (PubMed)
- Brasnyo P, Molnar GA, Mohas M, et al. Resveratrol improves insulin sensitivity, reduces oxidative stress and activates the Akt pathway in type 2 diabetic patients. Br J Nutr. 2011;106(3):383-389. (PubMed)
- Poulsen MM, Vestergaard PF, Clasen BF, et al. High-dose resveratrol supplementation in obese men: an investigator-initiated, randomized, placebo-controlled clinical trial of substrate metabolism, insulin sensitivity, and body composition. Diabetes. 2013;62(4):1186-1195. (PubMed)
- Timmers S, Konings E, Bilet L, et al. Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell Metab. 2011;14(5):612-622. (PubMed)
- Yoshino J, Conte C, Fontana L, et al. Resveratrol supplementation does not improve metabolic function in nonobese women with normal glucose tolerance. Cell Metab. 2012;16(5):658-664. (PubMed)
- Carpene C, Gomez-Zorita S, Deleruyelle S, Carpene MA. Novel strategies for preventing diabetes and obesity complications with natural polyphenols. Curr Med Chem. 2015;22(1):150-164. (PubMed)
- Hausenblas HA, Schoulda JA, Smoliga JM. Resveratrol treatment as an adjunct to pharmacological management in type 2 diabetes mellitus-systematic review and meta-analysis. Mol Nutr Food Res. 2015;59(1):147-159. (PubMed)
- Liu K, Zhou R, Wang B, Mi MT. Effect of resveratrol on glucose control and insulin sensitivity: a meta-analysis of 11 randomized controlled trials. Am J Clin Nutr. 2014;99(6):1510-1519. (PubMed)
- Burns J, Yokota T, Ashihara H, Lean ME, Crozier A. Plant foods and herbal sources of resveratrol. J Agric Food Chem. 2002;50(11):3337-3340. (PubMed)
- Rimando AM, Kalt W, Magee JB, Dewey J, Ballington JR. Resveratrol, pterostilbene, and piceatannol in vaccinium berries. J Agric Food Chem. 2004;52(15):4713-4719. (PubMed)
- Sanders TH, McMichael RW, Jr., Hendrix KW. Occurrence of resveratrol in edible peanuts. J Agric Food Chem. 2000;48(4):1243-1246. (PubMed)
- Hurst WJ, Glinski JA, Miller KB, Apgar J, Davey MH, Stuart DA. Survey of the trans-resveratrol and trans-piceid content of cocoa-containing and chocolate products. J Agric Food Chem. 2008;56(18):8374-8378. (PubMed)
- Creasey LL, Coffee M. Phytoalexin production potential of grape berries. J Am Soc Hortic Sci. 1988;113(2):230-234.
145. Fremont L. Biological effects of resveratrol. Life Sci. 2000;66(8):663-673. (PubMed) - Goldberg DM, Karumanchiri A, Ng E, Yan J, Eleftherios P, Soleas G. Direct gas chromatographic-mass spectrometric method to assay cis-resveratrol in wines: preliminary survey of its concentration in commercial wines. J Agric Food Chem. 1995;43(5):1245-1250.
- Guerrero RF, Garcia-Parrilla MC, Puertas B, Cantos-Villar E. Wine, resveratrol and health: a review. Nat Prod Commun. 2009;4(5):635-658. (PubMed)
- Stervbo U, Vang O, Bonnesen C. A review of the content of the putative chemopreventive phytoalexin resveratrol in red wine. Food Chemistry. 2007;101:449-457.
- Sobolev VS, Cole RJ. trans-resveratrol content in commercial peanuts and peanut products. J Agric Food Chem. 1999;47(4):1435-1439. (PubMed)
- Hendler SS, Rorvik DR, eds, eds. PDR for Nutritional Supplements. 2nd edition ed: Thomson Reuters; 2008.
- Williams LD, Burdock GA, Edwards JA, Beck M, Bausch J. Safety studies conducted on high-purity trans-resveratrol in experimental animals. Food Chem Toxicol. 2009;47(9):2170-2182. (PubMed)
- Johnson WD, Morrissey RL, Usborne AL, et al. Subchronic oral toxicity and cardiovascular safety pharmacology studies of resveratrol, a naturally occurring polyphenol with cancer preventive activity. Food Chem Toxicol. 2011;49(12):3319-3327. (PubMed)
- Gescher A, Steward WP, Brown K. Resveratrol in the management of human cancer: how strong is the clinical evidence? Ann N Y Acad Sci. 2013;1290:12-20. (PubMed)
- American Academy of Pediatrics. Committee on Substance Abuse and Committee on Children With Disabilities. Fetal alcohol syndrome and alcohol-related neurodevelopmental disorders. Pediatrics. 2000;106(2 Pt 1):358-361. (PubMed)
- Bertelli AA, Giovannini L, Giannessi D, et al. Antiplatelet activity of synthetic and natural resveratrol in red wine. Int J Tissue React. 1995;17(1):1-3. (PubMed)
- Koe XF, Tengku Muhammad TS, Chong AS, Wahab HA, Tan ML. Cytochrome P450 induction properties of food and herbal-derived compounds using a novel multiplex RT-qPCR in vitro assay, a drug-food interaction prediction tool. Food Sci Nutr. 2014;2(5):500-520. (PubMed)
- Piver B, Berthou F, Dreano Y, Lucas D. Inhibition of CYP3A, CYP1A and CYP2E1 activities by resveratrol and other non volatile red wine components. Toxicol Lett. 2001;125(1-3):83-91. (PubMed)
- Regev-Shoshani G, Shoseyov O, Kerem Z. Influence of lipophilicity on the interactions of hydroxy stilbenes with cytochrome P450 3A4. Biochem Biophys Res Commun. 2004;323(2):668-673. (PubMed)
- Detampel P, Beck M, Krahenbuhl S, Huwyler J. Drug interaction potential of resveratrol. Drug Metab Rev. 2012;44(3):253-265. (PubMed)
Disclaimer
The Linus Pauling Institute's Micronutrient Information Center provides scientific information on the health aspects of dietary factors and supplements, food, and beverages for the general public. The information is made available with the understanding that the author and publisher are not providing medical, psychological, or nutritional counseling services on this site. The information should not be used in place of a consultation with a competent health care or nutrition professional.
The information on dietary factors and supplements, food, and beverages contained on this website does not cover all possible uses, actions, precautions, side effects, and interactions. It is not intended as nutritional or medical advice for individual problems. Liability for individual actions or omissions based upon the contents of this site is expressly disclaimed.
You may not copy, modify, distribute, display, transmit, perform, publish or sell any of the copyrightable material on this website. You may hyperlink to this website but must include the following statement:
"This link leads to a website provided by the Linus Pauling Institute at Oregon State University. [Your name] is not affiliated or endorsed by the Linus Pauling Institute or Oregon State University."