Take our professional continuing education course, "Micronutrients and Bone Health."
Summary
- The skeleton is a dynamic organ, comprised of tissue and cells in a continual state of activity throughout a lifetime. (More information)
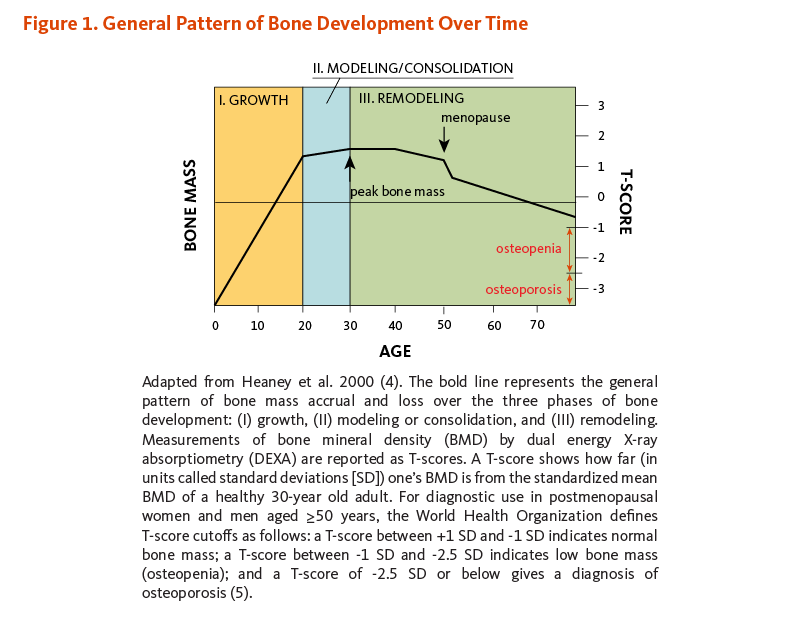
- Bone development occurs in three general phases that coincide with age: (i) growth, (ii) modeling or consolidation, and (iii) remodeling. The remodeling phase predominates during adulthood, with bone resorption and formation activities constantly occurring in linked succession. (More information)
- Accrual of bone mass is the product of both genetics and environmental factors. Diet and exercise can significantly affect the ability to achieve one’s genetically determined peak bone mass. (More information)
- Beginning around age 34, the rate of bone resorption outpaces that of bone formation, leading to an inevitable loss of bone mass with age. Clinical consequences of low bone mass include osteomalacia, osteopenia, and osteoporosis. (More information)
- Bone mass is usually measured by two-dimensional X-ray absorptiometry as an areal density called bone mineral density (BMD). Measurements of BMD are used to diagnose osteoporosis and predict osteoporotic fracture. The Fracture Risk Assessment Tool (FRAX) is designed to account for the impact of numerous risk factors and more accurately calculate the 10-year risk of fracture. (More information)
- In addition to the micronutrients calcium and vitamin D, several other minerals and vitamins have essential roles in bone health. (More information)
- While striving to achieve and maintain recommended intakes of calcium and vitamin D is essential to minimize age-related bone loss, there is no consensus regarding the use of supplements in the prevention of osteoporotic fracture and falls by community-dwelling older adults. (More information)
- In institutionalized elderly people and those with suboptimal calcium intakes, supplementation with calcium (with or without vitamin D) may reduce the risk of osteoporotic fracture. (More information)
- Although vitamin K is required for the calcium-binding activity of several bone matrix proteins, there is inconclusive evidence that vitamin K supplementation improves BMD or reduces the risk of osteoporotic fracture. (More information)
- Vitamin A excess, typically from supplemental sources, may have a negative effect on bone health and increase the risk of fracture. (More information)
- Vitamin C is essential for collagen synthesis and bone matrix quality, yet few trials have tested the efficacy of vitamin C supplementation on BMD and fracture risk. (More information)
- High concentrations of homocysteine in the blood have been associated with an increased risk of fracture. Yet, homocysteine lowering through B-vitamin supplementation failed to reduce the risk of fracture. (More information)
- Smoking, alcohol consumption, and physical activity significantly affect BMD and risk of osteoporotic fracture. (More information)
Other Articles
Overview of Bone Biology
Structure and physiology
Bone composition and structure
Our skeleton may seem an inert structure, but it is an active organ, made up of tissue and cells in a continual state of activity throughout a lifetime. Bone tissue is comprised of a mixture of minerals deposited around a protein matrix, which together contribute to the strength and flexibility of our skeleton.
Sixty-five percent of bone tissue is inorganic mineral, which provides the hardness of bone. The major minerals found in bone are calcium and phosphorus in the form of an insoluble salt called hydroxyapatite [chemical formula: (Ca)10(PO4)6(OH)2]. Hydroxyapatite crystals lie adjacent and bound to the organic protein matrix. Magnesium, sodium, potassium, and citrate ions are also present, conjugated to hydroxyapatite crystals rather than forming distinct crystals of their own (1).
The remaining 35% of bone tissue is an organic protein matrix, ~90% of which is type I collagen. Collagen fibers twist around each other and provide the interior scaffolding upon which bone minerals are deposited (1). The other ~10% of the organic bone matrix comprises various non-collagenous matrix proteins, including members of the vitamin K-dependent γ-carboxyglutamic acid (Gla)-containing protein family (see Vitamin K). Although their functions are not fully elucidated, non-collagenous proteins are thought to serve as anchor points for cells involved in bone (re)modeling and as regulators of the activities of these cells (2).
Types of bone tissue
There are two types of bone tissue: cortical (compact) bone and trabecular (spongy or cancellous) bone. Eighty percent of the skeleton is cortical bone, which forms the outer surface of all bones. The small bones of the wrists, hands, and feet are entirely cortical bone. Cortical bone looks solid but actually has microscopic openings that allow for the passage of blood vessels and nerves. The other 20% of skeleton is trabecular bone, found within the ends of long bones and inside flat bones (skull, pelvis, sternum, ribs, and scapula) and spinal vertebrae. Both cortical and trabecular bone have the same mineral and matrix components but differ in their porosity and microstructure: Trabecular bone is much less dense, has a greater surface area, and undergoes more rapid rates of turnover (see Bone remodeling/turnover) (2).
Bone formation and remodeling
There are three phases of bone development over time: growth, modeling (or consolidation), and remodeling (Figure 1). During the growth phase, the size of our bones increases. Bone growth is rapid from birth to age two, continues in spurts throughout childhood and adolescence, and eventually ceases in the late teens and early twenties. Although bones stop growing in length when we are about 20 years old, they change shape and thickness and continue accruing mass when stressed during the modeling phase. For example, weight training and body weight exert mechanical stresses that influence the shape of bones. Thus, acquisition of bone mass occurs during both the growth and modeling/consolidation phases of bone development. The remodeling phase consists of a constant process of bone resorption (breakdown) and formation that predominates during adulthood and continues throughout life. Beginning around age 34, the rate of bone resorption exceeds that of bone formation, leading to an inevitable loss of bone mass with age (3).
Peak bone mass
Bone mass refers to the quantity of bone present, both matrix and mineral. Bone mass increases through adolescence and peaks in the late teen years and into our twenties. The maximum amount of bone acquired by the time a stable skeletal state has been attained is known as peak bone mass (see Figure 1) (6). Achieving one’s genetically determined peak bone mass is influenced by several environmental factors, some of which are discussed below (see Determinants of adult bone health; reviewed in 6).
Bone cells
The cells responsible for bone formation and resorption are osteoblasts and osteoclasts, respectively.
Osteoblasts prompt the formation of new bone by secreting the collagen-containing component of bone that is subsequently mineralized (1). The enzyme alkaline phosphatase is secreted by osteoblasts while they are actively depositing bone matrix; alkaline phosphatase travels to the bloodstream and is used as a clinical marker of bone formation rate. Osteoblasts have receptors for vitamin D, estrogen, and parathyroid hormone (PTH). As a result, these hormones have potent effects on bone health through their regulation of osteoblastic activity. Once they have finished secreting matrix, osteoblasts either die, become lining cells, or transform into osteocytes, a type of bone cell embedded deep within the organic matrix (7, 8). Osteocytes make up 90%-95% of all bone cells and are very long-lived (up to decades) (7). They secrete soluble factors that influence osteoclastic and osteoblastic activity and play a central role in bone remodeling in response to mechanical stress (7-9).
Osteoclasts erode the surface of bones by secreting enzymes and acids that dissolve bone. More specifically, enzymes degrade the organic matrix and acids solubilize bone mineral salts (1). Osteoclasts work in small, concentrated masses and take approximately three weeks to dissolve bone, at which point they die and osteoblasts invade the space to form new bone tissue. In this way, bone resorption and formation are always "coupled." End products of bone matrix breakdown (hydroxyproline and amino-terminal collagen peptides) are excreted in the urine and can be used as convenient biochemical measures of bone resorption rates.
Bone remodeling/turnover
Bone tissue, both mineral and organic matrix, is continually being broken down and rebuilt in a process known as remodeling or turnover. During remodeling, bone resorption and formation are always coordinated: Osteoclasts first dissolve a section of bone and then osteoblasts invade the newly created "remodeling space" and secrete bone matrix (10). The activities of osteoblasts and osteoclasts are "coupled" together and form what is called the remodeling unit. The goal of remodeling is to repair and maintain a healthy skeleton, adapt bone structure to new loads, regulate calcium concentration in extracellular fluids, and provide a reserve supply of phosphorus (2). The bone remodeling cycle, which refers to the time required to complete the entire series of cellular events from resorption to final mineralization, generally lasts two to eight months (11).
Remodeling units cycle at staggered stages. The initiation of an intervention that influences bone remodeling (e.g., supplementation with calcium and vitamin D or administration of an anti-resorptive drug) will likely improve blood calcium and as a consequence reduce parathyroid hormone secretion (see Calcium). This will affect the frequency at which new remodeling cycles are initiated, likely leading to a reduction of the number of active remodeling units. Yet, units activated before the start of the treatment will still be progressing. Suppression of remodeling will lead to a transient retention of bone mineral at remodeling loci, which are largely demineralized, resulting in an increase in bone mass (called "remodeling transient"). This gain of bone at remodeling loci is not creating new bone as it will only usually persist for one remodeling cycle, until all remodeling cycles are synchronized to the treatment exposure and remodeling returns to its previous level. The lag time between treatment exposure and return to remodeling balance is known as the "bone remodeling transient" (12). Considering the bone remodeling transient, it may take a few months to a few years for the remodeling process to return to equilibrium and thus only then can an intervention be evaluated for its potential impact on bone density (2).
The rates of bone tissue turnover differ depending on the type of bone: Trabecular bone has a faster rate of turnover than cortical bone. Osteoporotic fracture manifests in trabecular bone, primarily as fractures of the hip and spine, and many osteoporotic therapies target remodeling activities in order to alter bone mass (13).
Determinants of adult bone health
Maximum attainment of peak bone mass
The majority of bone mass is acquired during the growth phase of bone development (see Figure 1) (4, 10). Attaining one’s peak bone mass (i.e., the maximum amount of bone) is the product of genetic, lifestyle, and environmental factors (14, 15). Sixty to 80% of peak bone mass is determined by genetics, while the remaining 20%-40% is influenced by lifestyle factors, primarily nutrition and physical activity (6). In other words, diet and exercise are known to contribute to bone mass acquisition but can only augment peak bone mass within an individual’s genetic potential.
Acquisition of bone mass during the growth phase is sometimes likened to a "bone bank account" (4, 15). As such, maximizing peak bone mass is important when we are young in order to protect against the consequences of age-related bone loss (16). However, improvements in BMD generally do not persist once a supplement or exercise intervention is terminated (17, 18). Thus, attention to diet and physical activity during all phases of bone development is beneficial for bone mass accrual and skeletal health (6).
Rate of bone loss with aging
Bone remodeling is a lifelong process, with resorption and formation linked in space and time. Yet the scales tip such that bone loss outpaces bone gain as we age. Beginning around age 34, the rate of bone resorption exceeds the rate of bone formation, leading to an inevitable loss of bone mass with age (see Figure 1) (19). Age-related estrogen reduction is associated with increased bone remodeling activity — both resorption and formation — in both sexes (9). However, the altered rate of bone formation does not match that of resorption; thus, estrogen deficiency contributes to loss of bone mass over time (8, 9). The first three to five years following the onset of menopause are associated with an accelerated, self-limiting loss of bone mass at a rate of two to six times the premenopausal rate. Subsequent postmenopausal bone loss occurs at a constant rate as we age and is then comparable between men and women (16). As we continue to lose bone, we near the threshold for osteoporosis and are at high risk for fractures of the hip and spine.
Loss of bone mineral and mass
Osteomalacia
Osteomalacia, also known as "adult rickets," is a failure to mineralize bone. Stereotypically, osteomalacia results from vitamin D deficiency (serum 25-hydroxyvitamin D concentrations ≤30 nmol/L or ≤12 ng/mL) and the associated inability to absorb dietary calcium and phosphorus across the small intestine. Blood calcium and phosphorus concentrations are tightly controlled (see Calcium). Briefly, parathyroid hormone (PTH) is quickly released in the circulation and vitamin D is activated following a slight drop in blood calcium. The increase in PTH stimulates bone remodeling activity — both resorption and formation, which are always coupled. Thus, osteoclasts release calcium and phosphorus from bone in order to restore blood calcium concentrations, and osteoblasts mobilize to replace the resorbed bone. During osteomalacia, however, the deficiency of calcium and phosphorus results in incomplete mineralization of the newly secreted bone matrix. In severe cases, newly formed, unmineralized bone loses its stiffness and can become deformed under the strain of body weight.
In addition to vitamin D deficiency, osteomalacia can result from extreme calcium deficiency, fluoride toxicity, cadmium poisoning, and genetic disorders of phosphate homeostasis (hypophosphatemia) (2).
Osteopenia
Simply put, osteopenia and osteoporosis are varying degrees of low bone mass. Whereas osteomalacia is characterized by low-mineral and high-matrix content, osteopenia and osteoporosis result from low levels of both. As defined by the World Health Organization (WHO), osteopenia precedes osteoporosis and occurs when one’s BMD is between 1 and 2.5 standard deviations (SD) below that of the average young adult (30 years of age) (see Figure 1).
Osteoporosis
Osteoporosis is a condition of increased bone fragility and susceptibility to fracture due to loss of bone mass. Clinically, osteoporosis is defined as a BMD that is greater than 2.5 SD below the mean of a young adult (see Figure 1). It has been estimated that fracture risk in adults is approximately doubled for each SD reduction in BMD (10). Common sites of osteoporotic fracture are the hip, femoral neck, and vertebrae of spinal column — skeletal sites rich in trabecular bone.
Bone mineral density and fracture risk
Technically, we cannot detect the matrix component of bone, so bone mass cannot be measured directly. We can, however, detect bone mineral by using dual energy X-ray absorptiometry (abbreviated DEXA or DXA). In this two-dimensional imaging technique, the absorption of photons from an X-ray is a function of the amount of mineral present in the path of the beam. Therefore, bone mineral density (BMD or aBMD for areal BMD) measures the quantity of mineral present in a given section of bone and is used as a proxy for bone mass (10). Although BMD is a convenient clinical marker to assess bone mass and is associated with risk for osteoporotic fracture, it is not the sole determinant of fracture risk. Bone quality (architecture, strength) and propensity to fall (balance, mobility) also factor into risk assessment and should be considered when deciding upon an intervention strategy. Over the last decade, high-resolution peripheral quantitative computed tomography (HRpQCT) has been increasingly used to better capture bone strength. HRpQCT is a three-dimensional imaging technique that uses attenuation of X-ray beams to image and measure cortical and trabecular volumetric bone mineral density (vBMD), bone geometry, and bone microarchitecture of the non-dominant distal femur, radius, and tibia (6, 20).
BMD, the quantity of mineral present per given area/volume of bone, is only a surrogate for bone strength. Although it is a convenient biomarker used in clinical and research settings to predict fracture risk, the likelihood of experiencing an osteoporotic fracture cannot be predicted solely by BMD (10). The risk of osteoporotic fracture is influenced by additional factors, including bone quality (microarchitecture, geometry) and propensity to fall (balance, mobility, muscular strength). Other modifiable and non-modifiable factors also play into osteoporotic fracture risk, and they are generally additive (21). The Fracture Risk Assessment Tool (FRAX) was designed to account for some of these additional risk factors (22). FRAX is available online. Once you have your BMD measurement, visit the website to calculate your 10-year probability of fracture, taking some of these additional risk factors into account.
Paying attention to modifiable risk factors for osteoporosis is an important component of fracture prevention strategies. For more details about individual dietary factors and osteoporosis, see the page on Osteoporosis or the Disease Index.
Micronutrients Important to Bone Health
Micronutrient supply plays a prominent role in bone health. Several minerals have direct roles in hydroxyapatite crystal formation and structure; other nutrients have indirect roles as cofactors or as regulators of cellular activity (23, 24).
Table 1 lists the dietary reference intakes (DRIs) for micronutrients important to bone health. The average dietary intake of Americans (aged ≥19 years) is also provided for comparative purposes (25).
| Micronutrient | RDA or AI* (≥19 y) | UL (≥19 y) | Mean Intakea (≥19 y, all food sources) (25) |
|---|---|---|---|
| Calcium | Men: 1,000 mg/day (19-70y) 1,200 mg/day (>70y) Women: 1,000 mg/day (19-50y) 1,200 mg/day (>50y) |
Men & Women: 2,500 mg/day (19-50y) 2,000 mg/day (>50y) |
910 mg/day |
| Phosphorus | Men & Women: 700 mg/day (≥19y) |
Men & Women: 4,000 mg/day (19-70y) 3,000 mg/day (>70y) |
1,300 mg/day |
| Fluoride | Men: 4 mg/day* Women: 3 mg/day* |
Men & Women: 10 mg/day |
Not reported |
| Magnesium | Men: 400 mg/day (19-30y) 420 mg/day (>31y) Women: 310 mg/day (19-30y) 320 mg/day (>31y) |
Men & Women: 350 mg/dayb |
290 mg/day |
| Sodium | Men & Women: 1,500 mg/day |
Not determinable | Not reported |
| Potassium | Men: 3,400 mg/day Women: 2,600 mg/day |
Not determinable | 2,700 mg/day |
| Vitamin D | Men & Women: 15 μg (600 IU)/day (19-70y) 20 μg (800 IU)/day (>70y) |
Men & Women: 100 μg (4,000 IU)/day |
4.5 μg (180 IU)/day |
| Vitamin K | Men: 120 μg/day* Women: 90 μg/day* |
Not determinable | 88.2 μg/day |
| Vitamin A | Men: 900 μg RAE (3,000 IU)/dayc Women: 700 μg RAE (2,333 IU)/dayc |
Men & Women: 3,000 μg RAE (10,000 IU)/dayd |
600 μg/day |
| Vitamin C | Men: 90 mg/day Women: 75 mg/day |
Men & Women: 2,000 mg/day |
85.4 mg/day |
| Vitamin B6 | Men: 1.3 mg/day (19-50y) 1.7 mg/day (>50y) Women: 1.3 mg/day (19-50y) 1.5 mg/day (>50y) |
Men & Women: 100 mg/day |
2.0 mg/day |
| Folate | Men & Women: 400 μg DFE/daye |
Men & Women: 1,000 μg/dayf |
540 μg DFE/day |
| Vitamin B12 | Men & Women: 2.4 μg/day |
Not determinable | 5.2 μg/day |
| Abbreviations: RDA, recommended dietary allowance; AI, adequate intake; UL, tolerable upper intake level; y, years; mg, milligram; μg, microgram; IU, international units aIncludes nutrients from enriched/fortified food bApplies only to magnesium in supplements cRetinol activity equivalent (RAE) is the international standard of measure that represents vitamin A activity as retinol. For example, it has been determined that 1 μg RAE would correspond to 1 μg of preformed retinol in animal products and 12 μg of β-carotene from plant-based food dApplies only to preformed retinol eDietary folate equivalent (DFE) is used to reflect the higher bioavailability of folic acid found in supplements: 1 μg of food folate = 1 μg DFE; 1 μg of folic acid taken with meals = 1.7 μg DFE; and 1 μg of folic acid taken on an empty stomach = 2 μg DFE fApplies to the synthetic form (i.e., folic acid) in fortified food and supplements |
|||
Minerals
Calcium
Calcium is the most common mineral in the human body. About 99% of the calcium in the body is found in bones and teeth, while the other 1% is found in blood and soft tissues. Calcium concentrations in the blood must be maintained within a very narrow concentration range to preserve normal physiological functions (e.g., muscle contraction, nerve impulse conduction, constriction and relaxation of blood vessels). Because these functions are essential, the body will demineralize bone to maintain normal blood calcium concentrations when calcium intake is inadequate. In response to low blood calcium, parathyroid hormone (PTH) is secreted by the parathyroid glands. PTH targets three main axes in order to restore blood calcium concentration: (i) vitamin D is activated (see the section on Vitamin D), (ii) filtered calcium is retained by the kidneys, and (iii) bone resorption is induced (1). It is thus critical to obtain enough calcium from food to limit bone resorption in response to fluctuating blood calcium concentrations.
A 2015 meta-analysis of 59 randomized, placebo-controlled trials in older adults suggested that calcium had a significant beneficial effect on bone mineral density (BMD) (26). Increased intakes of calcium, either from food or supplements, for at least one year resulted in significant 0.6%-1.8% increases in total body, lumbar spine, hip, and femoral neck BMD. Further analyses found no difference in BMD changes with the source of calcium, the dose of supplemental calcium, or the baseline level of dietary calcium intake among participants (26). Such modest increases may help limit the average rate of BMD loss after menopause and translate into meaningful fracture risk reductions (27). A 2015 meta-analysis of 20 randomized controlled trials that reported on total fracture risk found an overall 11% risk reduction associated with supplemental calcium with or without vitamin D (28). However, there was no effect when the analysis was restricted to the largest trials with the lowest risk of bias. Additionally, no reductions were found in risks of hip, vertebral, and forearm fractures with calcium supplementation (28). The most recent meta-analysis of 33 randomized controlled trials found no evidence of hip fracture risk reduction with calcium and/or vitamin D in community-dwelling older adults (29).
In 2011, the US Preventive Services Task Force (USPSTF) conducted a meta-analysis of 11 randomized, placebo-controlled trials that included 52,915 older people (of whom 69% were postmenopausal women) and reported that the supplementation of vitamin D (300-1,000 IU/day) and calcium (500-1,200 mg/day) for up to seven years resulted in a 12% reduction in the risk of any new fracture (30). While further analyses suggested a significant 29% risk reduction in institutionalized elderly people, supplementation did not result in substantial risk reduction in community-dwelling elderly people, postmenopausal women, or women with a history of fracture (30). Accordingly, the USPSTF advised against daily supplementation with calcium and vitamin D for the primary prevention of fractures in community-dwelling postmenopausal women (31). A recently updated meta-analysis of randomized placebo-controlled trials, commissioned by the National Osteoporosis Foundation (now the Bone Health & Osteoporosis Foundation), found a 15% reduction in risk of total fracture (eight studies) and a 30% reduction in risk of hip fractures (six studies) with calcium and vitamin D supplementation in older people (32). In contrast to the USPSTF’s recommendations, the Bone Health & Osteoporosis Foundation supports the use of supplemental calcium (1,000-1,200 mg/day) and vitamin D (800-1,000 IU/day) as a public health intervention to reduce the burden of fractures in all middle-aged men and women (32).
The current recommended dietary allowances (RDA) for calcium in the US are based on a combination of data from balance studies and clinical trials, and they appear to be set at levels that support bone health (see Table 1) (33, 34). Aside from the importance of meeting the RDA, calcium (and/or vitamin D) supplements are sometimes used as adjuvant during osteoporosis therapy. According to the latest guidelines from the American College of Physicians, calcium supplements are not a necessary component of osteoporosis treatment regimens, and dosages should be considered carefully as excess dosing has been linked to risks of hypercalcemia and kidney stones (35).
Finally, several prospective cohort studies and randomized controlled trials that primarily reported on bone health-related outcomes have raised concerns over the safety of calcium supplements, either alone or with vitamin D, on the risk of cardiovascular events. Although the issue requires further attention, studies designed to examine the effect of calcium supplements on cardiovascular risk as a primary outcome are lacking. Based on an updated review of the literature that included four randomized controlled trials, one nested case-control study, and 26 prospective cohort studies (36), an expert panel convened by the National Osteoporosis Foundation (now the Bone Health & Osteoporosis Foundation) and the American Society for Preventive Cardiology concluded that the use of supplemental calcium for generally healthy individuals was safe from a cardiovascular health standpoint when total calcium intakes did not exceed the tolerable upper intake level (UL) (37). The panel of experts supported the use of calcium supplements to correct shortfalls in dietary calcium intake and meet current recommendations (37). For a more extensive review of this issue, see the Safety section of the article on calcium.
Phosphorus
More than half the mass of bone mineral is comprised of phosphorus, which combines with calcium to form hydroxyapatite crystals. In addition to this structural role, osteoblastic activity relies heavily on local phosphate concentrations in the bone matrix (13, 38). Blood concentrations of both phosphorus and calcium are under the control of three endocrine hormones: PTH, vitamin D, and fibroblast growth factor-23 (FGF-23). A slight drop in blood calcium concentration (e.g., in the case of inadequate calcium intake) results in an increase in serum PTH, which rapidly decreases urinary excretion of calcium and increases urinary excretion of phosphorus. PTH also prompts bone resorption, which releases bone mineral (calcium and phosphate) and restores serum calcium concentrations. In the kidneys, PTH also stimulates vitamin D conversion into its active form, 1α,25-dihydroxyvitamin D3. In turn, 1α,25-dihydroxyvitamin D3 stimulates the intestinal absorption of both calcium and phosphorus. A third hormone, FGF-23, is secreted by osteocytes in response to increases in phosphorus intake. In a negative feedback loop, FGF-23 inhibits the formation of 1α,25-dihydroxyvitamin D3 and promotes an increase in urinary phosphorus excretion independently of PTH and 1α,25-dihydroxyvitamin D3 (39).
Given its prominent functions in bone, phosphorus deficiency could contribute to impaired bone mineralization (38). However, in healthy individuals, phosphorus deficiency is uncommon, and there is little evidence that phosphorus deficiency affects the incidence of osteoporosis (24). In contrast, phosphorus intakes in the US population tend to be much higher than the requirements and greater than calcium intakes (see Table 1). It is thought that high dietary phosphorus might interfere with calcium absorption and eventually promote bone resorption (40). However, there is currently no convincing evidence that dietary phosphorus levels experienced in the US adversely affect BMD and risk for osteoporosis (41). Nevertheless, the substitution of phosphoric acid-containing soft drinks and snack foods for milk and other calcium-rich food may represent a serious risk to bone health (40).
Fluoride
Fluoride has a high affinity for calcium, and most fluoride in the body is stored in calcified tissues, i.e., teeth and bones (42). In our teeth, very dense hydroxyapatite crystals are embedded in collagen fibers. The presence of fluoride in the hydroxyapatite crystals (forming fluorhydroxyapatite) enhances resistance to destruction by plaque bacteria (1, 42), and fluoride has been proven effective in the prevention of dental caries (43).
While fluoride may stimulate bone formation through direct effects on osteoblasts (44), the presence of fluoride in hydroxyapatite increases the crystal size but may contribute to bone fragility (45, 46). A 2008 meta-analysis of 25 randomized controlled studies (in 2,348 participants) investigating the effect of fluoride supplementation on bone health found significant increases in spine and hip BMD irrespective of the daily dose of fluoride used (47). Further, the odds of suffering from vertebral and non-vertebral fractures were found to be significantly reduced with daily doses of ≤20 mg fluoride but not with higher doses (47).
Some observational studies have examined chronic exposure of fluoride in drinking water in relation to bone health. In a cross-sectional study, structural and mechanical properties of bone from 91 femoral head specimens collected from older adults undergoing total hip arthroplasty were found to be similar whether subjects were living in an area with or without fluoridated water (48). Additionally, a meta-analysis that included 13 prospective cohort studies and one case-control study, conducted over three decades in more than 73 million subjects followed for up to 17 years, found no effect of fluoride exposure in water on hip fracture risk (49).
Fluoride supplementation is not currently approved by the US Food and Drug Administration for use in the prevention or treatment of osteoporosis (16).
Magnesium
Magnesium is a major mineral with essential structural and functional roles in the body. It is a critical component of our skeleton, with 50%-60% of total body magnesium found in bone where it colocalizes with hydroxyapatite, influencing the size and strength of hydroxyapatite crystals (24). Magnesium deficiency in rodents results in larger-than-normal hydroxyapatite crystals, ultimately affecting bone stiffness. It also reduced osteoblast activity and delayed bone differentiation and mineralization (reviewed in 50). Magnesium serves a role in mineral metabolism by regulating the PTH-vitamin D-FGF-23 axis. Magnesium deficiency has been associated with impaired secretion of PTH and end-organ resistance to the actions of PTH and 1α,25-dihydroxyvitamin D3 (51). Magnesium deficiency also increases serum FGF-23, thereby stimulating urinary phosphorus excretion and possibly inhibiting the production of 1α,25-dihydroxyvitamin D (the biologically active form of vitamin D) in the kidneys (see Phosphorus) (52, 53). In addition, magnesium is a cofactor to 25-hydroxyvitamin D-1α-hydroxylase enzyme that catalyzes 1α,25-dihydroxyvitamin D formation, suggesting that magnesium deficiency likely decreases the activity of this enzyme (50). Accordingly, a cross-sectional analysis of data from the US National Health and Nutrition Examination Survey (NHANES) found lower risks of vitamin D insufficiency and deficiency in participants with higher magnesium intakes (54). Low dietary intakes of magnesium are common in the US population (Table 1) (25), and it has been suggested that magnesium deficiency could impair bone mineralization and represent a risk factor for osteoporosis.
A 2016 meta-analysis of observational studies showed a positive association between magnesium intake and hip BMD (three studies) but no association with lumbar spine BMD (six studies), femoral neck BMD (three studies), or odds of hip or total fractures (three studies) (55). There was also no evidence of an association between dietary magnesium and risk of fracture in a recently published analysis of the Kuopio Ischemic Heart Disease Risk Factor Study that followed 2,245 middle-aged men for a median 25.6 years. Yet, the risk of any fracture (femoral, humeral, forearm) was ~2.5 times higher in subjects in the lowest quartile of serum magnesium concentrations compared to those in the highest quartile (<1.8 mg/dL versus >2 mg/dL) (56).
Few studies have addressed the effect of magnesium supplementation on BMD or osteoporosis in humans. In a small group of postmenopausal women with osteoporosis (mean age, 57.6±10.6 years), supplementation with 750 mg/day of magnesium hydroxide (depending on individual tolerance levels) for six months, followed by 250 mg/day for another 18 months, resulted in increased BMD at the wrist after one year with no further increase after two years of supplementation (57). Another very small study in postmenopausal women who were taking estrogen replacement therapy and also a multivitamin found that supplementation with an additional 400 mg/day of magnesium and 600 mg/day of calcium resulted in increased BMD at the heel compared to postmenopausal women receiving only estrogen replacement therapy (58). Evidence is not yet sufficient to recommend supplemental magnesium in the prevention of osteoporosis unless normalization of serum magnesium levels is required. In the Women’s Health Initiative (WHI) observational study that followed over 70,000 postmenopausal women for a mean 7.6 years, the lowest versus highest quintile of total magnesium intake (<206.5 mg/day versus ≥422.5 mg/day) was found to be associated with increased risks of falls and lower-arm and wrist fractures; no association was found with hip or total fracture (59). Moreover, abnormally elevated serum magnesium in subjects with chronic kidney disease or those undergoing dialysis interferes with the action of the calciotropic hormones, PTH and 1α,25-dihydroxyvitamin D, and leads to mineralization defects and skeletal disorders (renal osteodystrophy) (60). Presently, the potential for increased magnesium intake to influence calcium and bone metabolism warrants more research with particular attention to its role in the prevention and treatment of osteoporosis.
Sodium
Sodium is thought to influence skeletal health through its impact on urinary calcium excretion (38, 61). Indeed, high-sodium intake results in increased loss of calcium in the urine, possibly due to competition between sodium and calcium for reabsorption in the kidneys and/or by an effect of sodium on parathyroid hormone secretion. Every 1 gram of sodium (equivalent to 2.5 g of salt) excreted by the kidneys has been found to draw approximately 26.3 mg of calcium into the urine (61). Theoretically, if the urinary calcium loss is not compensated for by an increased intestinal absorption from dietary sources, bone calcium will be mobilized and could potentially affect skeletal health.
A number of cross-sectional and intervention studies have suggested that high-sodium intakes are deleterious to bone health, especially in older women (62). In particular, high-sodium intake in conjunction with low-calcium intake may be especially detrimental to bone health (63-65). A two-year longitudinal study in postmenopausal women found increased urinary sodium excretion (an indicator of increased sodium intake) to be associated with decreased BMD at the hip (66). Linear regression analysis estimated that BMD could be maintained by reducing sodium intake to recommended levels (≤2,300 mg/day) and by increasing calcium intake to 1,200 mg/day. A second longitudinal study in postmenopausal women found that habitual high-sodium intake of approximately 3,000 mg/day was not detrimental to BMD over three years of follow-up (67). Notably, the average calcium intake in this study population was 1,300 to 1,500 mg/day — slightly above the RDA for calcium in women over 50 years. Another study in 40 postmenopausal women found that adherence to a low-sodium diet (2,000 mg/day) for six months was associated with significant reductions in sodium excretion, calcium excretion, and aminoterminal propeptide of type I collagen (a biomarker of bone resorption). Yet, these associations were only observed in women with elevated baseline urinary sodium excretion (68). Although these results do not clarify the long-term impact of high-sodium intake on bone health, it is concerning that most Americans consume too much sodium and too little calcium (69). Thus, striving to lower sodium intake while increasing calcium intake to the recommended levels is a good strategy for supporting bone health.
Potassium
Potassium is thought to mitigate the impact that excessively acidic diets might have on the skeleton. Western diets tend to be relatively low in sources of alkali (fruit and vegetables) and high in sources of acid (fish, meat, and cheese). When the quantity of bicarbonate ions buffering acids in the body is insufficient to maintain normal pH, the body is capable of mobilizing alkaline calcium salts from bone in order to neutralize acids consumed in the diet and generated by metabolism (70). This acid-base theory suggests that alkaline potassium salts derived from consuming fruit and vegetables may reduce the net acid content of the diet and protect against bone resorption (71).
A 2015 meta-analysis examined the effect of alkaline potassium salts on calcium economy and bone turnover from seven short-term, metabolic cross-over studies and seven randomized controlled trials (72). The result suggested a significant decrease in calcium excretion by the kidneys with supplemental potassium bicarbonate or potassium citrate. Supplementation with alkaline potassium salts also decreased the level of resorption markers but had no effect on markers of bone formation (72). Further, a two-year, randomized, double-blind, placebo-controlled trial investigated the effect of potassium citrate supplementation (60 milliequivalent [mEq]/day = 2,340 mg/day) on BMD and fracture risk in 201 healthy older adults (mean age, 69 years) (73). All participants were supplemented with calcium (500 mg/day) and vitamin D (400 IU/day). Compared to placebo, potassium citrate reduced net renal acid excretion and led to significant increases in all-body and site-specific BMD. The authors of this study also predicted a reduction in fracture risk using a prediction score calculated with the FRAX assessment tool (73). However, another two-year trial in 276 postmenopausal women (mean age, 60 years) found no effect of either low potassium citrate (18.5 mEq/day = 721.5 mg/day), high potassium citrate (55.5 mEq/day = 2,165 mg/day), or 300 g/day of additional fruit and vegetables on spine and hip BMD compared to placebo (74). At present, whether dietary potassium has an important role in bone health is still debated. Yet, one cannot exclude the possibility that modern diets that are low in potassium and high in sodium might have far-reaching adverse health impacts beyond those linked to cardiovascular health (71). Thus, adopting a diet that will improve the potassium-to-sodium intake ratio might also help support bone health.
Fat-soluble vitamins
Vitamin D
The primary function of vitamin D is to control blood concentrations of calcium and phosphorus in order to preserve normal physiological function. In response to low blood calcium, the parathyroid hormone (PTH) is released in the circulation by the parathyroid glands. In the kidneys, PTH stimulates the conversion of 25-hydoxyvitamin D3 − the storage form of vitamin D3 − into 1α,25-dihydroxyvitamin D3, the biologically active form of vitamin D. Increased circulating 1α,25-dihydroxyvitamin D3 promotes the active absorption of calcium across the intestinal cell (75). In conjunction with PTH, 1α,25-dihydroxyvitamin D3 also stimulates the retention of filtered calcium by the kidneys. Both 1α,25-dihydroxyvitamin D3 and PTH stimulate bone resorption that leads to the release of minerals from bone. This contributes to restoring normal blood calcium concentration to the detriment of the skeleton. Vitamin D and PTH also regulate phosphorus homeostasis, and low circulating 25-hydoxyvitamin D3 can lead to hypophosphatemia (see Phosphorus).
Results from observational studies rather consistently suggest a relationship between poor vitamin D status (assessed using circulating concentrations of 25-hydroxyvitamin D) and adverse bone health outcomes (76). As mentioned in the Calcium section above, several meta-analyses of randomized controlled trials have shown that combined calcium and vitamin D supplementation decreases fracture incidence in older adults (30, 32, 77). However, results of trials investigating the effect of supplemental vitamin D alone found little-to-no protection against fractures. A meta-analysis of 15 trials found no reduction in risks of vertebral and non-vertebral fractures with supplemental vitamin D compared to placebo (77). In addition, there was no evidence of any additional benefits of vitamin D supplementation in a pooled analysis of 11 trials that compared combined supplementation with vitamin D and calcium to supplementation with calcium alone (77). Recently published results from the Vitamin D Assessment (ViDA) trial showed no significant difference in risk of non-vertebral fracture in men and women (ages, 50-84 years) supplemented with either vitamin D3 (initial dose of 200,000 IU followed by 100,000/month) or placebo for a mean 3.4 years (78).
Falls are the leading cause of injuries (including fractures) in older adults and have been linked to loss of muscle mass and strength. A pooled analysis of observational studies in older people found higher odds of inadequate vitamin D status in fallers compared to non-fallers (79). However, it is possible that vitamin D inadequacy in fallers results from restricted sun exposure following loss of mobility or residential care. Several randomized controlled trials have examined the impact of vitamin D supplementation on muscle strength, postural balance, or risk of fall in older subjects. A meta-analysis of these trials found limited evidence of an effect of vitamin D supplementation on muscle strength and mobility, based on only one type of measurement tool to assess each outcome (80). Nevertheless, in a 2011 meta-analysis of 26 randomized controlled trials in 45,782 participants (mean age, 76 years; 78% of women), vitamin D supplementation was found to reduce the risk of suffering from at least one fall by 14% (81). In a more recent randomized, double-blind, placebo-controlled study in 160 postmenopausal women (ages, 50-65 years) with suboptimal vitamin D status (mean serum 25-hydroxyvitamin D concentration <20 ng/mL), supplementation with 1,000 IU/day of vitamin D3 significantly improved vitamin D status, as well as upper and lower limb muscle strength and postural balance parameters (82, 83). The risks of fall and recurrent falls were found to be two- to three-fold greater in women in the control group than in those supplemented with vitamin D3 (83). In contrast, two other recent randomized controlled studies in older community-dwelling adults showed no benefits of high-dose vitamin D (monthly bolus doses corresponding to ≥2,000 IU/day) on odds of falling compared to either a lower daily dose of vitamin D (monthly bolus doses corresponding to 800 IU/day) or a placebo (78, 84). Further, in one of these studies, a 5.5-fold greater odds of falling was found in those reaching the highest (44.7-98.9 ng/mL) compared to the lowest (21.3-30.3 ng/mL) quartile of serum 25-hydroxyvitamin D concentrations after a 12-month follow-up (84). Why bolus doses of vitamin D resulting in serum 25-hydroxyvitamin D ≥45 ng/mL would increase the risk of falling remains unclear (85).
The RDAs set by the US Institute of Medicine were based on achieving circulating 25-hydroxyvitamin D concentrations of 20-50 ng/mL (equivalent to 50-125 nmol/L), considered to be adequate to maintain bone health across all age groups (33). Following a review of the evidence, the American Geriatrics Society (AGS) concluded in a 2014 statement report that serum 25-hydroxyvitamin D3 of ≥25 ng/mL (≥62.5 nmol/L) provided protection against fractures and falls in older adults. Accordingly, AGS advised clinicians to recommend vitamin D supplementation of at least 1,000 IU/day with calcium to all community-dwelling older adults (≥65 years) and older adults under institutionalized care (86). More specifically, AGS recommended a daily average input from all sources (sun, diet, and supplements) of 4,000 IU/day in older adults (86). However, the US Preventive Services Task Force (USPSTF) advises against vitamin D supplementation for fall prevention in community-dwelling older adults without osteoporosis (87). Moreover, there is currently little consensus regarding recommendations of vitamin D and calcium supplementation for the prevention of fractures in older adults (see also Calcium) (31, 32, 85, 86).
Vitamin K
Naturally occurring vitamin K includes phylloquinone (vitamin K1) and a family of molecules called menaquinones (vitamin K2; MKs). The major function of vitamin K is as a cofactor for a specific enzymatic reaction that modifies proteins to a form that facilitates calcium-binding (88). This reaction is catalyzed by γ-glutamylcarboxylase, which enables the carboxylation of the amino acid glutamic acid (Glu) to γ-carboxyglutamic acid (Gla). At least 16 vitamin K-dependent proteins have been identified to date, of which several are present in bone tissue: osteocalcin (also called bone Gla protein) (89), matrix Gla protein (MGP) (89), protein S (90), growth arrest specific protein 6 (Gas6) (91), Gla-rich protein (GRP) (92), and periostin (93). While the role of vitamin K in bone biology is primarily attributed to its function as cofactor in the carboxylation of these Gla-containing proteins, in vitro studies also suggest additional functions in the regulation of bone formation and bone resorption via γ-carboxylation-independent mechanisms (94, 95).
There is observational evidence suggesting that inadequate vitamin K status is associated with osteoarthritis (96) and increased osteoporotic fractures (97). A recent meta-analysis of four prospective cohort studies and one nested case-control study, which followed a total of 80,982 men and women for up to 10 years, found a 22% lower risk of fracture with the highest versus lowest intakes of phylloquinone (98). However, a high intake of phylloquinone — found in green leafy vegetables — is often a marker of a healthy lifestyle. Thus, one cannot exclude the possibility that the association between higher phylloquinone intakes and lower fracture risk might be due to other healthy lifestyle factors/habits (97, 99). Further, a protective effect of phylloquinone supplementation on bone loss has not been confirmed in randomized controlled trials (97, 100, 101).
Menaquinone-4 (MK-4) at therapeutic doses (45 mg/day) is used in Japan for second-line therapy to treat osteoporosis. Accordingly, most of the interventions to date have been conducted in postmenopausal Japanese women (102). A 2015 meta-analysis of 19 intervention studies reported a protective effect of supplemental menaquinones (primarily MK-4 and MK-7) on lumbar and forearm BMD and overall fracture risk in postmenopausal women with osteoporosis but not in those without osteoporosis. However, this result should be taken with caution because half of the intervention studies had methodological issues (selection bias and/or no blinding), especially those conducted in women with osteoporosis (102).
A double-blind, placebo-controlled intervention performed in 2009 observed no effect of either phylloquinone (1 mg/day) or MK-4 (45 mg/day) supplementation on markers of bone turnover or BMD among healthy, postmenopausal women (N=381) receiving calcium and vitamin D supplements (100). In the Postmenopausal Health Study II, the effect of supplemental calcium, vitamin D, and vitamin K (in fortified dairy products) and lifestyle counseling on bone health was examined in healthy, postmenopausal women (103, 104). One hundred fifty women (mean age, 62 years) were randomly assigned to one of four groups: (1) 800 mg calcium and 10 µg vitamin D3 (N=26); (2) 800 mg calcium, 10 µg vitamin D3, and 100 µg phylloquinone (N=26); (3) 800 mg calcium, 10 µg vitamin D3, and 100 µg MK-7 (N=24); or (4) control group receiving no dietary intervention or counseling. Supplemental nutrients were delivered via fortified milk and yogurt, and subjects were advised to consume one portion of each on a daily basis and to attend biweekly counseling sessions during the one-year intervention. BMD significantly increased in all three treatments compared to the control group. Between the three diet groups, a significant effect of phylloquinone or MK-7 on BMD remained only at the lumbar spine (not at hip and total body) after controlling for serum vitamin D and calcium intake. Overall, the positive influence on BMD was attributed to the combined effect of diet and lifestyle changes associated with the intervention, rather than with an isolated effect of phylloquinone or MK-7 (103). Even if phylloquinone and menaquinones have different effects on bone health, there is only limited evidence to suggest that supplementation with one form of vitamin K, rather than the other, would be more effective in preventing age-related bone loss in humans, especially since the body can convert phylloquinone into menaquinone (105).
The US Bone Health & Osteoporosis Foundation does not support the use of vitamin K supplements because the evidence of effectiveness in the prevention of osteoporosis and fracture is too limited and because their use may cause serious adverse effects in individuals at risk of blood clots and those taking blood-thinning medications (106).
Vitamin A
Both vitamin A deficiency and excess can negatively affect skeletal health. Vitamin A deficiency is a major public health concern in developing nations where it is the leading cause of preventable blindness. Early animal studies demonstrated that both vitamin A deficiency and excess at critical periods of development caused bone abnormalities due to impaired osteoclastic and osteoblastic activities (107).
Very high intakes of vitamin A as retinol from animal sources, fortified food, and supplements can be toxic. Accordingly, the US Institute of Medicine set a tolerable upper intake level (UL) for adults at 3,000 μg (3 mg) retinol activity equivalent (RAE) per day (10,000 IU/day) of preformed vitamin A (see Table 1). In animals, vitamin A toxicity (hypervitaminosis A) is associated with poor bone growth, loss of bone mineral content, and increased rate of fractures (23). Case reports in humans have indicated that vitamin A intakes several fold above the UL (≥100,000 IU/day) were associated with hypercalcemia and bone resorption (108-110).
However, there is some concern that long-term vitamin A intakes at levels not much higher than the RDA have a negative effect on bone (23, 111, 112). Some observational evidence suggests that high vitamin A intake (generally in supplement users and at intake levels about half the UL [~1,500 μg RAE/day]) is associated with an increased risk of osteoporosis and hip fracture (113-116). However, more recent results from large prospective cohort studies showed no association between high vitamin A intakes (~1,500 μg RAE/day) and low BMD or risk of fracture (117, 118). Methods to assess vitamin A intake and status are notoriously unreliable (119), and concerns regarding the potential impact of habitual high vitamin A intakes on bone health may require measuring vitamin A status using sensitive markers of body stores (120). At this time, striving for the recommended dietary intake (RDA) of vitamin A (see Table 1) is an important and safe goal for optimizing skeletal health.
Micronutrients needed for collagen formation
We often discuss the mineral aspect of bone, but the organic matrix is also an integral aspect of bone quality and health. Collagen makes up 90% of the organic matrix of bone. Type I collagen fibers twist around each other in a triple helix and become the scaffold upon which minerals are deposited.
Vitamin C
Vitamin C (ascorbic acid) is a required cofactor for the hydroxylation of lysine and proline during collagen synthesis by osteoblasts (121). In guinea pigs, vitamin C deficiency is associated with defective bone matrix production, both quantity and quality (122). Unlike humans and guinea pigs, rats can synthesize ascorbic acid on their own. Using a special strain of rats with a genetic defect in ascorbic acid synthesis (Osteogenic Disorder Shionogi [ODS] rats), researchers can mimic human scurvy by feeding these animals a vitamin C-deficient diet (123). Ascorbic acid-deficient ODS rats have a marked reduction in bone formation with no defect in bone mineralization (124). More specifically, ascorbic acid deficiency impairs collagen synthesis, the hydroxylation of collagenous proline and lysine residues, and osteoblastic adhesion to bone matrix (124).
In observational studies, vitamin C intake and status are inconsistently associated with bone mineral density and fracture risk (23). In addition, data from observational studies with a prospective design (i.e., prospective cohort studies) is scarce. An analysis of data from elderly adults (mean age, 75 years) followed for about 17 years in the Framingham osteoporosis study showed an inverse relationship between total (dietary plus supplemental) intake of vitamin C and the risk of hip fracture or non-vertebral osteoporotic fracture. However, the association ceased to be significant after adjusting for the confounding effect of potassium intakes (125). Another analysis of a younger sub-cohort (4,510 adults; mean age, 59.8 years) from the European Prospective Investigation into Cancer and nutrition-Norfolk study showed no correlations between plasma concentrations of ascorbic acid and heel ultrasound measures — used to quantify BMD. However, an inverse relationship was reported between plasma concentrations of ascorbic acid and risk of hip fracture in men but not in women (126).
A double-blind, placebo-controlled trial was performed with the premise that improving the collagenous bone matrix will enhance the efficacy of mineral supplementation to counteract bone loss (121). Sixty osteopenic women (35 to 55 years of age) received a placebo comprised of calcium and vitamin D (1,000 mg calcium carbonate and 250 IU vitamin D) or this placebo plus CB6Pro (500 mg vitamin C, 75 mg vitamin B6, and 500 mg proline) daily for one year. In contrast to controls receiving calcium plus vitamin D alone, there was no bone loss detected in the spine and femur in the CB6Pro group (118).
Micronutrients involved in homocysteine metabolism
High circulating concentrations of a metabolite known as homocysteine are an independent risk factor for cardiovascular disease (CVD) (see the Health & Disease page on High Homocysteine) and may also be a modifiable risk factor for osteoporotic fracture (23). A link between homocysteine and the skeleton was first noted in studies of hyperhomocysteinemia, a metabolic disorder characterized by exceedingly high concentrations of homocysteine in the plasma and urine. Individuals with hyperhomocysteinemia exhibit numerous skeletal defects, including reduced bone mineral density (BMD) and osteopenia (127). Moreover, in vitro and animal studies suggest that elevated homocysteine concentrations might decrease bone quality and increase bone resorption through various mechanisms, including (i) inhibiting the expression of lysyl oxidase, an enzyme involved in stabilization of collagen fibers; (ii) stimulating osteoclast formation and activity; and (iii) reducing bone blood flow (reviewed in 128).
The effect of more subtle elevations of plasma homocysteine on bone health is more difficult to demonstrate, and observational studies in humans report conflicting results (127, 129). Some report an association between elevated plasma homocysteine and fracture risk (130-132), while others find no relationship (133-135). A meta-analysis of eight prospective cohort studies reported a 4% higher risk of incident fracture with elevated plasma homocysteine (136).
B vitamins
Folate, vitamin B12, and vitamin B6 help regulate circulating homocysteine concentrations; thus, efforts to reduce plasma homocysteine concentrations by meeting recommended intake levels for these vitamins may benefit bone health (129). In addition, because vitamin B6 is a cofactor for lysyl oxidase, vitamin B6 deficiency may limit collagen cross-linking and reduce bone strength (137). A few intervention trials evaluating the homocysteine-lowering effect of B-vitamins on bone health outcomes have been conducted. For instance, 5,522 participants (ages ≥55 years) in the Heart Outcomes Prevention Evaluation (HOPE) 2 trial were randomized to receive daily homocysteine-lowering therapy (2.5 mg folic acid, 50 mg vitamin B6, and 1 mg vitamin B12) or placebo for a mean duration of five years (138). The results showed no significant difference between treatment and placebo on the risk of skeletal fracture. Incidence of fracture was the secondary outcome in the Vitamins to Prevent Strokes (VITATOPS) trial in 8,164 patients who recently suffered a transient ischemic attack (mini-stroke) or a stroke. Compared to placebo, daily consumption of 2 mg folic acid, 25 mg vitamin B6, and 0.5 mg vitamin B12 for 2.8 years did not lead to differences in the number of osteoporotic fractures and hip fractures or the time to first fracture at the end of the post-trial extended follow-up period of 3.4 years (139). Another randomized, double-blind, placebo-controlled intervention — the B-vitamins for the Prevention Of Osteoporotic Fracture (B-PROOF) trial — assessed the effect of supplemental vitamin B12 (0.4 mg/day) and folic acid (0.5 mg/day) on fracture incidence in older adults (both intervention and control arms also received 15 μg [600 IU] vitamin D daily) (140). Results from the B-PROOF trial in 2,919 subjects (≥65 years) with elevated plasma homocysteine (≥12 μmol/L) found that B-vitamin supplementation decreased plasma homocysteine concentrations by a mean 4.4 μmol/L but did not decrease risk of osteoporotic or any fracture compared to placebo (141). Supplementation with B vitamins also failed to increase the time before the first fall and to prevent the age-related decline in physical performance and handgrip strength (142). Finally, a pooled analysis of data from two randomized controlled interventions — the Norwegian Vitamin Trial (NORVIT; 3,749 participants) and the Western Norway B Vitamin Intervention Trial (WENBIT; 3,090 participants) — showed no difference in risk of hip fracture during trials (mean, 3 years) or extended follow-ups (mean, 10 years) between participants assigned to receive either folic acid (0.8 mg/day), vitamin B12 (0.4 mg/day), and vitamin B6 (40 mg); folic acid and vitamin B12; vitamin B6 alone; or a placebo (143). During the extended follow-ups, there was a significant increase in hip fracture risk in groups supplemented with vitamin B6 compared to groups without vitamin B6. However, this result should be considered with caution because these trials conducted in subjects with coronary heart disease were not designed or powered to assess bone fracture incidence (143).
Effects of Lifestyle Factors
It is important to note that the available data on the effects of lifestyle factors on bone health come primarily from observational studies; thus, interpretation of associations is limited by potential confounding. Specifically, factors that are associated with the exposure and have an effect on the outcome under study may not always be identified and accounted for (i.e., a confounder); residual confounding may remain after controlling for the potential (confounding) effect of confounders. For example, a study may find that coffee consumption is associated with lower bone mineral density (BMD); however, because smoking is often associated with coffee consumption, smoking may have confounded (distorted) the observed association between coffee intake and risk of low bone mass.
Smoking
Several meta-analyses have been conducted to assess the relationship between cigarette smoking and bone health. After pooling data from a number of similar studies, there is a consistent, significant reduction in bone mass and increased risk of fracture in smokers compared to non-smokers (144-146). The effects were dose-dependent and had a strong association with age. Beyond the direct effect of tobacco, unhealthy lifestyle habits and low body weight present in smokers may also contribute to the negative impact on bone health (147, 148). Additionally, smoking leads to alterations in hormone (e.g., 1,25-dihydroxyvitamin D3 and estrogen) production and metabolism that could affect bone cell activity and function (147, 148).
The deleterious effects of smoking on bone appear to be reversible. Recent meta-analyses of prospective cohort studies found a significant association between cigarette smoking and risk of hip fracture in current but not in former smokers (149, 150). Thus, efforts to stop smoking are likely to benefit many aspects of general health, including bone health.
Alcohol
Chronic light alcohol intake is associated with higher bone density and lower bone loss over time (16). This level of intake translates to one drink per day for women and two drinks per day for men, with one standard drink containing 10 g of ethanol. The effect of higher alcohol intakes (11-30 g ethanol/day) on BMD is more variable and may depend on age, gender, hormonal status, and type of alcoholic beverage consumed. At the other end of the spectrum, chronic alcoholism has a documented negative effect on bone and increases fracture risk (151). Alcoholics consuming 100 to 200 g of ethanol per day have low bone mineral density, impaired osteoblastic activity, and metabolic abnormalities that compromise bone health (151, 152). In addition, a meta-analysis of 18 prospective cohort studies, including more than 3.5 million participants, reported a 12% lower risk of hip fracture with light alcohol intake (0.01-12.5 g/day of ethanol) and a 71% higher risk of hip fracture with heavy alcohol intake (≥50 g/day of ethanol) compared to no consumption (153). Moderate alcohol consumption (12.6-50 g/day of ethanol) was found to be associated with a lower risk of hip fractures in men but not in women (153).
Coffee
The results of early controlled studies in humans indicated that coffee and caffeine consumption decreased the efficiency of calcium absorption, resulting in a loss of about 4 to 6 mg of calcium per cup of coffee (154, 155). This effect could be offset by as little as one or two tablespoons of milk (156). There is little evidence to suggest detrimental effects of coffee on bone health in populations with adequate calcium intakes (156). Results from observational studies that examined associations between coffee intakes and bone mineral density have been mixed (157-160). Two meta-analyses of observational studies reported no significant associations between coffee intake and risk of hip fracture (161, 162). A third meta-analysis of six case-control and nine prospective cohort studies found no overall association between coffee intake and total fracture, although a subgroup analysis of eight studies showed a 14% increased risk of fracture in women — but not in men — with the highest versus lowest intakes of coffee (163).
Current evidence is scarce to suggest that coffee consumption could increase the risk of bone loss and fracture. Limiting coffee consumption to ≤3 cups/day while ensuring adequate calcium and vitamin D intakes should prevent any potential adverse effects on calcium absorption and bone health (164).
Physical activity
Physical activity is highly beneficial to skeletal health across all stages of bone development. Regular resistance exercise helps to reduce risk of osteoporotic fracture for two reasons: It both directly and indirectly increases bone mass, and it reduces falling risk by improving strength, balance, and coordination (165).
Physical activity increases bone mass because mechanical forces imposed on bone induce an adaptive osteogenic (bone-forming) response. Bone adjusts its strength in proportion to the degree of bone stress (1), and the intensity and novelty of the load, rather than number of repetitions or sets, matter for building bone mass (166). Exercise recommendations for adults to maintain bone health are listed in Table 2. Additional guidance can be found in the 2008 Physical Activity Guidelines for Americans published by the US Department of Health and Human Services.
| Mode | Weight-bearing endurance activities | Tennis, stair climbing, jogging |
|---|---|---|
| Activities that involve jumping | Volleyball, basketball | |
| Resistance exercise | Weight lifting | |
| Intensity | ||
| Moderate to high | ||
| Frequency | Weight-bearing endurance activities | 3-5 times per week |
| Resistance exercise | 2-3 times per week | |
| Duration | 30-60 minutes per day | Combination of weight-bearing endurance activities, activities that involve jumping, and resistance exercise that targets all major muscle groups |
Finally, the ability of the skeleton to respond to physical activity can be either constrained or enabled by nutritional factors. For example, calcium insufficiency diminishes the effectiveness of mechanical loading to increase bone mass, and highly active people who are malnourished are at increased fracture risk (165, 167). Thus, exercise can be detrimental to bone health when the body is not receiving the nutrients it needs to remodel bone tissue in response to physical activity.
Conclusion
Micronutrients play a prominent role in bone health. The emerging theme with supplementation trials seems to be that habitual intake influences the efficacy of the intervention. In other words, correcting a deficiency and meeting the RDAs of micronutrients involved in bone health will improve bone mineral density (BMD) and benefit the skeleton (see Table 1). To realize lasting effects on bone, the intervention must persist throughout a lifetime. At all stages of life, high impact and resistance exercise in conjunction with adequate intakes of nutrients involved in bone health are critical factors in maintaining a healthy skeleton and minimizing age-related bone loss.
There is no consensus regarding supplementation with calcium and vitamin D in older adults as a preventive strategy against osteoporosis, falls, or fractures. Habitual, high intake of vitamin A at doses >1,500 μg RAE/day may negatively impact bone. Although low dietary vitamin K intake is associated with increased fracture risk, randomized controlled trials have generally not supported a direct role for vitamin K supplementation in fracture risk reduction. The other micronutrients important to bone health (phosphorus, fluoride, magnesium, potassium, sodium, and vitamin C) have essential roles in bone, but clinical evidence in support of supplementation beyond recommended levels of intake to improve BMD or reduce fracture incidence is lacking.
Many Americans, especially the elderly, are at risk of deficiencies for several micronutrients (25). Striving for the recommended dietary intakes of nutrients that are critical for bone health is an important and safe goal for optimizing skeletal health and limiting age-related bone loss.
Authors and Reviewers
Originally written in August 2012 by:
Giana Angelo, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in November 2017 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in December 2017 by:
Connie M. Weaver, Ph.D.
Distinguished Professor and Department Head
Department of Nutrition Science
Purdue University
In 2012, this article was underwritten, in part, by a grant from Bayer Consumer Care AG, Basel, Switzerland.
The 2017 update of this article was supported by a grant from Pfizer Inc.
Last updated 4/19/19 Copyright 2012-2026 Linus Pauling Institute
Figure Alternative Text
Figure 1. General pattern of bone development over time
The figure shows a line graph illustrating bone mass development from birth to older adulthood. Bone mass increases throughout childhood and adolescence, stabilizes in early adulthood, and then declines with advanced age, with a sharper decline after menopause. The x-axis is labeled “Age,” and the y-axis is labeled “Bone Mass.” A secondary y-axis on the right is labeled “T-score” from +3 to -3.
The graph is divided into three phases:
- Growth (shown in yellow) – from birth to age 20; the bone mass increases in linear fashion, illustrating accumulation of bone mass
- Modeling/consolidation (shown in blue) – from age 20 to 30; bone mass continues to increase more slowly, peaking around age 30
- Remodeling (shown in green) – age 30 onward; following the peak, the line slopes downward, indicating gradual bone mass throughout adulthood. A downward arrow at age 50 marks menopause, which is associated with a steep decline in bone loss among postmenopausal women.
On the right side of the graph, diagnostic categories based on T-scores are shown: osteopenia as T-scores between -1 and -2.5, and osteoporosis as a T-score below -2.5.
References
- Guyton AC, Hall JE. Textbook of medical physiology. 9th ed. Philadelphia: W.B. Saunders; 1996.
- Heaney RP. Bone biology in health and disease. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed: Lippincott Williams & Wilkins; 2014:1214-1226.
- Nordin BE, Need AG, Chatterton BE, Horowitz M, Morris HA. The relative contributions of age and years since menopause to postmenopausal bone loss. J Clin Endocrinol Metab. 1990;70(1):83-88. (PubMed)
- Heaney RP, Abrams S, Dawson-Hughes B, et al. Peak bone mass. Osteoporos Int. 2000;11(12):985-1009. (PubMed)
- Kanis JA, Melton LJ, 3rd, Christiansen C, Johnston CC, Khaltaev N. The diagnosis of osteoporosis. J Bone Miner Res. 1994;9(8):1137-1141. (PubMed)
- Weaver CM, Gordon CM, Janz KF, et al. The National Osteoporosis Foundation's position statement on peak bone mass development and lifestyle factors: a systematic review and implementation recommendations. Osteoporos Int. 2016;27(4):1281-1386. (PubMed)
- Bonewald LF. The amazing osteocyte. J Bone Miner Res. 2011;26(2):229-238. (PubMed)
- Raisz LG. Bone physiology: bone cells, modeling, and remodeling. In: Holick MF, Dawson-Hughes B, eds. Nutrition and Bone Health. Totowa, N.J.: Humana Press; 2004:43-62.
- Khosla S, Oursler MJ, Monroe DG. Estrogen and the skeleton. Trends Endocrinol Metab. 2012;23(11):576-81. (PubMed)
- Barker ME, Blumsohn A. Human nutrition. In: Geissler C, Powers HJ, eds. Human Nutrition. 12th ed. New York: Churchill Livingstone; 2011:p.473-490.
- Kini U, Nandeesh B. Physiology of bone formation, remodeling, and metabolism. Radionuclide and hybrid bone imaging: Springer; 2012:29-57.
- Heaney RP. The bone-remodeling transient: implications for the interpretation of clinical studies of bone mass change. J Bone Miner Res. 1994;9(10):1515-1523. (PubMed)
- Heaney RP. Constructive interactions among nutrients and bone-active pharmacologic agents with principal emphasis on calcium, phosphorus, vitamin D and protein. J Am Coll Nutr. 2001;20(5 Suppl):403S-409S; discussion 417S-420S. (PubMed)
- Marcus R. Osteoporosis. In: Coulston AM, Boushey C, eds. Nutrition in the Prevention and Treatment of Disease. 2nd ed. Amsterdam; Boston: Academic Press; 2008:853-869.
- Weaver CM. Osteoporosis: the early years. In: Coulston AM, Boushey C, eds. Nutrition in the Prevention and Treatment of Disease. 2nd ed. Amsterdam; Boston: Academic Press; 2008:833-851.
- Tucker KL, Rosen CJ. Prevention and management of osteoporosis. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed: Lippincott Williams & Wilkins; 2014:1227-1244.
- Gafni RI, Baron J. Childhood bone mass acquisition and peak bone mass may not be important determinants of bone mass in late adulthood. Pediatrics. 2007;119 Suppl 2:S131-136. (PubMed)
- Schonau E. The peak bone mass concept: is it still relevant? Pediatr Nephrol. 2004;19(8):825-831. (PubMed)
- Krolner B, Pors Nielsen S. Bone mineral content of the lumbar spine in normal and osteoporotic women: cross-sectional and longitudinal studies. Clin Sci. 1982;62(3):329-336. (PubMed)
- Cheung AM, Adachi JD, Hanley DA, et al. High-resolution peripheral quantitative computed tomography for the assessment of bone strength and structure: a review by the Canadian Bone Strength Working Group. Curr Osteoporos Rep. 2013;11(2):136-146. (PubMed)
- Dawson-Hughes B. Calcium and vitamin D for bone health in adults. In: Holick MF, Dawson-Hughes B, eds. Nutrition and Bone Health. Totowa, N.J.: Humana Press; 2004:197-210.
- Unnanuntana A, Gladnick BP, Donnelly E, Lane JM. The assessment of fracture risk. J Bone Joint Surg Am. 2010;92(3):743-753. (PubMed)
- Ahmadieh H, Arabi A. Vitamins and bone health: beyond calcium and vitamin D. Nut Rev. 2011;69(10):584-598. (PubMed)
- Palacios C. The role of nutrients in bone health, from A to Z. Crit Rev Food Sci Nutr. 2006;46(8):621-628. (PubMed)
- Fulgoni VL, 3rd, Keast DR, Bailey RL, Dwyer J. Foods, fortificants, and supplements: Where do Americans get their nutrients? J Nutr. 2011;141(10):1847-1854. (PubMed)
- Tai V, Leung W, Grey A, Reid IR, Bolland MJ. Calcium intake and bone mineral density: systematic review and meta-analysis. BMJ. 2015;351:h4183. (PubMed)
- Divittorio G, Jackson KL, Chindalore VL, Welker W, Walker JB. Examining the relationship between bone mineral density and fracture risk reduction during pharmacologic treatment of osteoporosis. Pharmacotherapy. 2006;26(1):104-114. (PubMed)
- Bolland MJ, Leung W, Tai V, et al. Calcium intake and risk of fracture: systematic review. BMJ. 2015;351:h4580. (PubMed)
- Zhao JG, Zeng XT, Wang J, Liu L. Association between calcium or vitamin D supplementation and fracture incidence in community-dwelling older adults: a systematic review and meta-analysis. JAMA. 2017;318(24):2466-2482. (PubMed)
- Chung M, Lee J, Terasawa T, Lau J, Trikalinos TA. Vitamin D with or without calcium supplementation for prevention of cancer and fractures: an updated meta-analysis for the U.S. Preventive Services Task Force. Ann Intern Med. 2011;155(12):827-838. (PubMed)
- Moyer VA. Vitamin D and calcium supplementation to prevent fractures in adults: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2013;158(9):691-696. (PubMed)
- Weaver CM, Alexander DD, Boushey CJ, et al. Calcium plus vitamin D supplementation and risk of fractures: an updated meta-analysis from the National Osteoporosis Foundation. Osteoporos Int. 2016;27(1):367-376. (PubMed)
- Food and Nutrition Board, Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D. Washington, D.C. : The National Academies Press; 2011. (The National Academies Press)
- Hunt CD, Johnson LK. Calcium requirements: new estimations for men and women by cross-sectional statistical analyses of calcium balance data from metabolic studies. Am J Clin Nutr. 2007;86(4):1054-1063. (PubMed)
- Qaseem A, Forciea MA, McLean RM, Denberg TD. Treatment of low bone density or osteoporosis to prevent fractures in men and women: a clinical practice guideline update from the American College of Physicians. Ann Intern Med. 2017;166(11):818-839. (PubMed)
- Chung M, Tang AM, Fu Z, Wang DD, Newberry SJ. Calcium intake and cardiovascular disease risk: an updated systematic review and meta-analysis. Ann Intern Med. 2016;165(12):856-866. (PubMed)
- Kopecky SL, Bauer DC, Gulati M, et al. Lack of evidence linking calcium with or without vitamin D supplementation to cardiovascular disease in generally healthy adults: a clinical guideline from the National Osteoporosis Foundation and the American Society for Preventive Cardiology. Ann Intern Med. 2016;165(12):867-868. (PubMed)
- Heaney RP. Sodium, potassium, phosphorus, and magnesium. In: Holick MF, Dawson-Hughes B, eds. Nutrition and Bone Health. Totowa, N.J.: Humana Press; 2004:327-344.
- O'Brien KO, Kerstetter JE, Insogna KL. Phosphorus. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed: Lippincott Williams & Wilkins; 2014:150-158.
- Calvo MS, Tucker KL. Is phosphorus intake that exceeds dietary requirements a risk factor in bone health? Ann N Y Acad Sci. 2013;1301:29-35. (PubMed)
- Lee AW, Cho SS. Association between phosphorus intake and bone health in the NHANES population. Nutr J. 2015;14:28. (PubMed)
- Ringe JD. Fluoride and bone health. In: Holick MF, Dawson-Hughes B, eds. Nutrition and Bone Health. Totowa, N.J.: Humana Press; 2004:345-362.
- Touger-Decker R, Rigassio Radler D, Depaola DP. Nutrition and dental medicine. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed: Lippincott Williams & Wilkins; 2014:1016-1040.
- Farley JR, Wergedal JE, Baylink DJ. Fluoride directly stimulates proliferation and alkaline phosphatase activity of bone-forming cells. Science. 1983;222(4621):330-332. (PubMed)
- Lundy MW, Stauffer M, Wergedal JE, et al. Histomorphometric analysis of iliac crest bone biopsies in placebo-treated versus fluoride-treated subjects. Osteoporos Int. 1995;5(2):115-129. (PubMed)
- Riggs BL, Hodgson SF, O'Fallon WM, et al. Effect of fluoride treatment on the fracture rate in postmenopausal women with osteoporosis. N Engl J Med. 1990;322(12):802-809. (PubMed)
- Vestergaard P, Jorgensen NR, Schwarz P, Mosekilde L. Effects of treatment with fluoride on bone mineral density and fracture risk--a meta-analysis. Osteoporos Int. 2008;19(3):257-268. (PubMed)
- Chachra D, Limeback H, Willett TL, Grynpas MD. The long-term effects of water fluoridation on the human skeleton. J Dent Res. 2010;89(11):1219-1223. (PubMed)
- Yin XH, Huang GL, Lin DR, et al. Exposure to fluoride in drinking water and hip fracture risk: a meta-analysis of observational studies. PLoS One. 2015;10(5):e0126488. (PubMed)
- Castiglioni S, Cazzaniga A, Albisetti W, Maier JA. Magnesium and osteoporosis: current state of knowledge and future research directions. Nutrients. 2013;5(8):3022-3033. (PubMed)
- Fatemi S, Ryzen E, Flores J, Endres DB, Rude RK. Effect of experimental human magnesium depletion on parathyroid hormone secretion and 1,25-dihydroxyvitamin D metabolism. J Clin Endocrinol Metab. 1991;73(5):1067-1072. (PubMed)
- Matsuzaki H, Kajita Y, Miwa M. Magnesium deficiency increases serum fibroblast growth factor-23 levels in rats. Magnes Res. 2013;26(1):18-23. (PubMed)
- Matsuzaki H, Katsumata S, Kajita Y, Miwa M. Magnesium deficiency regulates vitamin D metabolizing enzymes and type II sodium-phosphate cotransporter mRNA expression in rats. Magnes Res. 2013;26(2):83-86. (PubMed)
- Deng X, Song Y, Manson JE, et al. Magnesium, vitamin D status and mortality: results from US National Health and Nutrition Examination Survey (NHANES) 2001 to 2006 and NHANES III. BMC Med. 2013;11:187. (PubMed)
- Farsinejad-Marj M, Saneei P, Esmaillzadeh A. Dietary magnesium intake, bone mineral density and risk of fracture: a systematic review and meta-analysis. Osteoporos Int. 2016;27(4):1389-1399. (PubMed)
- Kunutsor SK, Whitehouse MR, Blom AW, Laukkanen JA. Low serum magnesium levels are associated with increased risk of fractures: a long-term prospective cohort study. Eur J Epidemiol. 2017; 32(7):593-603. (PubMed)
- Stendig-Lindberg G, Tepper R, Leichter I. Trabecular bone density in a two year controlled trial of peroral magnesium in osteoporosis. Magnes Res. 1993;6(2):155-163. (PubMed)
- Abraham GE, Grewal H. A total dietary program emphasizing magnesium instead of calcium. Effect on the mineral density of calcaneous bone in postmenopausal women on hormonal therapy. J Reprod Med. 1990;35(5):503-507. (PubMed)
- Orchard TS, Larson JC, Alghothani N, et al. Magnesium intake, bone mineral density, and fractures: results from the Women's Health Initiative Observational Study. Am J Clin Nutr. 2014;99(4):926-933. (PubMed)
- Navarro-Gonzalez JF, Mora-Fernandez C, Garcia-Perez J. Clinical implications of disordered magnesium homeostasis in chronic renal failure and dialysis. Semin Dial. 2009;22(1):37-44. (PubMed)
- Weaver CM. Calcium. In: Erdman JW, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition. 10th ed. Ames: Wiley-Blackwell; 2012:434-446.
- Frassetto LA, Morris RC, Jr., Sellmeyer DE, Sebastian A. Adverse effects of sodium chloride on bone in the aging human population resulting from habitual consumption of typical American diets. J Nutr. 2008;138(2):419S-422S. (PubMed)
- Bedford JL, Barr SI. Higher urinary sodium, a proxy for intake, is associated with increased calcium excretion and lower hip bone density in healthy young women with lower calcium intakes. Nutrients. 2011;3(11):951-961. (PubMed)
- Heaney RP. Role of dietary sodium in osteoporosis. J Am Coll Nutr. 2006;25(3 Suppl):271S-276S. (PubMed)
- Park SM, Jee J, Joung JY, et al. High dietary sodium intake assessed by 24-hour urine specimen increase urinary calcium excretion and bone resorption marker. J Bone Metab. 2014;21(3):189-194. (PubMed)
- Devine A, Criddle RA, Dick IM, Kerr DA, Prince RL. A longitudinal study of the effect of sodium and calcium intakes on regional bone density in postmenopausal women. Am J Clin Nutr. 1995;62(4):740-745. (PubMed)
- Ilich JZ, Brownbill RA, Coster DC. Higher habitual sodium intake is not detrimental for bones in older women with adequate calcium intake. Eur J Appl Physiol. 2010;109(4):745-755. (PubMed)
- Carbone LD, Barrow KD, Bush AJ, et al. Effects of a low sodium diet on bone metabolism. J Bone Miner Metab. 2005;23(6):506-513. (PubMed)
- Wallace TC, McBurney M, Fulgoni VL, 3rd. Multivitamin/mineral supplement contribution to micronutrient intakes in the United States, 2007-2010. J Am Coll Nutr. 2014;33(2):94-102. (PubMed)
- Lanham-New SA. The balance of bone health: tipping the scales in favor of potassium-rich, bicarbonate-rich foods. J Nutr. 2008;138(1):172s-177s. (PubMed)
- Weaver CM. Potassium and health. Adv Nutr. 2013;4(3):368s-377s. (PubMed)
- Lambert H, Frassetto L, Moore JB, et al. The effect of supplementation with alkaline potassium salts on bone metabolism: a meta-analysis. Osteoporos Int. 2015;26(4):1311-1318. (PubMed)
- ehle S, Hulter HN, Krapf R. Effect of potassium citrate on bone density, microarchitecture, and fracture risk in healthy older adults without osteoporosis: a randomized controlled trial. J Clin Endocrinol Metab. 2013;98(1):207-217. (PubMed)
- Macdonald HM, Black AJ, Aucott L, et al. Effect of potassium citrate supplementation or increased fruit and vegetable intake on bone metabolism in healthy postmenopausal women: a randomized controlled trial. Am J Clin Nutr. 2008;88(2):465-474. (PubMed)
- DeLuca HF. Overview of general physiologic features and functions of vitamin D. Am J Clin Nutr. 2004;80(6 Suppl):1689S-1696S. (PubMed)
- Hill TR, Aspray TJ. The role of vitamin D in maintaining bone health in older people. Ther Adv Musculoskelet Dis. 2017;9(4):89-95. (PubMed)
- Avenell A, Mak JC, O'Connell D. Vitamin D and vitamin D analogues for preventing fractures in post-menopausal women and older men. Cochrane Database Syst Rev. 2014(4):Cd000227. (PubMed)
- Khaw KT, Stewart AW, Waayer D, et al. Effect of monthly high-dose vitamin D supplementation on falls and non-vertebral fractures: secondary and post-hoc outcomes from the randomised, double-blind, placebo-controlled ViDA trial. Lancet Diabetes Endocrinol. 2017;5(6):438-447. (PubMed)
- Annweiler C, Beauchet O. Questioning vitamin D status of elderly fallers and nonfallers: a meta-analysis to address a 'forgotten step'. J Intern Med. 2015;277(1):16-44. (PubMed)
- Rosendahl-Riise H, Spielau U, Ranhoff AH, Gudbrandsen OA, Dierkes J. Vitamin D supplementation and its influence on muscle strength and mobility in community-dwelling older persons: a systematic review and meta-analysis. J Hum Nutr Diet. 2017;30(1):3-15. (PubMed)
- Murad MH, Elamin KB, Abu Elnour NO, et al. Clinical review: The effect of vitamin D on falls: a systematic review and meta-analysis. J Clin Endocrinol Metab. 2011;96(10):2997-3006. (PubMed)
- Cangussu LM, Nahas-Neto J, Orsatti CL, Bueloni-Dias FN, Nahas EA. Effect of vitamin D supplementation alone on muscle function in postmenopausal women: a randomized, double-blind, placebo-controlled clinical trial. Osteoporos Int. 2015;26(10):2413-2421. (PubMed)
- Cangussu LM, Nahas-Neto J, Orsatti CL, et al. Effect of isolated vitamin D supplementation on the rate of falls and postural balance in postmenopausal women fallers: a randomized, double-blind, placebo-controlled trial. Menopause. 2016;23(3):267-274. (PubMed)
- Bischoff-Ferrari HA, Dawson-Hughes B, Orav EJ, et al. Monthly high-dose vitamin D treatment for the prevention of functional decline: a randomized clinical trial. JAMA Intern Med. 2016;176(2):175-183. (PubMed)
- Gallagher JC. Vitamin D and falls - the dosage conundrum. Nat Rev Endocrinol. 2016;12(11):680-684. (PubMed)
- American Geriatrics Society Workgroup on Vitamin D Supplementation for Older Adults. Recommendations abstracted from the American Geriatrics Society consensus statement on vitamin D for prevention of falls and their consequences. J Am Geriatr Soc. 2014;62(1):147-152. (PubMed)
- US Preventive Services Task Force. Interventions to prevent falls in community-dwelling older adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319(16):1696-1704. (PubMed)
- Suttie JW. Vitamin K. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed: Lippincott Williams & Wilkins; 2014:305-316.
- Cancela ML, Laize V, Conceicao N. Matrix Gla protein and osteocalcin: from gene duplication to neofunctionalization. Arch Biochem Biophys. 2014;561:56-63. (PubMed)
- Maillard C, Berruyer M, Serre CM, Dechavanne M, Delmas PD. Protein-S, a vitamin K-dependent protein, is a bone matrix component synthesized and secreted by osteoblasts. Endocrinology. 1992;130(3):1599-1604. (PubMed)
- Nakamura YS, Hakeda Y, Takakura N, et al. Tyro 3 receptor tyrosine kinase and its ligand, Gas6, stimulate the function of osteoclasts. Stem Cells. 1998;16(3):229-238. (PubMed)
- Cancela ML, Conceicao N, Laize V. Gla-rich protein, a new player in tissue calcification? Adv Nutr. 2012;3(2):174-181. (PubMed)
- Bonnet N, Garnero P, Ferrari S. Periostin action in bone. Mol Cell Endocrinol. 2016;432:75-82. (PubMed)
- Atkins GJ, Welldon KJ, Wijenayaka AR, Bonewald LF, Findlay DM. Vitamin K promotes mineralization, osteoblast-to-osteocyte transition, and an anticatabolic phenotype by {gamma}-carboxylation-dependent and -independent mechanisms. Am J Physiol Cell Physiol. 2009;297(6):C1358-1367. (PubMed)
- Yamaguchi M, Weitzmann MN. Vitamin K2 stimulates osteoblastogenesis and suppresses osteoclastogenesis by suppressing NF-kappaB activation. Int J Mol Med. 2011;27(1):3-14. (PubMed)
- Harshman SG, Shea MK. The role of vitamin K in chronic aging diseases: inflammation, cardiovascular disease, and osteoarthritis. Curr Nutr Rep. 2016;5(2):90-98. (PubMed)
- Shea MK, Booth SL. Update on the role of vitamin K in skeletal health. Nutr Rev. 2008;66(10):549-557. (PubMed)
- Hao G, Zhang B, Gu M, et al. Vitamin K intake and the risk of fractures: A meta-analysis. Medicine (Baltimore). 2017;96(17):e6725. (PubMed)
- Booth SL, Charette AM. Vitamin K, oral anticoagulants, and bone health. In: Holick MF, Dawson-Hughes B, eds. Nutrition and Bone Health. Totowa, N.J.: Humana Press; 2004:457-478.
- Binkley N, Harke J, Krueger D, et al. Vitamin K treatment reduces undercarboxylated osteocalcin but does not alter bone turnover, density, or geometry in healthy postmenopausal North American women. J Bone Miner Res. 2009;24(6):983-991. (PubMed)
- Cheung AM, Tile L, Lee Y, et al. Vitamin K supplementation in postmenopausal women with osteopenia (ECKO trial): a randomized controlled trial. PLoS Med. 2008;5(10):e196. (PubMed)
- Huang ZB, Wan SL, Lu YJ, Ning L, Liu C, Fan SW. Does vitamin K2 play a role in the prevention and treatment of osteoporosis for postmenopausal women: a meta-analysis of randomized controlled trials. Osteoporos Int. 2015;26(3):1175-1186. (PubMed)
- Kanellakis S, Moschonis G, Tenta R, et al. Changes in Parameters of bone metabolism in postmenopausal women following a 12-month intervention period using dairy products enriched with calcium, vitamin D, and phylloquinone (vitamin K(1)) or menaquinone-7 (vitamin K (2)): the Postmenopausal Health Study II. Calcif Tissue Int. 2012;90(4):251-262. (PubMed)
- Moschonis G, Kanellakis S, Papaioannou N, Schaafsma A, Manios Y. Possible site-specific effect of an intervention combining nutrition and lifestyle counselling with consumption of fortified dairy products on bone mass: the Postmenopausal Health Study II. J Bone Miner Metab. 2011;29(4):501-506. (PubMed)
- Okano T, Shimomura Y, Yamane M, et al. Conversion of phylloquinone (Vitamin K1) into menaquinone-4 (Vitamin K2) in mice: two possible routes for menaquinone-4 accumulation in cerebra of mice. J Biol Chem. 2008;283(17):11270-11279. (PubMed)
- Tufts Health & Nutrition Letter. Finding new health benefits for vitamin K. Tufts University.
- Ross AC. Vitamin A. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease. 11th ed. Philadelphia: Lippincott Williams & Wilkins; 2014:260-277.
- Binkley N, Krueger D. Hypervitaminosis A and bone. Nutr Rev. 2000;58(5):138-144. (PubMed)
- Hathcock JN, Hattan DG, Jenkins MY, McDonald JT, Sundaresan PR, Wilkening VL. Evaluation of vitamin A toxicity. Am J Clin Nutr. 1990;52(2):183-202. (PubMed)
- McGuire J, Lawson JP. Skeletal changes associated with chronic isotretinoin and etretinate administration. Dermatologica. 1987;175 Suppl 1:169-181. (PubMed)
- Barker ME, Blumsohn A. Is vitamin A consumption a risk factor for osteoporotic fracture? Proc Nutr Soc. 2003;62(4):845-850. (PubMed)
- Genaro Pde S, Martini LA. Vitamin A supplementation and risk of skeletal fracture. Nutr Rev. 2004;62(2):65-67. (PubMed)
- Feskanich D, Singh V, Willett WC, Colditz GA. Vitamin A intake and hip fractures among postmenopausal women. JAMA. 2002;287(1):47-54. (PubMed)
- Melhus H, Michaelsson K, Kindmark A, et al. Excessive dietary intake of vitamin A is associated with reduced bone mineral density and increased risk for hip fracture. Ann Intern Med. 1998;129(10):770-778. (PubMed)
- Michaelsson K, Lithell H, Vessby B, Melhus H. Serum retinol levels and the risk of fracture. N Engl J Med. 2003;348(4):287-294. (PubMed)
- Promislow JH, Goodman-Gruen D, Slymen DJ, Barrett-Connor E. Retinol intake and bone mineral density in the elderly: the Rancho Bernardo Study. J Bone Miner Res. 2002;17(8):1349-1358. (PubMed)
- Caire-Juvera G, Ritenbaugh C, Wactawski-Wende J, Snetselaar LG, Chen Z. Vitamin A and retinol intakes and the risk of fractures among participants of the Women's Health Initiative Observational Study. Am J Clin Nutr. 2009;89(1):323-330. (PubMed)
- de Jonge EA, Kiefte-de Jong JC, Campos-Obando N, et al. Dietary vitamin A intake and bone health in the elderly: the Rotterdam Study. Eur J Clin Nutr. 2015;69(12):1375. (PubMed)
- Ribaya-Mercado JD, Blumberg JB. Vitamin A: is it a risk factor for osteoporosis and bone fracture? Nutr Rev. 2007;65(10):425-438. (PubMed)
- Tanumihardjo SA. Vitamin A and bone health: the balancing act. J Clin Densitom. 2013;16(4):414-419. (PubMed)
- Masse PG, Jougleux JL, C CT, Dosy J, Caissie M, S PC. Enhancement of calcium/vitamin d supplement efficacy by administering concomitantly three key nutrients essential to bone collagen matrix for the treatment of osteopenia in middle-aged women: a one-year follow-up. J Clin Biochem Nutr. 2010;46(1):20-29. (PubMed)
- Poal-Manresa J, Little K, Trueta J. Some observations on the effects of vitamin C deficiency on bone. Br J Exp Pathol. 1970;51(4):372-378. (PubMed)
- Mizushima Y, Harauchi T, Yoshizaki T, Makino S. A rat mutant unable to synthesize vitamin C. Experientia. 1984;40(4):359-361. (PubMed)
- Hasegawa T, Li M, Hara K, et al. Morphological assessment of bone mineralization in tibial metaphyses of ascorbic acid-deficient ODS rats. Biomed Res. 2011;32(4):259-269. (PubMed)
- Sahni S, Hannan MT, Gagnon D, et al. Protective effect of total and supplemental vitamin C intake on the risk of hip fracture--a 17-year follow-up from the Framingham Osteoporosis Study. Osteoporos Int. 2009;20(11):1853-1861. (PubMed)
- Finck H, Hart AR, Lentjes MA, et al. Cross-sectional and prospective associations between dietary and plasma vitamin C, heel bone ultrasound, and fracture risk in men and women in the European Prospective Investigation into Cancer in Norfolk cohort. Am J Clin Nutr. 2015;102(6):1416-1424. (PubMed)
- Herrmann M, Widmann T, Herrmann W. Homocysteine--a newly recognised risk factor for osteoporosis. Clinical chemistry and laboratory medicine : CCLM / FESCC.2005;43(10):1111-1117. (PubMed)
- Behera J, Bala J, Nuru M, Tyagi SC, Tyagi N. Homocysteine as a Pathological Biomarker for Bone Disease. J Cell Physiol. 2017;232(10):2704-2709. (PubMed)
- Levasseur R. Bone tissue and hyperhomocysteinemia. Joint Bone Spine. 2009;76(3):234-240. (PubMed)
- Gjesdal CG, Vollset SE, Ueland PM, Refsum H, Meyer HE, Tell GS. Plasma homocysteine, folate, and vitamin B12 and the risk of hip fracture: the hordaland homocysteine study. J Bone Miner Res. 2007;22(5):747-756. (PubMed)
- van Meurs JB, Dhonukshe-Rutten RA, Pluijm SM, et al. Homocysteine levels and the risk of osteoporotic fracture. N Engl J Med. 2004;350(20):2033-2041. (PubMed)
- McLean RR, Jacques PF, Selhub J, et al. Homocysteine as a predictive factor for hip fracture in older persons. N Engl J Med. 2004;350(20):2042-2049. (PubMed)
- Gerdhem P, Ivaska KK, Isaksson A, et al. Associations between homocysteine, bone turnover, BMD, mortality, and fracture risk in elderly women. J Bone Miner Res. 2007;22(1):127-134. (PubMed)
- Perier MA, Gineyts E, Munoz F, Sornay-Rendu E, Delmas PD. Homocysteine and fracture risk in postmenopausal women: the OFELY study. Osteoporos Int. 2007;18(10):1329-1336. (PubMed)
- Ravaglia G, Forti P, Maioli F, et al. Folate, but not homocysteine, predicts the risk of fracture in elderly persons. J Gerontol A Biol Sci Med Sci. 2005;60(11):1458-1462. (PubMed)
- van Wijngaarden JP, Doets EL, Szczecinska A, et al. Vitamin B12, folate, homocysteine, and bone health in adults and elderly people: a systematic review with meta-analyses. J Nutr Metab. 2013;2013:486186. (PubMed)
- Saito M, Marumo K. Collagen cross-links as a determinant of bone quality: a possible explanation for bone fragility in aging, osteoporosis, and diabetes mellitus. Osteoporos Int. 2010;21(2):195-214. (PubMed)
- Sawka AM, Ray JG, Yi Q, Josse RG, Lonn E. Randomized clinical trial of homocysteine level lowering therapy and fractures. Arch Int Med. 2007;167(19):2136-2139. (PubMed)
- Gommans J, Yi Q, Eikelboom JW, Hankey GJ, Chen C, Rodgers H. The effect of homocysteine-lowering with B-vitamins on osteoporotic fractures in patients with cerebrovascular disease: substudy of VITATOPS, a randomised placebo-controlled trial. BMC Geriatr. 2013;13:88. (PubMed)
- van Wijngaarden JP, Dhonukshe-Rutten RA, van Schoor NM, et al. Rationale and design of the B-PROOF study, a randomized controlled trial on the effect of supplemental intake of vitamin B12 and folic acid on fracture incidence. BMC Geriatr. 2011;11:80. (PubMed)
- van Wijngaarden JP, Swart KM, Enneman AW, et al. Effect of daily vitamin B-12 and folic acid supplementation on fracture incidence in elderly individuals with an elevated plasma homocysteine concentration: B-PROOF, a randomized controlled trial. Am J Clin Nutr. 2014;100(6):1578-1586. (PubMed)
- Swart KM, Ham AC, van Wijngaarden JP, et al. A randomized controlled trial to examine the effect of 2-year vitamin B12 and folic acid supplementation on physical performance, strength, and falling: additional findings from the B-PROOF study. Calcif Tissue Int. 2016;98(1):18-27. (PubMed)
- Garcia Lopez M, Bonaa KH, Ebbing M, et al. B vitamins and hip fracture: secondary analyses and extended follow-up of two large randomized controlled trials. J Bone Miner Res. 2017;32(10):1981-1989. (PubMed)
- Kanis JA, Johnell O, Oden A, et al. Smoking and fracture risk: a meta-analysis. Osteoporos Int. 2005;16(2):155-162. (PubMed)
- Law MR, Hackshaw AK. A meta-analysis of cigarette smoking, bone mineral density and risk of hip fracture: recognition of a major effect. BMJ. 1997;315(7112):841-846. (PubMed)
- Ward KD, Klesges RC. A meta-analysis of the effects of cigarette smoking on bone mineral density. Calcif Tissue Int. 2001;68(5):259-270. (PubMed)
- Wong PK, Christie JJ, Wark JD. The effects of smoking on bone health. Clin Sci. 2007;113(5):233-241. (PubMed)
- Yoon V, Maalouf NM, Sakhaee K. The effects of smoking on bone metabolism. Osteoporos Int. 2012;23(8):2081-2092. (PubMed)
- Shen GS, Li Y, Zhao G, et al. Cigarette smoking and risk of hip fracture in women: a meta-analysis of prospective cohort studies. Injury. 2015;46(7):1333-1340. (PubMed)
- Wu ZJ, Zhao P, Liu B, Yuan ZC. Effect of cigarette smoking on risk of hip fracture in men: a meta-analysis of 14 prospective cohort studies. PLoS One. 2016;11(12):e0168990. (PubMed)
- Maurel DB, Boisseau N, Benhamou CL, Jaffre C. Alcohol and bone: review of dose effects and mechanisms. Osteoporos Int. 2012;23(1):1-16. (PubMed)
- Kiel DP. Smoking, alcohol, and bone health. In: Holick MF, Dawson-Hughes B, eds. Nutrition and Bone Health. Totowa, N.J.: Humana Press; 2004:481-513.
- Zhang X, Yu Z, Yu M, Qu X. Alcohol consumption and hip fracture risk. Osteoporos Int. 2015;26(2):531-542. (PubMed)
- Barger-Lux MJ, Heaney RP. Caffeine and the calcium economy revisited. Osteoporos Int. 1995;5(2):97-102. (PubMed)
- Hasling C, Sondergaard K, Charles P, Mosekilde L. Calcium metabolism in postmenopausal osteoporotic women is determined by dietary calcium and coffee intake. J Nutr. 1992;122(5):1119-1126. (PubMed)
- Heaney RP. Effects of caffeine on bone and the calcium economy. Food Chem Toxicol. 2002;40(9):1263-1270. (PubMed)
- Choi E, Choi KH, Park SM, Shin D, Joh HK, Cho E. The benefit of bone health by drinking coffee among Korean postmenopausal women: a cross-sectional analysis of the Fourth & Fifth Korea National Health and Nutrition Examination Surveys. PLoS One. 2016;11(1):e0147762. (PubMed)
- Hallstrom H, Byberg L, Glynn A, Lemming EW, Wolk A, Michaelsson K. Long-term coffee consumption in relation to fracture risk and bone mineral density in women. Am J Epidemiol. 2013;178(6):898-909. (PubMed)
- Harris SS, Dawson-Hughes B. Caffeine and bone loss in healthy postmenopausal women. Am J Clin Nutr. 1994;60(4):573-578. (PubMed)
- Rapuri PB, Gallagher JC, Kinyamu HK, Ryschon KL. Caffeine intake increases the rate of bone loss in elderly women and interacts with vitamin D receptor genotypes. Am J Clin Nutr. 2001;74(5):694-700. (PubMed)
- Li S, Dai Z, Wu Q. Effect of coffee intake on hip fracture: a meta-analysis of prospective cohort studies. Nutr J. 2015;14:38. (PubMed)
- Sheng J, Qu X, Zhang X, et al. Coffee, tea, and the risk of hip fracture: a meta-analysis. Osteoporos Int. 2014;25(1):141-150. (PubMed)
- Lee DR, Lee J, Rota M, et al. Coffee consumption and risk of fractures: a systematic review and dose-response meta-analysis. Bone. 2014;63:20-28. (PubMed)
- Wikoff D, Welsh BT, Henderson R, et al. Systematic review of the potential adverse effects of caffeine consumption in healthy adults, pregnant women, adolescents, and children. Food Chem Toxicol. 2017;109(Pt 1):585-648. (PubMed)
- Kohrt WM, Bloomfield SA, Little KD, Nelson ME, Yingling VR. American College of Sports Medicine Position Stand: physical activity and bone health. Med Sci Sports Exerc. 2004;36(11):1985-1996. (PubMed)
- Singh MAF. Exercise and bone health. In: Holick MF, Dawson-Hughes B, eds. Nutrition and Bone Health. Totowa, N.J.: Humana Press; 2004:515-548.
- Thompson J, Manore M. Nutrition: an applied approach. 2nd ed. San Francisco, Calif.: Pearson/Benjamin Cummings; 2009.
Disclaimer
The Linus Pauling Institute's Micronutrient Information Center provides scientific information on the health aspects of dietary factors and supplements, food, and beverages for the general public. The information is made available with the understanding that the author and publisher are not providing medical, psychological, or nutritional counseling services on this site. The information should not be used in place of a consultation with a competent health care or nutrition professional.
The information on dietary factors and supplements, food, and beverages contained on this website does not cover all possible uses, actions, precautions, side effects, and interactions. It is not intended as nutritional or medical advice for individual problems. Liability for individual actions or omissions based upon the contents of this site is expressly disclaimed.
You may not copy, modify, distribute, display, transmit, perform, publish or sell any of the copyrightable material on this website. You may hyperlink to this website but must include the following statement:
"This link leads to a website provided by the Linus Pauling Institute at Oregon State University. [Your name] is not affiliated or endorsed by the Linus Pauling Institute or Oregon State University."