Summary
- Lipoic acid (often called α-lipoic acid), also known as thioctic acid, is a naturally occurring compound that is also synthesized by humans. (More information)
- Endogenously synthesized lipoic acid is bound to protein and functions as a cofactor for several important mitochondrial multienzyme complexes. (More information)
- Supplementation with lipoic acid transiently increases plasma and cellular concentrations of unbound lipoic acid. (More information)
- Lipoic acid is a direct antioxidant, although any increase in radical scavenging activity in vivo is likely transient. Lipoic acid may also trigger antioxidant defense, enhance cellular glucose uptake, and modulate the activity of various cell-signaling molecules and transcription factors. (More information)
- Some evidence from small randomized controlled studies suggests that high-dose lipoic acid may improve measures of glucose utilization in subjects with metabolic disorders. (More information)
- Available evidence suggests that treatment with intravenous or oral lipoic acid may help reduce symptoms of diabetic peripheral neuropathy. It is important to note that many of the studies examining the efficacy of lipoic acid for the treatment of diabetic neuropathy have been conducted by one German research group and funded by the manufacturer of lipoic acid in Germany. (More information)
- Lipoic acid was found to slow disease progression when administered to a mouse model of multiple sclerosis. A two-year clinical trial designed to assess the effect of lipoic acid on loss of mobility and changes in brain volume in patients with progressive multiple sclerosis is ongoing. (More information)
- Current evidence is too limited to suggest whether lipoic acid supplementation could benefit patients with Alzheimer’s disease. (More information)
- Supplementation with lipoic acid may show some benefits in terms of weight control for individuals with high body mass index. (More information)
- Lipoic acid occurs naturally in food covalently bound to protein, whereas supplements contain unbound (free) lipoic acid. (More information)
Other Articles
Introduction
Lipoic acid (often called α-lipoic acid), also known as thioctic acid, is a naturally occurring organosulfur compound that is synthesized by plants and animals, including humans (1, 2). Lipoic acid is covalently bound to certain proteins, which function as part of essential mitochondrial multienzyme complexes involved in energy and amino acid metabolism (see Biological Activities). In addition to the physiological functions of protein-bound lipoic acid, there is increasing scientific and medical interest in potential therapeutic uses of pharmacological doses of free (unbound) lipoic acid (3).
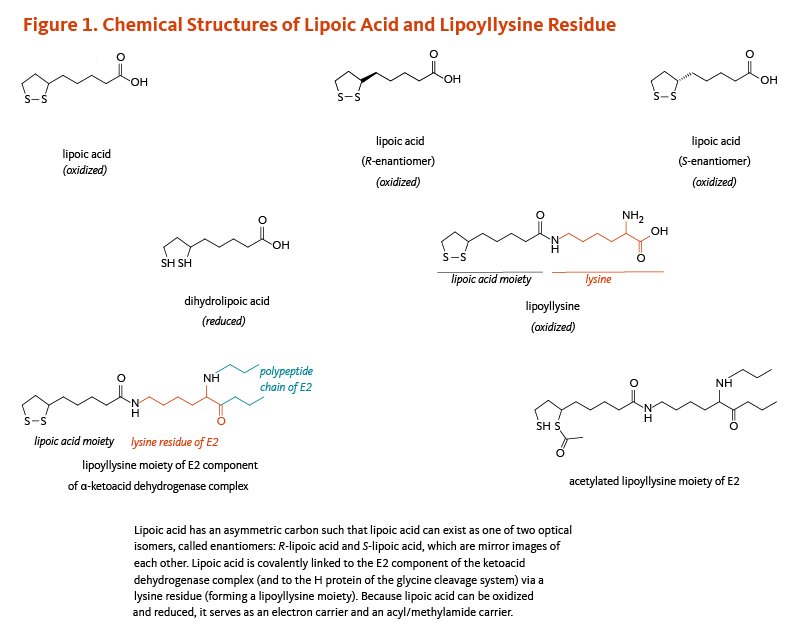
Lipoic acid contains two thiol (sulfur) groups, which may be oxidized or reduced; dihydrolipoic acid is the reduced form of lipoic acid (Figure 1) (4). Lipoic acid also contains an asymmetric carbon, which means that lipoic acid can exist as one of two possible optical isomers, also called enantiomers. These enantiomers are mirror images of each other: R-lipoic acid and S-lipoic acid (Figure 1). Only the R-enantiomer is endogenously synthesized and covalently bound to protein. R-lipoic acid occurs naturally in food (see Food sources). Free (unbound) lipoic acid supplements may contain either R-lipoic acid or a 50:50 (racemic) mixture of R-lipoic acid and S-lipoic acid (see Supplements).
Metabolism and Bioavailability
Endogenous biosynthesis
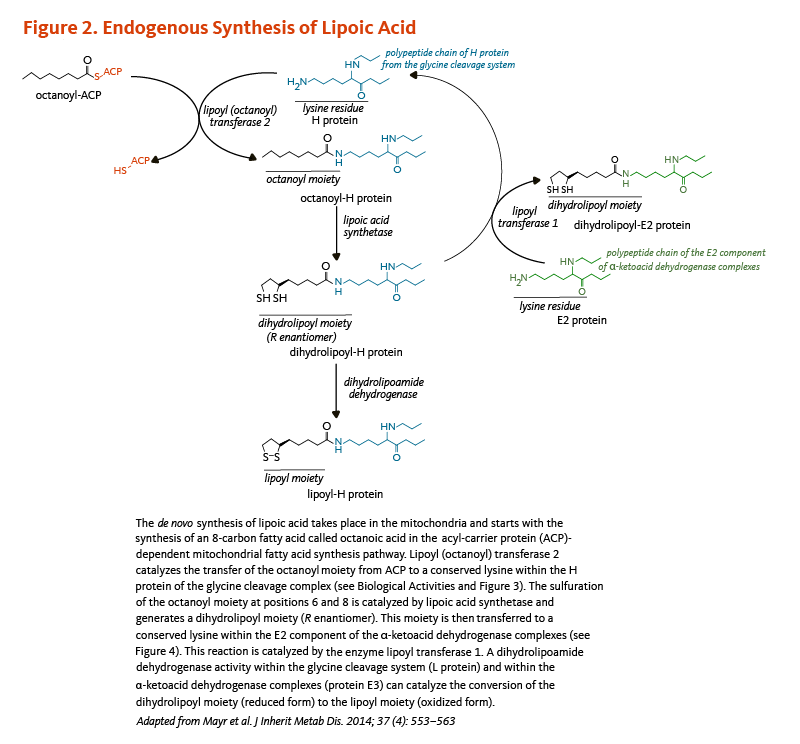
The synthesis of lipoic acid has been characterized in detail in the yeast Saccharomyces cerevisiae, but not all genes involved in the process have been identified in humans (5). Lipoic acid is synthesized de novo in mitochondria from octanoic acid, an 8-carbon fatty acid (C8:0), bound to the acyl-carrier protein (ACP; see article on Pantothenic Acid) during the process of fatty acid synthesis (Figure 2). An enzyme called lipoyl (octanoyl) transferase 2 catalyzes the transfer of the octanoyl moiety from octanoyl-ACP to a conserved lysine of the H protein of the glycine cleavage system (see also Biological Activities). The next reaction is the insertion of two sulfur atoms at positions 6 and 8 of the protein H-bound octanoyl moiety, thereby producing a dihydrolipoyl moiety. This step is catalyzed by the lipoic acid synthetase (also called lipoyl synthase), an enzyme containing iron-sulfur clusters that act as sulfur donors in the reaction (5). Finally, the enzyme lipoyl transferase 1 catalyzes the transfer of the dihydrolipoyl moiety from the H protein of the glycine cleavage system to conserved lysine residues of the E2 components of the α-ketoacid dehydrogenase multienzyme complexes (5). The oxidation of the dihydrolipoyl moiety is catalyzed by a dihydrolipoamide dehydrogenase (Figure 2).
Dietary and supplemental lipoic acid
Consumption of lipoic acid from food has not yet been found to result in detectable increases of free lipoic acid in human plasma or cells (3, 6). In contrast, high oral doses of free lipoic acid (≥50 mg) significantly, yet transiently, increase the concentration of free lipoic acid in plasma and cells. Pharmacokinetic studies in humans have found that about 30%-40% of an oral dose of a racemic mixture of R-lipoic acid and S-lipoic acid is absorbed (6, 7). Oral lipoic acid supplements are better absorbed on an empty stomach than with food: taking lipoic acid with food (versus without food) decreased peak plasma lipoic acid concentrations by about 30% and total plasma lipoic acid concentrations by about 20% (8). A liquid formulation of R-lipoic acid was found to be better absorbed and more stable in the plasma, suggesting that it might be more efficacious than the solid form in the management of a condition like diabetic neuropathy (9, 10).
There may also be differences in bioavailability of the two isomers of lipoic acid. Following single oral doses R,S-lipoic acid (racemic mixture), peak plasma concentrations of R-lipoic acid were found to be 40%-50% higher than S-lipoic acid, suggesting a differential absorption in favor of the R-enantiomer (6, 8, 11). Yet, following oral ingestion, both enantiomers are rapidly metabolized and excreted. Plasma lipoic acid concentrations generally peak within one hour or less and decline rapidly (6, 7, 11, 12). In cells, lipoic acid is swiftly reduced to dihydrolipoic acid, and in vitro studies indicate that dihydrolipoic acid is then rapidly exported from cells (3). Moreover, a pilot study in 19 healthy adults suggested that the bioavailability of R,S-lipoic acid and R-lipoic acid may vary with age and gender (13).
Finally, there is no evidence in humans that exogenous lipoic acid can be 'activated' with ATP or GTP and incorporated into lipoic acid-dependent enzymes by a lipoyl transferase (14). As a consequence, a loss of lipoic acid-dependent enzymatic activity caused by defects in endogenous lipoic acid synthesis (see Deficiency) cannot be rescued by the provision of exogenous lipoic acid (5).
Biological Activities
Protein-bound lipoic acid
Enzyme cofactor
R-lipoic acid is an essential cofactor for several mitochondrial multienzyme complexes that catalyze critical reactions related to the catabolism (breakdown) of amino acids and the production of energy (15). R-lipoic acid is covalently bound to a specific lysine residue in at least one of the proteins in each multienzyme complex. Such a non-protein cofactor is known as a "prosthetic group."
R-lipoic acid functions as a prosthetic group for the biological activity of the following multienzyme complexes:
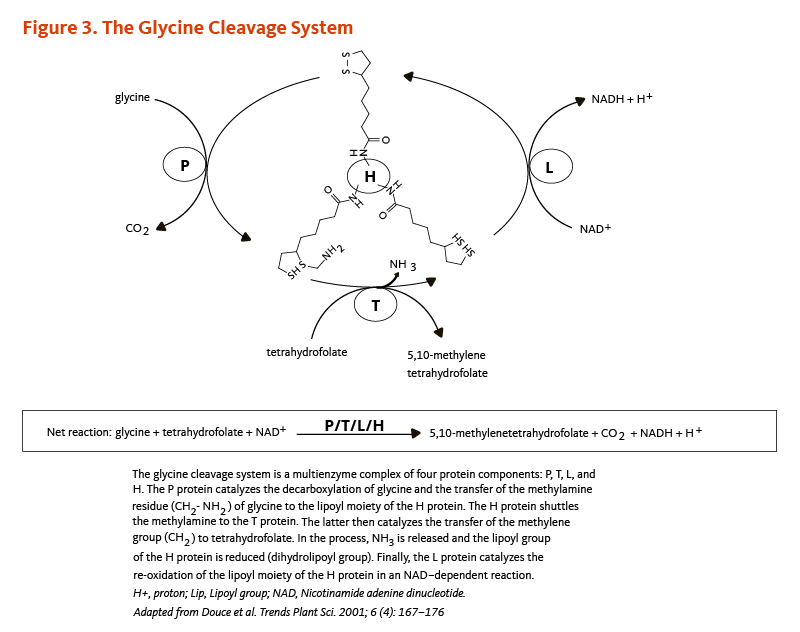
- The glycine cleavage system that catalyzes the decarboxylation of glycine coupled with the addition of a methylene group (-CH2) to tetrahydrofolate to form 5,10-methylene tetrahydrofolate, an important cofactor in the synthesis of nucleic acids (Figure 3). Within the glycine cleavage system, R-lipoic acid is covalently bound to a conserved lysine of the H protein (Figures 2 and 3).
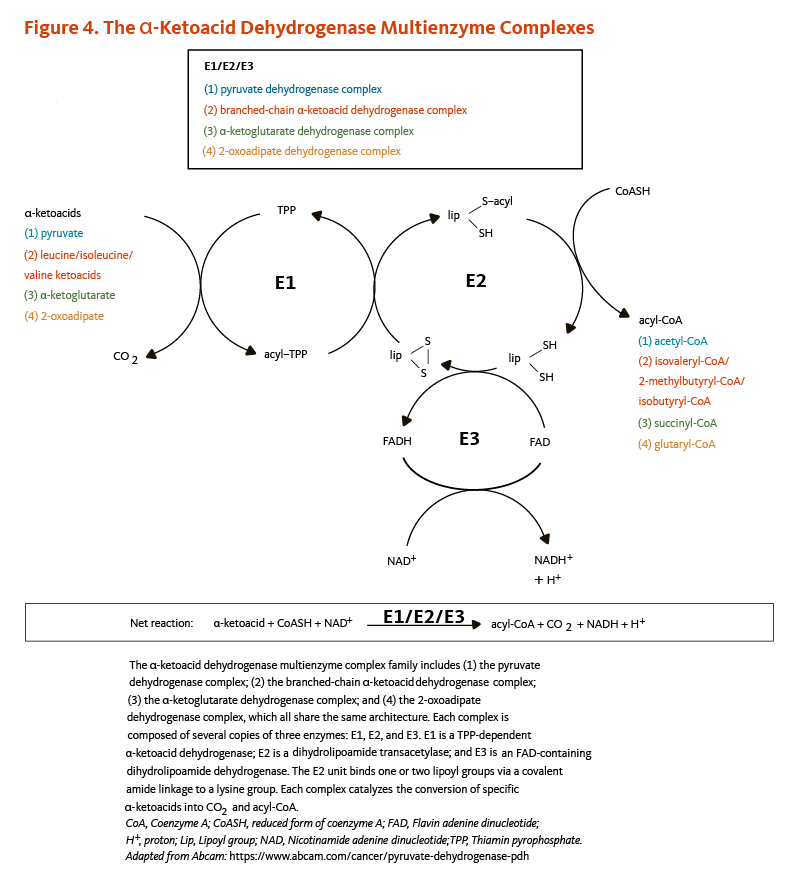
- Four α-ketoacid dehydrogenase complexes (Figure 4), including:
(i) the pyruvate dehydrogenase complex that catalyzes the conversion of pyruvate to acetyl-coenzyme A (CoA), an important substrate for energy production via the citric acid cycle;
(ii) the α-ketoglutarate dehydrogenase complex that catalyzes the conversion of α-ketoglutarate to succinyl CoA, another important intermediate of the citric acid cycle;
(iii) the branched-chain α-ketoacid dehydrogenase complex that is involved in the decarboxylation of ketoacids in the catabolic pathway of the branched-chain amino acids, namely leucine, isoleucine, and valine;
(iv) the 2-oxoadipate dehydrogenase complex that catalyzes the decarboxylation of 2-oxoadipate to glutaryl-CoA in the catabolic pathway of lysine, hydroxylysine, and tryptophan.
All four α-ketoacid dehydrogenase complexes contain three enzymatic activities, namely E1, E2, and E3. E1 is a thiamin pyrophosphate (TPP)-dependent α-ketoacid dehydrogenase, R-lipoic acid functions as a prosthetic group essential for E2 transacetylase activity, and E3 is a flavin adenine dinucleotide (FAD)-dependent dihydrolipoamide dehydrogenase (Figure 4). R-lipoic acid is also found in the E3-binding protein (protein X component) of the pyruvate dehydrogenase complex (5).
Unbound lipoic acid
When considering the biological activities of supplemental (unbound) lipoic acid, it is important to keep in mind the limited and transient nature of the increases in plasma and tissue lipoic acid (see Metabolism and Bioavailability) (3).
Antioxidant activities
Scavenging reactive oxygen and nitrogen species
Reactive oxygen species (ROS) and reactive nitrogen species (RNS) are highly reactive compounds with the potential to damage DNA, proteins, and lipids in cell membranes. Both lipoic acid and dihydrolipoic acid can directly scavenge (neutralize) physiologically relevant ROS and RNS in the test tube (reviewed in 3). However, whether direct quenching reactions occur in vivo is unknown. The highest tissue concentrations of free lipoic acid likely to be achieved through oral supplementation are at least 10 times lower than those of other intracellular antioxidants, such as vitamin C and glutathione. Moreover, free lipoic acid is rapidly eliminated from cells, so any increases in direct radical scavenging activity are unlikely to be sustained.
Regeneration of other antioxidants
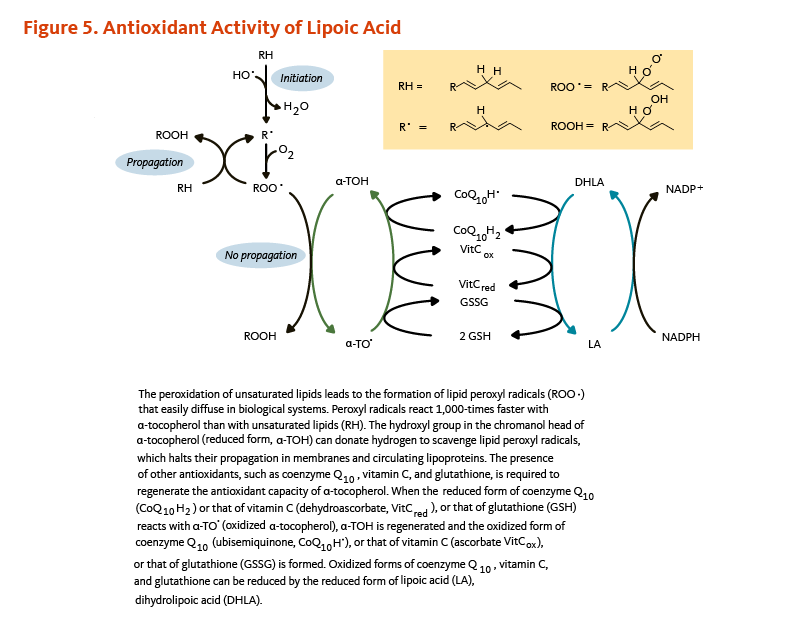
When an antioxidant scavenges a free radical, it becomes oxidized itself and is not able to scavenge additional ROS or RNS until it has been reduced. In the test tube, dihydrolipoic acid is a potent reducing agent with the capacity to reduce the oxidized forms of several important antioxidants, including coenzyme Q10, vitamin C, and glutathione (Figure 5) (16, 17). Dihydrolipoic acid may also reduce the oxidized form of α-tocopherol (vitamin E) directly or indirectly through regenerating oxidized vitamin C (see the article on Vitamin E) (18) or oxidized coenzyme Q10 (see the article on Coenzyme Q10) (19). Whether dihydrolipoic acid effectively regenerates antioxidants under physiological conditions is unclear (3).
Metal chelation
Redox-active metal ions, such as free iron and copper, can induce oxidative damage by catalyzing reactions that generate highly reactive free radicals (20). Compounds that chelate free metal ions in a way that prevents them from generating free radicals offer promise in the treatment of neurodegenerative diseases and other chronic diseases in which metal-induced oxidative damage may play a pathogenic role (21). Both lipoic acid and dihydrolipoic acid have been found to inhibit copper- and iron-mediated oxidative damage in the test tube (22, 23) and to inhibit excess iron and copper accumulation in animal models (24, 25). Lipoic acid may also be helpful as an adjunct treatment against heavy metal toxicity. No clinical trial has examined the use of lipoic acid as a chelating agent in mercury toxicity, yet it has proven to be effective in several mammalian species (26, 27).
Activation of antioxidant signaling pathways
Glutathione is an important intracellular antioxidant that also plays a role in the detoxification and elimination of potential carcinogens and toxins. Reductions in glutathione synthesis and tissue glutathione concentrations in aged animals (compared to younger ones) are suggestive of a potentially lower ability to respond to oxidative stress or toxin exposure (28). Lipoic acid has been found to increase glutathione concentrations in cultured cells and in the tissues of aged animals fed lipoic acid (29, 30). Lipoic acid might be able to increase glutathione synthesis in aged rats by up-regulating the expression of γ-glutamylcysteine ligase (γ-GCL), the rate-limiting enzyme in glutathione synthesis (31), and by increasing cellular uptake of cysteine, an amino acid required for glutathione synthesis (32). Lipoic acid was found to upregulate the expression of γ-GCL and other antioxidant enzymes via the activation of the nuclear factor E2-related factor 2 (Nrf2)-dependent pathway (31, 33).
Briefly, Nrf2 is a transcription factor that is bound to the protein Kelch-like ECH-associated protein 1 (Keap1) in the cytosol. Keap1 responds to oxidative stress signals by freeing Nrf2. Upon release, Nrf2 translocates to the nucleus where it can bind to the antioxidant response element (ARE) located in the promoter region of genes coding for antioxidant enzymes and scavengers. Lipoic acid — but not dihydrolipoic acid — can react with specific sulfhydryl residues of Keap1, causing the release of Nrf2 (34). Nrf2/ARE target genes code for several mediators of the antioxidant response, including γ-GCL, NAD(P)H quinone oxidoreductase 1 (NQO-1), heme oxygenase-1 (HO-1), catalase, and superoxide dismutase (SOD). For example, the upregulation of the Nrf2 pathway by lipoic acid in cultured hepatocytes and in the liver of obese or diabetic rats prevented lipid overload-induced steatosis (35) and cell death (36). Lipoic acid also protected liver from oxidative stress-induced liver injury in methotrexate-treated rats through the activation of Nrf-2 pathway and other anti-inflammatory pathways (37). Pre-treatment and post-treatment with lipoic acid, respectively, prevented and reversed lipopolysaccharide (LPS)-induced lung histopathological alterations in rats through Nrf2-mediated HO-1 upregulation (38).
Inhibition of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (NOX)
NOX is a plasma membrane-bound enzymatic complex that catalyzes the production of superoxide from oxygen and NADPH and has been involved in innate immune defense against microbes (39). Lipoic acid prevented NOX-induced superoxide production in a rat model of cerebral ischemia and limited infarct volume and neurological deficiencies through upregulating the insulin-phosphatidylinositide-3 kinase (PI3K)-protein kinase B (PKB/Akt) signaling pathway (40). Treatment of gastric cancer cells with lipoic acid limited NOX-generated ROS production and reduced cancer cell proliferation induced by Helicobacter pylori (H. pylori) infection (41).
Regulation of cellular glucose uptake
The binding of insulin to the insulin receptor stimulates a cascade of protein phosphorylations leading to the translocation of glucose transporters (GLUT4) to the cell membrane and an increased cellular uptake of glucose (3, 42). Lipoic acid has been found to activate the insulin signaling cascade in cultured cells (3, 42, 43), increase GLUT4 translocation to cell membranes, and increase glucose uptake in cultured adipose and muscle cells (44, 45). A computer modeling study suggested that lipoic acid might bind to the intracellular tyrosine kinase domain of the insulin receptor and stabilize the active form of the enzyme (43).
Regulation of other signaling pathways
In addition to Nrf2 and insulin signaling pathways, lipoic acid was found to target other cell-signaling molecules thereby affecting a variety of cellular processes, including metabolism, stress responses, proliferation, and survival. For example, in cultured endothelial cells, lipoic acid was found to inhibit IKK-β, an enzyme that promotes the translocation of redox-sensitive and pro-inflammatory transcription factor, nuclear factor-kappa B (NFκB) from the cytosol to the nucleus (46). Lipoic acid has also been shown to improve nitric oxide (NO)-dependent vasodilation in aged rats by increasing PKB/Akt-dependent phosphorylation of endothelial NO synthase (eNOS) and eNOS-catalyzed NO production (47). Additionally, lipoic acid increased mitochondrial biogenesis through triggering AMP-activated protein kinase (AMPK)-induced transcription factor PGC-1α activation in skeletal muscle of aged mice (48). Several reviews of the literature have described pathways that are potential targets of lipoic acid in various models and under different experimental conditions (49-52).
Deficiency
Lipoic acid deficiency has been described in rare cases of inherited mutations in the lipoic acid biosynthetic pathway. Mutations identified in patients with defective lipoic acid metabolism affect genes involved in the synthesis of iron-sulfur clusters and genes coding for lipoic acid synthetase (LIAS), lipoyl transferase 1 (LIPT1), and dihydrolipoamide dehydrogenase (E3 component of α-ketoacid dehydrogenase complexes; DLD) (5, 53, 54).
Disease Treatment
Diabetes mellitus
Chronically elevated blood glucose concentration is the hallmark of diabetes mellitus. Type 1 diabetes is caused by the autoimmune destruction of the insulin-producing β-cells of the pancreas, leading to an insufficient production of insulin. Exogenous insulin is required to maintain a normal blood glucose concentration (i.e., fasting blood glucose <100 milligram per deciliter [mg/dL]). In contrast, impaired tissue glucose uptake in response to insulin (a phenomenon called insulin resistance) plays a key role in the development of type 2 diabetes (55). Although patients with type 2 diabetes may eventually require insulin, interventions that enhance insulin sensitivity may be used to maintain normal blood glucose concentrations. The term 'prediabetes' is sometimes used to describe early metabolic abnormalities that place individuals at high risk of developing type 2 diabetes. Of note, these patients are also at high risk for cardiovascular disease. According to the American Diabetes Association, prediabetes can be defined by a condition of impaired fasting glucose, characterized by a fasting blood glucose concentration between 100 mg/dL and 125 mg/dL and/or a condition of impaired glucose tolerance, characterized by a 2-hour blood glucose concentration ≥140 mg/dL following an oral glucose tolerance test (56).
Glucose utilization
The effect of high-dose lipoic acid on glucose utilization has been primarily examined in individuals with type 2 diabetes. An early clinical trial in 13 patients with type 2 diabetes found that a single intravenous infusion of 1,000 mg of lipoic acid improved insulin-stimulated glucose disposal (i.e., insulin sensitivity) by 50% compared to a placebo infusion (57). A placebo-controlled study of 72 patients with type 2 diabetes found that oral administration of lipoic acid at doses of 600 mg/day, 1,200 mg/day or 1,800 mg/day improved insulin sensitivity by 25% after four weeks of treatment (58). There were no significant differences among the three doses of lipoic acid, suggesting that 600 mg/day may be the maximum effective dose (55). However, in a more recent randomized, placebo-controlled study in 102 subjects, daily supplementation with 600 mg of lipoic acid (+/- 800 mg of vitamin E [α-tocopherol]) for 16 weeks had no effect on fasting blood glucose, fasting blood insulin, or a measure of insulin resistance called the homeostatic model assessment of insulin resistance (HOMA-IR) index (59). A 2018 systematic review and meta-analysis identified 20 randomized controlled trials (published between 2007 and 2017) that examined the effect of supplemental lipoic acid on markers of glucose utilization in 1,245 subjects with metabolic disorders (not limited to type 2 diabetes) (60). Administration of lipoic acid (200 to 1,800 mg/day for 2 weeks to 1 year), alone or together with other nutrients, was found to lower fasting blood glucose and insulin concentrations, insulin resistance, and blood HbA1c concentration — a marker of glycemic control over the past few months (60).
Endothelial function
The inner lining of blood vessels, known as the vascular endothelium, plays an important role in the maintenance of cardiovascular health. In particular, nitric oxide (NO) regulates vascular tone and blood flow by promoting the relaxation of all types of blood vessels, including arteries — a phenomenon called vasodilation. Alterations in NO-mediated endothelium-dependent vasodilation results in widespread vasoconstriction and coagulation abnormalities and is considered to be an early step in the development of atherosclerosis. The presence of chronic hyperglycemia, insulin resistance, oxidative stress, and pro-inflammatory mechanisms contribute to endothelial dysfunction in patients with diabetes mellitus (61).
The measurement of brachial flow-mediated dilation (FMD) is often used as a surrogate marker of endothelial function. Two techniques are being used to measure endothelium-dependent vasodilation. One technique measures the forearm blood flow by venous occlusion plethysmography during infusion of acetylcholine. Using this invasive technique, intra-arterial infusion of lipoic acid was found to improve endothelium-dependent vasodilation in 39 subjects with type 2 diabetes but not in 11 healthy controls (62). A more recent randomized, double-blind, placebo-controlled study in 30 patients with type 2 diabetes found that intravenous infusion of 600 mg of lipoic acid improved the response to the endothelium-dependent vasodilator acetylcholine but not to the endothelium-independent vasodilator, glycerol trinitrate (63). Another noninvasive technique using ultrasound to measure flow-mediated vasodilation was used in two additional studies conducted by Xiang et al. (64, 65). The results of these randomized, placebo-controlled studies showed that intravenous lipoic acid could improve endothelial function in patients with impaired fasting glucose (64) or impaired glucose tolerance (65).
One randomized placebo-controlled trial that assessed the effect of oral lipoic acid supplementation in 58 patients diagnosed with metabolic syndrome, a condition characterized by abnormal glucose and lipid metabolism, showed that flow-mediated vasodilation improved by 44% with 300 mg/day of lipoic acid for four weeks (66).
Diabetic neuropathy
Peripheral neuropathy
Up to 50% of diabetic patients develop peripheral neuropathy, a type of nerve damage that may result in pain, loss of sensation, and weakness, particularly in the lower extremities (67). Peripheral neuropathy is also a leading cause of lower limb amputation in diabetic patients (68). Several mechanisms have been proposed to explain chronic hyperglycemia-induced nerve damage, such as intracellular accumulation of sorbitol, glycation reactions, and oxidative and nitrosative stress (reviewed in 69). The results of several large randomized controlled trials indicated that maintaining blood glucose at near normal concentrations was the most important step in limiting the risk of diabetic neuropathy and lower extremity amputation (70-72). However, evidence of the efficacy of enhanced control of glycemia in preventing neuropathy is stronger in patients with type 1 diabetes than in those with type 2 diabetes (73). Moreover, this glucose control intervention increased the risk of hypoglycemic episodes (73).
The efficacy of lipoic acid, administered either intravenously or orally, in the management of neuropathic symptoms has been examined in patients with diabetes. Meta-analyses of randomized controlled trials suggest that infusion of 300 to 600 mg/day of lipoic acid for two to four weeks significantly reduced the symptoms of diabetic neuropathy to a clinically meaningful degree (55, 74). Regarding the efficacy of oral lipoic acid supplementation, an initial short-term study in 24 patients with type 2 diabetes mellitus found that the symptoms of peripheral neuropathy improved in those who took 600 mg of lipoic acid three times a day for three weeks compared to those who took a placebo (75). A larger clinical trial randomly assigned more than 500 patients with type 2 diabetes and symptomatic peripheral neuropathy to one of the following treatments: (i) 600 mg/day of intravenous lipoic acid for three weeks followed by 1,800 mg/day of oral lipoic acid for six months, (ii) 600 mg/day of intravenous lipoic acid for three weeks followed by oral placebo for six months, or (iii) intravenous placebo for three weeks followed by oral placebo for six months (76). Evidence of improvements in sensory and motor deficits — assessed by physicians — could be observed after three weeks of intravenous lipoic acid therapy, yet not at the end of six months of oral lipoic acid therapy. However, another randomized, double-blind, placebo-controlled trial in 181 patients with diabetic neuropathy found that oral supplementation with either 600 mg/day, 1,200 mg/day, or 1,800 mg/day of lipoic acid for five weeks significantly improved neuropathic symptoms (77). In this study, the 600 mg/day dose was as effective as the higher doses. Finally, a four-year, multicenter, clinical trial in 421 diabetic patients with distal symmetric sensorimotor polyneuropathy found no difference between oral administration of 600 mg/day of lipoic and placebo on the primary endpoint, a composite score that assessed neuropathic impairment of the lower limbs and nerve conduction (78). Yet, measures of specific neuropathic impairments (secondary outcomes) improved with lipoic acid supplementation (78). A post-hoc analysis suggested that oral lipoic acid supplementation may reduce neuropathic symptoms particularly in subjects with a high burden of cardiovascular disease, diabetes, and neuropathy yet with normal body mass index (BMI) and blood pressure (79).
Autonomic neuropathy
Another neuropathic complication of diabetes mellitus is cardiac autonomic neuropathy (CAN), which occurs in as many as 25% of diabetic patients (55). CAN is characterized by damage to the nerve fibers that innervate the heart and blood vessels, leading to reduced heart rate variability (variability in the time interval between heartbeats) and increased risk of mortality (80). In a randomized controlled trial of 72 patients with type 2 diabetes and reduced heart rate variability, oral supplementation with 800 mg/day of lipoic acid for four months resulted in significant improvement in two out of four measures of heart rate variability compared to placebo (81).
Overall, the available research suggests that treatment with intravenous or oral lipoic acid may help reduce symptoms of diabetic peripheral neuropathy. The use of lipoic acid is currently approved for the treatment of diabetic neuropathy in Germany (4). It is important to note that many of the studies that examined the efficacy of lipoic acid in the treatment of diabetic neuropathy have been primarily conducted by one German research group and funded by the manufacturer of lipoic acid in Germany (82).
Diabetic retinopathy
Chronic hyperglycemia can damage blood vessels in the retina and cause a potentially sight-threatening condition called diabetic retinopathy (83). One placebo-controlled study examined the effect of lipoic acid on the visual capability of 80 participants of whom 12 had type 1 diabetes, 48 had type 2 diabetes, and 20 were diabetes-free. The result showed that daily oral administration of 300 mg of lipoic acid for three months prevented the deterioration of contrast sensitivity in patients with diabetes and improved it in healthy patients compared to placebo (84).
Multiple sclerosis
Multiple sclerosis is an autoimmune disease of unknown etiology that is characterized by the progressive destruction of myelin and nerve fibers in the central nervous system, causing neurological symptoms in affected individuals (85). There are four main types of multiple sclerosis defined according to the disease course: (i) clinically isolated syndrome, (ii) relapsing-remitting multiple sclerosis, (iii) secondary progressive multiple sclerosis, and (iv) primary progressive multiple sclerosis (for more information, visit the National Multiple Sclerosis Society website) (86). Lipoic acid was found to effectively slow disease progression when administered either orally (87), intraperitoneally (88), or subcutaneously (89) to mice with experimental autoimmune encephalomyelitis (EAE), a model of multiple sclerosis. In vitro and animal studies have found that lipoic acid exhibits immunomodulatory properties through mechanisms that stimulate the production of cyclic AMP (cAMP) (90, 91) — a central regulator of innate immune functions — and inhibits the migration of immune cells into the brain and spinal cord (92), possibly by decreasing endothelial expression of cell adhesion molecules, inhibiting expression of enzymes like matrix metalloproteinases (MMP), and/or reducing the permeability of the blood-brain barrier (87, 89, 93, 94).
Only a few studies have examined lipoic acid supplementation in humans. A small pilot study designed to evaluate the safety of lipoic acid in 30 people with relapsing or progressive multiple sclerosis found that treatment with 1,200 to 2,400 mg/day of oral lipoic acid for two weeks was generally well tolerated (see Safety) (95). In this study, higher serum concentrations of lipoic acid were associated with the lowest serum concentrations of MMP-9 — a marker of inflammation (95). Another study suggested that an oral dose of 1,200 mg of lipoic acid in subjects with multiple sclerosis could help achieve serum lipoic acid concentrations similar to those found to be therapeutic in mice (96). A randomized, placebo-controlled study in 52 subjects (mean age, 30 years) with relapsing-remitting multiple sclerosis found an increase in total antioxidant capacity in blood with lipoic acid supplementation (1,200 mg/day for 12 weeks) yet not in the activity of specific antioxidant enzymes (superoxide dismutase and glutathione peroxidase) (97). Supplemental lipoic acid also decreased the serum concentrations of some (IFN-γ, ICAM-1, TGF-γ, IL-4), but not all markers (TNF-γ, IL-6, MMP-9), cytokines and other inflammation (98). In addition, lipoic acid supplementation did not reduce the severity of multiple sclerosis symptoms, as assessed by the Expanded Disability Status Scale (EDSS) scoring system (98, 99).
A two-year clinical trial designed to assess the effect of lipoic acid (1,200 mg/day) on loss of mobility and changes in brain volume in patients with progressive multiple sclerosis is ongoing (85).
Cognitive impairments and dementia
Studies in animal models of aging and neurodegenerative disease have indicated that lipoic acid administration might improve measures of spatial memory, learning capacity, and/or motor function (reviewed in 100).
It is not known whether oral lipoic acid supplementation can slow cognitive decline related to aging or pathological conditions in humans. An uncontrolled, open-label trial in nine patients with probable Alzheimer’s disease and related dementias, who were also taking acetylcholinesterase inhibitors, reported that oral supplementation with 600 mg/day of lipoic acid appeared to stabilize cognitive function over a one-year period (101). A subsequent study that followed 43 patients for up to four years found those with mild dementia or moderate-early dementia who took lipoic acid (600 mg/day), in addition to acetylcholinesterase inhibitors, experienced slower cognitive decline compared to the typical cognitive decline of Alzheimer’s patients as reported in the literature (102). However, the significance of these findings is difficult to assess without a control group for comparison. A randomized controlled trial found that oral supplementation with 1,200 mg/day of lipoic acid for 10 weeks was of no benefit in treating HIV-associated cognitive impairment (103). The results of another randomized trial in 39 patients with Alzheimer’s disease suggested that supplementation with fish oil concentrate (high in omega-3 fatty acids) with or without lipoic acid (600 mg/day) for one year could delay the progression in cognitive and functional impairments assessed by the Instrumental Activities of Daily Living (IADL) scoring system compared to placebo (104). Interestingly, patients who took fish oil concentrate together with lipoic acid showed no worsening of global cognitive function (as assessed by the Mini-Mental State Examination [MMSE] score system) over 12 months as opposed to those who took either the fish oil concentrate alone or a placebo (104). Larger trials are needed to confirm these preliminary findings and further evaluate the usefulness of supplemental lipoic acid in the prevention and/or management of neurodegenerative diseases.
Weight management
A 2018 meta-analysis of randomized, placebo-controlled trials found that lipoic acid supplementation in those with high body mass index (BMI) resulted in significant, yet modest, reductions in weight (9 studies) and BMI (11 studies) in the absence of caloric restriction (except in one study) (105). Subgroup analyses revealed that weight loss was greater in overweight versus obese participants, in unhealthy versus healthy participants, with daily doses ≤600 mg, and for intervention period shorter than 10 weeks. There was no reduction in waist circumference with supplemental lipoic acid (5 studies) (105). Substantial weight and BMI reductions with lipoic acid supplementation in overweight or obese subjects were also reported in a prior meta-analysis (106).
Sources
Endogenous biosynthesis
R-lipoic acid is synthesized endogenously by humans (see Metabolism and Bioavailability).
Food sources
R-lipoic acid occurs naturally in food covalently bound to lysine in proteins (lipoyllysine; see Figure 1). Although lipoic acid is found in a wide variety of foods from plant and animal sources, quantitative information on the lipoic acid or lipoyllysine content of food is limited; published databases are lacking. Animal tissues with high lipoyllysine content (~1-3 μg/g dry wt) include kidney, heart, and liver, while lipoyllysine-rich vegetables include spinach and broccoli (107). Somewhat lower amounts of lipoyllysine (~0.5 μg/g dry wt) have been measured in tomatoes, peas, and Brussels sprouts.
Supplements
Unlike lipoic acid in foods, lipoic acid in supplements is not bound to protein. Moreover, the amounts of lipoic acid available in dietary supplements (50-600 mg) are likely as much as 1,000 times greater than the amounts that could be obtained from the diet. In Germany, lipoic acid is approved for the treatment of diabetic neuropathies and is available by prescription (108). Lipoic acid is available as a dietary supplement without a prescription in the US. Most lipoic acid supplements contain a racemic mixture of R-lipoic acid and S-lipoic acid (sometimes noted d,l-lipoic acid). Supplements that claim to contain only R-lipoic acid are usually more expensive, and information regarding their purity is not publicly available (109). Since taking lipoic acid with a meal decreases its bioavailability, it is generally recommended that lipoic acid be taken 30 min prior to a meal (see also Metabolism and Bioavailability) (8).
Racemic mixture versus R-lipoic acid only
R-lipoic acid is the isomer that is synthesized by plants and animals and functions as a cofactor for mitochondrial enzymes in its protein-bound form (see Biological Activities). Direct comparisons of the bioavailability of the oral racemic mixture and R-lipoic acid supplements have not been published. Following the ingestion of R,S-lipoic acid, peak plasma concentrations of R-lipoic acid were found to be 40%-50% higher than S-lipoic acid, suggesting better absorption of R-lipoic acid. Both isomers were nonetheless rapidly metabolized and eliminated (6, 8, 11). In rats, R-lipoic acid was more effective than S-lipoic acid in enhancing insulin-stimulated glucose transport and metabolism in skeletal muscle (110), and R-lipoic acid was more effective than R,S-lipoic acid and S-lipoic acid in preventing cataracts (111). However, all of the published human studies have used R,S-lipoic acid (racemic mixture). It has been suggested that the presence of S-lipoic acid in the racemic mixture may limit the polymerization of R-lipoic acid and enhance its bioavailability (52). At present, it remains unclear which supplemental form is best to use in clinical trials.
Safety
Adverse effects
In general, high-dose lipoic acid administration has been found to have few serious side effects. Intravenous administration of lipoic acid at doses of 600 mg/day for three weeks (112) and oral lipoic acid at doses as high as 1,800 mg/day for six months (113) and 1,200 mg/day for two years (76) did not result in serious adverse effects when used to treat diabetic peripheral neuropathy. There was no significant difference in the incidence of adverse events and serious adverse events in patients with diabetic neuropathy who took 600 mg/day of lipoic acid for four years compared to those in the placebo group (78). Oral intake of 2,400 mg/day for two weeks was also found to be safe in a pilot study that included participants with multiple sclerosis (95). Two mild anaphylactoid reactions and one severe anaphylactic reaction, including laryngospasm, were reported after intravenous lipoic acid administration (55). The most frequently reported side effects of oral lipoic acid supplementation are allergic reactions affecting the skin, including rashes, hives, and itching. Abdominal pain, nausea, vomiting, diarrhea, and vertigo have also been reported, and one trial found that the incidence of nausea, vomiting, and vertigo was dose-dependent (77). Further, malodorous urine has been noted by people taking 1,200 mg/day of lipoic acid orally (95).
Pregnancy and lactation
A retrospective observational study reported that daily oral supplementation with 600 mg of lipoic acid (racemic mixture) during pregnancy and without interruption from a period spanning between week 10 and week 30 of gestation and until the end of week 37 was not associated with any adverse effect in mothers and their newborns (114). In absence of further evidence, lipoic acid supplementation during pregnancy should only be considered under strict medical supervision. The safety of lipoic acid supplements in lactating women has not been established and should thus be discouraged (115).
Children
A case of intoxication was reported in a 20-month old child (10.5 kg bw) after the accidental ingestion of four 600-mg tablets of lipoic acid (116). The child was admitted to hospital with seizure, acidosis, and unconsciousness. Symptomatic management and rapid elimination of lipoic acid led to a full recovery without sequelae within five days. The non-accidental ingestion of a very high dose of lipoic acid led to multi-organ failure and subsequent death of an adolescent girl (117).
Drug interactions
In theory, because lipoic acid supplementation may improve insulin-mediated glucose utilization (see Diabetes mellitus), there is a potential risk of hypoglycemia in diabetic patients using insulin or oral anti-diabetic agents (118). Consequently, blood glucose concentrations should be monitored closely when lipoic acid supplementation is added to diabetes treatment regimens. Yet, one study in 24 healthy volunteers reported no significant drug interactions with the co-administration of a single oral dose of lipoic acid (600 mg) and the oral anti-diabetic agents, glyburide (also called glybenclamide) or acarbose (Precose/Prandase/Glucobay) (119).
Nutrient interactions
Biotin
The chemical structure of biotin is similar to that of lipoic acid, and there is some evidence that high concentrations of lipoic acid can compete with biotin for transport across cell membranes (120, 121). The administration of high doses of lipoic acid by injection to rats decreased the activity of two biotin-dependent enzymes by about 30%-35% (122), but it is not known whether oral or intravenous lipoic acid supplementation substantially increases the requirement for biotin in humans (123).
Authors and Reviewers
Originally written in 2002 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in July 2003 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in April 2006 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in January 2012 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in October 2018 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in January 2019 by:
Tory M. Hagen, Ph.D.
Principal Investigator, Linus Pauling Institute
Professor, Dept. of Biochemistry and Biophysics
Helen P. Rumbel Professor for Healthy Aging Research
Oregon State University
Copyright 2002-2026 Linus Pauling Institute
Figure Alternative Text
Figure 1. Chemical structures of lipoic acid and lipoyllysine residue
The figure illustrates the chemical structures of lipoic acid in its oxidized and reduced forms, its two enantiomers (R- and S-lipoic acid), and its covalent attachment to a lysine residue of the E2 component of the α-ketoacid dehydrogenase complex (and the H protein of the glycine cleavage system), forming a lipoyllysine moiety.
Because lipoic acid can undergo reversible oxidation and reduction, it functions as both an electron carrier and an acyl/methylamide carrier. The figure also shows an acetylated lipoyllysine moiety, highlighting its role in acyl group transfer.
Figure 2. Endogenous synthesis of lipoic acid
The figure illustrates the mitochondrial de novo synthesis of lipoic acid, starting with the formation of octanoic acid in the acyl-carrier protein (ACP)-dependent fatty acid synthesis pathway. Lipoyl (octanoyl) transferase 2 catalyzes the transfer of the octanoyl moiety from ACP to a conserved lysine in the H protein of the glycine cleavage complex (see Biological Activities and Figure 3). Lipoic acid synthetase then catalyzes sulfuration at positions 6 and 8 of the octanoyl moiety, forming a dihydrolipoyl moiety (R enantiomer). This moiety is subsequently transferred to a conserved lysine in the E2 component of the α-ketoacid dehydrogenase complexes by lipoyl transferase 1 (see Figure 4). Finally, dihydrolipoamide dehydrogenase activity in the glycine cleavage system (L protein) and in the α-ketoacid dehydrogenase complexes (protein E3) catalyzes the conversion of the dihydrolipoyl moiety (reduced form) to the lipoyl moiety (oxidized form). The figure was adapted from Mayr et al. J Inherit Metab Dis. 2014;37(4):553-563.
Figure 3. The glycine cleavage system
The figure illustrates the glycine cleavage system, a multienzyme complex of four protein components: P, T, L, and H. The P protein catalyzes the decarboxylation of glycine and transfers the methylamine residue (CH2-NH2) to the lipoyl moiety of the H protein. The H protein then shuttles this methylamine group to the T protein, which catalyzes the transfer of the methylene group (CH2) to tetrahydrofolate, forming 5,10-methylenetetrahydrofolate. During this process, NH3 is released and the lipoyl group of the H protein becomes reduced (dihydrolipoyl form). Finally, the L protein catalyzes the re-oxidation of the lipoyl moiety of the H protein in an NAD+-dependent reaction, generating NADH and a proton (H+).
The figure also shows the net reaction: glycine + tetrahydrofolate + NAD+ → 5,10-methylenetetrahydrofolate + CO2 + NADH + H+.
Adapted from Douce et al. Trends Plant Sci. 2001;6(4):167-176.
Figure 4. The α-ketoacid dehydrogenase multienzyme complexes
The α-ketoacid dehydrogenase multienzyme complex family includes (1) the pyruvate dehydrogenase complex; (2) the branched-chain α-ketoacid dehydrogenase complex; (3) the α-ketoglutarate dehydrogenase complex; and (4) the 2-oxoadipate dehydrogenase complex, all of which share the same architecture. Each complex is composed of several copies of three enzymes: E1, E2, and E3. E1 is a TPP-dependent α-ketoacid dehydrogenase, E2 is a dihydrolipoamide transacetylase that binds one or two lipoyl groups via covalent amide linkages to lysine residues, and E3 is an FAD-containing dihydrolipoamide dehydrogenase that re-oxidizes dihydrolipoamide using NAD+. Each complex catalyzes the conversion of specific α-ketoacids into carbon dioxide and acyl-CoA.
The figure also shows the net reaction: α-ketoacid + CoASH (coenzyme A) + NAD+ → acyl-CoA + CO2 + NADH + H+.
Figure 5. Antioxidant activity of lipoic acid
The figure illustrates how lipoic acid supports antioxidant defense by facilitating the regeneration of other antioxidants. Lipid peroxidation of unsaturated fatty acids produces lipid peroxyl radicals (ROO•), which diffuse easily in biological systems. These radicals react about 1,000 times faster with α-tocopherol (vitamin E) than with unsaturated lipids. The hydroxyl group in the chromanol head of α-tocopherol donates a hydrogen atom to neutralize lipid peroxyl radicals, halting their propagation in membranes and circulating lipoproteins. To regenerate α-tocopherol, other antioxidants, including coenzyme Q10, vitamin C, and glutathione, donate electrons. When reduced forms of coenzyme Q10 (CoQ10H2), vitamin C (ascorbate), or glutathione (GSH) reduce oxidized α-tocopherol (α-TO•), they themselves become oxidized (ubisemiquinone, dehydroascorbate, or GSSG). Dihydrolipoic acid (DHLA), the reduced form of lipoic acid, restores these antioxidants by reducing their oxidized forms, maintaining redox balance through antioxidant regeneration.
References
- Reed LJ. A trail of research from lipoic acid to alpha-keto acid dehydrogenase complexes. J Biol Chem. 2001;276(42):38329-38336. (PubMed)
- Carreau JP. Biosynthesis of lipoic acid via unsaturated fatty acids. Methods Enzymol. 1979;62:152-158. (PubMed)
- Smith AR, Shenvi SV, Widlansky M, Suh JH, Hagen TM. Lipoic acid as a potential therapy for chronic diseases associated with oxidative stress. Curr Med Chem. 2004;11(9):1135-1146. (PubMed)
- Kramer K, Packer L. R-alpha-lipoic acid. In: Kramer K, Hoppe P, Packer L, eds. Nutraceuticals in Health and Disease Prevention. New York: Marcel Dekker, Inc.; 2001:129-164.
- Mayr JA, Feichtinger RG, Tort F, Ribes A, Sperl W. Lipoic acid biosynthesis defects. J Inherit Metab Dis. 2014;37(4):553-563. (PubMed)
- Hermann R, Niebch G, Borbe H, et al. Enantioselective pharmacokinetics and bioavailability of different racemic alpha-lipoic acid formulations in healthy volunteers. Eur J Pharm Sci. 1996;4(3):167-174.
- Teichert J, Hermann R, Ruus P, Preiss R. Plasma kinetics, metabolism, and urinary excretion of alpha-lipoic acid following oral administration in healthy volunteers. J Clin Pharmacol. 2003;43(11):1257-1267. (PubMed)
- Gleiter CH, Schug BS, Hermann R, Elze M, Blume HH, Gundert-Remy U. Influence of food intake on the bioavailability of thioctic acid enantiomers. Eur J Clin Pharmacol. 1996;50(6):513-514. (PubMed)
- Brufani M, Figliola R. (R)-alpha-lipoic acid oral liquid formulation: pharmacokinetic parameters and therapeutic efficacy. Acta Biomed. 2014;85(2):108-115. (PubMed)
- Maglione E, Marrese C, Migliaro E, et al. Increasing bioavailability of (R)-alpha-lipoic acid to boost antioxidant activity in the treatment of neuropathic pain. Acta Biomed. 2015;86(3):226-233. (PubMed)
- Breithaupt-Grogler K, Niebch G, Schneider E, et al. Dose-proportionality of oral thioctic acid--coincidence of assessments via pooled plasma and individual data. Eur J Pharm Sci. 1999;8(1):57-65. (PubMed)
- Evans JL, Heymann CJ, Goldfine ID, Gavin LA. Pharmacokinetics, tolerability, and fructosamine-lowering effect of a novel, controlled-release formulation of alpha-lipoic acid. Endocr Pract. 2002;8(1):29-35. (PubMed)
- Keith DJ, Butler JA, Bemer B, et al. Age and gender dependent bioavailability of R- and R,S-alpha-lipoic acid: a pilot study. Pharmacol Res. 2012;66(3):199-206. (PubMed)
- Hiltunen JK, Autio KJ, Schonauer MS, Kursu VA, Dieckmann CL, Kastaniotis AJ. Mitochondrial fatty acid synthesis and respiration. Biochim Biophys Acta. 2010;1797(6-7):1195-1202. (PubMed)
- Bustamante J, Lodge JK, Marcocci L, Tritschler HJ, Packer L, Rihn BH. Alpha-lipoic acid in liver metabolism and disease. Free Radic Biol Med. 1998;24(6):1023-1039. (PubMed)
- Jones W, Li X, Qu ZC, Perriott L, Whitesell RR, May JM. Uptake, recycling, and antioxidant actions of alpha-lipoic acid in endothelial cells. Free Radic Biol Med. 2002;33(1):83-93. (PubMed)
- Kozlov AV, Gille L, Staniek K, Nohl H. Dihydrolipoic acid maintains ubiquinone in the antioxidant active form by two-electron reduction of ubiquinone and one-electron reduction of ubisemiquinone. Arch Biochem Biophys. 1999;363(1):148-154. (PubMed)
- May JM, Qu ZC, Mendiratta S. Protection and recycling of alpha-tocopherol in human erythrocytes by intracellular ascorbic acid. Arch Biochem Biophys. 1998;349(2):281-289. (PubMed)
- Upston JM, Terentis AC, Stocker R. Tocopherol-mediated peroxidation of lipoproteins: implications for vitamin E as a potential antiatherogenic supplement. Faseb J. 1999;13(9):977-994. (PubMed)
- Valko M, Morris H, Cronin MT. Metals, toxicity and oxidative stress. Curr Med Chem. 2005;12(10):1161-1208. (PubMed)
- Doraiswamy PM, Finefrock AE. Metals in our minds: therapeutic implications for neurodegenerative disorders. Lancet Neurol. 2004;3(7):431-434. (PubMed)
- Ou P, Tritschler HJ, Wolff SP. Thioctic (lipoic) acid: a therapeutic metal-chelating antioxidant? Biochem Pharmacol. 1995;50(1):123-126. (PubMed)
- Suh JH, Zhu BZ, deSzoeke E, Frei B, Hagen TM. Dihydrolipoic acid lowers the redox activity of transition metal ions but does not remove them from the active site of enzymes. Redox Rep. 2004;9(1):57-61. (PubMed)
- Suh JH, Moreau R, Heath SH, Hagen TM. Dietary supplementation with (R)-alpha-lipoic acid reverses the age-related accumulation of iron and depletion of antioxidants in the rat cerebral cortex. Redox Rep. 2005;10(1):52-60. (PubMed)
- Yamamoto H, Watanabe T, Mizuno H, et al. The antioxidant effect of DL-alpha-lipoic acid on copper-induced acute hepatitis in Long-Evans Cinnamon (LEC) rats. Free Radic Res. 2001;34(1):69-80. (PubMed)
- Patrick L. Mercury toxicity and antioxidants: Part 1: role of glutathione and alpha-lipoic acid in the treatment of mercury toxicity. Altern Med Rev. 2002;7(6):456-471. (PubMed)
- Rooney JP. The role of thiols, dithiols, nutritional factors and interacting ligands in the toxicology of mercury. Toxicology. 2007;234(3):145-156. (PubMed)
- Hagen TM, Vinarsky V, Wehr CM, Ames BN. (R)-alpha-lipoic acid reverses the age-associated increase in susceptibility of hepatocytes to tert-butylhydroperoxide both in vitro and in vivo. Antioxid Redox Signal. 2000;2(3):473-483. (PubMed)
- Busse E, Zimmer G, Schopohl B, Kornhuber B. Influence of alpha-lipoic acid on intracellular glutathione in vitro and in vivo. Arzneimittelforschung. 1992;42(6):829-831. (PubMed)
- Monette JS, Gomez LA, Moreau RF, et al. (R)-alpha-Lipoic acid treatment restores ceramide balance in aging rat cardiac mitochondria. Pharmacol Res. 2011;63(1):23-29. (PubMed)
- Suh JH, Shenvi SV, Dixon BM, et al. Decline in transcriptional activity of Nrf2 causes age-related loss of glutathione synthesis, which is reversible with lipoic acid. Proc Natl Acad Sci U S A. 2004;101(10):3381-3386. (PubMed)
- Suh JH, Wang H, Liu RM, Liu J, Hagen TM. (R)-alpha-lipoic acid reverses the age-related loss in GSH redox status in post-mitotic tissues: evidence for increased cysteine requirement for GSH synthesis. Arch Biochem Biophys. 2004;423(1):126-135. (PubMed)
- Zhang J, Zhou X, Wu W, Wang J, Xie H, Wu Z. Regeneration of glutathione by alpha-lipoic acid via Nrf2/ARE signaling pathway alleviates cadmium-induced HepG2 cell toxicity. Environ Toxicol Pharmacol. 2017;51:30-37. (PubMed)
- Fratantonio D, Speciale A, Molonia MS, et al. Alpha-lipoic acid, but not di-hydrolipoic acid, activates Nrf2 response in primary human umbilical-vein endothelial cells and protects against TNF-alpha induced endothelium dysfunction. Arch Biochem Biophys. 2018;655:18-25. (PubMed)
- Sena CM, Cipriano MA, Botelho MF, Seica RM. Lipoic acid prevents high-fat diet-induced hepatic steatosis in Goto Kakizaki rats by reducing oxidative stress through Nrf2 activation. Int J Mol Sci. 2018;19(9). (PubMed)
- Pilar Valdecantos M, Prieto-Hontoria PL, Pardo V, et al. Essential role of Nrf2 in the protective effect of lipoic acid against lipoapoptosis in hepatocytes. Free Radic Biol Med. 2015;84:263-278. (PubMed)
- Fayez AM, Zakaria S, Moustafa D. Alpha lipoic acid exerts antioxidant effect via Nrf2/HO-1 pathway activation and suppresses hepatic stellate cells activation induced by methotrexate in rats. Biomed Pharmacother. 2018;105:428-433. (PubMed)
- Lin YC, Lai YS, Chou TC. The protective effect of alpha-lipoic Acid in lipopolysaccharide-induced acute lung injury is mediated by heme oxygenase-1. Evid Based Complement Alternat Med. 2013;2013:590363. (PubMed)
- Segal AW. The function of the NADPH oxidase of phagocytes and its relationship to other NOXs in plants, invertebrates, and mammals. Int J Biochem Cell Biol. 2008;40(4):604-618. (PubMed)
- Dong Y, Wang H, Chen Z. Alpha-lipoic acid attenuates cerebral ischemia and reperfusion injury via insulin receptor and PI3K/Akt-dependent inhibition of NADPH oxidase. Int J Endocrinol. 2015;2015:903186. (PubMed)
- Byun E, Lim JW, Kim JM, Kim H. alpha-Lipoic acid inhibits Helicobacter pylori-induced oncogene expression and hyperproliferation by suppressing the activation of NADPH oxidase in gastric epithelial cells. Mediators Inflamm. 2014;2014:380830. (PubMed)
- Konrad D. Utilization of the insulin-signaling network in the metabolic actions of alpha-lipoic acid-reduction or oxidation? Antioxid Redox Signal. 2005;7(7-8):1032-1039. (PubMed)
- Diesel B, Kulhanek-Heinze S, Holtje M, et al. Alpha-lipoic acid as a directly binding activator of the insulin receptor: protection from hepatocyte apoptosis. Biochemistry. 2007;46(8):2146-2155. (PubMed)
- Estrada DE, Ewart HS, Tsakiridis T, et al. Stimulation of glucose uptake by the natural coenzyme alpha-lipoic acid/thioctic acid: participation of elements of the insulin signaling pathway. Diabetes. 1996;45(12):1798-1804. (PubMed)
- Yaworsky K, Somwar R, Ramlal T, Tritschler HJ, Klip A. Engagement of the insulin-sensitive pathway in the stimulation of glucose transport by alpha-lipoic acid in 3T3-L1 adipocytes. Diabetologia. 2000;43(3):294-303. (PubMed)
- Ying Z, Kampfrath T, Sun Q, Parthasarathy S, Rajagopalan S. Evidence that alpha-lipoic acid inhibits NF-kappaB activation independent of its antioxidant function. Inflamm Res. 2011;60(3):219-225. (PubMed)
- Smith AR, Hagen TM. Vascular endothelial dysfunction in aging: loss of Akt-dependent endothelial nitric oxide synthase phosphorylation and partial restoration by (R)-alpha-lipoic acid. Biochem Soc Trans. 2003;31(Pt 6):1447-1449. (PubMed)
- Wang Y, Li X, Guo Y, Chan L, Guan X. alpha-Lipoic acid increases energy expenditure by enhancing adenosine monophosphate-activated protein kinase-peroxisome proliferator-activated receptor-gamma coactivator-1alpha signaling in the skeletal muscle of aged mice. Metabolism. 2010;59(7):967-976. (PubMed)
- Moura FA, de Andrade KQ, dos Santos JC, Goulart MO. Lipoic acid: its antioxidant and anti-inflammatory role and clinical applications. Curr Top Med Chem. 2015;15(5):458-483. (PubMed)
- Packer L, Cadenas E. Lipoic acid: energy metabolism and redox regulation of transcription and cell signaling. J Clin Biochem Nutr. 2011;48(1):26-32. (PubMed)
- Rochette L, Ghibu S, Richard C, Zeller M, Cottin Y, Vergely C. Direct and indirect antioxidant properties of alpha-lipoic acid and therapeutic potential. Mol Nutr Food Res. 2013;57(1):114-125. (PubMed)
- Shay KP, Moreau RF, Smith EJ, Smith AR, Hagen TM. Alpha-lipoic acid as a dietary supplement: molecular mechanisms and therapeutic potential. Biochim Biophys Acta. 2009;1790(10):1149-1160. (PubMed)
- Mayr JA, Zimmermann FA, Fauth C, et al. Lipoic acid synthetase deficiency causes neonatal-onset epilepsy, defective mitochondrial energy metabolism, and glycine elevation. Am J Hum Genet. 2011;89(6):792-797. (PubMed)
- Tort F, Ferrer-Cortes X, Thio M, et al. Mutations in the lipoyltransferase LIPT1 gene cause a fatal disease associated with a specific lipoylation defect of the 2-ketoacid dehydrogenase complexes. Hum Mol Genet. 2014;23(7):1907-1915. (PubMed)
- Ziegler D. Thioctic acid for patients with symptomatic diabetic polyneuropathy: a critical review. Treat Endocrinol. 2004;3(3):173-189. (PubMed)
- Nathan DM, Davidson MB, DeFronzo RA, et al. Impaired fasting glucose and impaired glucose tolerance: implications for care. Diabetes Care. 2007;30(3):753-759. (PubMed)
- Jacob S, Henriksen EJ, Schiemann AL, et al. Enhancement of glucose disposal in patients with type 2 diabetes by alpha-lipoic acid. Arzneimittelforschung. 1995;45(8):872-874. (PubMed)
- Jacob S, Rett K, Henriksen EJ, Haring HU. Thioctic acid--effects on insulin sensitivity and glucose-metabolism. Biofactors. 1999;10(2-3):169-174. (PubMed)
- de Oliveira AM, Rondo PH, Luzia LA, D'Abronzo FH, Illison VK. The effects of lipoic acid and alpha-tocopherol supplementation on the lipid profile and insulin sensitivity of patients with type 2 diabetes mellitus: a randomized, double-blind, placebo-controlled trial. Diabetes Res Clin Pract. 2011;92(2):253-260. (PubMed)
- Akbari M, Ostadmohammadi V, Lankarani KB, et al. The effects of alpha-lipoic acid supplementation on glucose control and lipid profiles among patients with metabolic diseases: A systematic review and meta-analysis of randomized controlled trials. Metabolism. 2018;87:56-69. (PubMed)
- Roberts AC, Porter KE. Cellular and molecular mechanisms of endothelial dysfunction in diabetes. Diab Vasc Dis Res. 2013;10(6):472-482. (PubMed)
- Heitzer T, Finckh B, Albers S, Krohn K, Kohlschutter A, Meinertz T. Beneficial effects of alpha-lipoic acid and ascorbic acid on endothelium-dependent, nitric oxide-mediated vasodilation in diabetic patients: relation to parameters of oxidative stress. Free Radic Biol Med. 2001;31(1):53-61. (PubMed)
- Heinisch BB, Francesconi M, Mittermayer F, et al. Alpha-lipoic acid improves vascular endothelial function in patients with type 2 diabetes: a placebo-controlled randomized trial. Eur J Clin Invest. 2010;40(2):148-154. (PubMed)
- Xiang G, Pu J, Yue L, Hou J, Sun H. alpha-lipoic acid can improve endothelial dysfunction in subjects with impaired fasting glucose. Metabolism. 2011;60(4):480-485. (PubMed)
- Xiang GD, Sun HL, Zhao LS, Hou J, Yue L, Xu L. The antioxidant alpha-lipoic acid improves endothelial dysfunction induced by acute hyperglycaemia during OGTT in impaired glucose tolerance. Clin Endocrinol (Oxf). 2008;68(5):716-723. (PubMed)
- Sola S, Mir MQ, Cheema FA, et al. Irbesartan and lipoic acid improve endothelial function and reduce markers of inflammation in the metabolic syndrome: results of the Irbesartan and Lipoic Acid in Endothelial Dysfunction (ISLAND) study. Circulation. 2005;111(3):343-348. (PubMed)
- National Institute of Diabetes and Digestive and Kidney Diseases. Diabetic Neuropathy. Available at: https://www.niddk.nih.gov/health-information/diabetes/overview/preventing-problems/nerve-damage-diabetic-neuropathies. Accessed 9/23/18.
- Malik RA, Tesfaye S, Ziegler D. Medical strategies to reduce amputation in patients with type 2 diabetes. Diabet Med. 2013;30(8):893-900. (PubMed)
- Obrosova IG. Diabetes and the peripheral nerve. Biochim Biophys Acta. 2009;1792(10):931-940. (PubMed)
- Dy SM, Bennett WL, Sharma R, et al. AHRQ Comparative Effectiveness Reviews. Preventing complications and treating symptoms of diabetic peripheral neuropathy. Rockville (MD): Agency for Healthcare Research and Quality (US); 2017. (PubMed)
- The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med. 1993;329(14):977-986. (PubMed)
- UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352(9131):837-853. (PubMed)
- Callaghan BC, Little AA, Feldman EL, Hughes RA. Enhanced glucose control for preventing and treating diabetic neuropathy. Cochrane Database Syst Rev. 2012(6):Cd007543. (PubMed)
- Han T, Bai J, Liu W, Hu Y. A systematic review and meta-analysis of alpha-lipoic acid in the treatment of diabetic peripheral neuropathy. Eur J Endocrinol. 2012;167(4):465-471. (PubMed)
- Ruhnau KJ, Meissner HP, Finn JR, et al. Effects of 3-week oral treatment with the antioxidant thioctic acid (alpha-lipoic acid) in symptomatic diabetic polyneuropathy. Diabet Med. 1999;16(12):1040-1043. (PubMed)
- Ziegler D, Hanefeld M, Ruhnau KJ, et al. Treatment of symptomatic diabetic polyneuropathy with the antioxidant alpha-lipoic acid: a 7-month multicenter randomized controlled trial (ALADIN III Study). ALADIN III Study Group. Alpha-lipoic acid in diabetic neuropathy. Diabetes Care. 1999;22(8):1296-1301. (PubMed)
- Ziegler D, Ametov A, Barinov A, et al. Oral treatment with alpha-lipoic acid improves symptomatic diabetic polyneuropathy: the SYDNEY 2 trial. Diabetes Care. 2006;29(11):2365-2370. (PubMed)
- Ziegler D, Low PA, Litchy WJ, et al. Efficacy and safety of antioxidant treatment with alpha-lipoic acid over 4 years in diabetic polyneuropathy: the NATHAN 1 trial. Diabetes Care. 2011;34(9):2054-2060. (PubMed)
- Ziegler D, Low PA, Freeman R, Tritschler H, Vinik AI. Predictors of improvement and progression of diabetic polyneuropathy following treatment with alpha-lipoic acid for 4 years in the NATHAN 1 trial. J Diabetes Complications. 2016;30(2):350-356. (PubMed)
- Balcioglu AS, Muderrisoglu H. Diabetes and cardiac autonomic neuropathy: Clinical manifestations, cardiovascular consequences, diagnosis and treatment. World J Diabetes. 2015;6(1):80-91. (PubMed)
- Ziegler D, Schatz H, Conrad F, Gries FA, Ulrich H, Reichel G. Effects of treatment with the antioxidant alpha-lipoic acid on cardiac autonomic neuropathy in NIDDM patients. A 4-month randomized controlled multicenter trial (DEKAN Study). Deutsche Kardiale Autonome Neuropathie. Diabetes Care. 1997;20(3):369-373. (PubMed)
- Nguyen N, Takemoto JK. A case for alpha-lipoic acid as an alternative treatment for diabetic polyneuropathy. J Pharm Pharm Sci. 2018;21(1s):177s-191s. (PubMed)
- National Institute of Diabetes and Digestive and Kidney Diseases. Diabetic Eye Disease. Available at: https://www.niddk.nih.gov/health-information/diabetes/overview/preventing-problems/diabetic-eye-disease. Accessed 9/24/18.
- Gebka A, Serkies-Minuth E, Raczynska D. Effect of the administration of alpha-lipoic acid on contrast sensitivity in patients with type 1 and type 2 diabetes. Mediators Inflamm. 2014;2014:131538. (PubMed)
- National Multiple Sclerosis Society. Definition of Multiple Sclerosis (MS). Available at: https://www.nationalmssociety.org/What-is-MS/Definition-of-MS. Accessed 9/28/18.
- National Multiple Sclerosis Society. Types of Multiple Sclerosis (MS). Available at: https://www.nationalmssociety.org/What-is-MS/Types-of-MS. Accessed 9/28/18.
- Marracci GH, Jones RE, McKeon GP, Bourdette DN. Alpha lipoic acid inhibits T cell migration into the spinal cord and suppresses and treats experimental autoimmune encephalomyelitis. J Neuroimmunol. 2002;131(1-2):104-114. (PubMed)
- Morini M, Roccatagliata L, Dell'Eva R, et al. Alpha-lipoic acid is effective in prevention and treatment of experimental autoimmune encephalomyelitis. J Neuroimmunol. 2004;148(1-2):146-153. (PubMed)
- Schreibelt G, Musters RJ, Reijerkerk A, et al. Lipoic acid affects cellular migration into the central nervous system and stabilizes blood-brain barrier integrity. J Immunol. 2006;177(4):2630-2637. (PubMed)
- Salinthone S, Schillace RV, Marracci GH, Bourdette DN, Carr DW. Lipoic acid stimulates cAMP production via the EP2 and EP4 prostanoid receptors and inhibits IFN gamma synthesis and cellular cytotoxicity in NK cells. J Neuroimmunol. 2008;199(1-2):46-55. (PubMed)
- Schillace RV, Pisenti N, Pattamanuch N, et al. Lipoic acid stimulates cAMP production in T lymphocytes and NK cells. Biochem Biophys Res Commun. 2007;354(1):259-264. (PubMed)
- George JD, Kim E, Spain R, Bourdette D, Salinthone S. Effects of lipoic acid on migration of human B cells and monocyte-enriched peripheral blood mononuclear cells in relapsing remitting multiple sclerosis. J Neuroimmunol. 2018;315:24-27. (PubMed)
- Chaudhary P, Marracci GH, Bourdette DN. Lipoic acid inhibits expression of ICAM-1 and VCAM-1 by CNS endothelial cells and T cell migration into the spinal cord in experimental autoimmune encephalomyelitis. J Neuroimmunol. 2006;175(1-2):87-96. (PubMed)
- Marracci GH, McKeon GP, Marquardt WE, Winter RW, Riscoe MK, Bourdette DN. Alpha lipoic acid inhibits human T-cell migration: implications for multiple sclerosis. J Neurosci Res. 2004;78(3):362-370. (PubMed)
- Yadav V, Marracci G, Lovera J, et al. Lipoic acid in multiple sclerosis: a pilot study. Mult Scler. 2005;11(2):159-165. (PubMed)
- Yadav V, Marracci GH, Munar MY, et al. Pharmacokinetic study of lipoic acid in multiple sclerosis: comparing mice and human pharmacokinetic parameters. Mult Scler. 2010;16(4):387-397. (PubMed)
- Khalili M, Eghtesadi S, Mirshafiey A, et al. Effect of lipoic acid consumption on oxidative stress among multiple sclerosis patients: a randomized controlled clinical trial. Nutr Neurosci. 2014;17(1):16-20. (PubMed)
- Khalili M, Azimi A, Izadi V, et al. Does lipoic acid consumption affect the cytokine profile in multiple sclerosis patients: a double-blind, placebo-controlled, randomized clinical trial. Neuroimmunomodulation. 2014;21(6):291-296. (PubMed)
- Khalili M, Soltani M, Moghadam SA, Dehghan P, Azimi A, Abbaszadeh O. Effect of alpha-lipoic acid on asymmetric dimethylarginine and disability in multiple sclerosis patients: A randomized clinical trial. Electron Physician. 2017;9(7):4899-4905. (PubMed)
- Molz P, Schroder N. Potential therapeutic effects of lipoic acid on memory deficits related to aging and neurodegeneration. Front Pharmacol. 2017;8:849. (PubMed)
- Hager K, Marahrens A, Kenklies M, Riederer P, Munch G. Alpha-lipoic acid as a new treatment option for Azheimer type dementia. Arch Gerontol Geriatr. 2001;32(3):275-282. (PubMed)
- Hager K, Kenklies M, McAfoose J, Engel J, Munch G. Alpha-lipoic acid as a new treatment option for Alzheimer's disease--a 48 months follow-up analysis. J Neural Transm Suppl. 2007(72):189-193. (PubMed)
- Dana Consortium on the Therapy of HIV Dementia and Related Cognitive Disorders. A randomized, double-blind, placebo-controlled trial of deprenyl and thioctic acid in human immunodeficiency virus-associated cognitive impairment. Neurology. 1998;50(3):645-651. (PubMed)
- Shinto L, Quinn J, Montine T, et al. A randomized placebo-controlled pilot trial of omega-3 fatty acids and alpha lipoic acid in Alzheimer's disease. J Alzheimers Dis. 2014;38(1):111-120. (PubMed)
- Namazi N, Larijani B, Azadbakht L. Alpha-lipoic acid supplement in obesity treatment: A systematic review and meta-analysis of clinical trials. Clin Nutr. 2018;37(2):419-428. (PubMed)
- Kucukgoncu S, Zhou E, Lucas KB, Tek C. Alpha-lipoic acid (ALA) as a supplementation for weight loss: results from a meta-analysis of randomized controlled trials. Obes Rev. 2017;18(5):594-601. (PubMed)
- Lodge JK, Youn HD, Handelman GJ, et al. Natural sources of lipic acid: determination of lipoyllysine released from protease-digested tissues by high performance liquid chromatography incorporating electrochemical detection. J Appl Nutr. 1997;49(1 & 2):3-11.
- Biewenga GP, Haenen GR, Bast A. The pharmacology of the antioxidant lipoic acid. Gen Pharmacol. 1997;29(3):315-331. (PubMed)
- ConsumerLab.com. Alpha-Lipoic Acid Supplements Review July 2017. https://www.consumerlab.com/reviews/Alpha-Lipoic_Acid_Supplements/alphalipoic/. Accessed 9/27/18.
- Streeper RS, Henriksen EJ, Jacob S, Hokama JY, Fogt DL, Tritschler HJ. Differential effects of lipoic acid stereoisomers on glucose metabolism in insulin-resistant skeletal muscle. Am J Physiol. 1997;273(1 Pt 1):E185-191. (PubMed)
- Maitra I, Serbinova E, Tritschler HJ, Packer L. Stereospecific effects of R-lipoic acid on buthionine sulfoximine-induced cataract formation in newborn rats. Biochem Biophys Res Commun. 1996;221(2):422-429. (PubMed)
- Ziegler D, Nowak H, Kempler P, Vargha P, Low PA. Treatment of symptomatic diabetic polyneuropathy with the antioxidant alpha-lipoic acid: a meta-analysis. Diabet Med. 2004;21(2):114-121. (PubMed)
- Reljanovic M, Reichel G, Rett K, et al. Treatment of diabetic polyneuropathy with the antioxidant thioctic acid (alpha-lipoic acid): a two year multicenter randomized double-blind placebo-controlled trial (ALADIN II). Alpha Lipoic Acid in Diabetic Neuropathy. Free Radic Res. 1999;31(3):171-179. (PubMed)
- Parente E, Colannino G, Picconi O, Monastra G. Safety of oral alpha-lipoic acid treatment in pregnant women: a retrospective observational study. Eur Rev Med Pharmacol Sci. 2017;21(18):4219-4227. (PubMed)
- Natural Medicines. Alpha-Lipoic Acid/Safety - Professional Handout. Available at: https://naturalmedicines.therapeuticresearch.com. Accessed 9/26/18.
- Karaarslan U, Isguder R, Bag O, Kisla M, Agin H, Unal N. Alpha lipoic acid intoxication, treatment and outcome. Clin Toxicol (Phila). 2013;51(6):522. (PubMed)
- Hadzik B, Grass H, Mayatepek E, Daldrup T, Hoehn T. Fatal non-accidental alpha-lipoic acid intoxication in an adolescent girl. Klin Padiatr. 2014;226(5):292-294. (PubMed)
- Natural Medicines. Alpha-Lipoic acid/Interactions with Drugs - Professional handout. Available at: https://naturalmedicines.therapeuticresearch.com. Accessed 9/25/18.
- Gleiter CH, Schreeb KH, Freudenthaler S, et al. Lack of interaction between thioctic acid, glibenclamide and acarbose. Br J Clin Pharmacol. 1999;48(6):819-825. (PubMed)
- Prasad PD, Wang H, Huang W, et al. Molecular and functional characterization of the intestinal Na+-dependent multivitamin transporter. Arch Biochem Biophys. 1999;366(1):95-106. (PubMed)
- Balamurugan K, Vaziri ND, Said HM. Biotin uptake by human proximal tubular epithelial cells: cellular and molecular aspects. Am J Physiol Renal Physiol. 2005;288(4):F823-831. (PubMed)
- Zempleni J, Trusty TA, Mock DM. Lipoic acid reduces the activities of biotin-dependent carboxylases in rat liver. J Nutr. 1997;127(9):1776-1781. (PubMed)
- Zempleni J, Mock DM. Biotin biochemistry and human requirements. J Nutr Biochem. 1999;10(3):128-138. (PubMed)
Disclaimer
The Linus Pauling Institute's Micronutrient Information Center provides scientific information on the health aspects of dietary factors and supplements, food, and beverages for the general public. The information is made available with the understanding that the author and publisher are not providing medical, psychological, or nutritional counseling services on this site. The information should not be used in place of a consultation with a competent health care or nutrition professional.
The information on dietary factors and supplements, food, and beverages contained on this website does not cover all possible uses, actions, precautions, side effects, and interactions. It is not intended as nutritional or medical advice for individual problems. Liability for individual actions or omissions based upon the contents of this site is expressly disclaimed.
You may not copy, modify, distribute, display, transmit, perform, publish or sell any of the copyrightable material on this website. You may hyperlink to this website but must include the following statement:
"This link leads to a website provided by the Linus Pauling Institute at Oregon State University. [Your name] is not affiliated or endorsed by the Linus Pauling Institute or Oregon State University."