Summary
- Plant sterols and plant stanols, known commonly as phytosterols, are plant-derived compounds that are structurally related to cholesterol. (More information)
- Early human diets were likely rich in phytosterols, providing as much as 1 g/day; however, the typical Western diet today is relatively low in phytosterols. (More information)
- Although phytosterols are present in the diet in amounts similar to cholesterol, they are poorly absorbed and blood concentrations tend to be low. After absorption into enterocytes, phytosterols are actively excreted back into the intestinal lumen by the ATP-binding cassette transporter, ABCG5/G8. (More information)
- Phytosterols interfere with the intestinal absorption of dietary cholesterol by displacing cholesterol from micelles; they also facilitate the excretion of biliary cholesterol in the feces. (More information)
- Numerous clinical trials have demonstrated that daily consumption of phytosterols from phytosterol-enriched foods can significantly lower serum low-density lipoprotein (LDL)-cholesterol. An average phytosterol intake of 2 g/day lowers serum LDL-cholesterol by 8%-10%. (More information)
- The effect of long-term use of foods enriched with phytosterols on cardiovascular risk is not known. (More information)
- The results of a few clinical trials suggested that phytosterol supplementation at relatively low doses can improve urinary tract symptoms related to benign prostatic hyperplasia, but further research is needed to confirm these findings. (More information)
- Good food sources of phytosterols include unrefined vegetable oils, whole grains, nuts, seeds, and legumes. (More information)
- Foods and beverages with added phytosterols are now available in many countries throughout the world, and some countries allow health claims on such commercial products. (More information)
- Consumption of phytosterol-enriched foods may have undesirable effects, such as a reduction in plasma carotenoid concentrations. (More information)
Other Articles
Introduction
Throughout much of human evolution, it is likely that large amounts of plant foods were consumed (1). In addition to being rich in fiber and plant protein, the diets of our ancestors were also rich in phytosterols — plant-derived compounds that are structurally very similar to cholesterol (Figure 1). There is increasing evidence to suggest that the reintroduction of plant foods providing phytosterols into the modern diet could improve serum lipid (cholesterol) profiles and help reduce the risk of cardiovascular disease (1).
Cholesterol in human blood and tissues is derived from the diet, as well as from endogenous cholesterol synthesis. In contrast, all phytosterols in human blood and tissues are derived from the diet because humans cannot synthesize phytosterols (2). While cholesterol is the predominant sterol in animals, including humans, a variety of sterols are found in plants (3). Nutritionists recognize two classes of phytosterols:
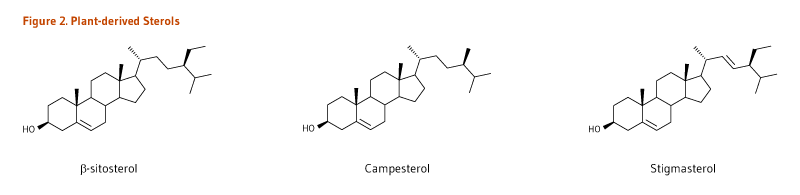
- Plant sterols have a double bond in the sterol ring. The most abundant sterols in plants and the human diet are β-sitosterol, campesterol, and stigmasterol (Figure 2).
- Plant stanols lack a double bond in the sterol ring. Stanols, especially sitostanol and campestanol, comprise only about 10% of total dietary phytosterols (Figure 3).
Definitions
Phytosterols: a collective term for plant-derived sterols and stanols.
Plant sterols or stanols: terms generally applied to plant-derived sterols or stanols; these phytochemicals are added to food or supplements.
Plant sterol or stanol esters: plant sterols or stanols that have been esterified by creating an ester bond between a fatty acid and the sterol or stanol. Esterification occurs in intestinal cells and is also an industrial process. Esterification makes plant sterols and stanols more fat-soluble so they are easily incorporated into fat-containing foods, including margarines and salad dressings. In this article, the weights of plant sterol and stanol esters are expressed as the equivalent weights of free (unesterified) sterols and stanols.
Metabolism and Bioavailability
Absorption and metabolism of dietary cholesterol
Dietary cholesterol must be incorporated into mixed micelles in order to be absorbed by the cells that line the intestine (enterocytes) (4). Mixed micelles are mixtures of bile salts, lipids, and sterols formed in the small intestine after a fat-containing meal is consumed. Transport across the apical membrane of enterocytes is mediated by intestinal cholesterol transporter, Niemann Pick C1-Like 1 (NPC1L1), which is also involved in the uptake of phytosterols (5). Inside the enterocyte, cholesterol is esterified in a reaction catalyzed by intestinal acyl-coenzyme A (CoA) cholesterol acyltransferases (ACATs; also present in the liver) and incorporated into triglyceride-rich lipoproteins known as chylomicrons, which are secreted into the intestinal lymphatics. The thoracic lymphatic duct then collects most of the lymph before draining into the systemic blood circulation (6). As circulating chylomicrons become depleted of triglycerides, they become chylomicron remnants, which are taken up by the liver. In the liver, cholesterol from chylomicron remnants may be repackaged into other lipoproteins for transport throughout the circulation or, alternatively, secreted into bile, which is released into the small intestine.
Absorption and metabolism of dietary phytosterols
Although varied diets typically contain similar amounts of phytosterols and cholesterol, serum phytosterol concentrations are usually several hundred times lower than serum cholesterol concentrations in humans (7). Less than 5% of dietary plant sterols and less than 0.5% of dietary plant stanols are systemically absorbed, in contrast to about 50%-60% of dietary cholesterol (8, 9). Like cholesterol, phytosterols must be incorporated into mixed micelles before they are taken up by enterocytes. Once inside the enterocyte, systemic absorption of phytosterols is inhibited by the activity of an efflux transporter, consisting of a pair of ATP-binding cassette (ABC) proteins known as ABCG5 and ABCG8. ABCG5 and ABCG8 each form one half of a transporter that secretes phytosterols and unesterified cholesterol from the enterocyte into the intestinal lumen. Phytosterols are secreted back into the intestine by ABCG5/G8 transporters at a much greater rate than cholesterol, resulting in much lower intestinal absorption of dietary phytosterols than cholesterol (10).
Within the enterocyte, phytosterols are not as readily esterified as cholesterol, so they are incorporated into chylomicrons at much lower concentrations. Those phytosterols that are incorporated into chylomicrons enter the circulation and are taken up by the liver. Once inside the liver, phytosterols are rapidly secreted into bile by hepatic ABCG5/G8 transporters. Although cholesterol is also secreted into bile, the rate of phytosterol secretion into bile is much greater than cholesterol secretion (11). Thus, the low serum concentrations of phytosterols relative to cholesterol can be explained by decreased intestinal absorption and increased excretion of phytosterols into bile.
Biological Activities
Effects on cholesterol absorption and excretion
It is well established that high intakes of plant sterols or stanols can lower serum total and low-density lipoprotein (LDL)-cholesterol concentrations in humans (see Cardiovascular disease). Different mechanisms appear to underlie the cholesterol-lowering effect of phytosterols (reviewed in 12). In the intestinal lumen, phytosterols displace cholesterol from mixed micelles and reduce cholesterol absorption (13). It is also suggested that phytosterols might interfere with the esterification and incorporation of cholesterol into chylomicrons inside the enterocytes (12). In a placebo-controlled, cross-over trial, the consumption of moderate (0.46 g/day) and high (2.1 g/day) phytosterol-enriched beverages reduced cholesterol absorption by about 10% and 25%, respectively (14). Moderate and high phytosterol intakes also significantly increased the excretion of biliary and dietary cholesterol in the feces by 36% and 74%, respectively (14). Although the mechanisms are currently not clear, phytosterols might facilitate cholesterol efflux from peripheral tissues and macrophages lining vessel walls. Cholesterol is then transported to the liver and incorporated into bile stored in the gallbladder. While plant sterols may promote the hepatobiliary secretion of cholesterol into the intestinal lumen, they are also hypothesized to facilitate the disposal of cholesterol via a nonbiliary route called transintestinal cholesterol efflux (TICE) (12).
Effects on cholesterol metabolism
A decrease in intestinal-derived cholesterol entering the circulation as chylomicrons triggers the endogenous production of cholesterol in order to maintain cholesterol homeostasis (14). Cell surface LDL-receptor expression is also up-regulated to enhance a receptor-mediated uptake of circulating LDL-cholesterol into cells (15). This process results in an increased clearance of circulating LDL from the blood. Within the cells, LDL particles are dismantled in lysosomes and cholesterol becomes available for metabolic needs. Through inhibiting the sterol regulatory element-binding protein (SREBP) pathway, LDL and LDL-derived cholesterol then suppress the transcription of the genes coding for 3-hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductase and other enzymes involved in the synthesis of cholesterol and of the LDL-receptor (16). The net result is the maintenance of cellular cholesterol homeostasis within tissues (especially in the liver) and a reduction in serum LDL-cholesterol concentration.
Of note, some individuals with low intestinal cholesterol absorption efficiency (17) and/or high basal cholesterol synthesis rate (18) have been found to be poorly responsive to phytosterol therapy (reviewed in 19).
Other biological activities
Experiments in cell culture and animal models have suggested that phytosterols might have biological activities unrelated to cholesterol lowering. However, their significance in humans is not yet known.
Alterations in cell membrane properties
Cholesterol is an important structural component of mammalian cell membranes (20). Displacement of cholesterol with phytosterols has been found to alter the physical properties of cell membranes in vitro (21), which could potentially affect signal transduction or membrane-bound enzyme activity (22, 23). Limited evidence from an animal model of hemorrhagic stroke suggested that very high intakes of phytosterols could displace cholesterol in red blood cell membranes, resulting in decreased deformability and potentially increased fragility (24, 25). However, daily phytosterol supplementation (1 g/1,000 kcal) for four weeks did not alter red blood cell fragility in humans (26).
Alterations in testosterone metabolism
Limited evidence from animal studies suggests that very high phytosterol intake could alter testosterone metabolism by inhibiting 5-α-reductase, a membrane-bound enzyme that converts testosterone to dihydrotestosterone, a more potent metabolite (27, 28). It is not known whether phytosterol consumption alters testosterone metabolism in humans. No significant changes in free or total serum testosterone concentrations were observed in men who consumed 1.6 g/day of plant sterol esters for one year (29).
Anticancer effects
Phytosterols have been found to inhibit proliferation, induce apoptosis, and reduce invasiveness of cancer cells in culture (reviewed in 30). There is currently little evidence to suggest that phytosterol consumption could substantially contribute to lower the risk of cancer in humans (see Cancer).
Anti-inflammatory effects
Limited data from cell culture and animal studies suggest that phytosterols may attenuate the inflammatory activity of immune cells, including macrophages and neutrophils (31, 32). The result of a recent meta-analysis of 20 randomized controlled trials found that reductions in total cholesterol and LDL-cholesterol concentrations with phytosterol-enriched foods were not associated with changes in plasma concentration of C-reactive protein (CRP), a surrogate marker of chronic low-grade inflammation (33).
Disease Prevention
Cardiovascular disease
Typical diets across different populations have been estimated to provide 150 to 450 mg/day of naturally occurring phytosterols. Nevertheless, the consumption of vegetarian diets and of food products enriched with phytosterols can help achieve much greater intakes of phytosterols (see Food sources). Relatively few studies have considered the effects of naturally occurring dietary phytosterol intakes on serum LDL-cholesterol concentrations, while an abundance of studies have examined the lipid-lowering effect of phytosterol-enriched foods.
Foods enriched with plant sterols or stanols
Lipid-lowering effect
Elevated LDL-cholesterol concentration is a well-established risk factor in the development of atherosclerosis and coronary heart disease (34, 35). Numerous clinical trials have found that daily consumption of foods enriched with free or esterified forms of plant sterols or stanols lowers concentrations of serum total and LDL-cholesterol (36-40). This wealth of evidence has been summarized in several meta-analyses combining the results of randomized controlled trials (41-46). A dose-dependent relationship was reported between total phytosterol intake levels (from less than 1 g/day to 4 g/day) and LDL-cholesterol reduction in a recent meta-analysis of 124 human studies (47). When analyzed separately, plant sterols and stanols showed similar dose-response effects on LDL-cholesterol concentrations for average doses ranging from 0.6 g/day to 3.3 g/day. Average doses of phytosterols between 0.6 and 1.1 g/day were found to significantly lower LDL-cholesterol concentrations by at least 5%, while an average intake of 3.3 g/day resulted in reductions of about 12.4% (47).
Another meta-analysis that analyzed the results of 59 randomized controlled trials suggested that reductions in LDL-cholesterol were greater in those with higher baseline concentrations of LDL-cholesterol (41). Interestingly, a recent meta-analysis of 15 randomized controlled trials investigating the effects of phytosterol-enriched food intake (1.8 to 6 g/day of phytosterols) in patients treated with statins (drugs that inhibit endogenous cholesterol synthesis) found that co-administration of phytosterols and statins significantly reduced total cholesterol and LDL-cholesterol concentrations compared to statin therapy alone (48). The concentrations of HDL-cholesterol and triglycerides were unaffected by the combination of phytosterols and statins compared to statin alone. In subgroup analyses, the effect of combining phytosterols and statins on blood lipid profile was not found to be significantly influenced by lipid baseline values, phytosterol dosage, or study duration (48).
Effect on vascular health
Impairment of vascular endothelial function is considered to be an early step in the development of atherosclerosis and cardiovascular disease (49). A recent 12-week randomized, double-blind, placebo-controlled study in 240 subjects with hypercholesterolemia (serum cholesterol ≥5 mmol/L [≥193 mg/dL]) found no effect of consuming 3 g/day of phytosterols added to low-fat spread on brachial artery flow-mediated dilation (FMD), a surrogate marker of endothelial health (50). Assessment of arterial stiffness — using measures of aortic pulse wave velocity (PWV) and augmentation index (AI) — and blood pressure also showed no difference between supplemented and placebo groups, despite a significant 6.7% reduction in total and LDL-cholesterol. Other trials in individuals with hypercholesterolemia (51, 52) and type 1 diabetes mellitus (53) also failed to find an effect of phytosterol-enriched spread consumption on brachial artery diameter, FMD, and/or arterial stiffness. Nonetheless, the results of a randomized controlled trial in 92 individuals of whom 72% had serum cholesterol ≥5 mmol/L suggested beneficial effects of plant stanol-enriched spread consumption (corresponding to 3 g/day of stanols for six months) on arterial stiffness and endothelial function, as assessed by cardio-ankle vascular index (CAVI) and reactive hyperemia index (RHI) measures, respectively (54). Finally, a 21-month randomized controlled trial used retinal photography to examine the effect of phytosterol-enriched margarine consumption on retinal microcirculation in 43 statin-treated subjects (55). Reductions in LDL-cholesterol concentration by 9.7% and 11.2% with plant sterols (2.5 g/day) and plant stanols (2.5 g/day), respectively, were not accompanied by significant changes in the diameter of retinal arterioles and venules, a proxy measure to assess microvascular health (55). At present, whether phytosterols can improve vascular health in individuals with endothelial dysfunction is unclear. The lack of an effect of phytosterols in most of the abovementioned trials may be due to the inclusion of apparently healthy participants who may have normal endothelial function (50).
Effect on the risk of coronary heart disease
Elevated LDL-cholesterol is an established risk factor for coronary heart disease (CHD) (56). The pooled analysis of 27 randomized controlled trials of statin drug therapy found a 24% decrease in the risk of major coronary events and a 12% decrease in vascular mortality per 1 millimol/L (1 mM) reduction in LDL-cholesterol concentration, irrespective of gender and level of cardiovascular risk (57). Yet, at present, the effect of long-term use of foods enriched with plant sterols or stanols on CHD risk is not known.
The addition of plant sterol- or stanol-enriched foods to a heart-healthy diet that is low in saturated fat and rich in fruit and vegetables, whole grains, and fiber offers the potential for additive effects in CHD risk reduction. For example, following a diet that substituted monounsaturated and polyunsaturated fats for saturated fat resulted in a 9% reduction in serum LDL-cholesterol after 30 days, but the addition of 1.7 g/day of plant sterols to the same diet resulted in a 24% reduction (58). In addition, one-month adherence to a diet providing a portfolio of cholesterol-lowering foods, including plant sterols (1 g/1,000 kcal), soy protein, almonds, and viscous fibers, lowered serum LDL-cholesterol concentrations by an average of 30% — a decrease that was not significantly different from that induced by statin therapy (59).
The National Cholesterol Education Program (NCEP) Adult Treatment Panel III included the use of plant sterol or stanol esters (2 g/day) as a component of maximal dietary therapy for elevated LDL-cholesterol (60). The 2013 report of the American College of Cardiology (ACC) task force advised clinicians to consider the use of phytosterol-enriched foods as dietary adjuncts for high-risk patients with insufficient LDL-cholesterol response to statin therapy (61). However, stepping back from a general recommendation, the ACC and American Heart Association (AHA) did not include phytosterols in their 2013 report on lifestyle management guidelines to reduce cardiovascular risk (62). Likewise, the 2015-2020 Dietary Guidelines for Americans — issued jointly by the US Department of Health and Human Services and the US Department of Agriculture — does not mention phytosterols in the composition of healthy eating patterns (63).
The US Food and Drug Administration (FDA) has authorized the use of health claims on food labels indicating that regular consumption of foods enriched with plant sterol or stanol esters, as part of a diet low in saturated fat and cholesterol, may reduce the risk of heart disease (see Foods enriched with plant sterols and plant stanols) (61, 64). In the EU, disease risk reduction claims for phytosterols are restricted to certain fortified food products and include a number of mandatory statements such as the fact that these products are not intended for people who do not need to control their blood cholesterol level (65).
Dietary phytosterols
Clinical trials finding daily consumption of foods enriched with plant sterols or stanols can significantly lower LDL-cholesterol concentrations do not account for naturally occurring phytosterols in the diet (66). Relatively few studies have considered the effects of dietary phytosterol intakes on serum LDL-cholesterol concentrations. Limited evidence, primarily from cross-sectional studies, suggests that dietary phytosterols may play an important role in decreasing cholesterol absorption. A cross-sectional study in the UK found that dietary phytosterol intakes were inversely related to serum total and LDL-cholesterol concentrations even after adjusting for saturated fat and fiber intake (67). Similarly, an analysis in a Swedish population found that dietary intake of phytosterols was inversely associated with total cholesterol in both men and women and with LDL-cholesterol in women (68). Dietary phytosterol intakes were also found to be inversely associated with LDL-cholesterol concentrations in another cross-sectional study in healthy Spanish participants (69). In single-meal tests, removal of 150 mg of phytosterols from corn oil increased cholesterol absorption by 38% (70), and removal of 328 mg of phytosterols from wheat germ increased cholesterol absorption by 43% (71). Although these findings suggest that moderate intakes of phytosterols could have an important impact on cardiovascular health, the intake of phytosterols (83 to 966 mg/day) from natural sources was not found to be associated with reduced risks of CHD, myocardial infarction, or total cardiovascular disease during the 12.2-year follow-up of 35,597 participants of the European Prospective Investigation into Cancer and Nutrition-The Netherlands (EPIC-NL) (72).
Cancer
Limited data from animal studies suggest that very high intakes of phytosterols, particularly sitosterol, may inhibit the growth of breast and prostate cancer (reviewed in 73). Only a few observational studies have examined associations between dietary phytosterol intakes and cancer risk in humans (30). A series of case-control studies in Uruguay found that dietary phytosterol intakes were lower in people diagnosed with stomach, lung, or breast cancer than in cancer-free control groups (74-76). Case-control studies in the US found that women diagnosed with breast or endometrial (uterine) cancer had lower dietary phytosterol intakes than women who did not have cancer (77, 78). In contrast, another case-control study in the US found that men diagnosed with prostate cancer had higher dietary campesterol intakes than cancer-free men, but total phytosterol consumption was not associated with prostate cancer risk (79). Although higher intakes of plant foods containing phytosterols may be associated with lower cancer risk, it is not clear whether potential anticancer health benefits can be attributed to phytosterols or to other compounds in plant foods (e.g., other phytochemicals, vitamins, minerals, and fiber).
Disease Treatment
Benign prostatic hyperplasia
Benign prostatic hyperplasia (BPH) is the term used to describe a noncancerous enlargement of the prostate. The enlarged prostate may exert pressure on the urethra, resulting in difficulty urinating. Plant extracts that provide a mixture of phytosterols (marketed as β-sitosterol) are often included in herbal therapies for urinary symptoms related to BPH. However, relatively few controlled studies have examined the efficacy of phytosterol supplements in men with symptomatic BPH. In a six-month study of 200 men with symptomatic BPH, 60 mg/day of a β-sitosterol preparation improved symptom scores, increased peak urinary flow, and decreased post-void residual urine volume compared to placebo (80). A follow-up study reported that these improvements were maintained for up to 18 months in the 38 participants who continued β-sitosterol treatment (81). Similarly, in a six-month study of 177 men with symptomatic BPH, 130 mg/day of a different β-sitosterol preparation improved urinary symptom scores, increased peak urinary flow, and decreased post-void residual urine volume compared to placebo (82). A systematic review that combined the results of these and two other controlled clinical trials found that β-sitosterol extracts increased peak urinary flow by an average of 3.9 mL/second and decreased post-void residual volume by an average of 29 mL (83). Although the results of a few clinical trials suggest that relatively low doses of phytosterols can improve lower urinary tract symptoms related to BPH, further research is needed to confirm these findings (84).
Sources
Food
Unlike the typical diet in most developed countries today, the diets of our ancestors were rich in phytosterols, likely providing as much as 1 g/day (1). Present-day dietary phytosterol intakes have been estimated to vary from 150 to 450 mg/day in different populations (85). Vegetarians, particularly vegans, generally have the highest intakes of dietary phytosterols (86). Phytosterols are found in all plant foods, but the highest concentrations are found in unrefined plant oils, including vegetable, nut, and olive oils (3). Nuts, seeds, whole grains, and legumes are also good dietary sources of phytosterols (4). The phytosterol content of selected foods are presented in Table 1. For information on the nutrient content of specific foods, search USDA's FoodData Central.
| Food | Serving | Phytosterols* (mg) |
|---|---|---|
| Soybeans, mature seeds, raw | ½ cup | 149 |
| Peas, green, mature seeds, raw | ½ cup | 133 |
| Sesame oil | 1 tablespoon (14 g) | 118 |
| Kidney beans, mature seeds, raw | ½ cup | 117 |
| Pistachio nuts | 1 ounce (49 kernels) | 61 |
| Safflower oil | 1 tablespoon (14 g) | 60 |
| Lentils, pink or red, mature seeds, raw | ½ cup | 54 |
| Cashew nuts | 1 ounce | 45 |
| Soybeans, green, cooked, boiled | ½ cup | 45 |
| Cottonseed oil | 1 tablespoon (14 g) | 44 |
| Orange, raw | 1 fruit | 34 |
| Macadamia nuts | 1 ounce (10-12 kernels) | 33 |
| Almonds, blanched | 1 ounce | 32 |
| Olive oil | 1 tablespoon (14 g) | 30 |
| Banana, raw | 1 large | 24 |
| Brussels sprouts, raw | 1 cup | 21 |
|
*In the USDA food composition database, the values of phytosterol content of foods are likely to be underestimates since they account only for major sterols (sitosterol, campesterol, and stigmasterol). In addition, the values correspond to the amounts of free and esterified phytosterols in foods, because phytosterol glycosides are not quantified by the current method unless glycosides (sugars) are removed before quantification (87). |
||
Food enriched with plant sterols and plant stanols
Clinical trials that demonstrated a cholesterol-lowering effect have primarily used plant sterol or stanol esters solubilized in fat-containing foods, such as margarine or mayonnaise (44). Additional studies indicate that low-fat or even nonfat foods can effectively deliver plant sterols or stanols if they are adequately solubilized (37, 66). Plant sterols or stanols added to low-fat yogurt (88-91), low-fat milk (92-94), low-fat cheese (95), dark chocolate (96), and orange juice (97, 98) have been reported to lower LDL-cholesterol in randomized controlled trials. A variety of foods containing added plant sterols or stanols, including margarines, mayonnaises, vegetable oils, salad dressings, yogurt, milk, soy milk, orange juice, snack bars, and meats, are available in the US, Europe, Asia, Australia, and New Zealand (37). A 2008 meta-analysis found that phytosterols added to fat spreads, mayonnaise, salad dressings, milk, or yogurt more effectively reduced LDL-cholesterol concentrations compared to phytosterols incorporated into chocolate, orange juice, cheese, meats, and cereal bars (41). In most clinical trials, dividing the daily dose of phytosterols among two or three meals appeared to effectively lower LDL-cholesterol (41). Nevertheless, consumption of the daily dose of plant sterols or stanols with a single meal has also been found to lower LDL-cholesterol in a few clinical trials (89-91, 99, 100).
In the US, FDA-authorized health claims on food labels specify that the daily dietary intake of plant sterol (≥1.3 g/day) or stanol esters (≥3.4 g/day) that has been associated with a reduced risk of heart disease should be consumed in two servings eaten at different times of the day with other foods, as part of a diet low in saturated fat and cholesterol (61, 64). In the EU, food labels must indicate that the beneficial effect of phytosterols is obtained with a daily intake of 1.5 to 3 g of plant sterols/stanols in order to use the following European Food Safety Authority (EFSA)-approved statement: "Plant sterol and stanol esters have been shown to lower blood cholesterol. High cholesterol is a risk factor in the development of coronary heart disease" (65).
Supplements
Available without a prescription in the US, β-sitosterol supplements typically contain a mixture of β-sitosterol with other phytosterols and/or with substances like pumpkin seed oil and saw palmetto extract (101). Doses of 60 to 130 mg/day of β-sitosterol have been found to alleviate the symptoms of benign prostatic hyperplasia in a few clinical trials (see Benign prostatic hyperplasia). Phytosterol and phytostanol supplements should be taken with a meal that contains fat.
Safety
In the US, plant sterols and stanols added to a variety of food products are generally recognized as safe (GRAS) by the FDA (102). Additionally, the Scientific Committee on Foods of the EU concluded that plant sterols and stanols added to various food products are safe for human use (103). However, the Committee recommended that intakes of plant sterols and stanols from food products should not exceed 3 g/day because there is no evidence of health benefits at higher intakes and there might be undesirable effects at high intakes (65).
Adverse effects
Few adverse effects have been associated with regular consumption of plant sterols or stanols for up to one year. People who consumed a plant sterol-enriched spread providing 1.6 g/day did not report any more adverse effects than those consuming a control spread for up to one year (29), and people consuming a plant stanol-enriched spread providing 1.8 to 2.6 g/day for one year did not report any adverse effects (104). Consumption of up to 8.6 g/day of phytosterols in margarine for three to four weeks was well tolerated by healthy men and women and did not adversely affect intestinal bacteria or female hormone levels (105). Although phytosterols are usually well tolerated, nausea, indigestion, diarrhea, and constipation have occasionally been reported (106).
Sitosterolemia
Sitosterolemia, also known as phytosterolemia, is a very rare hereditary disease that results from inheriting a mutation in both copies of the ABCG5 or ABCG8 gene (107). Individuals who are homozygous for a mutation in either transporter protein have dramatically elevated serum phytosterol concentrations due to increased intestinal absorption and decreased biliary excretion of phytosterols. Although serum cholesterol concentrations may be normal or only mildly elevated, individuals with sitosterolemia are at high risk for premature atherosclerosis. Other clinical symptoms include tuberous and tendon xanthomas (i.e., cutaneous lipid depositions), hematological abnormalities, and sometimes joint pain and arthritis.
People with sitosterolemia should avoid foods or supplements with added plant sterols (37). Two studies have examined the effect of plant sterol consumption in heterozygous carriers of sitosterolemia, a more common condition. Consumption of 3 g/day of plant sterols for four weeks by two heterozygous carriers (108) and consumption of 2.2 g/day of plant sterols for 6 to 12 weeks by 12 heterozygous carriers did not result in abnormally elevated serum phytosterols (109). Because atherosclerosis has been reported in subjects with sitosterolemia, phytosterols have been attributed atherogenic effects. However, no relationship between serum concentrations of sitosterol and campesterol and risk of cardiovascular disease has been identified in a recent meta-analysis of 17 observational studies in 11,182 participants (110).
Pregnancy and lactation
Phytosterol-enriched foods and supplements are not recommended for pregnant or breastfeeding women because their safety has not been studied (106). At present, there is no evidence that high dietary intakes of naturally occurring phytosterols, such as those consumed by vegetarian women, adversely affects pregnancy or lactation.
Drug interactions
Statins
There is some evidence showing that statin administration initially reduces plant sterol concentrations in blood. This might be attributed to the reduction of circulating LDL, the major transport lipoprotein of plant sterols, due to enhanced hepatic uptake of LDL. However, statin therapy appears to increase the absorption of plant sterols that can be then transported by the remaining LDL particles (111). Further, the LDL-cholesterol-lowering effect of plant sterols or stanols may be additive to that of statins. The result of a recent meta-analysis of controlled clinical trials suggested that consumption of 2 to 3 g/day of plant sterols or stanols by individuals on statin therapy may lower both total cholesterol and LDL-cholesterol by an additional 0.30 mmol/L (11.6 mg/dL), compared to statin alone (48).
Ezetimibe
Ezetimibe (marketed as Zetia) is another cholesterol-lowering drug that may interfere with the intestinal absorption of phytosterols, thus significantly reducing phytosterol concentration in blood (112).
Nutrient interactions
Fat-soluble vitamins (vitamins A, D, E, and K)
Because plant sterols and stanols decrease cholesterol absorption and serum LDL-cholesterol concentrations, their effects on fat-soluble vitamin status have also been studied in clinical trials. Plasma vitamin A (retinol) concentrations were not affected by consumption of plant sterol esters or plant stanol esters for up to one year (29, 44). Although the majority of studies found no changes in plasma vitamin D (25-hydroxyvitamin D3) concentrations, one placebo-controlled study in individuals consuming 1.6 g/day of sterol esters for one year observed a small (7%) but statistically significant decrease in plasma 25-hydroxyvitamin D3 concentrations (29). There is little evidence that plant sterol or stanol consumption adversely affects vitamin K status. Consumption of 1.6 g/day of sterol esters for six months was associated with a nonsignificant, 14% decrease in plasma vitamin K1 (phylloquinone) concentrations, and the level of carboxylated osteocalcin, a functional indicator of vitamin K status, was unchanged (29). Other studies of shorter duration also found no change in plasma concentrations of phylloquinone (113, 114) or vitamin K-dependent clotting factors with the consumption of plant sterol and stanol esters (115). Consumption of phytosterol-enriched foods has been found to decrease plasma vitamin E (α-tocopherol) concentration in a number of studies (44, 114). However, those decreases generally do not persist when plasma α-tocopherol concentrations are standardized to LDL-cholesterol concentrations, suggesting that observed reductions in plasma α-tocopherol are due in part to reductions in its lipoprotein carrier, LDL.
A recent meta-analysis of intervention studies found no adverse effects of phytosterol-enriched food consumption (average dose of 2.5 g/day) on fat-soluble vitamin status in well-nourished people (116).
Carotenoids
Dietary carotenoids are fat-soluble phytochemicals that circulate in lipoproteins. A recent meta-analysis of randomized controlled studies reported about 5 to 20% reductions in plasma hydrocarbon carotenoids after consumption of plant sterol- or stanol-enriched foods for one month to one year (116). Even when standardized to serum total cholesterol concentrations, decreases in α-carotene, β-carotene, and lycopene may persist, suggesting that phytosterols could inhibit the absorption of these carotenoids. Total cholesterol-standardized concentrations of xanthophyll carotenoids, zeaxanthin and β-cryptoxanthin, but not lutein, were also found to be significantly reduced by 5 to 15% with the consumption of phytosterol-enriched foods (116).
Although it is not clear whether reductions in plasma carotenoid concentrations confer any health risks (see the article on Carotenoids), a few studies showed that increasing intakes of carotenoid-rich fruit and vegetables would prevent phytosterol-induced decreases in plasma concentrations of carotenoids (117). In one randomized controlled study, advice to consume five daily servings of fruit and vegetables, including one serving of carotenoid-rich vegetables, was enough to maintain plasma carotenoid levels in people consuming 2.5 g/day of plant sterol or stanol esters (118).
Authors and Reviewers
Originally written in 2005 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in September 2008 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in November 2016 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in March 2017 by:
Susan B. Racette, Ph.D.
Professor, Program in Physical Therapy
and Department of Medicine
Washington University in St. Louis
Copyright 2005-2026 Linus Pauling Institute
Figure Alternative Text
Figure 1. Chemical structure of cholesterol
The figure shows the chemical structure of cholesterol.
Figure 2. Plant-derived sterols
The figure shows the chemical structures of three plant-derived sterols: β-sitosterol, campesterol, and stigmasterol.
Figure 3. Plant-derived stanols
The figure shows the chemical structures of two plant-derived stanols: sitostanol and campestanol.
References
- Jew S, AbuMweis SS, Jones PJ. Evolution of the human diet: linking our ancestral diet to modern functional foods as a means of chronic disease prevention. J Med Food. 2009;12(5):925-934. (PubMed)
- Sudhop T, Lutjohann D, von Bergmann K. Sterol transporters: targets of natural sterols and new lipid lowering drugs. Pharmacol Ther. 2005;105(3):333-341. (PubMed)
- Ostlund RE, Jr. Phytosterols in human nutrition. Annu Rev Nutr. 2002;22:533-549. (PubMed)
- de Jong A, Plat J, Mensink RP. Metabolic effects of plant sterols and stanols (Review). J Nutr Biochem. 2003;14(7):362-369. (PubMed)
- Davis HR, Zhu LJ, Hoos LM, et al. Niemann-Pick C1 like 1 (NPC1L1) is the intestinal phytosterol and cholesterol transporter and a key modulator of whole-body cholesterol homeostasis. Journal of Biological Chemistry. 2004;279(32):33586-33592. (PubMed)
- Howles PN. Cholesterol absorption and metabolism. Methods Mol Biol. 2016;1438:177-197. (PubMed)
- von Bergmann K, Sudhop T, Lutjohann D. Cholesterol and plant sterol absorption: recent insights. Am J Cardiol. 2005;96(1A):10D-14D. (PubMed)
- Ostlund RE, Jr., McGill JB, Zeng CM, et al. Gastrointestinal absorption and plasma kinetics of soy Delta(5)-phytosterols and phytostanols in humans. Am J Physiol Endocrinol Metab. 2002;282(4):E911-916. (PubMed)
- Weingartner O, Bohm M, Laufs U. Controversial role of plant sterol esters in the management of hypercholesterolaemia. Eur Heart J. 2009;30(4):404-409. (PubMed)
- Jones PJ, Rideout T. Lipids, sterols, and their metabolites. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease: Lippincott Williams & Wilkins; 2014:65-87.
- Sudhop T, Sahin Y, Lindenthal B, et al. Comparison of the hepatic clearances of campesterol, sitosterol, and cholesterol in healthy subjects suggests that efflux transporters controlling intestinal sterol absorption also regulate biliary secretion. Gut. 2002;51(6):860-863. (PubMed)
- De Smet E, Mensink RP, Plat J. Effects of plant sterols and stanols on intestinal cholesterol metabolism: suggested mechanisms from past to present. Mol Nutr Food Res. 2012;56(7):1058-1072. (PubMed)
- Nissinen M, Gylling H, Vuoristo M, Miettinen TA. Micellar distribution of cholesterol and phytosterols after duodenal plant stanol ester infusion. Am J Physiol Gastrointest Liver Physiol. 2002;282(6):G1009-1015. (PubMed)
- Racette SB, Lin X, Lefevre M, et al. Dose effects of dietary phytosterols on cholesterol metabolism: a controlled feeding study. Am J Clin Nutr. 2010;91(1):32-38. (PubMed)
- Plat J, Mensink RP. Plant stanol and sterol esters in the control of blood cholesterol levels: mechanism and safety aspects. Am J Cardiol. 2005;96(Suppl):15D-22D. (PubMed)
- Goldstein JL, Brown MS. The LDL receptor. Arterioscler Thromb Vasc Biol. 2009;29(4):431-438. (PubMed)
- Zhao HL, Houweling AH, Vanstone CA, et al. Genetic variation in ABC G5/G8 and NPC1L1 impact cholesterol response to plant sterols in hypercholesterolemic men. Lipids. 2008;43(12):1155-1164. (PubMed)
- Rideout TC, Harding SV, Mackay D, Abumweis SS, Jones PJ. High basal fractional cholesterol synthesis is associated with nonresponse of plasma LDL cholesterol to plant sterol therapy. Am J Clin Nutr. 2010;92(1):41-46. (PubMed)
- Rideout TC, Harding SV, Mackay DS. Metabolic and genetic factors modulating subject specific LDL-C responses to plant sterol therapy. Can J Physiol Pharmacol. 2012;90(5):509-514. (PubMed)
- Mouritsen OG, Zuckermann MJ. What's so special about cholesterol? Lipids. 2004;39(11):1101-1113. (PubMed)
- Halling KK, Slotte JP. Membrane properties of plant sterols in phospholipid bilayers as determined by differential scanning calorimetry, resonance energy transfer and detergent-induced solubilization. Biochim Biophys Acta. 2004;1664(2):161-171. (PubMed)
- Awad AB, Chen YC, Fink CS, Hennessey T. beta-Sitosterol inhibits HT-29 human colon cancer cell growth and alters membrane lipids. Anticancer Res. 1996;16(5A):2797-2804. (PubMed)
- Leikin AI, Brenner RR. Fatty acid desaturase activities are modulated by phytosterol incorporation in microsomes. Biochim Biophys Acta. 1989;1005(2):187-191. (PubMed)
- Ratnayake WM, L'Abbe MR, Mueller R, et al. Vegetable oils high in phytosterols make erythrocytes less deformable and shorten the life span of stroke-prone spontaneously hypertensive rats. J Nutr. 2000;130(5):1166-1178. (PubMed)
- Ratnayake WM, Plouffe L, L'Abbe MR, Trick K, Mueller R, Hayward S. Comparative health effects of margarines fortified with plant sterols and stanols on a rat model for hemorrhagic stroke. Lipids. 2003;38(12):1237-1247. (PubMed)
- Jones PJ, Raeini-Sarjaz M, Jenkins DJ, et al. Effects of a diet high in plant sterols, vegetable proteins, and viscous fibers (dietary portfolio) on circulating sterol levels and red cell fragility in hypercholesterolemic subjects. Lipids. 2005;40(2):169-174. (PubMed)
- Awad AB, Hartati MS, Fink CS. Phytosterol feeding induces alteration in testosterone metabolism in rat tissues. J Nutr Biochem. 1998;9(12):712-717.
- Cabeza M, Bratoeff E, Heuze I, Ramirez E, Sanchez M, Flores E. Effect of beta-sitosterol as inhibitor of 5 alpha-reductase in hamster prostate. Proc West Pharmacol Soc. 2003;46:153-155. (PubMed)
- Hendriks HF, Brink EJ, Meijer GW, Princen HM, Ntanios FY. Safety of long-term consumption of plant sterol esters-enriched spread. Eur J Clin Nutr. 2003;57(5):681-692. (PubMed)
- Woyengo TA, Ramprasath VR, Jones PJ. Anticancer effects of phytosterols. Eur J Clin Nutr. 2009;63(7):813-820. (PubMed)
- Awad AB, Toczek J, Fink CS. Phytosterols decrease prostaglandin release in cultured P388D1/MAB macrophages. Prostaglandins Leukot Essent Fatty Acids. 2004;70(6):511-520. (PubMed)
- Navarro A, De las Heras B, Villar A. Anti-inflammatory and immunomodulating properties of a sterol fraction from Sideritis foetens Clem. Biol Pharm Bull. 2001;24(5):470-473. (PubMed)
- Rocha VZ, Ras RT, Gagliardi AC, Mangili LC, Trautwein EA, Santos RD. Effects of phytosterols on markers of inflammation: A systematic review and meta-analysis. Atherosclerosis. 2016;248:76-83. (PubMed)
- Catapano AL, Graham I, De Backer G, et al. 2016 ESC/EAS Guidelines for the Management of Dyslipidaemias: The Task Force for the Management of Dyslipidaemias of the European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS). Developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur Heart J. 2016;37(39):2999-3058. (PubMed)
- Stone NJ, Robinson JG, Lichtenstein AH, et al. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(25 Suppl 2):S1-45. (PubMed)
- St-Onge MP, Jones PJ. Phytosterols and human lipid metabolism: efficacy, safety, and novel foods. Lipids. 2003;38(4):367-375. (PubMed)
- Berger A, Jones PJ, Abumweis SS. Plant sterols: factors affecting their efficacy and safety as functional food ingredients. Lipids Health Dis. 2004;3:5. (PubMed)
- Moruisi KG, Oosthuizen W, Opperman AM. Phytosterols/stanols lower cholesterol concentrations in familial hypercholesterolemic subjects: a systematic review with meta-analysis. J Am Coll Nutr. 2006;25(1):41-48. (PubMed)
- Ellegard LH, Andersson SW, Normen AL, Andersson HA. Dietary plant sterols and cholesterol metabolism. Nutr Rev. 2007;65(1):39-45. (PubMed)
- Van Horn L, McCoin M, Kris-Etherton PM, et al. The evidence for dietary prevention and treatment of cardiovascular disease. J Am Diet Assoc. 2008;108(2):287-331. (PubMed)
- AbuMweis SS, Barake R, Jones P. Plant sterols/stanols as cholesterol lowering agents: A meta-analysis of randomized controlled trials. Food & Nutrition Research. 2008; 52. doi: 10.3402/fnr.v52i0.1811. (PubMed)
- Chen JT, Wesley R, Shamburek RD, Pucino F, Csako G. Meta-analysis of natural therapies for hyperlipidemia: plant sterols and stanols versus policosanol. Pharmacotherapy. 2005;25(2):171-183. (PubMed)
- Demonty I, Ras RT, van der Knaap HC, et al. Continuous dose-response relationship of the LDL-cholesterol-lowering effect of phytosterol intake. J Nutr. 2009;139(2):271-284. (PubMed)
- Katan MB, Grundy SM, Jones P, Law M, Miettinen T, Paoletti R. Efficacy and safety of plant stanols and sterols in the management of blood cholesterol levels. Mayo Clin Proc. 2003;78(8):965-978. (PubMed)
- Law M. Plant sterol and stanol margarines and health. BMJ. 2000;320(7238):861-864. (PubMed)
- Musa-Veloso K, Poon TH, Elliot JA, Chung C. A comparison of the LDL-cholesterol lowering efficacy of plant stanols and plant sterols over a continuous dose range: results of a meta-analysis of randomized, placebo-controlled trials. Prostaglandins Leukot Essent Fatty Acids. 2011;85(1):9-28. (PubMed)
- Ras RT, Geleijnse JM, Trautwein EA. LDL-cholesterol-lowering effect of plant sterols and stanols across different dose ranges: a meta-analysis of randomised controlled studies. Br J Nutr. 2014;112(2):214-219. (PubMed)
- Han S, Jiao J, Xu J, et al. Effects of plant stanol or sterol-enriched diets on lipid profiles in patients treated with statins: systematic review and meta-analysis. Sci Rep. 2016;6:31337. (PubMed)
- Landmesser U, Drexler H. The clinical significance of endothelial dysfunction. Curr Opin Cardiol. 2005;20(6):547-551. (PubMed)
- Ras RT, Fuchs D, Koppenol WP, et al. The effect of a low-fat spread with added plant sterols on vascular function markers: results of the Investigating Vascular Function Effects of Plant Sterols (INVEST) study. Am J Clin Nutr. 2015;101(4):733-741. (PubMed)
- Hallikainen M, Lyyra-Laitinen T, Laitinen T, et al. Endothelial function in hypercholesterolemic subjects: Effects of plant stanol and sterol esters. Atherosclerosis. 2006;188(2):425-432. (PubMed)
- Raitakari OT, Salo P, Gylling H, Miettinen TA. Plant stanol ester consumption and arterial elasticity and endothelial function. Br J Nutr. 2008;100(3):603-608. (PubMed)
- Hallikainen M, Lyyra-Laitinen T, Laitinen T, Moilanen L, Miettinen TA, Gylling H. Effects of plant stanol esters on serum cholesterol concentrations, relative markers of cholesterol metabolism and endothelial function in type 1 diabetes. Atherosclerosis. 2008;199(2):432-439. (PubMed)
- Gylling H, Halonen J, Lindholm H, et al. The effects of plant stanol ester consumption on arterial stiffness and endothelial function in adults: a randomised controlled clinical trial. BMC Cardiovasc Disord. 2013;13:50. (PubMed)
- Kelly ER, Plat J, Mensink RP, Berendschot TT. Effects of long term plant sterol and -stanol consumption on the retinal vasculature: a randomized controlled trial in statin users. Atherosclerosis. 2011;214(1):225-230. (PubMed)
- McCormack T, Dent R, Blagden M. Very low LDL-C levels may safely provide additional clinical cardiovascular benefit: the evidence to date. Int J Clin Pract. 2016;70(11):886-897. (PubMed)
- Cholesterol Treatment Trialists Collaboration. Fulcher J, O'Connell R, et al. Efficacy and safety of LDL-lowering therapy among men and women: meta-analysis of individual data from 174,000 participants in 27 randomised trials. Lancet. 2015;385(9976):1397-1405. (PubMed)
- Jones PJ, Ntanios FY, Raeini-Sarjaz M, Vanstone CA. Cholesterol-lowering efficacy of a sitostanol-containing phytosterol mixture with a prudent diet in hyperlipidemic men. Am J Clin Nutr. 1999;69(6):1144-1150. (PubMed)
- Jenkins DJ, Kendall CW, Marchie A, et al. Direct comparison of a dietary portfolio of cholesterol-lowering foods with a statin in hypercholesterolemic participants. Am J Clin Nutr. 2005;81(2):380-387. (PubMed)
- Grundy SM. Stanol esters as a component of maximal dietary therapy in the National Cholesterol Education Program Adult Treatment Panel III Report. Am J Cardiol. 2005;96(1A):47D-50D. (PubMed)
- Writing C, Lloyd-Jones DM, Morris PB, et al. 2016 ACC expert consensus decision pathway on the role of non-statin therapies for LDL-cholesterol lowering in the management of atherosclerotic cardiovascular disease risk: a report of the American College of Cardiology Task Force on Clinical Expert Consensus Documents. J Am Coll Cardiol. 2016;68(1):92-125. (PubMed)
- Eckel RH, Jakicic JM, Ard JD, et al. 2013 AHA/ACC guideline on lifestyle management to reduce cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(25 Suppl 2):S76-99. (PubMed)
- US Department of Health and Human Services and US Department of Agriculture. 2015 – 2020 Dietary Guidelines for Americans; 2015. Available at: https://www.dietaryguidelines.gov/about-dietary-guidelines/previous-editions/2015-dietary-guidelines. Accessed 8/13/25.
- Food and Drug Administration. Health claims: plant sterol/stanol esters and risk of coronary heart disease (CHD). U. S. Government Printing Office [Code of Federal Regulations]. April 1, 2002. Available at: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=101.83. Accessed 6/29/05.
- Shortt C. Authorised EU health claims for phytosterols. In: Sadler M, ed. Foods, Nutrients and Food Ingredients with Authorised EU Health Claims: Elsevier Ltd; 2015:31-47.
- Ostlund RE, Jr. Phytosterols and cholesterol metabolism. Curr Opin Lipidol. 2004;15(1):37-41. (PubMed)
- Andersson SW, Skinner J, Ellegard L, et al. Intake of dietary plant sterols is inversely related to serum cholesterol concentration in men and women in the EPIC Norfolk population: a cross-sectional study. Eur J Clin Nutr. 2004;58(10):1378-1385. (PubMed)
- Klingberg S, Ellegard L, Johansson I, et al. Inverse relation between dietary intake of naturally occurring plant sterols and serum cholesterol in northern Sweden. Am J Clin Nutr. 2008;87(4):993-1001. (PubMed)
- Sanclemente T, Marques-Lopes I, Fajo-Pascual M, et al. Naturally-occurring phytosterols in the usual diet influence cholesterol metabolism in healthy subjects. Nutr Metab Cardiovasc Dis. 2012;22(10):849-855. (PubMed)
- Ostlund RE, Jr., Racette SB, Okeke A, Stenson WF. Phytosterols that are naturally present in commercial corn oil significantly reduce cholesterol absorption in humans. Am J Clin Nutr. 2002;75(6):1000-1004. (PubMed)
- Ostlund RE, Jr., Racette SB, Stenson WF. Inhibition of cholesterol absorption by phytosterol-replete wheat germ compared with phytosterol-depleted wheat germ. Am J Clin Nutr. 2003;77(6):1385-1389. (PubMed)
- Ras RT, van der Schouw YT, Trautwein EA, et al. Intake of phytosterols from natural sources and risk of cardiovascular disease in the European Prospective Investigation into Cancer and Nutrition-the Netherlands (EPIC-NL) population. Eur J Prev Cardiol. 2015;22(8):1067-1075. (PubMed)
- Ramprasath VR, Awad AB. Role of phytosterols in cancer prevention and treatment. J AOAC Int. 2015;98(3):735-738. (PubMed)
- De Stefani E, Boffetta P, Ronco AL, et al. Plant sterols and risk of stomach cancer: a case-control study in Uruguay. Nutr Cancer. 2000;37(2):140-144. (PubMed)
- Mendilaharsu M, De Stefani E, Deneo-Pellegrini H, Carzoglio J, Ronco A. Phytosterols and risk of lung cancer: a case-control study in Uruguay. Lung Cancer. 1998;21(1):37-45. (PubMed)
- Ronco A, De Stefani E, Boffetta P, Deneo-Pellegrini H, Mendilaharsu M, Leborgne F. Vegetables, fruits, and related nutrients and risk of breast cancer: a case-control study in Uruguay. Nutr Cancer. 1999;35(2):111-119. (PubMed)
- McCann SE, Freudenheim JL, Marshall JR, Brasure JR, Swanson MK, Graham S. Diet in the epidemiology of endometrial cancer in western New York (United States). Cancer Causes Control. 2000;11(10):965-974. (PubMed)
- McCann SE, Freudenheim JL, Marshall JR, Graham S. Risk of human ovarian cancer is related to dietary intake of selected nutrients, phytochemicals and food groups. J Nutr. 2003;133(6):1937-1942. (PubMed)
- Strom SS, Yamamura Y, Duphorne CM, et al. Phytoestrogen intake and prostate cancer: a case-control study using a new database. Nutr Cancer. 1999;33(1):20-25. (PubMed)
- Berges RR, Windeler J, Trampisch HJ, Senge T. Randomised, placebo-controlled, double-blind clinical trial of beta-sitosterol in patients with benign prostatic hyperplasia. Beta-sitosterol Study Group. Lancet. 1995;345(8964):1529-1532. (PubMed)
- Berges RR, Kassen A, Senge T. Treatment of symptomatic benign prostatic hyperplasia with beta-sitosterol: an 18-month follow-up. BJU Int. 2000;85(7):842-846. (PubMed)
- Klippel KF, Hiltl DM, Schipp B. A multicentric, placebo-controlled, double-blind clinical trial of beta-sitosterol (phytosterol) for the treatment of benign prostatic hyperplasia. German BPH-Phyto Study group. Br J Urol. 1997;80(3):427-432. (PubMed)
- Wilt TJ, MacDonald R, Ishani A. Beta-sitosterol for the treatment of benign prostatic hyperplasia: a systematic review. BJU Int. 1999;83(9):976-983. (PubMed)
- Dreikorn K. The role of phytotherapy in treating lower urinary tract symptoms and benign prostatic hyperplasia. World J Urol. 2002;19(6):426-435. (PubMed)
- Othman RA, Myrie SB, Jones PJ. Non-cholesterol sterols and cholesterol metabolism in sitosterolemia. Atherosclerosis. 2013;231(2):291-299. (PubMed)
- Nair PP, Turjman N, Kessie G, et al. Diet, nutrition intake, and metabolism in populations at high and low risk for colon cancer. Dietary cholesterol, beta-sitosterol, and stigmasterol. Am J Clin Nutr. 1984;40(4 Suppl):927-930. (PubMed)
- Racette SB, Spearie CA, Phillips KM, Lin X, Ma L, Ostlund RE, Jr. Phytosterol-deficient and high-phytosterol diets developed for controlled feeding studies. J Am Diet Assoc. 2009;109(12):2043-2051. (PubMed)
- Mensink RP, Ebbing S, Lindhout M, Plat J, van Heugten MM. Effects of plant stanol esters supplied in low-fat yoghurt on serum lipids and lipoproteins, non-cholesterol sterols and fat soluble antioxidant concentrations. Atherosclerosis. 2002;160(1):205-213. (PubMed)
- Volpe R, Niittynen L, Korpela R, et al. Effects of yoghurt enriched with plant sterols on serum lipids in patients with moderate hypercholesterolaemia. Br J Nutr. 2001;86(2):233-239. (PubMed)
- Plana N, Nicolle C, Ferre R, et al. Plant sterol-enriched fermented milk enhances the attainment of LDL-cholesterol goal in hypercholesterolemic subjects. Eur J Nutr. 2008;47(1):32-39. (PubMed)
- Doornbos AM, Meynen EM, Duchateau GS, van der Knaap HC, Trautwein EA. Intake occasion affects the serum cholesterol lowering of a plant sterol-enriched single-dose yoghurt drink in mildly hypercholesterolaemic subjects. Eur J Clin Nutr. 2006;60(3):325-333. (PubMed)
- Noakes M, Clifton PM, Doornbos AME, Trautwein EA. Plant sterol ester-enriched milk and yoghurt effectively reduce serum cholesterol in modestly hypercholesterolemic subjects. Eur J Clin Nutr. 2005;44(4):214-222. (PubMed)
- Thomsen AB, Hansen HB, Christiansen C, Green H, Berger A. Effect of free plant sterols in low-fat milk on serum lipid profile in hypercholesterolemic subjects. Eur J Clin Nutr. 2004;58(6):860-870. (PubMed)
- Seppo L, Jauhiainen T, Nevala R, Poussa T, Korpela R. Plant stanol esters in low-fat milk products lower serum total and LDL cholesterol. Eur J Nutr. 2007;46(2):111-117. (PubMed)
- Jauhiainen T, Salo P, Niittynen L, Poussa T, Korpela R. Effects of low-fat hard cheese enriched with plant stanol esters on serum lipids and apolipoprotein B in mildly hypercholesterolaemic subjects. Eur J Clin Nutr. 2006;60(11):1253-1257. (PubMed)
- Allen RR, Carson L, Kwik-Uribe C, Evans EM, Erdman JW, Jr. Daily consumption of a dark chocolate containing flavanols and added sterol esters affects cardiovascular risk factors in a normotensive population with elevated cholesterol. J Nutr. 2008;138(4):725-731. (PubMed)
- Devaraj S, Jialal I, Vega-Lopez S. Plant sterol-fortified orange juice effectively lowers cholesterol levels in mildly hypercholesterolemic healthy individuals. Arterioscler Thromb Vasc Biol. 2004;24(3):e25-28. (PubMed)
- Devaraj S, Autret BC, Jialal I. Reduced-calorie orange juice beverage with plant sterols lowers C-reactive protein concentrations and improves the lipid profile in human volunteers. Am J Clin Nutr. 2006;84(4):756-761. (PubMed)
- Matvienko OA, Lewis DS, Swanson M, et al. A single daily dose of soybean phytosterols in ground beef decreases serum total cholesterol and LDL cholesterol in young, mildly hypercholesterolemic men. Am J Clin Nutr. 2002;76(1):57-64. (PubMed)
- Plat J, van Onselen EN, van Heugten MM, Mensink RP. Effects on serum lipids, lipoproteins and fat soluble antioxidant concentrations of consumption frequency of margarines and shortenings enriched with plant stanol esters. Eur J Clin Nutr. 2000;54(9):671-677. (PubMed)
- Hendler SS, Rorvik DM. Beta-sitosterol. In: Reuters T, ed. PDR for Nutritional Supplements. 2nd ed. Montvale: Physicians' Desk Reference Inc.; 2008:78-80.
- Food and Drug Administration. GRAS Notice No. GRN 000112. February 4, 2003. http://www.cfsan.fda.gov/~rdb/opa-g112.html. Accessed 7/11/05.
- Scientific Committee on Food. Opinion on Applications for Approval of a Variety of Plant Sterol-Enriched Foods. March 5, 2003. http://europa.eu.int/comm/food/fs/sc/scf/out174_en.pdf. Accessed 7/11/05.
- Miettinen TA, Puska P, Gylling H, Vanhanen H, Vartiainen E. Reduction of serum cholesterol with sitostanol-ester margarine in a mildly hypercholesterolemic population. N Engl J Med. 1995;333(20):1308-1312. (PubMed)
- Ayesh R, Weststrate JA, Drewitt PN, Hepburn PA. Safety evaluation of phytosterol esters. Part 5. Faecal short-chain fatty acid and microflora content, faecal bacterial enzyme activity and serum female sex hormones in healthy normolipidaemic volunteers consuming a controlled diet either with or without a phytosterol ester-enriched margarine. Food Chem Toxicol. 1999;37(12):1127-1138. (PubMed)
- Hendler SS, Rorvik DM. Phytosterols. In: Reuters T, ed. PDR for Nutritional Supplements. 2nd ed. Montvale: Physicians' Desk Reference Inc.; 2008:500-503.
- Berge KE. Sitosterolemia: a gateway to new knowledge about cholesterol metabolism. Ann Med. 2003;35(7):502-511. (PubMed)
- Stalenhoef AF, Hectors M, Demacker PN. Effect of plant sterol-enriched margarine on plasma lipids and sterols in subjects heterozygous for phytosterolaemia. J Intern Med. 2001;249(2):163-166. (PubMed)
- Kwiterovich PO, Jr., Chen SC, Virgil DG, Schweitzer A, Arnold DR, Kratz LE. Response of obligate heterozygotes for phytosterolemia to a low-fat diet and to a plant sterol ester dietary challenge. J Lipid Res. 2003;44(6):1143-1155. (PubMed)
- Genser B, Silbernagel G, De Backer G, et al. Plant sterols and cardiovascular disease: a systematic review and meta-analysis. Eur Heart J. 2012;33(4):444-451. (PubMed)
- Miettinen TA, Gylling H. Effect of statins on noncholesterol sterol levels: implications for use of plant stanols and sterols. Am J Cardiol. 2005;96(1A):40D-46D. (PubMed)
- Assmann G, Kannenberg F, Ramey DR, Musliner TA, Gutkin SW, Veltri EP. Effects of ezetimibe, simvastatin, atorvastatin, and ezetimibe-statin therapies on non-cholesterol sterols in patients with primary hypercholesterolemia. Curr Med Res Opin. 2008;24(1):249-259. (PubMed)
- Raeini-Sarjaz M, Ntanios FY, Vanstone CA, Jones PJ. No changes in serum fat-soluble vitamin and carotenoid concentrations with the intake of plant sterol/stanol esters in the context of a controlled diet. Metabolism. 2002;51(5):652-656. (PubMed)
- Korpela R, Tuomilehto J, Hogstrom P, et al. Safety aspects and cholesterol-lowering efficacy of low fat dairy products containing plant sterols. Eur J Clin Nutr. 2006;60(5):633-642. (PubMed)
- Plat J, Mensink RP. Vegetable oil based versus wood based stanol ester mixtures: effects on serum lipids and hemostatic factors in non-hypercholesterolemic subjects. Atherosclerosis. 2000;148(1):101-112. (PubMed)
- Baumgartner S, Ras RT, Trautwein EA, Mensink RP, Plat J. Plasma fat-soluble vitamin and carotenoid concentrations after plant sterol and plant stanol consumption: a meta-analysis of randomized controlled trials. Eur J Nutr. 2017;56(3):909-923. (PubMed)
- Fardet A, Morise A, Kalonji E, Margaritis I, Mariotti F. Influence of phytosterol and phytostanol food supplementation on plasma liposoluble vitamins and provitamin A carotenoid levels in humans: an updated review of the evidence. Crit Rev Food Sci Nutr. 2015;57(9):1906-1921. (PubMed)
- Noakes M, Clifton P, Ntanios F, Shrapnel W, Record I, McInerney J. An increase in dietary carotenoids when consuming plant sterols or stanols is effective in maintaining plasma carotenoid concentrations. Am J Clin Nutr. 2002;75(1):79-86. (PubMed)
Disclaimer
The Linus Pauling Institute's Micronutrient Information Center provides scientific information on the health aspects of dietary factors and supplements, food, and beverages for the general public. The information is made available with the understanding that the author and publisher are not providing medical, psychological, or nutritional counseling services on this site. The information should not be used in place of a consultation with a competent health care or nutrition professional.
The information on dietary factors and supplements, food, and beverages contained on this website does not cover all possible uses, actions, precautions, side effects, and interactions. It is not intended as nutritional or medical advice for individual problems. Liability for individual actions or omissions based upon the contents of this site is expressly disclaimed.
You may not copy, modify, distribute, display, transmit, perform, publish or sell any of the copyrightable material on this website. You may hyperlink to this website but must include the following statement:
"This link leads to a website provided by the Linus Pauling Institute at Oregon State University. [Your name] is not affiliated or endorsed by the Linus Pauling Institute or Oregon State University."