Summary
- Vitamin D can be synthesized in the skin upon exposure to sunlight and is then metabolized in the liver and kidney to the metabolically active form called 1α,25-dihydroxyvitamin D. Through binding to the vitamin D receptor (VDR), 1α,25-dihydroxyvitamin D can regulate the expression of hundreds of genes involved in skeletal and other biological functions. (More information)
- Vitamin D is essential for maintenance of bone mineralization through the regulation of calcium and phosphorus homeostasis. Vitamin D also exhibits many non-skeletal effects, particularly on the immune, endocrine, and cardiovascular systems. (More information)
- Vitamin D is important for normal bone development and maintenance. Severe vitamin D deficiency causes rickets in children and osteomalacia in adults. (More information)
- Secondary hyperparathyroidism due to vitamin D insufficiency can increase bone breakdown and precipitate osteoporosis. Randomized clinical trials indicate that supplementation with at least 800 IU/day of vitamin D may reduce the risk of falls and fractures in older individuals. (More information)
- Vitamin D can regulate cell differentiation and growth by binding to the vitamin D receptor found in most body cells. Observational studies have reported associations between low sun exposure, poor vitamin D status, and increased risk of developing colorectal and breast cancer. Randomized controlled trials are needed to evaluate whether cancer prevention may benefit from vitamin D supplementation. (More information)
- Various observational studies have reported inverse associations between vitamin D status and the susceptibility or severity of autoimmune diseases, including type 1 diabetes mellitus, multiple sclerosis, rheumatoid arthritis, and systemic lupus erythematosus. (More information)
- Current evidence from observational studies suggests an inverse relationship between circulating vitamin D concentrations and risk of type 2 diabetes mellitus. It is not yet known whether correcting vitamin D deficiency in individuals with glucose intolerance can decrease the risk of progression to type 2 diabetes mellitus. (More information)
- Randomized clinical trials are currently investigating whether vitamin D supplementation can limit cognitive deterioration and disease progression in subjects with neurodegenerative disease. (More information)
- Vitamin D insufficiency in pregnant women may be associated with several adverse effects for the mother and newborn. Safety and benefits of vitamin D supplementation during pregnancy both need to be evaluated in clinical trials. (More information)
- Observational studies have documented an association between vitamin D deficiency and increased incidence and severity of the coronavirus disease, COVID-19. (More information)
- Preliminary studies have shown that vitamin D supplementation may offer promising improvements in the management of atopic dermatitis (eczema) and Crohn’s disease. (More information)
Other Articles
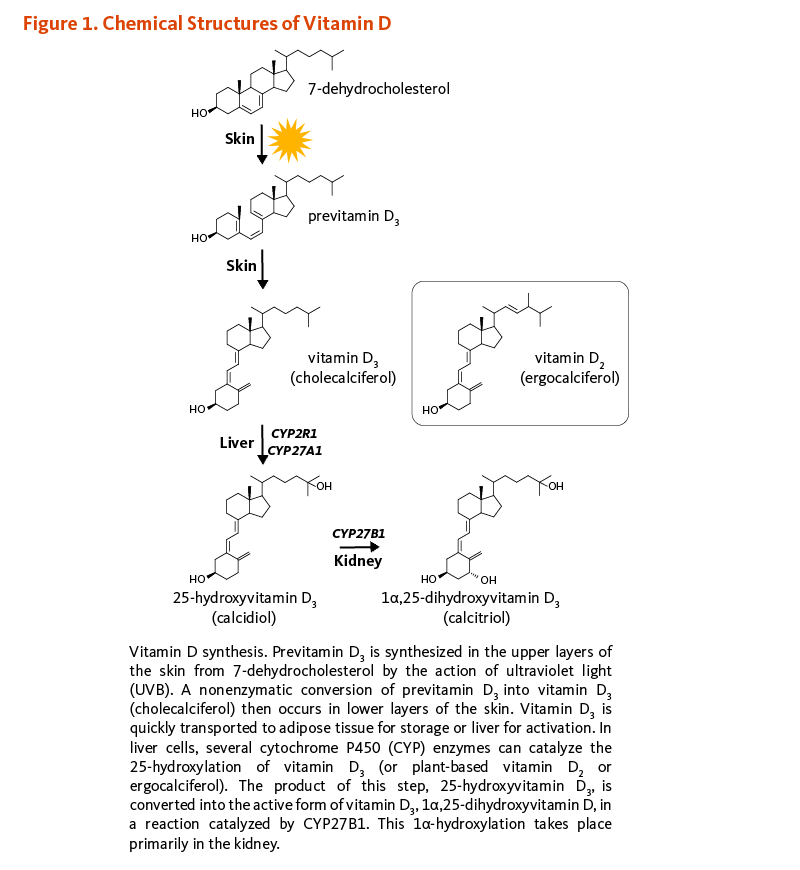
Vitamin D is a fat-soluble vitamin that regulates calcium homeostasis and is vital for bone health (1). While it can also be obtained from dietary sources or supplements, vitamin D3 (cholecalciferol) is synthesized in the human skin from 7-dehydrocholesterol upon exposure to ultraviolet-B (UVB) radiation from sunlight (see the separate article on Vitamin D and Skin Health). Vitamin D2 (ergocalciferol) is a vitamin D analog photosynthesized in plants, mushrooms, and yeasts; vitamin D2 is also sometimes used in vitamin D food fortification (2). When vitamin D3 in skin is inadequate due to insufficient exposure to UVB radiation, oral intake of vitamin D is necessary to meet vitamin D requirements.
Function
Vitamin D metabolism
Cholecalciferol and ergocalciferol are biologically inactive precursors of vitamin D and must be converted to biologically active forms in the liver and kidneys (Figure 1). Indeed, following dietary intake or synthesis in the epidermis of skin after UVB exposure, both forms of vitamin D enter the circulation and are transported to the liver by the vitamin D-binding protein (and to a lesser extent by albumin). In hepatocytes (liver cells), vitamin D is hydroxylated to form 25-hydroxyvitamin D (calcidiol; calcifediol). Exposure to sunlight or dietary intake of vitamin D increases serum concentrations of 25-hydroxyvitamin D. 25-Hydroxyvitamin D constitutes the major circulating form of vitamin D, and the sum of 25-hydroxyvitamin D2 and 25-hydroxyvitamin D3 concentrations in serum is used as an indicator of vitamin D nutritional status (3). The renal 25-hydroxyvitamin D3-1α-hydroxylase enzyme (also known as CYP27B1) eventually catalyzes a second hydroxylation that converts 25-hydroxyvitamin D to 1α,25-dihydroxyvitamin D (calcitriol). The production of 1α,25-dihydroxyvitamin D in the kidneys is regulated by several factors, including serum phosphorus, calcium, parathyroid hormone (PTH), fibroblast growth factor-23 (FGF-23), and 1α,25-dihydroxyvitamin D itself. While the kidney is the main source of 1α-hydroxylase activity, extra-renal production of 1α,25-dihydroxyvitamin D has also been demonstrated in a variety of tissues, including skin, parathyroid gland, breast, colon, prostate, as well as cells of the immune system and bone cells (2). Most of the physiological effects of vitamin D in the body are related to the activity of 1α,25-dihydroxyvitamin D (4). Various forms of vitamin D are listed in Figure 1.
Mechanisms of action
Most, if not all, actions of vitamin D are mediated through a nuclear transcription factor known as the vitamin D receptor (VDR) (Figure 2) (5). Upon entering the nucleus of a cell, 1α,25-dihydroxyvitamin D binds to the VDR and recruits another nuclear receptor known as retinoid X receptor (RXR). In the presence of 1α,25-dihydroxyvitamin D, the VDR/RXR complex binds small sequences of DNA known as vitamin D response elements (VDREs) and initiates a cascade of molecular interactions that modulate the transcription of specific genes. Thousands of VDREs have been identified throughout the genome, and VDR activation by 1α,25-dihydroxyvitamin D is thought to directly and/or indirectly regulate 100 to 1,250 genes (6).
Calcium balance
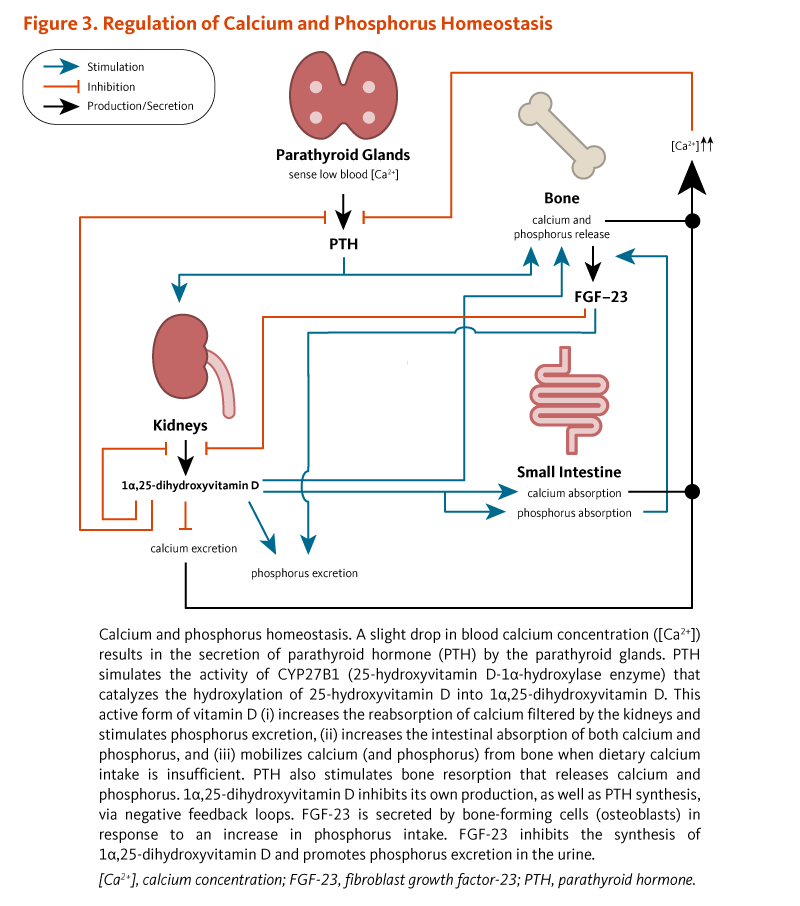
Maintenance of serum calcium concentrations within a narrow range is vital for normal functioning of the nervous system, as well as for bone growth and maintenance of bone density. Vitamin D is essential for the efficient utilization of calcium by the body (1). The parathyroid glands sense serum calcium concentrations and secrete parathyroid hormone (PTH) if calcium concentrations decrease below normal (Figure 3). Elevations in PTH stimulate the activity of the 25-hydroxyvitamin D3-1α-hydroxylase enzyme in the kidney, resulting in the increased production of 1α,25-dihydroxyvitamin D. The active vitamin D form, 1α,25-dihydroxyvitamin D, is released into the circulation and transported to target tissues. Within target cells, 1α,25-dihydroxyvitamin D binds to and induces the activation of VDR, which leads to changes in gene expression that normalize serum calcium by (1) increasing the intestinal absorption of dietary calcium, (2) increasing the reabsorption of calcium filtered by the kidneys, and (3) mobilizing calcium from bone when there is insufficient dietary calcium to maintain normal serum calcium concentrations (7).
Phosphorus balance
The regulations of calcium and phosphorus homeostasis are closely related, and the calciotropic hormones, PTH and 1α,25-dihydroxyvitamin D, can also control serum phosphorus. Specifically, 1α,25-dihydroxyvitamin D increases intestinal phosphorus absorption by stimulating the expression of a sodium-phosphate cotransporter in the small intestine. While PTH increases urinary excretion of phosphorus by reducing reabsorption in the kidney, it is not yet clear whether 1α,25-dihydroxyvitamin D can directly regulate renal phosphorus transport. The phosphaturic hormone fibroblast growth factor-23 (FGF-23), secreted by osteoblasts (bone-forming cells), limits the production of 1α,25-dihydroxyvitamin D by inhibiting 25-hydroxyvitamin D-1α-hydroxylase (Figure 3) (reviewed in 8).
Cell differentiation
Cells that are dividing rapidly are said to be proliferating. Differentiation results in the specialization of cells for specific functions. In general, differentiation of cells leads to a decrease in proliferation. While cellular proliferation is essential for growth and wound healing, uncontrolled proliferation of cells with certain mutations may lead to cancer. The active form of vitamin D, 1α,25-dihydroxyvitamin D, inhibits proliferation and stimulates the differentiation of cells through binding to the VDR (1).
Immunity
Acting through the VDR, 1α,25-dihydroxyvitamin D is a potent immune system modulator. The VDR is expressed by most cells of the immune system, including regulatory T cells and antigen-presenting cells, such as dendritic cells and macrophages (9). Under specific circumstances, monocytes, macrophages, and T cells can express the 25-hydroxyvitamin D3-1α-hydroxylase enzyme and produce 1α,25-dihydroxyvitamin D, which acts locally to regulate the immune response (10, 11). There is considerable scientific evidence that 1α,25-dihydroxyvitamin D has a variety of effects on immune system function, which may enhance innate immunity and inhibit the development of autoimmunity (12). Conversely, vitamin D deficiency may compromise the integrity of the immune system and lead to inappropriate immune responses (see Autoimmune diseases).
Insulin secretion
The VDR is expressed by insulin-secreting cells of the pancreas, and the results of animal studies suggest that 1α,25-dihydroxyvitamin D plays a role in insulin secretion under conditions of increased insulin demand (13, 14). Cross-sectional and prospective studies suggest that insufficient vitamin D status may have an adverse effect on insulin secretion and glucose tolerance in type 2 diabetes mellitus (noninsulin-dependent diabetes mellitus) (reviewed in 15).
Blood pressure regulation
The renin-angiotensin system plays an important role in the regulation of blood pressure (16). Renin is an enzyme that catalyzes the cleavage (splitting) of a small peptide (angiotensin I) from a larger protein (angiotensinogen) produced in the liver. Angiotensin-converting enzyme (ACE) catalyzes the cleavage of angiotensin I to form angiotensin II, a peptide that can increase blood pressure by inducing the constriction of small arteries and by increasing sodium and water retention. The rate of angiotensin II synthesis is dependent on renin (17). Research in mice lacking the gene encoding the VDR indicates that 1α,25-dihydroxyvitamin D decreases the expression of the gene encoding renin through its interaction with the VDR (18). Since inappropriate activation of the renin-angiotensin system can contribute to the development of hypertension, achieving adequate vitamin D status may be important for decreasing the risk of high blood pressure (see Hypertension).
Deficiency
In vitamin D deficiency, calcium absorption cannot be increased enough to satisfy the body’s calcium needs (4). Consequently, PTH production by the parathyroid glands is increased and calcium is mobilized from the skeleton to maintain normal serum calcium concentrations — a condition known as secondary hyperparathyroidism. Although it has long been known that severe vitamin D deficiency has serious consequences for bone health, research suggests that less obvious states of vitamin D deficiency are common and increase the risk of osteoporosis and various other health problems (see Disease Prevention).
Severe vitamin D deficiency
Rickets
In infants and children, severe vitamin D deficiency results in the failure of bone to mineralize. The process of mineralization, which involves the production of crystals of calcium phosphate by bone-forming cells, determines the hardness and strength of bones. Vitamin D deficiency severely affects rapidly growing bones. The growth plates of bones continue to enlarge, but in the absence of adequate mineralization, weight-bearing limbs (arms and legs) become bowed. In infants, rickets may result in delayed closure of the fontanels (soft spots) in the skull, and the rib cage may become deformed due to the pulling action of the diaphragm. In severe cases, low serum calcium concentrations (hypocalcemia) may cause seizures. Although fortification of food has led to complacency regarding vitamin D deficiency, nutritional rickets is still being reported throughout the world (19, 20).
Osteomalacia
Although adult bones are no longer growing, they are in a constant state of turnover, or "remodeling." In adults with severe vitamin D deficiency, the collagenous bone matrix is preserved, but bone mineral is progressively lost, resulting in a softening of bones (osteomalacia), bone pain, and increased risk of osteoporosis (21).
Muscle weakness and pain
Vitamin D deficiency causes muscle weakness and pain in children and adults. Muscle pain and weakness were prominent symptoms of vitamin D deficiency in a study of Arab and Danish Muslim women living in Denmark (22). In a cross-sectional study of 150 consecutive patients referred to a clinic in Minnesota for the evaluation of persistent, nonspecific musculoskeletal pain, 93% had serum 25-hydroxyvitamin D concentrations equal to or below 20 ng/mL, with a mean concentration of 12.1 ng/mL, which is indicative of vitamin D insufficiency (23). Loss of muscle strength greatly contributes to increased risk of falling and bone fractures, especially in older people (24). In addition, long-term vitamin D insufficiency may be a contributing factor to osteoporosis in the elderly (see Osteoporosis).
Risk factors for vitamin D deficiency
Both environmental factors and cultural practices result in variations in vitamin D status:
- Environmental conditions: Geographical locations, including latitude and altitudes, and atmospheric conditions (e.g., air pollution, presence of clouds) greatly influence the intensity of UVB radiation that reaches the ground. Seasonal changes also affect the quality and quantity of UVB rays and thus vitamin D production in skin (25-27).
- Concealed clothing style: In a study of 2,032 Middle Eastern women, who wore a headscarf or covered all skin for religious or cultural reasons, 96% had serum 25-hydroxyvitamin D concentrations less than 20 ng/mL, and 60% had vitamin D concentrations below 12 ng/mL (28). Rickets and osteomalacia are not uncommon in the Middle East and North African regions where children and women cover the majority or all of their skin whenever outside (29).
- Sun safety measures: Sun protection practices, including limiting sun exposure, wearing protective clothing and hats, and applying sunscreens, hinder skin exposure to sunlight and thus result in lower vitamin D3 production and circulating vitamin D metabolites unless there is adequate oral intake. Of note, the application of sunscreen (2 mg/cm2) with a sun protection factor (SPF) of 10 reduces UVB radiation by 90% (30).
- Exclusively breastfed infants: Infants who are exclusively breastfed and do not receive vitamin D supplementation are at high risk for vitamin D deficiency, particularly if they have dark skin and/or receive little sun exposure (19). Human milk generally provides 10 to 80 IU of vitamin D per liter (L), which corresponds to 0.2 to 1.5 g/day (8 to 60 IU/day) when using an average daily milk intake of 0.75 L (25 oz) (31). The American Academy of Pediatrics recommends that all breastfed and partially breastfed infants be given an oral vitamin D supplement of 400 IU/day (19). Maternal vitamin D supplementation during breastfeeding may contribute to improved vitamin D status of the breastfed infant, especially in populations with a high prevalence of vitamin D deficiency (32). Older infants and toddlers exclusively fed milk substitutes (e.g., soy-based formulas) and weaning foods that are not vitamin D fortified are at risk for vitamin D deficiency (33).
The efficiency of vitamin D synthesis, absorption, and metabolism also depends on a variety of biological factors:
- Skin pigmentation: People with a dark complexion synthesize less vitamin D on exposure to sunlight than those with light-colored skin (34). A national US survey reported average serum 25-hydroxyvitamin D concentrations of 28.1 ng/mL, 21.6 ng/mL, and 16.9 ng/mL in Caucasian, Mexican American, and African American adults aged ≥20 years old, respectively (25).
- Genetic variations: An international, multicenter, genome-wide association study (GWAS) of 15 cohorts, including ~30,000 participants of European descent — known as the SUNLIGHT [Study of Underlying Genetic Determinants of Vitamin D and Highly Related Traits] consortium — identified common variations (called polymorphisms) in genes involved in cholesterol synthesis, hydroxylation, and vitamin D transport that influence vitamin D status (35). While genetic determinants of low vitamin D status are being identified in populations of European (36, 37) and Asian descent (38, 39), genome-wide association studies are needed in populations of African descent.
- Older age: The elderly have reduced capacity to synthesize vitamin D in skin when exposed to UVB radiation and are more likely to stay indoors or use sunscreen, which prevents vitamin D synthesis. It has been estimated that across Canada, the US, and Europe, the prevalence of vitamin D deficiency ranges between 20%-100% in free-living elderly (40). Moreover, institutionalized adults who are not supplemented with vitamin D are at extremely high risk of vitamin D deficiency (41, 42).
- Chronic kidney disease (CKD): Vitamin D deficiency in patients with impaired renal function is due to a reduced synthesis of 1α,25-dihydroxyvitamin D and an increased loss of 25-hydroxyvitamin D in urine (43).
- Fat malabsorption syndromes: Vitamin D deficiency is common among people with cystic fibrosis and both cholestatic and non-cholestatic liver diseases due to decreased absorption of dietary vitamin D and impaired conversion of vitamin D to 25-hydroxyvitamin D (reviewed in 44).
- Inflammatory bowel disease: People with inflammatory bowel disease like Crohn’s disease appear to be at increased risk of vitamin D deficiency, especially those who have had small bowel resections (45).
- Obesity: Obesity (body mass index ≥30 kg/m2) increases the risk of vitamin D deficiency (46). Once vitamin D is synthesized in the skin or ingested, it can be sequestered in body fat stores, making it less bioavailable to people with higher body fat mass. Moreover, vitamin D supplementation trials have shown that obese people reached much lower serum 25-hydroxyvitamin D concentrations compared to normal weight (BMI <25 kg/m2) participants with equivalent oral dosages (47).
- Magnesium deficiency: Recent findings suggest that high magnesium intakes may reduce the risk of vitamin D insufficiency. Magnesium regulates the activity of critical enzymes in vitamin D metabolism, which would explain how magnesium deficiency negatively affects vitamin D status (48).
Assessing vitamin D nutritional status
Growing awareness that vitamin D insufficiency has serious health consequences beyond rickets and osteomalacia highlights the need for accurate assessment of vitamin D nutritional status. It is currently agreed that the measurement of total serum 25-hydroxyvitamin D concentration (1 ng/mL corresponding to 2.5 nmol/L) is the best indicator to evaluate vitamin D status. However, high-quality evidence is still needed to ensure that the current cutoff values are optimal to define states of insufficiency and deficiency (40). Moreover, only recently, efforts have been made toward the standardization of commercially and laboratory-developed 25-hydroxyvitamin D assays, such that guidelines have been developed using largely unstandardized research data (49). Although laboratory reference values for sufficient vitamin D status were initially based on serum 25-hydroxyvitamin D concentrations from cohorts of healthy individuals, additional studies have suggested that health-based cutoff values aimed at preventing secondary hyperparathyroidism and bone loss should be considerably higher. Indeed, while it is considered that serum 25-hydroxyvitamin D concentrations less than 8 to 10 ng/mL (20 to 25 nmol/L) indicate severe deficiency associated with rickets and osteomalacia, several studies have observed that PTH concentrations (50, 51) and calcium absorption (52) were not optimized with serum 25-hydroxyvitamin D concentrations below 32 ng/mL (80 nmol/L).
Yet, more recent studies have failed to find threshold values of serum 25-hydroxyvitamin D concentrations in relation to PTH suppression and optimal calcium absorption. On the one hand, a cross-sectional analysis of 312,962 clinical samples did not find any evidence of threshold for PTH suppression in the well-fitted curve displaying the inverse association between paired measurements of serum PTH and 25-hydroxyvitamin D, even with 25-hydroxyvitamin D concentrations beyond 70 ng/mL (175 nmol/L) (53). This contradicted an analysis of the US National Health and Nutrition Examination Survey (NHANES 2003-2006) that estimated maximum PTH suppression for 25-hydroxyvitamin D concentrations of 40 ng/mL (100 nmol/L) and above (54). In addition, both studies identified evidence of mild hyperparathyroidism (serum PTH >65 pg/mL) in individuals with serum 25-hydroxyvitamin D concentrations well beyond 20 ng/mL (50 nmol/L), questioning the use of serum PTH as a sensible indicator of vitamin D insufficiency (53, 54). On the other hand, a randomized, placebo-controlled trial in postmenopausal women with vitamin D insufficiency (serum 25-hydroxyvitamin D <20 ng/mL) supplemented with daily vitamin D3 doses from 400 to 4,800 IU found little change (6%) in calcium absorption over a normal 25-hydroxyvitamin D concentration range of 20 to 66 ng/mL (55).
The current cutoffs proposed by the Institute of Medicine (IOM) are as follows: deficiency as serum 25-hydroxyvitamin D values <12 ng/mL (<30 nmol/L), insufficiency as serum 25-hydroxyvitamin D values of 12 to 19 ng/mL (30 to 49 nmol/L), and sufficiency as serum 25-hydroxyvitamin D values of 20 to 50 ng/mL (50 to 125 nmol/L) (56). The dietary reference intakes (EAR, RDA) set by the IOM are based on achieving circulating 25-hydroxyvitamin D concentrations (20 to 50 ng/mL) that are adequate to maintain bone health and optimal calcium absorption (57).
Yet, considering the potential role of circulating concentrations lower than 30 ng/mL in the burden of many chronic diseases (6), the US Endocrine Society has suggested to define cutoff values as follows: deficiency as serum 25-hydroxyvitamin D values ≤20 ng/mL (≤50 nmol/L), insufficiency as serum 25-hydroxyvitamin D values of 21 to 29 ng/mL (51 to 74 nmol/L), and sufficiency as serum 25-hydroxyvitamin D values of 30 to 100 ng/mL (75 to 250 nmol/L) (40). This alternate target range is supported by some observational studies, but it is not based on data from randomized controlled trials (see Disease Prevention) (47). With these latter cutoff values, studies from across the world have estimated that hypovitaminosis D is widespread and that children and adults of all ages are equally at risk of insufficiency and deficiency (58). Data from supplementation studies indicate that vitamin D intakes of at least 800 to 1,000 IU/day are required by adults living in temperate latitudes to achieve serum 25-hydroxyvitamin D concentrations of at least 30 ng/mL (75 nmol/L) (40).
Finally, total serum 25-hydroxyvitamin D concentrations may not always adequately reflect vitamin D bioavailability (59), and additional evidence is needed to improve the determination of vitamin D status in different ethnic populations.
The RDA
In 2010, the Food and Nutrition Board (FNB) of the IOM set a recommended dietary allowance (RDA) based on the amount of vitamin D needed for bone health. While the RDA was increased from the adequate intake (AI) set in 1997, the optimal levels of recommended intakes and serum 25-hydroxyvitamin D to minimize hyperthyroidism and maximize bone health in the general population remain controversial (40). The RDA for vitamin D is listed in Table 1 by life stage and gender.
| Life Stage | Age | Males | Females | ||
|---|---|---|---|---|---|
| μg/day | IU/day | μg/day | IU/day | ||
| Infants (AI) | 0-6 months |
10
|
400
|
10
|
400
|
| Infants (AI) | 6-12 months |
10
|
400
|
10
|
400
|
| Children | 1-3 years |
15
|
600
|
15
|
600
|
| Children | 4-8 years |
15
|
600
|
15
|
600
|
| Children | 9-13 years |
15
|
600
|
15
|
600
|
| Adolescents | 14-18 years |
15
|
600
|
15
|
600
|
| Adults | 19-70 years |
15
|
600
|
15
|
600
|
| Adults | 71 years and older |
20
|
800
|
20
|
800
|
| Pregnancy | all ages |
-
|
-
|
15
|
600
|
| Breastfeeding | all ages |
-
|
-
|
15
|
600
|
Disease Prevention
Mortality
In a nine-year follow-up analysis of the Third US National Health and Nutrition Examination Survey (NHANES III) that included 15,099 participants (of which 77% were Caucasians), serum concentrations of 25-hydroxyvitamin D — standardized as per the methodology developed by the Vitamin D Standardization Program [VDSP] — were examined in relation to mortality. The analysis suggested an increase in all-cause mortality with decreasing serum 25-hydroxyvitamin D concentrations <16 ng/mL (60). In contrast, the risk of all-cause mortality varied little for baseline serum 25-hydroxyvitamin D concentrations in the range of 16 to 40 ng/mL (60). Similar results were obtained in a meta-analysis of eight prospective cohort studies that considered the relationship between standardized 25-hydroxyvitamin D concentrations and mortality during a median follow-up period of 10.5 years. The risk of death was found to be 19% higher with 25-hydroxyvitamin D concentrations between 12 and 15.99 ng/mL and 56% higher with concentrations <12 ng/mL compared to the risk associated with concentrations between 30 and 39.99 ng/mL (61). A meta-analysis of 73 prospective cohort studies, including >800,000 participants, found that the lowest versus highest tertile of serum 25-hydroxyvitamin D concentrations was associated with greater risks of all-cause mortality (+35%), mortality due to cardiovascular disease (+35%), and mortality due to cancer (+14%) (62). Yet, a Mendelian randomization analysis — which limits bias due to confounding and reverse causation (63) — of data from three large Danish cohorts of 95,766 adults found a significant association of genetically low plasma 25-hydroxyvitamin D concentrations with all-cause and cancer-related mortality, but not with cardiovascular disease-related mortality (64). Finally, two meta-analyses of randomized controlled trials have suggested a modest reduction in all-cause mortality in older people supplemented with vitamin D and calcium, but not vitamin D alone (62, 65). Additional placebo-controlled trials need to examine further whether supplementation with vitamin D alone or in combination with calcium might help prevent premature death in replete individuals.
Osteoporosis
Vitamin D status, osteoporosis, and risk of fracture
Although the causes of osteoporosis are multifactorial, vitamin D insufficiency can be an important etiological factor in older adults. Osteoporosis affects one-third of women aged 60 to 70 years and two-thirds of women aged 80 years and above (66). A multinational (18 different countries with latitudes ranging from 64 degrees north to 38 degrees south) survey of more than 2,600 postmenopausal women with osteoporosis revealed that 31% of subjects had 25-hydroxyvitamin D concentrations <20 ng/mL (50 nmol/L) (67). In addition, a case-control study that included 111 hip fracture patients and 73 controls (median age, 83 years) found that lower serum concentrations of both 25-hydroxyvitamin D and vitamin K1 in patients compared to controls were associated with an increased risk of hip fracture (68). Without sufficient vitamin D from sun exposure or dietary intake, intestinal calcium absorption can be significantly reduced. This increases PTH secretion by the parathyroid glands; sustained PTH elevation may result in increased bone resorption, which in turn may increase the risk of osteoporotic fracture (69).
Vitamin D supplementation and bone mineral density
The progressive loss of bone mineral density (BMD) leading to osteopenia (pre-osteoporosis) and osteoporosis is commonly observed in older adults, especially the elderly. The results of a meta-analysis of 23 randomized controlled trials with more than 4,000 participants (mean age, 59 years) showed little evidence for an effect of vitamin D supplementation on BMD at any of the five skeletal sites examined, including lumbar spine, femoral neck, trochanter, forearm, and total body. A significant increase in BMD was reported only at the femoral neck (70). It was, however, suggested that individuals in this age group would have adequate calcium intake and thus normal bone metabolism, explaining the lack of an effect of vitamin D in strengthening bone mass (71). Conversely, in older individuals, vitamin D supplementation is essential to correct and maintain adequate concentrations of serum 25-hydroxyvitamin D and to prevent secondary hyperparathyroidism and BMD loss (72).
Vitamin D supplementation and risk of fracture
A prospective cohort study that followed more than 72,000 postmenopausal women in the US for 18 years found that those who consumed at least 600 IU/day of vitamin D from diet and supplements had a 37% lower risk of osteoporotic hip fracture than women who consumed less than 140 IU/day of vitamin D (73). However, daily supplementation with 400 IU of vitamin D3, in combination with 1,000 mg calcium, did not significantly reduce risk of hip fracture compared to a placebo in 36,282 postmenopausal women from the Women's Health Initiative trial (74), suggesting that there might be a threshold of vitamin D intake that is necessary to observe reductions in fracture risk. Results of a genetic analysis of data from this trial also suggested that beneficial effects of vitamin D and calcium supplementation on fracture risk might be limited to women with the lowest genetic risk of low BMD (75). Yet, this trial has been questioned for reasons that include poor adherence and the fact that participants were allowed to take additional vitamin D and calcium supplements that might have confounded the results. In addition, use of hormone replacement therapy was not considered in the study of the effect of vitamin D and calcium on skeletal health in postmenopausal women despite being a major confounding factor in this population (57, 76).
Another trial, the Randomised Evaluation of Calcium Or vitamin D (RECORD) study, reported that oral supplemental vitamin D3 (800 IU/day) alone, or in combination with calcium (1,000 mg/day), did not prevent the occurrence of osteoporotic fractures in elderly adults who had already experienced a low-trauma, osteoporotic fracture (77). In this latter study as well, a number of limitations, including poor adherence and/or the fact that vitamin D supplementation did not raise serum 25-hydroxyvitamin D concentrations to a level that would protect against fractures, might explain the lack of an effect (78). Despite high adherence to treatment, the incidence of non-vertebral fracture was similar in postmenopausal women supplemented with vitamin D3 (initial dose of 200,000 IU followed by 100,000/month) or placebo for over three years in the Vitamin D Assessment (ViDA) trial (79).
Nevertheless, the US Preventive Services Task Force that conducted the meta-analysis of 11 randomized, placebo-controlled trials, including 52,915 older people (of whom 69% were postmenopausal women), found that the supplementation of vitamin D (300-1,000 IU/day) and calcium (500-1,200 mg/day) for up to seven years resulted in a 12% reduction in the risk of any new fracture (80). Another meta-analysis of 11 randomized, double-blind, placebo-controlled trials on the effect of vitamin D supplementation in 31,022 individuals (91% women) 65 years and older indicated that those with the highest vitamin D intake (792-2,000 IU/day) had a 30% lower risk of hip fracture and a 14% lower risk of any non-spine fracture (81). Finally, a third meta-analysis of trials that examined the effect of combined vitamin D and calcium in preventing fractures in older men and postmenopausal women also concluded that the risk of new fractures, including hip fractures, was significantly reduced in those supplemented compared to controls (82). Interestingly, the three meta-analyses have found that the prevention of fractures by supplemental vitamin D and calcium was limited to institutionalized, older people. Indeed, the risk of fracture was not significantly reduced by vitamin D in community-dwelling seniors (80-82).
Vitamin D supplementation and postural balance, muscle strength, and risk of fall
A meta-analysis of seven observational studies in 840 fallers and 1,330 non-fallers found significantly lower serum 25-hydroxyvitamin D concentrations in fallers than in non-fallers (83). Moreover, another meta-analysis of four cohorts from three observational studies reported a modest yet significant inverse association between vitamin D status and the risk of fall (83). Several randomized controlled trials have examined the impact of vitamin D supplementation on muscle strength, postural balance, or risk of fall in older subjects. A meta-analysis of these trials found limited evidence of an effect of vitamin D supplementation on muscle strength and mobility, based on only one type of test for each outcome. Nevertheless, in a recent randomized, double-blind, placebo-controlled study in 160 postmenopausal women (ages, 50-65 years) with suboptimal vitamin D status (mean serum 25-hydroxyvitamin D concentration <20 ng/mL), supplementation with 1,000 IU/day of vitamin D3 significantly improved vitamin D status, as well as upper and lower limb muscle strength and postural balance parameters (84, 85). The risks of fall and recurrent falls were found to be two- to three-fold greater in women in the control group than in those supplemented with vitamin D3 (85). In contrast, another 12-month randomized controlled study in 200 older adults (of which 58% had a baseline serum 25-hydroxyvitamin D concentration <20 ng/mL) showed no benefits regarding lower extremity function or odds of falling in those supplemented with 2,000 IU/day (+/- 10 µg of calcidiol) compared to those who received 800 IU/day (86). The recently published post-hoc analysis of the ViDA trial found no differences in odds of falling and number of falls reported by 5,108 community-dwelling participants (ages, 50-84 years) regardless of whether they were randomized to receive supplemental vitamin D (100,000 IU/month, i.e. ~3,350 IU/day) or a placebo for a mean 3.4 years (79). Most ViDA participants had serum 25-hydroxyvitamin D concentrations ≥20 ng/mL, which might at least partly explain the lack of an effect of vitamin D on falls (87).
Overall, the current evidence suggests that vitamin D3 supplements of 800-1,000 IU/day may be helpful in reducing falls and fracture rates in older adults. In order for vitamin D supplementation to be effective in preserving bone health, adequate dietary calcium (1,000 to 1,200 mg/day) should be consumed (see the article on Calcium) (88).
Cancer
Ecologic studies first suggested an association between Northern latitudes, vitamin D deficiency, and cancer incidence (89). Since the 1980s, several prospective cohort studies have examined the association of vitamin D intake or status and various types of cancer. A 2013 systematic review and meta-analysis of 16 prospective studies, including 137,567 subjects, reported an 11% reduction in total cancer incidence and a 17% reduction in cancer mortality with each 20 ng/mL (50 nmol/L) increase in circulating 25-hydroxyvitamin D concentrations. Yet, a sex-based subgroup analysis of eight studies found an inverse association between circulating vitamin D and cancer mortality in women, but not in men (90). In addition, increasing evidence suggests that a few variations in the gene coding for the vitamin D receptor (VDR) might influence individual vitamin D status and subsequently modify the susceptibility to site-specific cancers (91) and influence cancer survival (92). Finally, many malignant tumors have been found to express the VDR, including breast, lung, skin (melanoma), colon, and bone (93), suggesting that they might be susceptible to the effects of vitamin D. Numerous experimental studies have demonstrated that biologically active forms of vitamin D, such as 1α,25-dihydroxyvitamin D and its analogs, upon binding to the VDR, can control cell fate by inhibiting proliferation and/or inducing cell differentiation or death (apoptosis) of a number of cancerous cell types (94).
Colorectal cancer
The geographic distribution of colon cancer mortality resembles the historical geographic distribution of rickets (95), providing circumstantial evidence that decreased sunlight exposure and diminished vitamin D nutritional status may be related to an increased risk of colon cancer. Evidence from observational studies has largely supported this hypothesis. A recent meta-analysis of four prospective cohort studies, four cross-sectional studies, and seven case-control studies found an inverse relationship between circulating vitamin D and incidence of colorectal adenoma — a benign tumor that may transform to become malignant (96). The analysis identified a 32% risk reduction between top versus bottom quantiles of serum 25-hydroxyvitamin D concentrations (96). Additionally, there is strong evidence from meta-analyses of prospective cohort studies to suggest that higher vitamin D intakes and serum 25-hydroxyvitamin D concentrations are associated with reductions in colorectal cancer risk (97-99). The most recent meta-analysis of four prospective cohort, 17 nested case-control, and three case-control studies found a 38% reduced risk of colorectal cancer with high versus low quantiles of circulating 25-hydroxyvitamin D concentrations (100). A dose-response analysis estimated that serum 25-hydroxyvitamin D concentrations of ~20 to 30 ng/mL (compared to ≤12 ng/mL) were associated with a 17% lower risk of colorectal cancer, and the risk was even lower (-35%) with a serum concentration of 55 ng/mL (100). An earlier dose-response analysis based on five nested case-control studies had estimated that serum 25-hydroxyvitamin D concentrations ≥33 ng/mL (compared to ≤12 ng/mL) were associated with a 50% lower risk of colorectal cancer (101).
However, in a seven-year, randomized, double-blind, placebo-controlled trial in 36,282 postmenopausal women participating in the Women's Health Initiative study, a combination of supplemental vitamin D3 (400 IU/day) and calcium (1,000 mg/day) did not lower incidence of colorectal cancer (102). Another randomized controlled trial of vitamin D3 supplementation (1,000 IU/day), with or without calcium supplementation (1,200 mg/day), found no reduction in the risk of colorectal adenoma recurrence over a three-to-five year period, compared to placebo, after initial adenoma removal in participants (103). Whether these daily vitamin D doses are too low to detect any effect on cancer incidence is not clear (101, 104). Additional randomized clinical trials are needed to assess whether vitamin D supplementation could help prevent colorectal cancer. Moreover, it is uncertain whether genetic variations (polymorphisms) in the sequence of genes involved in vitamin D metabolism and function can influence the relationship between vitamin D status and risk of colorectal adenoma or colorectal cancer (105-107).
Finally, growing evidence suggests that adequate vitamin D status may be linked to better survival of colorectal cancer patients. A meta-analysis of five prospective studies found a 35% reduced risk of colorectal cancer-specific mortality in cancer patients with higher serum 25-hydroxyvitamin D concentrations. A dose-response analysis estimated that every 8 ng/mL increase in 25-hydroxyvitamin D concentration was associated with a 10% decrease in colorectal cancer mortality (108).
Breast cancer
Although ecologic evidence suggests that breast cancer mortality rises with increasing latitudes and decreasing sunlight exposure (109), the most current observational data provide little support for an association between vitamin D nutritional status and risk of breast cancer. An early prospective study of women who participated in the First US National Health and Nutrition Examination Survey (NHANES I) found that Caucasian women with adequate sunlight exposure and dietary vitamin D intake had a significantly reduced risk of breast cancer 20 years later (110). Nonetheless, when this study was included in a meta-analysis with nine more recent prospective studies, there was no significant difference in the risk of developing breast cancer between the highest and lowest levels of vitamin D intakes (111). Moreover, whether an association exists between circulating vitamin D concentrations and risk of breast cancer is unclear. One meta-analysis of 14 observational studies (9,110 cases and 16,244 controls) reported an overall risk reduction of 16% when the highest quantile of serum 25-hydroxyvitamin D concentrations was compared to the lowest. This inverse association was statistically significant in postmenopausal women but not in premenopausal women (112). Yet, another meta-analysis that included a similar set of 14 prospective studies (two studies were different) found no overall association (111). Additionally, a meta-analysis of studies conducted in patients in the early stage of breast cancer identified associations between inadequate vitamin D status and increased risks of recurrence and death (113). Evidence from randomized controlled trials is currently too limited to conclude whether vitamin D supplementation may reduce breast cancer incidence (reviewed in 114).
Nonetheless, three meta-analyses have found an inverse association between circulating vitamin D concentrations and breast cancer-related mortality (111, 115, 116). In one meta-analysis of one retrospective and five prospective cohort studies, the highest versus lowest categories of serum vitamin D concentrations was associated with a 33% reduction in mortality; a dose-response analysis found a 12% reduction per 8 ng/mL increase in serum vitamin D (115).
Finally, current evidence does not suggest that specific genetic variations in the gene coding for the VDR may influence the risk of breast cancer (117, 118).
Other types of cancer
Evidence associating vitamin D status with other types of cancer is currently limited. While incidence of prostate cancer appears to be inversely associated with the availability of sunlight, prospective cohort studies have not generally found significant relationships between serum 25-hydroxyvitamin D concentrations and subsequent risk of developing prostate cancer (119, 120). In fact, some studies have suggested an increased risk of prostate cancer with higher circulating vitamin D concentrations. For example, a nested case-control study of men (622 cases and 1,451 controls) from Scandinavia found a U-shaped relationship between serum 25-hydroxyvitamin D concentrations and prostate cancer risk. In that study, serum 25-hydroxyvitamin D concentrations of 7.6 ng/mL or lower, or 32 ng/mL or higher, were associated with increased prostate cancer risk (121). A meta-analysis of 17 nested case-control studies, three prospective cohort studies, and one retrospective cohort study found a 17% increased risk of prostate cancer in individuals in the highest versus lowest categories of blood 25-hydroxyvitamin D concentrations (122). Potential confounding factors that might explain the detection of a slight increase in prostate cancer cases in men with high circulating vitamin D concentrations have been highlighted in a recent publication (123).
Finally, recent meta-analyses of observational studies found an inverse relationship between vitamin D status and risk of lung cancer (124, 125) and bladder cancer (126, 127). Yet, in the few and often heterogeneous studies published to date, serum 25-hydroxyvitamin D concentrations were not associated with other cancer types, including non-Hodgkin’s lymphoma (128), ovarian cancer (129), gastric cancer (130), or skin cancers (131).
Autoimmune diseases
Insulin-dependent diabetes mellitus (type 1 diabetes mellitus), multiple sclerosis (MS), rheumatoid arthritis (RA), and systemic lupus erythematosus (SLE) are examples of autoimmune diseases. Autoimmune diseases occur when the body mounts an immune response against its own tissue, rather than a foreign pathogen. In type 1 diabetes mellitus, insulin-producing β-cells of the pancreas are the target of an inappropriate immune response. In MS, the targets are the myelin-producing cells of the central nervous system, and in RA, the targets are the collagen-producing cells of the joints (132). SLE is characterized by the presence of a large spectrum of autoantibodies resulting in potential damage to multiple tissues (133). Autoimmune responses are mediated by immune cells called T cells. The biologically active form of vitamin D, 1α,25-dihydroxyvitamin D, has been found to modulate T cell responses, such that the autoimmune responses are diminished. Ecologic studies have found that the prevalence of autoimmune diseases (particularly for MS; 134) increases as latitude increases, suggesting that lower exposure to UVB radiation and associated decreases in skin vitamin D synthesis may play a role in the pathology of these diseases. Results of several prospective cohort studies also suggest that adequate vitamin D status at different ages (including in utero, early childhood, and during adolescence) could possibly decrease the risk of autoimmune diseases.
Type 1 diabetes mellitus
Lower levels of circulating vitamin D have been reported in patients newly diagnosed with type 1 diabetes mellitus compared to age- and sex-matched non-diabetic subjects (135, 136). A greater prevalence of vitamin D insufficiency and deficiency has also been observed in prediabetic children who developed multiple islet autoantibodies (antibodies against insulin-secreting pancreatic cells) compared to autoantibody-negative children. However, a prospective study that followed the cohort of prediabetic children found that their vitamin D status, defined as either insufficient, deficient, or sufficient, was not associated with rate of progression to type 1 diabetes after 5 or 10 years of follow up (137). An earlier prospective cohort study of children born in Finland during the year 1966 and followed for 30 years found that children supplemented with vitamin D during the first year of life had an 88% lower risk of developing type 1 diabetes compared to those receiving no supplementation. Moreover, children suspected of having had rickets (severe vitamin D deficiency) during the first year of life showed a significantly higher risk of developing type 1 diabetes (138). Thus, vitamin D supplementation appears protective against type 1 diabetes onset, and suboptimal vitamin D status in infancy may have long-term effects on immune responses later in life.
There are also limited data suggesting that maternal vitamin D insufficiency during pregnancy may influence the risk of type 1 diabetes in offspring. In a recent case-control study, the risk of childhood onset of type 1 diabetes was more than two-fold greater in children whose mothers had serum 25-hydroxyvitamin D concentrations <21.6 ng/mL (54 nmol/L) during the last trimester of pregnancy compared to children born from women with serum 25-hydroxyvitamin D >35.6 ng/mL (89 nmol/L) (139). Other case-control studies have found that vitamin D supplementation during pregnancy was associated with a lower risk of their children developing diabetes-related autoantibodies (140, 141). However, a larger study conducted in mothers of children at increased genetic risk for diabetes reported no association between the appearance of islet autoantibodies and/or diabetes onset in offspring in the first year of life and maternal vitamin D intake during pregnancy (142). Another case-control study failed to observe a relationship between serum 25-dihydroxyvitamin D during early pregnancy and type 1 diabetes diagnosis in offspring (143). Large prospective studies are needed to establish whether maternal vitamin D status during pregnancy can influence the risk of type 1 diabetes in offspring.
Finally, the relationship of polymorphisms in vitamin D metabolism-related genes and type 1 diabetes is currently under investigation. It has been proposed that specific polymorphisms in genes, such as CYP27B1 (coding for 25-hydroxyvitamin D3-1α-hydroxylase) and VDR, may be functionally relevant to the action of vitamin D and may thus affect disease susceptibility. In a study conducted in 8,517 children and adolescents with type 1 diabetes and 7,320 control subjects, polymorphisms in genes involved in cholesterol synthesis and vitamin D hydroxylation were linked to circulating vitamin D concentrations and diabetic status (26).
Multiple sclerosis
Low levels of sun exposure and vitamin D deficiency appear to be associated with the development of multiple sclerosis (MS). Poor vitamin D status may compromise the function of specific immune cells critical in the regulation of various immune responses and help trigger autoimmunity in MS (144). Genetic determinants of low vitamin D status have been recently linked to an increased susceptibility to adult-onset MS in a Mendelian randomization analysis of data from the Multiple Sclerosis Genetics Consortium (145). This echoed the results of several observational studies that suggested an association between vitamin D sufficiency and decreased MS risk. A retrospective study of levels of ambient UV radiation and cases of MS conducted in Australia revealed that MS incidence in offspring was inversely correlated to maternal exposure to UV during early pregnancy (146). Sun exposure was also used as a surrogate marker for vitamin D exposure in a recent case-control study that included 1,660 MS patients and 3,050 controls. The authors found that infrequent outdoor activities and the use of sunscreen during early childhood and adolescence were associated with an increased risk of developing MS later in life (147). In a cross-sectional study, sun exposure and intake of cod liver oil (rich in vitamin D) during childhood were linked to later symptom onset among veterans with relapsing MS (148). Additionally, a case-control study in US military personnel, including 257 cases of diagnosed MS, found that Caucasian subjects in the highest quintile of serum 25-hydroxyvitamin D (>39.6 ng/mL) had a 62% lower risk of developing MS compared to the lowest quintile (<25.3 ng/mL) (149). Further, in two large cohorts of over 187,000 US women followed for at least 10 years, vitamin D supplement use (≥400 IU/day) was associated with a 41% reduction in the risk of developing MS (150). Another prospective, uncontrolled study monitored incidence of relapse in relation to vitamin D status in 156 patients with relapsing-remitting MS before and after they were given supplemental vitamin D (100,000 IU/month; 6-42 months, median of 31 months), in addition to first-line immunomodulatory therapy (151). Each 4 ng/mL increase in serum 25-hydroxyvitamin D concentration was associated with a 14.9% decrease in incidence of relapse (151). In a multicenter study conducted in patients newly diagnosed with a clinically isolated syndrome (CIS) and treated with interferon (IFN)-β, vitamin D status was predictive of MS disease activity and progression. Higher serum 25-hydroxyvitamin D concentrations (≥20 ng/mL or ≥50 nmol/L) in the first year following CIS diagnosis predicted a longer time to MS diagnosis, lower number of new lesions, and lower changes in lesion and brain volume during the subsequent four years of follow-up (152). However, a retrospective study suggested that vitamin D status in patients with relapsing-remitting MS had no predictive value regarding the time to conversion to secondary progressive MS, which is characterized by a worsening of disability (153).
Clinical trials have failed to demonstrate any benefit of vitamin D supplementation, alone or in combination with IFN-β treatment, with respect to relapse rates and disability-related symptoms in MS patients (154, 155). In other trials, supplemental vitamin D3 also failed to demonstrate immunomodulary activities (156-159). In a recent randomized, placebo-controlled trial in 53 IFN-β-treated patients with relapsing-remitting MS, supplementation with vitamin D3 (7,000 IU/day for four weeks, followed by 14,000 IU/day until week 48) showed little effect on the proportion of some regulatory T and B lymphocytes over the 48-week study period. Vitamin D3 only appeared to help maintain the proportion of anti-inflammatory CD4+ T cells — which decreased in patients given placebo — but failed to enhance their reactivity when stimulated with 1,25-dihydroxyvitamin D in vitro (157). In another trial, supplementation with vitamin D3 (10,400 IU/day for three months) to patients with relapsing-remitting MS was found to reduce the proportion of pro-inflammatory IL-17-producing CD4+ T cells, which are thought to play a central role in MS development (160).
Rheumatoid arthritis
Vitamin D deficiency may also be implicated in the etiology and/or progression of rheumatoid arthritis (RA), although evidence is mainly from animal studies. The absence of vitamin D receptors (VDR) in genetically modified mice has been linked to higher levels of inflammation and increased susceptibility to autoimmunity (161). When transgenic mice that spontaneously develop inflammatory arthritis are also deficient in VDR, they develop a more aggressive form of chronic arthritis (162). Also, specific polymorphisms in the VDR gene have been linked to an increased susceptibility to RA in certain populations, although how these genetic variants influence vitamin D functionality is not fully understood (163-165). The current data, however, point to a role for vitamin D in modulating the inflammatory process that underlies many chronic diseases, including RA. Several cross-sectional studies in individuals with moderate-to-high levels of inflammation have reported either no association or an inverse association between circulating 25-hydroxyvitamin D concentration and markers of inflammation. Nonetheless, there is a lack of intervention trials to show whether vitamin D supplementation could limit inflammation and reduce disease risk (including RA) in subjects with high inflammation levels (166).
At this time, it remains unclear whether the prevalence of vitamin D deficiency is linked to RA incidence. In a large cohort study of nearly 30,000 postmenopausal US women, subjects with the highest total vitamin D intakes (≥467.7 IU/day) had a 33% lower risk of developing RA after 11 years of follow-up than those with the lowest intakes (<221.4 IU/day) (167). Yet, more recent analyses of two large cohorts of nearly 200,000 US women followed for several decades found no association between reported vitamin D dietary intakes (using food frequency questionnaires) during adolescence or adulthood and incidence of RA later in life (168, 169). Moreover, several studies that explored the relationship between circulating vitamin D and disease activity in RA patients have reported mixed results (reviewed in 170). Yet, two recent meta-analyses of observational studies found an inverse relationship between vitamin D status and disease activity in RA patients, assessed using the Disease Activity Score 28 (DAS28) (171, 172). Finally, there is a dearth of studies exploring the effect of vitamin D supplementation on disease activity in arthritis subjects. A small randomized, double-blind, placebo-controlled study in 22 RA patients failed to demonstrate improvements in disease activity and inflammation level in subjects supplemented with calcium (1,500 mg/day) and high doses of vitamin D2 (ergocalciferol; averaging over 4,500 IU/day) for a year compared to placebo (173). Another three-month, randomized controlled trial in 41 women with early RA found no additional benefits of supplemental vitamin D3 (one bolus dose of 300,000 IU) to standard care (methotrexate and glucocorticoids) regarding T-helper lymphocyte enumeration, cytokine production, or clinical parameters including disease activity (174). Supplemental vitamin D also failed to reduce disease recurrence rate in RA patients enrolled in two small randomized controlled trials (175, 176). Since these studies have several limitations, including small sample size, additional research is warranted.
Systemic lupus erythematosus
More prevalent and severe in non-Caucasian populations (Hispanics, African descendants, and Asians) (177), systemic lupus erythematosus (SLE) is an autoimmune disease with heterogeneous clinical manifestations. The disease can potentially affect most tissues and organs, including skin (skin rash and photosensitivity), kidneys (nephritis), and joints (arthritis). There is evidence of a role for vitamin D in the prevention of SLE in animal models (178). Interestingly, a recent meta-analysis of 11 case-control studies found that specific VDR polymorphisms were linked to SLE in Asians particularly (179). However, the functional relevance of such genetic variants is not known (180). Analyses of two large prospective cohort studies of nearly 200,000 US women failed to show an association between dietary vitamin D intake (measured by food frequency questionnaire) during adolescence or adulthood and incidence of SLE later in life (168, 169).
Yet, a suboptimal vitamin D status is commonly observed in subjects with SLE, and this is partly explained by the lack of sunlight exposure, which tends to aggravate disease symptoms (181, 182). Serum concentrations of 25-hydroxyvitamin D were inversely correlated with measures of disease activity in a cohort of 378 patients with SLE (183). The correction of vitamin D insufficiency with high levels of vitamin D3 (100,000 IU/week for one month followed by 100,000 IU/month for six months) in 20 subjects with SLE was linked to a reduction in signs of immune imbalance and in levels of autoantibodies typically detected in SLE, suggesting a therapeutic value for vitamin D in disease treatment (184). Another prospective study conducted in 52 vitamin D-deficient patients with cutaneous lupus erythematosus (a type of lupus with skin disorders only) reported a reduction in disease severity in the group supplemented with vitamin D3 (1,400 IU/day initially, followed by 800 IU/day) and calcium for one year compared to untreated patients (185). Supplementation with vitamin D3 (200 IU/day for one year) was also able to reduce the level of inflammatory cytokines in a randomized, placebo-controlled study conducted in 267 patients with SLE (186). In another randomized, placebo-controlled trial, supplementation with vitamin D3 (50,000 IU/week for six months) improved SLE disease activity index (SLEDAI) and European Consensus Lupus Activity Measurement (ECLAM) scores, as well as some measures of fatigue in young adults with juvenile-onset SLE (187). However, in two other recent studies, supplementation with vitamin D3 (weekly/monthly bolus doses equivalent to ~800 to 7,000 IU/day for 6 to 24 months) improved vitamin D status in SLE patients but failed to show any benefit regarding disease activity (188, 189). While oral vitamin D administration to SLE patients is well tolerated, its efficacy remains questionable and deserves further investigation in clinical trials.
Summary
Thus, evidence from human epidemiological studies suggests that while it cannot yet be concluded that vitamin D supplementation is beneficial in prevention or treatment of autoimmune disease, it is reasonable to assume that correcting vitamin D insufficiency and maintaining sufficient levels could possibly help decrease disease risk (190).
Cardiovascular disease
Hypertension (high blood pressure)
Hypertension is a well-known risk factor for cardiovascular disease (CVD) (191). The results of observational and clinical studies suggest a role for vitamin D in lowering blood pressure, which may be partly explained by the fact that 1α,25-dihydroxyvitamin D inhibits renin synthesis (see Function). Thus, vitamin D deficiency and subsequent upregulation of the renin-angiotensin system may contribute to high blood pressure and CVD risk. It has also been suggested that elevated PTH concentrations may increase the risk of hypertension and CVD (6). Yet, in a recent prospective cohort study of 3,002 individuals (mean age, 59 years at baseline), the incidence of hypertension, which affected 41% of participants during the nine year follow-up period, was not higher in those with serum 25-hydroxyvitamin D concentrations lower than 20 ng/mL and was only marginally associated with elevated PTH concentrations (192). Nevertheless, a meta-analysis of seven prospective studies, including a total of 48,633 participants with nearly 5,000 incident hypertension cases, found a 30% lower risk of hypertension in those in the top versus bottom tertiles of serum 25-hydroxyvitamin D concentrations. The dose-response analysis estimated that every 10 ng/mL increase in serum 25-hydroxyvitamin D concentration was associated with a 12% lower risk of hypertension (193). Another meta-analysis of four prospective and 14 cross-sectional studies also reported an inverse relationship between circulating 25-hydroxyvitamin D and hypertension (194).
Endothelial dysfunction
Vascular endothelium dysfunction, which contributes to an increased risk of cardiovascular disease (CVD), is common in patients with chronic kidney disease (CKD) (195). In CKD patients, abnormal endothelial function is associated with low values of flow-mediated dilation (FMD) of the brachial artery, a surrogate marker of vascular health. In a recent study conducted in subjects with mild-to-moderate CKD, serum 25-hydroxyvitamin D concentrations were positively associated with FMD values, suggesting a link between suboptimal vitamin D status and endothelial dysfunction (196). In a preliminary intervention study, 26 patients with moderate CKD and vitamin D insufficiency (mean value, 17.2 ng/mL) were supplemented twice with 300,000 IU of vitamin D3 (at weeks 1 and 8) and followed for a total of 16 weeks. Vitamin D supplementation nearly doubled serum 25-hydroxyvitamin D concentrations and decreased PTH concentrations by 68.5%; improved vitamin D status was accompanied by increased FMD values and reduced levels of endothelial dysfunction markers (197). A recent meta-analysis of 12 small randomized controlled trials in participants at high risk for CVD found a significant increase in FMD with vitamin D supplementation (daily doses, 2,500-5,000 IU; weekly dose, 50,000 IU; monthly dose, 60,000 IU; single-bolus doses, 100,000-200,000 IU) for eight weeks to six months (198).
Cardiovascular events in observational studies and clinical trials
To date, the many epidemiological studies investigating the relationship between vitamin D and outcomes of CVD have provided mixed results (reviewed in 199). Recent Mendelian randomization studies found no association between genetically low serum 25-hydroxyvitamin D concentrations and risks of coronary heart disease, ischemic heart disease, or myocardial infarction (200, 201), suggesting that associations reported in observational studies may be due to confounding or reverse causation. In the RECORD trial in 5,292 older people (see Osteoporosis), supplementation with 800 IU/day of vitamin D3 (± calcium) reduced the risk of first cardiac failure but had no effect on the risk of myocardial infarction and stroke compared to supplementation with calcium alone or placebo (202). Data on the effect of vitamin D supplementation on cardiovascular events were collected from 21 randomized controlled studies (including the RECORD trial) in 13,033 participants (≥60 years old) and combined in a meta-analysis (202). No effect of vitamin D (including vitamin D analogs) was found for major cardiovascular events, including heart failure, myocardial infarction, and stroke over follow-up periods of 1 to 6.2 years (202). However, caution is advised when interpreting these results since the trials were initially designed to evaluate the effect of vitamin D on bone health, and cardiovascular outcomes were not primary endpoints. Several randomized controlled trials exploring the effect of vitamin D supplementation on CVD risk are currently underway (203), including two large trials, the Vitamin D and Omega-3 Trial (VITAL) in the US (204) and the D-Health trial in Australia (205). The results of one randomized controlled trial, the Vitamin D Assessment (ViDA) trial in New Zealand, were recently published. The total number of CVD events and time to first CVD event during follow-up did not differ between those supplemented with vitamin D3 (initial dose of 200,000 IU for the first month followed by monthly doses of 100,000 IU) and those given a placebo for a median 3.3 years (206).
Type 2 diabetes mellitus
People with metabolic syndrome are at increased risk for type 2 diabetes mellitus (noninsulin-dependent diabetes mellitus) and cardiovascular disease (CVD). Metabolic syndrome refers to several metabolic disorders, including dyslipidemia, hypertension, insulin resistance, and obesity. A recent study found that the prevalence of type 2 diabetes was associated with low levels of serum 25-hydroxyvitamin D (<30 ng/mL) in 1,801 patients with metabolic syndrome. During an eight-year follow-up period, lower risks of all-cause mortality (72% lower risk) and CVD-specific mortality (64% lower risk) were reported in individuals with serum 25-hydroxyvitamin D concentrations over 30 ng/mL (75 nmol/L) when compared to those with concentrations below 10 ng/mL (25 nmol/L) (207).
In healthy people, vitamin D sufficiency is positively correlated with insulin sensitivity and adequate pancreatic β-cell function. Conversely, vitamin D deficiency might affect glucose homeostasis and cause impaired glucose tolerance and insulin resistance (208). In a cross-sectional study conducted in 12,719 adults, of whom 4,057 had prediabetes (i.e., an increased risk of developing type 2 diabetes), the prevalence of prediabetes was associated with lower concentrations of serum 25-hydroxyvitamin D (≤32.4 ng/mL). Subjects with the lowest concentrations of serum 25-hydroxyvitamin D (≤17.7 ng/mL) were more likely to be current smokers, obese, and have hypertension (209). Vitamin D insufficiency in high-risk individuals may accelerate the progression to overt diabetes. In a prospective study of 2,378 middle-aged men and women followed for 8 to 10 years, the risk for progression to type 2 diabetes from prediabetes was 62% lower in women and 60% lower in men in the highest compared to the lowest quartile of circulating vitamin D (>28.4 ng/mL vs. <18.5 ng/mL). A dose-response analysis measured an average 23% reduction in the risk of progression to type 2 diabetes for every 4 ng/mL (10 nmol/L) increment in serum 25-hydroxyvitamin D concentration (210). A recent review and meta-analysis of 18 prospective cohort studies, including over 210,000 participants followed for a median period of 10 years, found that individuals in the top third of vitamin D levels (reported as either circulating vitamin D or dietary intakes) had lower risks of developing type 2 diabetes (19% lower risk) and metabolic syndrome (14% lower risk) compared to those in the bottom third (211). In another meta-analysis of nine prospective studies, including 28,258 older people (mean age, 67.7 years), lower versus higher circulating vitamin D concentrations at baseline were found to be associated with a 17% higher risk of developing type 2 diabetes over a median follow-up period of 7.3 years (212). Currently, limited evidence suggests that vitamin D supplementation may improve insulin sensitivity in individuals with glucose intolerance or manifest type 2 diabetes (213-216). There is a need for well-designed clinical trials to examine whether maintaining adequate vitamin D levels can prevent adverse metabolic outcomes in healthy and at-risk individuals.
Neurodegenerative diseases
Cognitive impairment and Alzheimer's disease
Alzheimer’s disease (AD) is the most common form of dementia, characterized by the presence of extra-neuronal β-amyloid plaques and intra-neuronal Tau protein aggregates (known as neurofibrillary tangles) in the brain. Mechanistic models currently investigated in animal research suggest that vitamin D deficiency or disorders of vitamin D metabolism and/or the disruption of the vitamin D-VDR pathway in the cerebral regions of the cortex and hippocampus may be involved in the degeneration of neurons and loss of cognitive functions (217). Experimental evidence supporting a role for vitamin D in calcium channel regulation, neuroprotection, and immunomodulation in the central nervous system also implies that low vitamin D status may precede or contribute to cognitive dysfunction with age (218).
A number of observational studies have examined cognitive decline and degenerative brain disease in the elderly in relation to dietary intake of vitamin D and serum 25-hydroxyvitamin D concentration. In a large French cohort study on osteoporosis and hip fractures in postmenopausal women, impairments in global cognitive performance, assessed with the Pfeiffer Short Portable Mental State Questionnaire (SPMSQ), were associated with lower dietary intakes of vitamin D (<1,400 IU/week vs. ≥1,400 IU/week) in 5,596 elderly women (mean age, 80.5 years) (219). A seven-year follow-up study of a subgroup of 498 women indicated that the risk of Alzheimer’s disease (but not other types of dementia) was 77% lower in those in the highest versus the lowest quintiles of vitamin D dietary intakes at baseline (220). Some, but not all, observational studies have found an association between low serum 25-hydroxyvitamin D concentrations and mild cognitive impairment in older adults (219, 221, 222). The cross-sectional and longitudinal analysis of two prospective studies, which included 1,604 men (223) and 6,257 women (224) aged 65 and over, reported a 60% greater odds of cognitive impairment at baseline and a 58% increased risk of cognitive decline during a four-year follow-up period in women, but not in men, with vitamin D deficiency (circulating 25-hydroxyvitamin D <10 ng/mL vs. ≥30 ng/mL). In the nested case-control, multiethnic, Singapore Kidney Eye Study, which included 2,273 individuals (mean age, 70.4 years), serum 25-hydroxyvitamin D concentrations were inversely correlated with cognitive deficits affecting retrograde episodic memory, semantic memory, and orientation in time, as assessed by the Abbreviated Mental Test (AMT) (225).
Yet, systematic reviews and meta-analyses of observational studies have given mixed results regarding the association of vitamin D status with cognitive performance and AD (226-230). Moreover, the recent analysis of data from 1,182 men followed for 18 years in the Uppsala Longitudinal Study of Adult Men (ULSAM) failed to find associations of genetic determinants of vitamin D synthesis, vitamin D intakes, and plasma 25-hydroxyvitamin D concentrations with risks of cognitive impairments, AD, vascular dementia, or all-cause dementia (231). In contrast, another Mendelian randomization study associated genetic determinants of low vitamin D status to higher risk of AD in the International Genomics of Alzheimer’s Project dataset (17,008 AD cases and 37,154 healthy cases) (232).
Nevertheless, the prevalence of vitamin D insufficiency/deficiency ranges between 70% and 90% in older adults, and correcting low concentrations of serum 25-hydroxyvitamin D may help improve cognitive processes, in particular executive functions (233). In a small, non-randomized controlled study in an outpatient clinic, global cognitive function was assessed at baseline and after 16 months in 20 patients supplemented with 800 IU/day (or 100,000 IU/month) of vitamin D and in 24 control subjects. The supplementation of outpatients with vitamin D resulted in the correction of low vitamin D status (average serum 25-hydroxyvitamin D concentration was 16.8 ng/mL at baseline and 30 ng/mL at 16 months) and was associated with a significantly improved scoring in cognition tests compared to the non-supplemented group (234). In a small randomized, placebo-controlled clinical trial in 32 mild-to-moderate AD patients receiving nasal insulin, high doses of vitamin D2 supplementation for eight weeks (up to 36,000 IU/day) did not significantly improve cognitive performance compared to low doses (1,000 IU/day) (235). More research is needed to investigate a causal relationship between vitamin D repletion and potential long-term cognitive benefits in older adults. Further, it is of great importance to evaluate whether correcting vitamin D deficiency in cognitively impaired subjects can improve the impact of anti-dementia therapy (236).
Parkinson's disease
Parkinson’s disease (PD) has been associated with a high prevalence of vitamin D insufficiency among patients, especially those with greater mobility problems (237). A case-control study of 296 outpatients with a mean age of 65 years indicated that 23% of PD subjects had serum 25-hydroxyvitamin D concentrations lower than 20 ng/mL compared to 16% and 10% of AD and healthy individuals, respectively (238). In a prospective cohort study conducted among 3,173 men and women aged 50-79 years and free of PD at baseline, individuals in the highest quartile of serum 25-hydroxyvitamin D (≥20 ng/mL for women and ≥22.8 ng/mL for men) had a 67% lower risk of PD compared to those in the lowest quartile (≤10 ng/mL for women and ≤11.2 ng/mL for men) (239). Meta-analyses pooling data from observational studies all showed that vitamin D inadequacy was more likely reported in subjects with PD than in healthy controls (240-242).
In a randomized, double-blind, placebo-controlled study, 112 PD patients (mean age, 72 years) on standard PD treatment were supplemented with 1,200 IU/day of vitamin D or a placebo for 12 months. Vitamin D supplementation nearly doubled serum 25-hydroxyvitamin D concentration (from mean of 22.5 ng/mL to 41.7 ng/mL) in supplemented subjects and limited the progression of PD, as indicated by a greater proportion of patients who showed no worsening (as assessed by the Hoehn and Yahr stage and the United Parkinson Disease Rating Scale part II) in the supplemented group compared to the placebo group (243). It is not known whether vitamin D insufficiency has a role in the pathogenesis of the disease, but the repletion of vitamin D may provide health benefits that go beyond the prevention and/or the treatment of PD. For example, vitamin D deficiency may contribute to the increased risk of osteoporosis and bone fracture in individuals with neurologic disorders, including PD and multiple sclerosis (244-246). Interestingly, sunlight exposure was found to be associated with improved vitamin D status, higher bone mineral density of the second metacarpal bone, and lower incidence of hip fracture in a prospective study conducted in 324 elderly people with PD (247).
Adverse pregnancy outcomes
A systematic review and meta-analysis of 31 observational studies on maternal vitamin D status and pregnancy outcomes indicated that vitamin D insufficiency may be associated with gestational diabetes mellitus, preeclampsia, and bacterial vaginosis in pregnant women. Low maternal serum vitamin D during pregnancy was also linked to an increased risk for small-for-gestational age infants and low-birth-weight infants, but not for Cesarean section (248). However, the number of intervention trials is currently too limited to draw conclusions as to whether vitamin D supplementation during pregnancy might reduce the incidence of the above-mentioned adverse outcomes (249).
Gestational diabetes mellitus
Abnormal hyperglycemia due to pancreatic β-cell dysfunction characterizes the onset of gestational diabetes mellitus (GDM) in pregnant women without known type 2 diabetes mellitus. This condition is associated with serious adverse maternal outcomes, including preeclampsia, high risk of Cesarean delivery, and life-long increased risk of developing metabolic syndrome and type 2 diabetes mellitus. GDM may also contribute to increased risks of fetal macrosomia (excessive birth weight), neonatal hypoglycemia, infant respiratory distress, and increased life-long risk for obesity, glucose intolerance, type 2 diabetes mellitus, and cardiovascular disease in the offspring (reviewed in 250).
A recent prospective study conducted in 655 pregnant women found that the mean serum 25-hydroxyvitamin D concentration during the first trimester of pregnancy was significantly lower in 54 women who developed incident GDM compared to the rest of the cohort (23 ng/mL vs. 25.4 ng/mL). After multiple adjustments for confounding factors of vitamin D status and GDM risk (including overweight/obesity and prior history of type 2 diabetes and GDM), the study found each 7.5 ng/mL decrease in serum 25-hydroxyvitamin D concentration during early pregnancy was associated with a 48% higher risk of developing GDM (251). Low serum 25-hydroxyvitamin D concentrations (<29.4 ng/mL) during the second trimester of pregnancy were also associated with GDM incidence in a nested case-control study of 118 women with GDM and 219 matched control subjects (252). Five meta-analyses (248, 253-256), including observational studies of moderate-to-high quality, also reported that maternal serum vitamin D concentrations during pregnancy were inversely related to the risk of developing GDM despite evidence of bias amongst studies, such as the use of different methods for serum 25-hydroxyvitamin D measurement, measures done in different trimesters, and different criteria to assess GDM (reviewed in 257).
Further, evidence for the role of vitamin D in glucose regulation during pregnancy was reported in a small randomized, double-blind, placebo-controlled trial in 54 pregnant women diagnosed with GDM. The supplementation with 50,000 IU of vitamin D3 twice during a six-week period (at day 1 and day 21) resulted in significantly lower fasting plasma glucose and serum insulin concentration, reduced insulin resistance, and improved insulin sensitivity compared to placebo (258). This suggests that vitamin D deficiency may adversely affect glucose tolerance during pregnancy and contribute to the onset of GDM. Yet, the potential benefits of vitamin D supplementation in the prevention of glucose intolerance and GDM during pregnancy have not been assessed. A multicentered, randomized controlled trial (DALI) is ongoing in Europe to evaluate the effects of vitamin D and lifestyle interventions (healthy eating and physical activity) on the metabolic status of pregnant women at risk of GDM (inclusion criteria: pre-pregnancy BMI ≥29 kg/m2) (259). Preliminary findings suggest that healthy eating and physical activity can help lower gestational weight gain, when compared to standard-of-care; however, these lifestyle changes are unlikely to reduce the risk of GDM among obese pregnant women (260). The results regarding the effect of vitamin D supplementation in the DALI study are yet to be published.
Health outcomes in offspring
During pregnancy, increased intestinal calcium absorption and mobilization of calcium from the skeleton allows accretion of calcium within the fetal skeleton. Yet, observational studies that examined the relationship between maternal vitamin D status and measures of fetal bone growth have not provided consistent results (261, 262). In addition, recent data from the Maternal Vitamin D Osteoporosis Study (MAVIDOS) suggested no difference in whole-body bone mineral content (BMC) of newborns from mothers randomized to daily supplementation with either vitamin D3 (1,000 IU) or placebo from <17 weeks’ gestation until delivery (263). Further, the risk of fracture in Danish children ages 10 to 18 years was similar regardless of whether their mothers were exposed to extra vitamin D from fortification during pregnancy (264). While maternal vitamin D supplementation during pregnancy effectively prevents the neonate’s risk of vitamin D deficiency at birth (265), there is little evidence that neonatal vitamin D status influences the risk of fracture later during childhood (266).
A few observational studies have given rather weak evidence in support of a relationship between maternal vitamin D sufficiency during pregnancy and incidence of respiratory conditions and allergies in children (267). A randomized controlled trial found that the supplementation of 108 pregnant women in the third trimester (at week 27 of gestation until delivery) with either 800 IU/day or a bolus dose of 200,000 IU of vitamin D3 did not decrease the risk of wheezing, allergic rhinitis, food allergy diagnosis, lower respiratory tract infections, or eczema in offspring at three years of age compared to placebo (N=50) (268). A more recent double-blind, randomized controlled trial found that vitamin D3 supplementation of 295 Danish pregnant women, from weeks 22 to 26 of gestation until delivery, with 2,800 IU/day (70 μg/day) — compared to 400 IU/day (10 μg/day) of vitamin D3 (i.e., the current recommendation in Denmark) (N=286) — reduced the risk of troublesome episodes of lung symptoms by 17% in offspring during the first three years of life (269). However, no differences were reported regarding the risk of persistent wheeze, asthma, allergic sensitization, respiratory tract infections, or eczema between treatment and control groups (269). In a similar randomized, double-blind, controlled trial — the Vitamin D Antenatal Asthma Reduction Trial — conducted in 777 US pregnant women with a history of asthma, allergic rhinitis, or eczema (or whose partner had such an history), supplementation with 2,400 IU/day (60 μg/day) or 400 IU/day (10 μg/day) did not result in differences in the risk of developing asthma or recurrent wheezing in their children at age three years (270). Despite the lack of significance reported in individual studies, the pooled analysis of the three trials found a 19% reduction in the risk of recurrent wheeze in children whose mothers received high-dose versus low-dose vitamin D supplementation during pregnancy (271). In a cohort of 378 mother-child pairs, high serum 25-hydroxyvitamin D concentrations measured in the 34th week of pregnancy were associated with an increase in food allergy of the child during the first two years of life, warranting careful evaluation of the safety of vitamin D supplementation during pregnancy (272).
Since vitamin D insufficiency has been linked to autoimmunity (see Autoimmune diseases), it has also been proposed that poor maternal vitamin D status during pregnancy may contribute to an increased risk of autoimmune diabetes (insulin-dependent type 1 diabetes mellitus) in the offspring. Yet, the results of a study of 3,723 children at high genetic risk for type 1 diabetes and followed for a mean 4.3 year-period found that maternal intake of vitamin D (from food and/or supplements) during the third trimester of pregnancy (assessed through food frequency questionnaires) was not associated with advanced β-cell autoimmunity or clinical diabetes (142). In a nested case-control study, there was no difference in mean serum concentration of 25-hydroxyvitamin D during the first trimester of pregnancy between 343 mothers of children with type 1 diabetes and 343 control mothers (143). A follow-up study suggested that specific maternal VDR polymorphisms, rather than vitamin D status, may be linked to an increased susceptibility to developing type 1 diabetes in children (273). Another nested case-control study (119 mothers of children with type 1 diabetes and 129 control mothers) found an inverse association between maternal vitamin D-binding protein — but not 25-hydroxyvitamin D — concentration during the third trimester of pregnancy and the risk of type 1 diabetes in children (274). At present, there is no established causality between maternal vitamin D status during pregnancy and risk of autoimmune disease in offspring.
Acute respiratory infections
More than 200 viruses are responsible for causing familiar infections of the upper respiratory tract (URT), known as the common cold, resulting in symptoms of nasal congestion and discharge, cough, sore throat, and sneezing (275). The analysis of cross-sectional data from 18,883 participants (ages 12 years and older) of the Third US National Health and Nutrition Examination Survey (NHANES III) reported an inverse relationship between serum 25-hydroxyvitamin D concentrations and recent (self-reported) URT infection (URTI). Compared to levels of circulating vitamin D of 30 ng/mL or above, the risk of URTI was 24% higher in individuals with concentrations between 10 and 29 ng/mL and 36% higher in those with levels below 10 ng/mL (276). A subgroup analysis indicated that low concentrations of serum 25-hydroxyvitamin D in subjects with asthma and chronic obstructive pulmonary disease (COPD) were linked to a greater susceptibility to URTI when compared to people without pulmonary disease.
In a randomized, double-blind, placebo-controlled trial conducted in 322 healthy adults (ages ≥18 years), supplementation with monthly doses of vitamin D3 (200,000 IU for the first two months and 100,000 IU for the following 16 months) significantly raised the mean serum 25-hydroxyvitamin D concentration (from 29 ng/mL to 48 ng/mL) in the intervention group but did not decrease the occurrence of URTI compared to placebo (277). Moreover, in a larger, multicenter, four-arm clinical trial in 2,259 subjects (aged 45-75 years) with a history of colorectal adenoma, daily vitamin D3 supplementation of 1,000 IU did not reduce the number or the duration of URTI episodes during winter or the rest of the year, even among participants with the lowest serum 25-hydroxyvitamin D concentrations at baseline (278). In addition, the post-hoc analysis of data from a randomized, placebo-controlled trial in 644 individuals (ages 60-84 years) found that monthly supplementation with 30,000 IU or 60,000 IU of vitamin D3 for a maximum period of one year did not significantly decrease the rate of antibiotic prescriptions for bacterial airway infections. The stratified analysis, however, found that doses of 60,000 IU/month reduced the risk of using antibiotics in participants ≥70 years old by 47% (279). In addition, compared to placebo, the supplementation of pregnant women with vitamin D3 (with 2,000 IU/day) for three months until birth followed by the supplementation of their infants (800 IU/day) from birth to six months of age significantly reduced the number of acute respiratory infections after the intervention period in children 6 to 18 months old (280). Interestingly, despite significant heterogeneity among trials, the pooled analysis of these data with that of 21 additional trials suggested an overall 12% reduction in incidence of URTI with vitamin D3 given as bolus doses (every week, month, or every three months), daily doses, or a combination of bolus and daily doses (281). Subgroup analyses revealed a 42% reduction in URTI risk with vitamin D3 supplementation in subjects with baseline serum 25-hydroxyvitamin D concentrations <10 ng/mL, while there was no protective effect of vitamin D3 in those with concentrations ≥10 ng/mL. Moreover, large bolus doses (≥30,000 IU) were found to be ineffective compared to daily or weekly doses such that, in subgroup analyses that excluded bolus doses, daily and weekly vitamin D3 regimens appeared to be protective against URTI regardless of baseline vitamin D status. Finally, the effect of vitamin D3 supplementation on URTI did not appear to vary with age, BMI, the presence of asthma or COPD, and flu vaccination status (281).
The findings of a large clinical trial in New Zealand, the Vitamin D Assessment (ViDA) Study, confirmed the ineffectiveness of large bolus doses of vitamin D in reducing the risk of acute respiratory infections (282). In this double-blind, placebo-controlled trial in 5,110 older adults (ages 50-84 at baseline), supplementation with an initial dose of 200,000 IU of vitamin D3 followed by 100,000 IU/month of vitamin D3 for a median of 3.3 years had no effect on the incidence of (self-reported) acute respiratory infections (HR, 1.01; 95% CI, 0.94-1.07). Upon data stratification by vitamin D status, a similar null result (HR, 1.08; 95% CI, 0.95-1.23) was seen in study participants with blood vitamin D concentrations ≤20 ng/mL (≤50 nmol/L; 282).
The recently completed VITAL (Vitamin D and Omega-3 Trial) may provide additional evidence of an effect of daily dosing (2,000 IU/day of vitamin D) on risk of airway infections in older adults, although the prevention of infectious disease is a secondary outcome in the trial (204, 283, 284).
In a trial in preschool-age children, vitamin D supplementation with 2,000 IU/day for at least four months did not decrease the incidence (285) or severity (286) of URTI in winter compared to supplementation with 400 IU/day for at least four months. Supplementation with 2,000 IU/day led to significantly higher mean serum 25-hydroxyvitamin D concentrations compared to the lower dose (48.7 ng/mL vs. 36.8 ng/mL), but the 400 IU dose may have been sufficient to prevent URTI in these young children (285). In a placebo-controlled trial in 1,300 healthy children and adolescents in rural Vietnam (baseline 25-hydroxyvitamin D concentrations of 26 ng/mL for both the control and intervention group), supplementation with 14,000 IU/week of vitamin D for eight months significantly reduced the incidence of non-influenza viral respiratory infections by 24%, but not the incidence of influenza A or B infection (287).
Coronavirus disease, COVID-19
The coronavirus disease, COVID-19, is caused by infection with the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). This virus originated in Wuhan, China in late 2019 and rapidly spread throughout the world causing a global pandemic. Flu-like symptoms, including cough, fever, fatigue, and difficulty breathing, as well as several other diverse symptoms characterize COVID-19 (see the websites of the CDC and the WHO). The effects of the disease vary widely, with the most severe resulting in pneumonia, respiratory distress, and death. Some individuals infected with SARS-CoV-2, however, are asymptomatic but can transmit virus to others (288). Moreover, SARS-CoV-2 infected individuals can spread disease before they experience symptoms (i.e., presymptomatic transmission) (289), placing importance on public health measures like hand washing, face mask wearing, social distancing, and testing and contact tracing to curtail the pandemic.
A number of observational studies have examined the correlation of vitamin D status and COVID-19 incidence, with most — but not all (290-292) — finding vitamin D deficiency or insufficiency associated with an increased risk of SARS-CoV-2 infection. In a retrospective cohort study of more than 191,000 US residents administered a SARS-CoV-2 test during a three-month period in spring 2020, SARS-CoV-2 positivity was strongly associated with a lower serum 25-hydroxyvitamin D concentration, measured at a single point in the 12 months prior to the viral test (293). In this study, each 1 ng/mL increment of serum 25-hydroxyvitamin D was linked to a 1.6% lower risk of SARS-CoV-2 positivity, with the risk being the lowest at serum concentrations of 55 ng/mL and above (293). In a retrospective, population-based study among 7,807 people in Israel, including 782 SARS-CoV-2 positive cases, plasma 25-hydroxyvitamin D concentrations below 30 ng/mL were associated with a 50% higher risk of SARS-CoV-2 infection compared to higher vitamin D concentrations (294). The vitamin D status assessment in this study was done prior to the viral testing, but details on the time frame are lacking. The temporal relationship of the vitamin D status measure and COVID diagnostic test is also a concern in an analysis of a large dataset from the UK Biobank, which found vitamin D status was not associated with SARS-CoV-2 infection: the study utilized vitamin D status assessments done 10 to 14 years prior to the COVID-19 diagnostic test (291). Consistent with findings of most available studies, a small retrospective cohort study (295) and five case-control studies (296-300) reported an inverse association of vitamin D status and SARS-CoV-2 infection. A 2021 meta-analysis of 10 observational studies, including 361,934 participants, reported an association between vitamin D insufficiency or deficiency and increased risk of COVID-19 (OR, 1.43; 95% CI: 1.00-2.05), although heterogeneity across studies was high (301). Further, in an analysis of data from the UK Biobank, regular use of vitamin D supplements was found to be associated with a 34% lower risk of SARS-CoV-2 infection compared nonusers of vitamin D supplements (292).
Additionally, most of the available observational research suggests an association between low vitamin D status and COVID-19 severity, with studies finding vitamin D deficiency linked to an increased risk of disease severity, as measured by need for hospitalization, intensive care unit admission (302), stage of pneumonia (in men but not in women; 303), need for non-invasive ventilation (304), need for invasive mechanical ventilation (305), or a combination of these and other indicators (297). Another study found that insufficient vitamin D status, defined as plasma 25-hydroxyvitamin D concentrations below 30 ng/mL, was associated with an increased likelihood of COVID-related hospitalization, but the association failed to reach statistical significance (p=0.061) except in a subanalysis of individuals over 50 years of age (OR, 2.71; 95% CI: 1.55-4.78; p<0.001) (294). In contrast to most of the evidence to date, one case-control study found vitamin D deficiency was not linked to COVID-19 severity (298). Further, a few studies have looked at the relationship of vitamin D status and COVID-19 mortality, with four finding vitamin D deficiency to be associated with an increased risk of death (303, 305-307) and two finding no association (304, 308), although one of the studies finding no association used vitamin D status measurements more than 10 years prior to the COVID-19 diagnostic testing (308).
Since there are several known risk factors for severe illness from COVID-19, including advanced age, obesity, and preexisting type 2 diabetes or cardiovascular disease (309), it is important for observational studies to control for potential confounders. Two quasi-experimental studies reported that bolus supplementation with vitamin D3 (either 50,000 IU per month or 80,000-100,000 IU every two to three months) prior to or during SARS-CoV-2 infection was associated with a less severe illness and improved survival in frail elderly COVID-19 patients (310, 311). A number of randomized controlled trials of vitamin D supplementation in COVID-19 prevention and treatment are currently underway; the results of these trials will inform on the causality of the association. Nevertheless, the currently available data indicate that improving vitamin D status through supplementation represents a modifiable risk factor for COVID-19.
Disease Treatment
Atopic dermatitis
Atopic dermatitis or eczema is particularly prevalent in industrialized countries, affecting 10%-20% of children and 1%-3% of adults. Atopic dermatitis is a chronic inflammatory skin disorder characterized by dry and pruritus (itchy) areas of the skin in affected subjects. Local skin inflammation and immune dysfunction can damage the epidermal barrier and increase the susceptibility to skin infections and atopic reactions in affected individuals. The disease is often associated with other atopic diseases, including food allergies, asthma, and allergic rhinitis (312).
While the etiology of the disease is not fully elucidated, it has been suggested that vitamin D deficiency might contribute to the onset and/or the severity of the disease (313). Recently, using large-scale datasets from Caucasian people of European descent, including the UK Biobank resource (314) and the SUNLIGHT (35), the GABRIEL asthma (315), and EAGLE eczema (316) consortia, a Mendelian randomization study found no association between genetically low serum 25-hydroxyvitamin D concentrations and risks of atopic dermatitis, asthma, or high serum immunoglobulin (Ig)-E concentrations (317).
A number of randomized controlled studies have examined whether vitamin D might be an effective adjunct tool in disease management, possibly through regulating local inflammatory reactions and stimulating antimicrobial activities in the skin. Moreover, the beneficial effect of phototherapy observed in specific cases of atopic dermatitis may be partly mediated by the action of vitamin D (312). In a small randomized, double-blind, placebo-controlled study in 45 patients with atopic dermatitis and low vitamin D status (70% of subjects had serum 25-hydroxyvitamin D concentrations <20 ng/mL), daily administration of 1,600 IU of oral vitamin D3, alone or together with 600 IU/day of vitamin E, for a period of 60 days significantly reduced the extent and intensity of eczema, as assessed by the SCORAD (SCORing Atopic Dermatitis) score (318). Vitamin D3 (1,600 IU/day for 60 days) also improved vitamin D status and reduced disease severity in 53 patients with atopic dermatitis in another small randomized trial (319). More recently, vitamin D3 (1,000 IU/day for one month) improved the severity of winter-related atopic dermatitis in Mongolian children, as shown by changes in Eczema Area and Severity Index (EASI) and Investigator’s Global Assessment (IGA) scores (320). A meta-analysis of four small trials (including those cited above) confirmed that supplemental vitamin D can lead to measurable clinical improvements in affected individuals (321). Larger trials are needed to strengthen these preliminary findings and determine the most appropriate and effective supplementation regimen. Of note, topical treatment of psoriasis with vitamin D analogs has been approved by the US Food and Drug Administration (FDA) and may be effective in the management of other skin disorders (322).
Inflammatory bowel diseases
Several ill-defined environmental and genetic factors are thought to contribute to the development of the inappropriate immune response to the intestinal microbiota that causes ulcerative colitis (UC) and Crohn’s disease (CD). While specific VDR polymorphisms may be linked to an increased susceptibility to developing UC and CD (323), higher vitamin D intakes and predicted circulating levels were found to be associated with a reduced incidence of UC and CD in a large cohort of 72,719 women (324). A meta-analysis of six observational studies found an inverse association between vitamin D status and severity of CD (325). Three studies have investigated whether vitamin D3 could benefit patients with CD, possibly through reducing intestinal inflammation. In one multicenter, double-blind, placebo-controlled study, the relapse rate in CD patients in remission after one year of treatment was significantly lower in those supplemented daily with 1,200 IU of vitamin D3 and 1,200 mg of calcium compared to those who received calcium alone (13% vs. 29%) (326). In a second pilot study, incremental daily doses of vitamin D3, from 1,000 IU up to 5,000 IU, were administrated over a 24-week period to 18 CD patients in order to achieve circulating 25-hydroxyvitamin D concentrations >40 ng/mL. Although half of the patients failed to achieve 40 ng/mL, the mean 25-hydroxyvitamin D concentration was raised to 45 ng/mL (from a baseline mean of 16 ng/mL), and the overall improvement in vitamin D status was associated with a significant decrease in disease severity as assessed by Crohn’s Disease Activity (CDAI) scores (327). In a three-month, randomized, double-blind, placebo-controlled trial in 27 CD patients in remission, daily supplementation with vitamin D3 (2,000 IU) improved vitamin D status but had no significant effect on intestinal permeability ('leaky gut') or measures of inflammation and disease activity (328). Yet, the study suggested that achieving serum 25-hydroxyvitamin D concentrations ≥30 ng/mL might help reduce intestinal inflammation and improve patients’ quality of life. Additional studies are needed to confirm the therapeutic efficacy of vitamin D in inflammatory bowel diseases.
Cardiovascular disease
The prospective analysis of 41,504 electronic medical records in the Intermountain Heart Collaborative study found that only one-third of patients had adequate serum 25-hydroxyvitamin D concentrations (>30 ng/mL); vitamin D insufficiency (serum 25-hydroxyvitamin D concentrations ≤30 ng/mL) was associated with increased prevalence and incidence of many cardiovascular conditions, including hypertension, coronary artery disease, heart failure, and stroke (329). Suboptimal vitamin D status has also been linked to arterial stiffness and vascular endothelial dysfunction — strong determinants of incident hypertension and adverse cardiovascular outcomes (330).
Hypertension
Several intervention studies have evaluated the effect of vitamin D supplementation on blood pressure. An early controlled clinical trial in 18 men and women with untreated mild hypertension living in the Netherlands found that exposure to UVB radiation three times weekly for six weeks during the winter increased serum 25-hydroxyvitamin D concentrations by 162%, lowered PTH concentrations by 15%, and decreased 24-hour ambulatory systolic and diastolic blood pressure measurements by an average of 6 mm Hg (331). A recent meta-analysis of 16 randomized controlled trials involving 1,879 participants, either healthy or with pre-existing cardiometabolic conditions (including hypertension), found no significant reduction in systolic and diastolic blood pressures with vitamin D supplementation (800-8,571 IU/day for five weeks to one year). However, a subgroup analysis of six trials found a significant reduction of 1.31 mm Hg in diastolic blood pressure in individuals with preexisting conditions. While improvements in blood pressure may be expected in cases of vitamin D insufficiency/deficiency, the authors noted that suboptimal vitamin D status in participants was not exclusively observed in those with cardiometabolic conditions (332). In contrast, in two recent intervention studies — the Styrian vitamin D hypertension trial (333) and the Vitamin D therapy in individuals at high risk of hypertension trial [DAYLIGHT] (334) — subjects with (pre)hypertension supplemented with vitamin D3 (400 to 4,000 IU/day for two to six months) showed no evidence of blood pressure lowering, regardless of their baseline vitamin D status (insufficient or adequate, according to the IOM’s current cutoffs).
Conditions that decrease vitamin D synthesis in the skin, such as having dark-colored skin, living in temperate latitudes, and aging, are associated with increased prevalence of hypertension (335), suggesting that vitamin D may reduce blood pressure levels in selected groups of individuals. In the above-mentioned meta-analysis, one four-arm, double-blind, placebo-controlled clinical trial was conducted in 283 African Americans randomized to receive daily vitamin D3 supplements of 1,000 IU, 2,000 IU, or 4,000 IU for a period of three months. Systolic blood pressure was decreased by 0.66 mm Hg with 1,000 IU/day, 3.4 mm Hg with 2,000 IU/day, and by 4 mm Hg with 4,000 IU/day while it increased by 1.7 mm Hg in the placebo group when compared to baseline. A significant reduction of 0.2 mm Hg in systolic blood pressure was detected per 1 ng/mL incremental increase in 25-hydroxyvitamin D concentration. However, there was no statistical difference on three-month change in blood pressure between vitamin D3 and placebo (336). Another randomized, placebo-controlled study in 150 elderly participants (mean age, 77 years) showed that supplementation with 100,000 IU of vitamin D3 every three months for one year did not significantly lower blood pressure compared to placebo (337).
Further research is needed to determine whether vitamin D supplementation is helpful in the prevention or management of hypertension.
Congestive heart failure
Congestive heart failure (also called cardiac insufficiency) is characterized by increased heart rate and subsequent hypertrophy of the left heart ventricle. Cardiac insufficiency is associated with a reduced left ventricular ejection fraction (LVEF), assessed by echocardiography. Inhibitors of angiotensin-converting enzyme (ACE) (see Blood pressure regulation) are currently used as first-line therapy for patients with heart failure. In a cross-sectional study in healthy patients who underwent coronary angiography, serum 25-hydroxyvitamin D concentrations <30 ng/mL were associated with poorer coronary flow rates (338). Suboptimal vitamin D status has also been linked to poorer prognosis in patients with heart failure (339). Over the past several years, a number of intervention studies have examined the effect of vitamin D supplementation in those with cardiac insufficiency. In a 12-week randomized, double-blind, placebo-controlled study, daily supplementation with 1,200 IU of vitamin D3 in children with chronic congestive heart failure led to a significant increase in vitamin D status accompanied by an improved heart muscle performance (increased LVEF), as well as by lower levels of PTH and pro-inflammatory cytokines (340). In another randomized, double-blind, placebo-controlled trial in 64 elderly patients with heart failure, participants receiving 800 mg/day of calcium and 50,000 IU/week of vitamin D3 did not perform significantly better at physical performance tasks (used as proxy to assess aerobic capacity and skeletal muscle strength) compared to those supplemented with calcium only (341). A recent meta-analysis of seven small randomized placebo-controlled trials in 573 subjects with heart failure showed that vitamin D supplementation (from 1,000 IU/day to 50,000 IU/week) for six weeks to nine months could reduce serum concentrations of PTH, tumor necrosis factor-α (TNF-α), and C-reactive protein (CRP). Yet, there were no significant differences in LVEF, circulating interleukin-10 (IL-10) concentration, and renin concentration between patients treated with vitamin D and those given a placebo (342). Finally, in the EVITA (Effect of vitamin D on all-cause mortality) trial in patients with end-stage heart failure and inadequate vitamin D status (baseline serum 25-hydroxyvitamin D values, 8.6-19.7 ng/mL), supplementation with 4,000 IU/day for three years did not reduce the risk of mortality compared to placebo (343).
Sources
Sunlight
Solar ultraviolet-B radiation (UVB; wavelengths of 290 to 315 nanometers) stimulates the production of vitamin D3 in the epidermis of the skin (344). Sunlight exposure can provide most people with their entire vitamin D requirement. Children and young adults who spend a short time outside two or three times a week will generally synthesize all the vitamin D they need to prevent deficiency. One study reported that serum vitamin D concentrations following exposure to one minimal erythemal dose of simulated sunlight (the amount required to cause a slight pinkness of the skin) to the whole body was equivalent to ingesting approximately 10,000 to 25,000 IU of vitamin D (345). People with dark-colored skin synthesize markedly less vitamin D on exposure to sunlight than those with lighter complexion (34). Additionally, older adults have diminished capacity to synthesize vitamin D from sunlight exposure and frequently use sunscreen or protective clothing in order to prevent skin cancer and sun damage. The application of sunscreen with an SPF factor of 10 reduces production of vitamin D by 90% (30). In latitudes around 40 degrees north or 40 degrees south (Boston is 42 degrees north), there is insufficient UVB radiation available for vitamin D synthesis from November to early March. Ten degrees farther north or south (Edmonton, Canada), the "vitamin D winter" extends from mid-October to mid-March. It has been estimated that up to 15 minutes of daily sun exposure on the hands, arms, and face around 12 pm throughout the year at 25 degrees latitude (Miami, FL) and during the spring, summer, and fall at 42 degrees (Boston, MA) latitude may provide a light-skinned individual with 1,000 IU of vitamin D (346).
Food sources
Vitamin D is found naturally in only a few foods, such as some fatty fish (mackerel, salmon, sardines), fish liver oils, eggs from hens that have been fed vitamin D, and mushrooms exposed to sunlight or UV light. In the US, milk and infant formula are fortified with vitamin D so that they contain 400 IU (10 μg) per quart. However, other dairy products, such as cheese and yogurt, are not always fortified with vitamin D. Some cereal, bread, and fruit juices may also be fortified with vitamin D. Accurate estimates of average dietary intakes of vitamin D are difficult because of the high variability of the vitamin D content of fortified foods (347). The vitamin D content (sum of vitamin D2 and vitamin D3) of some vitamin D-rich foods is listed in Table 2 in both international units (IU) and micrograms (μg). For more information on the nutrient content of specific foods, search USDA's FoodData Central. The 25-hydroxyvitamin D metabolite is also present at low levels is certain foods, including meats, dairy products, and eggs (348, 349).
| Food | Serving | Vitamin D (IU) | Vitamin D (μg) |
|---|---|---|---|
| Pink salmon, canned | 3 ounces | 465 | 11.6 |
| Mackerel, canned (boneless) | 3 ounces | 248 | 6.2 |
| Sardines, canned | 3 ounces | 164 | 4.1 |
| Milk, low-fat (1%), fortified with vitamin D | 8 fluid ounces | 108 | 2.7 |
| Orange juice, fortified with vitamin D | 8 fluid ounces | 100 | 2.5 |
| Cereal, fortified | 1 serving (usually 1 cup) | 40-50 | 1.0-1.3 |
| Egg yolk | 1 large | 37 | 0.9 |
Supplements
Most vitamin D supplements available without a prescription contain cholecalciferol (vitamin D3). Multivitamin supplements generally provide 400 IU-1,000 IU (10 μg-25 μg) of vitamin D2 or vitamin D3. Single-ingredient vitamin D supplements may provide 400 to 50,000 IU of vitamin D3, but 400 IU is the most commonly available dose (66). A number of calcium supplements may also provide vitamin D. A meta-analysis of randomized controlled trials suggested that bolus doses of vitamin D2 (ergocalciferol) may not always be as effective as vitamin D3 in raising serum 25-hydroxyvitamin D concentrations, yet no difference in efficacy was found with daily supplementation with vitamin D2 or vitamin D3 (350). Nonetheless, a 25-week, randomized, double-blind, placebo-controlled trial found daily supplementation with 1,000 IU of vitamin D3 initiated at the end of summer to be more efficacious than vitamin D2 in maintaining summertime concentrations of 25-hydroxyvitamin D during fall and winter months (351).
There is growing interest in using the hydroxylated form of cholecalciferol, 25-hydroxyvitamin D3 (calcidiol; calcifediol), as a supplement. This vitamin D metabolite is synthesized by the liver of humans and animals from cholecalciferol produced in skin or consumed in the diet, and is present in some foods (e.g., meats, milk, eggs) at low levels (348, 349). In general, clinical studies have found calcifediol is two to five times more potent at increasing blood concentrations of 25-hydroxyvitamin D compared to supplementation with equivalent doses of cholecalciferol (352-360). Supplementation with calcifediol may thus represent a means to improve vitamin D status rapidly and consistently with lower doses than cholecalciferol and may benefit those individuals with conditions that decrease intestinal absorption of cholecalciferol (360). However, the 25-hydroxyvitamin D3 form is presently not available as an over-the-counter supplement in the United States.
Safety
Toxicity
Vitamin D toxicity (hypervitaminosis D) has not been observed to result from sun exposure. The reason is that excessive sunlight exposure generates a number of biologically inert photoproducts from 7-dehydrocholesterol and cholecalciferol (3). Vitamin D toxicity induces abnormally high serum calcium concentration (hypercalcemia), which could result in bone loss, kidney stones, and calcification of organs like the heart and kidneys if untreated over a long period of time. Hypercalcemia has been observed following daily doses of greater than 50,000 IU of vitamin D (361). Overall, research suggests that vitamin D toxicity is very unlikely in healthy people at intake levels lower than 10,000 IU/day (362-364). However, the Food and Nutrition Board of the IOM conservatively set the tolerable upper intake level (UL) at 4,000 IU/day (100 μg/day) for all adults (Table 3). Certain medical conditions can increase the risk of hypercalcemia in response to vitamin D, including primary hyperparathyroidism, sarcoidosis, tuberculosis, and lymphoma (362). People with these conditions may develop hypercalcemia in response to any increase in vitamin D nutrition and should consult a qualified health care provider regarding any increase in vitamin D intake.
| Age Group | μg/day | IU/day |
|---|---|---|
| Infants 0-6 months | 25 | 1,000 |
| Infants 6-12 months | 37.5 | 1,500 |
| Children 1-3 years | 62.5 | 2,500 |
| Children 4-8 years | 75 | 3,000 |
| Children 9-13 years | 100 | 4,000 |
| Adolescents 14-18 years | 100 | 4,000 |
| Adults 19 years and older | 100 | 4,000 |
Drug interactions
The following medications should not be taken at the same time as vitamin D because they can decrease the intestinal absorption of vitamin D: cholestyramine (Questran), colestipol (Colestid), orlistat (Xenical), and mineral oil (365, 366). The following medications increase the metabolism of vitamin D and may decrease serum 25-hydroxyvitamin D concentrations: phenytoin (Dilantin), fosphenytoin (Cerebyx), phenobarbital (Luminal), carbamazepine (Tegretol), and rifampin (Rimactane) (6). Cimetidine, a H2 blocker that suppresses stomach acid secretion, inhibits the hydroxylation of vitamin D in the liver (367). Treating acid reflux, gastroesophageal reflux disease (GERD), or ulcers with proton-pump inhibitors (omeprazole, lansoprazole) might interfere with calcium absorption and increase the risk of fracture such that patients are advised to take calcium and vitamin D supplements (368). The oral antifungal medication, ketoconazole, inhibits the 25-hydroxyvitamin D3-1α-hydroxylase enzyme and has been found to reduce serum 1α,25-dihydroxyvitamin D concentrations in healthy men (369). The Endocrine Society also recommends monitoring vitamin D status of patients on glucocorticoids and HIV treatment drugs because these medications increase the catabolism of 25-hydroxyvitamin D (40). The use of some cytostatic agents (cell growth inhibitors) may also increase the degradation of 25-hydroxyvitamin D and 1α,25-dihydroxyvitamin D in cancer patients under chemotherapy (6). The induction of hypercalcemia by toxic levels of vitamin D may precipitate cardiac arrhythmia in patients on digoxin (Lanoxin) (367). Hypercalcemia may also reduce the effectiveness of verapamil (Calan) and diltiazem (Cardizem) in atrial fibrillation (367).
LPI Recommendation
The Linus Pauling Institute (LPI) recommends that generally healthy adults take 2,000 IU (50 μg) of supplemental vitamin D daily. Most multivitamins contain 400 IU (10 μg) of vitamin D, and single-ingredient vitamin D supplements are available for additional supplementation. Sun exposure, diet, skin color, and body mass index (BMI) have variable, substantial impact on body vitamin D levels. To adjust for individual differences and ensure adequate body vitamin D status, the Linus Pauling Institute recommends aiming for a serum 25-hydroxyvitamin D concentration of at least 30 ng/mL (75 nmol/L). Observational studies suggest that serum 25-hydroxyvitamin D concentrations between 30 ng/mL and 60 ng/mL are associated with lower risks of adverse health outcomes, including cancers and autoimmune diseases.
The American Academy of Pediatrics currently suggests that all infants, children, and adolescents receive 400 IU of supplemental vitamin D daily (19). Consistent with the recommendations of the Endocrine Society (40), the Linus Pauling Institute recommends daily intakes of 400 to 1,000 IU (10 to 25 μg) of vitamin D in infants and 600 to 1,000 IU (15 to 25 μg) of vitamin D in children and adolescents. Given the average vitamin D content of breast milk, infant formula, and the diets of children and adolescents, supplementation may be necessary to meet these recommendations.
Older adults (>50 years)
Daily supplementation with 2,000 IU (50 μg) of vitamin D is especially important for older adults because aging is associated with a reduced capacity to synthesize vitamin D in the skin upon sun exposure.
Authors and Reviewers
Originally written in 2000 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in March 2003 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in March 2004 by:
Jane Higdon, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in January 2008 by:
Victoria J. Drake, Ph.D.
Linus Pauling Institute
Oregon State University
Updated in July 2014 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
The 2014 update of this article was supported by a grant from Bayer Consumer Care AG, Basel, Switzerland.
Updated in July 2017 by:
Barbara Delage, Ph.D.
Linus Pauling Institute
Oregon State University
Reviewed in October 2017 by:
Adrian F. Gombart, Ph.D.
Principal Investigator, Linus Pauling Institute
Associate Professor, Department of Biochemistry and Biophysics
Oregon State University
The 2017 update of this article was supported by a grant from Pfizer Inc.
Last updated 2/11/21 Copyright 2000-2026 Linus Pauling Institute
Figure Alternative Text
Figure 1. Chemical structures of vitamin D
The figure shows the chemical structures and synthesis pathway of vitamin D. In the upper layers of the skin, ultraviolet-B light converts 7-dehydrocholesterol into previtamin D3. Previtamin D3 is then non-enzymatically converted into vitamin D3 (cholecalciferol) in the lower layers of the skin. Vitamin D3 is transported to adipose tissue for storage or to the liver for activation.
In liver cells, cytochrome P450 enzymes hydroxylate vitamin D3, forming 25-hydroxyvitamin D3 (calcidiol). In the kidneys (and to a limited extent, other tissues), CYP27B1 converts 25-hydroxyvitamin D3 to the active form: 1α,25-dihydroxyvitamin D3 (calcitriol). Plant-derived vitamin D2 (ergocalciferol) follows a similar activation pathway.
Figure 2. Conversion to the active form of vitamin D and VDR-mediated gene regulation
The figure shows the conversion of vitamin D to its active form and vitamin D receptor (VDR)-mediated gene regulation. Both 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D circulate in the blood bound to vitamin D-binding protein (DBP) and enter cells via the megalin/cubulin complex.
On the left: In kidney cells, CYP27B1 hydroxylates 25-hydroxyvitamin D to form the active form of vitamin D: 1,25-dihydroxyvitamin D. This reaction is stimulated by parathyroid hormone, estradiol, and low blood phosphorus, whereas the reaction is inhibited by 1,25-dihydroxyvitamin D, FGF-23, and high blood calcium.
On the right: 1,25-Dihydroxyvitamin D enters the circulation and reaches extra-renal target tissues, where it regulates gene expression. In the nucleus, 1,25-dihydroxyvitamin D binds to the VDR, which heterodimerizes with the retinoid X receptor (RXR). The VDR-RXR complex binds to vitamin D response elements (VDRE) in the promoter regions of vitamin D target genes, recruiting RNA polymerase II to initiate transcription.
Figure 3. Regulation of calcium and phosphorus homeostasis
The figure shows mechanisms of calcium and phosphorus homeostasis. When blood calcium concentration decreases, the parathyroid glands release parathyroid hormone (PTH). PTH acts on the kidney to stimulate CYP27B1 activity, which hydroxylates 25-hydroxyvitamin D to 1,25-dihydroxyvitamin D. This active form of vitamin D (1) increases reabsorption of calcium by the kidneys and stimulates phosphorus excretion, (2) increases the intestinal absorption of both calcium and phosphorus, and (3) mobilizes calcium and phosphorus from bone when dietary calcium intake is insufficient. PTH also stimulates bone resorption, releasing calcium and phosphorus into the blood. 1α,25-dihydroxyvitamin D inhibits its own production, as well as PTH synthesis, via negative feedback loops. FGF-23, secreted by bone-forming cells (osteoblasts) in response to increased phosphorus intake, inhibits the synthesis of 1α,25-dihydroxyvitamin D and promotes urinary excretion of phosphorus.
References
- Holick MF. Vitamin D: importance in the prevention of cancers, type 1 diabetes, heart disease, and osteoporosis. Am J Clin Nutr. 2004;79(3):362-371. (PubMed)
- Bikle DD. Vitamin D metabolism, mechanism of action, and clinical applications. Chem Biol. 2014;21(3):319-329. (PubMed)
- Volmer DA, Mendes LR, Stokes CS. Analysis of vitamin D metabolic markers by mass spectrometry: Current techniques, limitations of the "gold standard" method, and anticipated future directions. Mass Spectrom Rev. 2015;34(1):2-23. (PubMed)
- Holick MF. Vitamin D: A millenium perspective. J Cell Biochem. 2003;88(2):296-307. (PubMed)
- Sutton AL, MacDonald PN. Vitamin D: more than a "bone-a-fide" hormone. Mol Endocrinol. 2003;17(5):777-791. (PubMed)
- Grober U, Spitz J, Reichrath J, Kisters K, Holick MF. Vitamin D: Update 2013: From rickets prophylaxis to general preventive healthcare. Dermatoendocrinol. 2013;5(3):331-347. (PubMed)
- Lieben L, Carmeliet G. The delicate balance between vitamin D, calcium and bone homeostasis: lessons learned from intestinal- and osteocyte-specific VDR null mice. J Steroid Biochem Mol Biol. 2013;136:102-106. (PubMed)
- Fukumoto S. Phosphate metabolism and vitamin D. Bonekey Rep. 2014;3:497. (PubMed)
- Lin R, White JH. The pleiotropic actions of vitamin D. Bioessays. 2004;26(1):21-28. (PubMed)
- Edfeldt K, Liu PT, Chun R, et al. T-cell cytokines differentially control human monocyte antimicrobial responses by regulating vitamin D metabolism. Proc Natl Acad Sci U S A. 2010;107(52):22593-22598. (PubMed)
- Smolders J, Thewissen M, Damoiseaux J. Control of T cell activation by vitamin D. Nat Immunol. 2011;12(1):3; author reply 3-4. (PubMed)
- Aranow C. Vitamin D and the immune system. J Investig Med. 2011;59(6):881-886. (PubMed)
- Zeitz U, Weber K, Soegiarto DW, Wolf E, Balling R, Erben RG. Impaired insulin secretory capacity in mice lacking a functional vitamin D receptor. Faseb J. 2003;17(3):509-511. (PubMed)
- Bourlon PM, Billaudel B, Faure-Dussert A. Influence of vitamin D3 deficiency and 1,25 dihydroxyvitamin D3 on de novo insulin biosynthesis in the islets of the rat endocrine pancreas. J Endocrinol. 1999;160(1):87-95. (PubMed)
- Heer M, Egert S. Nutrients other than carbohydrates: their effects on glucose homeostasis in humans. Diabetes Metab Res Rev. 2015;31(1):14-35. (PubMed)
- Sheng H-W. Sodium, chloride, and potassium. In: Stipanuk M, ed. Biochemical and Physiological Aspects of Human Nutrition. Philadelphia: W.B. Saunders Company; 2000:686-710.
- Sigmund CD. Regulation of renin expression and blood pressure by vitamin D(3). J Clin Invest. 2002;110(2):155-156. (PubMed)
- Li YC, Kong J, Wei M, Chen ZF, Liu SQ, Cao LP. 1,25-Dihydroxyvitamin D(3) is a negative endocrine regulator of the renin-angiotensin system. J Clin Invest. 2002;110(2):229-238. (PubMed)
- Wagner CL, Greer FR, American Academy of Pediatrics Section on B, American Academy of Pediatrics Committee on N. Prevention of rickets and vitamin D deficiency in infants, children, and adolescents. Pediatrics. 2008;122(5):1142-1152. (PubMed)
- Goldacre M, Hall N, Yeates DG. Hospitalisation for children with rickets in England: a historical perspective. Lancet. 2014;383(9917):597-598. (PubMed)
- Jones AN, Hansen KE. Recognizing the musculoskeletal manifestations of vitamin D deficiency. J Musculoskelet Med. 2009;26(10):389-396. (PubMed)
- Bringhurst FR, Demay MB, Kronenberg HM. Mineral Metabolism. In: Larson PR, Kronenberg HM, Melmed S, Polonsky KS, eds. Williams Textbook of Endocrinology. 10th ed. Philadelphia: Saunders Book Company; 2003:1317-1320.
- Plotnikoff GA, Quigley JM. Prevalence of severe hypovitaminosis D in patients with persistent, nonspecific musculoskeletal pain. Mayo Clin Proc. 2003;78(12):1463-1470. (PubMed)
- Deandrea S, Lucenteforte E, Bravi F, Foschi R, La Vecchia C, Negri E. Risk factors for falls in community-dwelling older people: a systematic review and meta-analysis. Epidemiology. 2010;21(5):658-668. (PubMed)
- Al-Khalidi B, Kimball SM, Rotondi MA, Ardern CI. Standardized serum 25-hydroxyvitamin D concentrations are inversely associated with cardiometabolic disease in US adults: a cross-sectional analysis of NHANES, 2001-2010. Nutr J. 2017;16(1):16. (PubMed)
- Cooper JD, Smyth DJ, Walker NM, et al. Inherited variation in vitamin D genes is associated with predisposition to autoimmune disease type 1 diabetes. Diabetes. 2011;60(5):1624-1631. (PubMed)
- Webb AR, Kline L, Holick MF. Influence of season and latitude on the cutaneous synthesis of vitamin D3: exposure to winter sunlight in Boston and Edmonton will not promote vitamin D3 synthesis in human skin. J Clin Endocrinol Metab. 1988;67(2):373-378. (PubMed)
- Nichols EK, Khatib IM, Aburto NJ, et al. Vitamin D status and determinants of deficiency among non-pregnant Jordanian women of reproductive age. Eur J Clin Nutr. 2012;66(6):751-756. (PubMed)
- Bassil D, Rahme M, Hoteit M, Fuleihan Gel H. Hypovitaminosis D in the Middle East and North Africa: prevalence, risk factors and impact on outcomes. Dermatoendocrinol. 2013;5(2):274-298. (PubMed)
- Balk SJ, Council on Environmental H, Section on D. Ultraviolet radiation: a hazard to children and adolescents. Pediatrics. 2011;127(3):e791-817. (PubMed)
- Dawodu A, Tsang RC. Maternal vitamin D status: effect on milk vitamin D content and vitamin D status of breastfeeding infants. Adv Nutr. 2012;3(3):353-361. (PubMed)
- Thiele DK, Senti JL, Anderson CM. Maternal vitamin D supplementation to meet the needs of the breastfed infant: a systematic review. J Hum Lact. 2013;29(2):163-170. (PubMed)
- Wharton B, Bishop N. Rickets. Lancet. 2003;362(9393):1389-1400. (PubMed)
- Chen TC, Chimeh F, Lu Z, et al. Factors that influence the cutaneous synthesis and dietary sources of vitamin D. Arch Biochem Biophys. 2007;460(2):213-217. (PubMed)
- Wang TJ, Zhang F, Richards JB, et al. Common genetic determinants of vitamin D insufficiency: a genome-wide association study. Lancet. 2010;376(9736):180-188. (PubMed)
- Ahn J, Yu K, Stolzenberg-Solomon R, et al. Genome-wide association study of circulating vitamin D levels. Hum Mol Genet. 2010;19(13):2739-2745. (PubMed)
- Wang W, Ingles SA, Torres-Mejia G, et al. Genetic variants and non-genetic factors predict circulating vitamin D levels in Hispanic and non-Hispanic White women: the Breast Cancer Health Disparities Study. Int J Mol Epidemiol Genet. 2014;5(1):31-46. (PubMed)
- Elkum N, Alkayal F, Noronha F, et al. Vitamin D insufficiency in Arabs and South Asians positively associates with polymorphisms in GC and CYP2R1 genes. PLoS One. 2014;9(11):e113102. (PubMed)
- Zhang Y, Yang S, Liu Y, Ren L. Relationship between polymorphisms in vitamin D metabolism-related genes and the risk of rickets in Han Chinese children. BMC Med Genet. 2013;14:101. (PubMed)
- Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911-1930. (PubMed)
- Harris SS, Soteriades E, Coolidge JA, Mudgal S, Dawson-Hughes B. Vitamin D insufficiency and hyperparathyroidism in a low income, multiracial, elderly population. J Clin Endocrinol Metab. 2000;85(11):4125-4130. (PubMed)
- Allain TJ, Dhesi J. Hypovitaminosis D in older adults. Gerontology. 2003;49(5):273-278. (PubMed)
- Doorenbos CR, van den Born J, Navis G, de Borst MH. Possible renoprotection by vitamin D in chronic renal disease: beyond mineral metabolism. Nat Rev Nephrol. 2009;5(12):691-700. (PubMed)
- Pappa HM, Bern E, Kamin D, Grand RJ. Vitamin D status in gastrointestinal and liver disease. Curr Opin Gastroenterol. 2008;24(2):176-183. (PubMed)
- Jahnsen J, Falch JA, Mowinckel P, Aadland E. Vitamin D status, parathyroid hormone and bone mineral density in patients with inflammatory bowel disease. Scand J Gastroenterol. 2002;37(2):192-199. (PubMed)
- Arunabh S, Pollack S, Yeh J, Aloia JF. Body fat content and 25-hydroxyvitamin D levels in healthy women. J Clin Endocrinol Metab. 2003;88(1):157-161. (PubMed)
- Gallagher JC, Yalamanchili V, Smith LM. The effect of vitamin D supplementation on serum 25(OH)D in thin and obese women. J Steroid Biochem Mol Biol. 2013;136:195-200. (PubMed)
- Deng X, Song Y, Manson JE, et al. Magnesium, vitamin D status and mortality: results from US National Health and Nutrition Examination Survey (NHANES) 2001 to 2006 and NHANES III. BMC Med. 2013;11:187. (PubMed)
- Sempos CT, Durazo-Arvizu RA, Binkley N, Jones J, Merkel JM, Carter GD. Developing vitamin D dietary guidelines and the lack of 25-hydroxyvitamin D assay standardization: The ever-present past. J Steroid Biochem Mol Biol. 2016;164:115-119. (PubMed)
- Chapuy MC, Preziosi P, Maamer M, et al. Prevalence of vitamin D insufficiency in an adult normal population. Osteoporos Int. 1997;7(5):439-443. (PubMed)
- Thomas MK, Lloyd-Jones DM, Thadhani RI, et al. Hypovitaminosis D in medical inpatients. N Engl J Med. 1998;338(12):777-783. (PubMed)
- Heaney RP, Dowell MS, Hale CA, Bendich A. Calcium absorption varies within the reference range for serum 25-hydroxyvitamin D. J Am Coll Nutr. 2003;22(2):142-146. (PubMed)
- Valcour A, Blocki F, Hawkins DM, Rao SD. Effects of age and serum 25-OH-vitamin D on serum parathyroid hormone levels. J Clin Endocrinol Metab. 2012;97(11):3989-3995. (PubMed)
- Ginde AA, Wolfe P, Camargo CA, Jr., Schwartz RS. Defining vitamin D status by secondary hyperparathyroidism in the US population. J Endocrinol Invest. 2012;35(1):42-48. (PubMed)
- Gallagher JC, Yalamanchili V, Smith LM. The effect of vitamin D on calcium absorption in older women. J Clin Endocrinol Metab. 2012;97(10):3550-3556. (PubMed)
- Looker AC, Johnson CL, Lacher DA, Pfeiffer CM, Schleicher RL, Sempos CT. Vitamin D status: United States, 2001-2006. NCHS Data Brief. 2011(59):1-8. (PubMed)
- Food and Nutrition Board, Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D. Washington, D.C.: The National Academies Press; 2011. (The National Academies Press)
- Mithal A, Wahl DA, Bonjour JP, et al. Global vitamin D status and determinants of hypovitaminosis D. Osteoporos Int. 2009;20(11):1807-1820. (PubMed)
- Powe CE, Evans MK, Wenger J, et al. Vitamin D-binding protein and vitamin D status of black Americans and white Americans. N Engl J Med. 2013;369(21):1991-2000. (PubMed)
- Durazo-Arvizu RA, Dawson-Hughes B, Kramer H, et al. The reverse J-shaped association between serum total 25-hydroxyvitamin D concentration and all-cause mortality: the impact of assay standardization. Am J Epidemiol. 2017;185(8):720-726. (PubMed)
- Gaksch M, Jorde R, Grimnes G, et al. Vitamin D and mortality: Individual participant data meta-analysis of standardized 25-hydroxyvitamin D in 26916 individuals from a European consortium. PLoS One. 2017;12(2):e0170791. (PubMed)
- Chowdhury R, Kunutsor S, Vitezova A, et al. Vitamin D and risk of cause specific death: systematic review and meta-analysis of observational cohort and randomised intervention studies. BMJ. 2014;348:g1903. (PubMed)
- Gupta V, Walia GK, Sachdeva MP. 'Mendelian randomization': an approach for exploring causal relations in epidemiology. Public Health. 2017;145:113-119. (PubMed)
- Afzal S, Brondum-Jacobsen P, Bojesen SE, Nordestgaard BG. Genetically low vitamin D concentrations and increased mortality: Mendelian randomisation analysis in three large cohorts. BMJ. 2014;349:g6330. (PubMed)
- Bjelakovic G, Gluud LL, Nikolova D, et al. Vitamin D supplementation for prevention of mortality in adults. Cochrane Database Syst Rev. 2014(1):Cd007470. (PubMed)
- Wacker M, Holick MF. Vitamin D - effects on skeletal and extraskeletal health and the need for supplementation. Nutrients. 2013;5(1):111-148. (PubMed)
- Lips P, Hosking D, Lippuner K, et al. The prevalence of vitamin D inadequacy amongst women with osteoporosis: an international epidemiological investigation. J Intern Med. 2006;260(3):245-254. (PubMed)
- Torbergsen AC, Watne LO, Wyller TB, et al. Vitamin K1 and 25(OH)D are independently and synergistically associated with a risk for hip fracture in an elderly population: A case control study. Clin Nutr. 2015;34(1):101-106. (PubMed)
- Lips P, van Schoor NM. The effect of vitamin D on bone and osteoporosis. Best Pract Res Clin Endocrinol Metab. 2011;25(4):585-591. (PubMed)
- Reid IR, Bolland MJ, Grey A. Effects of vitamin D supplements on bone mineral density: a systematic review and meta-analysis. Lancet. 2014;383(9912):146-155. (PubMed)
- Rosen CJ. Vitamin D supplementation: bones of contention. Lancet. 2014;383(9912):108-110. (PubMed)
- Mocanu V, Vieth R. Three-year follow-up of serum 25-hydroxyvitamin D, parathyroid hormone, and bone mineral density in nursing home residents who had received 12 months of daily bread fortification with 125 mug of vitamin D3. Nutr J. 2013;12:137. (PubMed)
- Feskanich D, Willett WC, Colditz GA. Calcium, vitamin D, milk consumption, and hip fractures: a prospective study among postmenopausal women. Am J Clin Nutr. 2003;77(2):504-511. (PubMed)
- Jackson RD, LaCroix AZ, Gass M, et al. Calcium plus vitamin D supplementation and the risk of fractures. N Engl J Med. 2006;354(7):669-683. (PubMed)
- Wang Y, Wactawski-Wende J, Sucheston-Campbell LE, et al. The influence of genetic susceptibility and calcium plus vitamin D supplementation on fracture risk. Am J Clin Nutr. 2017;105(4):970-979. (PubMed)
- Gurney EP, Nachtigall MJ, Nachtigall LE, Naftolin F. The Women's Health Initiative trial and related studies: 10 years later: a clinician's view. J Steroid Biochem Mol Biol. 2014;142:4-11. (PubMed)
- Grant AM, Avenell A, Campbell MK, et al. Oral vitamin D3 and calcium for secondary prevention of low-trauma fractures in elderly people (Randomised Evaluation of Calcium Or vitamin D, RECORD): a randomised placebo-controlled trial. Lancet. 2005;365(9471):1621-1628. (PubMed)
- Bischoff-Ferrari HA, Giovannucci E, Willett WC, Dietrich T, Dawson-Hughes B. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am J Clin Nutr. 2006;84(1):18-28. (PubMed)
- Khaw KT, Stewart AW, Waayer D, et al. Effect of monthly high-dose vitamin D supplementation on falls and non-vertebral fractures: secondary and post-hoc outcomes from the randomised, double-blind, placebo-controlled ViDA trial. Lancet Diabetes Endocrinol. 2017;5(6):438-447. (PubMed)
- Chung M, Lee J, Terasawa T, Lau J, Trikalinos TA. Vitamin D with or without calcium supplementation for prevention of cancer and fractures: an updated meta-analysis for the US Preventive Services Task Force. Ann Intern Med. 2011;155(12):827-838. (PubMed)
- Bischoff-Ferrari HA, Willett WC, Orav EJ, et al. A pooled analysis of vitamin D dose requirements for fracture prevention. N Engl J Med. 2012;367(1):40-49. (PubMed)
- Avenell A, Mak JC, O'Connell D. Vitamin D and vitamin D analogues for preventing fractures in post-menopausal women and older men. Cochrane Database Syst Rev. 2014;4:CD000227. (PubMed)
- Annweiler C, Beauchet O. Questioning vitamin D status of elderly fallers and nonfallers: a meta-analysis to address a 'forgotten step'. J Intern Med. 2015;277(1):16-44. (PubMed)
- Cangussu LM, Nahas-Neto J, Orsatti CL, Bueloni-Dias FN, Nahas EA. Effect of vitamin D supplementation alone on muscle function in postmenopausal women: a randomized, double-blind, placebo-controlled clinical trial. Osteoporos Int. 2015;26(10):2413-2421. (PubMed)
- Cangussu LM, Nahas-Neto J, Orsatti CL, et al. Effect of isolated vitamin D supplementation on the rate of falls and postural balance in postmenopausal women fallers: a randomized, double-blind, placebo-controlled trial. Menopause. 2016;23(3):267-274. (PubMed)
- Bischoff-Ferrari HA, Dawson-Hughes B, Orav EJ, et al. Monthly high-dose vitamin D treatment for the prevention of functional decline: a randomized clinical trial. JAMA Intern Med. 2016;176(2):175-183. (PubMed)
- Murad MH, Elamin KB, Abu Elnour NO, et al. Clinical review: The effect of vitamin D on falls: a systematic review and meta-analysis. J Clin Endocrinol Metab. 2011;96(10):2997-3006. (PubMed)
- Boonen S, Lips P, Bouillon R, Bischoff-Ferrari HA, Vanderschueren D, Haentjens P. Need for additional calcium to reduce the risk of hip fracture with vitamin d supplementation: evidence from a comparative metaanalysis of randomized controlled trials. J Clin Endocrinol Metab. 2007;92(4):1415-1423. (PubMed)
- Grant WB. Update on evidence that support a role of solar ultraviolet-B irradiance in reducing cancer risk. Anticancer Agents Med Chem. 2013;13(1):140-146. (PubMed)
- Yin L, Ordonez-Mena JM, Chen T, Schottker B, Arndt V, Brenner H. Circulating 25-hydroxyvitamin D serum concentration and total cancer incidence and mortality: a systematic review and meta-analysis. Prev Med. 2013;57(6):753-764. (PubMed)
- Gandini S, Gnagnarella P, Serrano D, Pasquali E, Raimondi S. Vitamin D receptor polymorphisms and cancer. Adv Exp Med Biol. 2014;810:69-105. (PubMed)
- Vaughan-Shaw PG, O'Sullivan F, Farrington SM, et al. The impact of vitamin D pathway genetic variation and circulating 25-hydroxyvitamin D on cancer outcome: systematic review and meta-analysis. Br J Cancer. 2017;116(8):1092-1110. (PubMed)
- Gombart AF, Luong QT, Koeffler HP. Vitamin D compounds: activity against microbes and cancer. Anticancer Res. 2006;26(4A):2531-2542. (PubMed)
- Thorne J, Campbell MJ. The vitamin D receptor in cancer. Proc Nutr Soc. 2008;67(2):115-127. (PubMed)
- Garland CF, Garland FC, Gorham ED. Calcium and vitamin D. Their potential roles in colon and breast cancer prevention. Ann N Y Acad Sci. 1999;889:107-119. (PubMed)
- Choi YJ, Kim YH, Cho CH, Kim SH, Lee JE. Circulating levels of vitamin D and colorectal adenoma: A case-control study and a meta-analysis. World J Gastroenterol. 2015;21(29):8868-8877. (PubMed)
- Gandini S, Boniol M, Haukka J, et al. Meta-analysis of observational studies of serum 25-hydroxyvitamin D levels and colorectal, breast and prostate cancer and colorectal adenoma. Int J Cancer. 2011;128(6):1414-1424. (PubMed)
- Ma Y, Zhang P, Wang F, Yang J, Liu Z, Qin H. Association between vitamin D and risk of colorectal cancer: a systematic review of prospective studies. J Clin Oncol. 2011;29(28):3775-3782. (PubMed)
- Touvier M, Chan DS, Lau R, et al. Meta-analyses of vitamin D intake, 25-hydroxyvitamin D status, vitamin D receptor polymorphisms, and colorectal cancer risk. Cancer Epidemiol Biomarkers Prev. 2011;20(5):1003-1016. (PubMed)
- Ekmekcioglu C, Haluza D, Kundi M. 25-Hydroxyvitamin D status and risk for colorectal cancer and type 2 diabetes mellitus: a systematic review and meta-analysis of epidemiological studies. Int J Environ Res Public Health. 2017;14(2). (PubMed)
- Gorham ED, Garland CF, Garland FC, et al. Optimal vitamin D status for colorectal cancer prevention: a quantitative meta analysis. Am J Prev Med. 2007;32(3):210-216. (PubMed)
- Cauley JA, Chlebowski RT, Wactawski-Wende J, et al. Calcium plus vitamin D supplementation and health outcomes five years after active intervention ended: the Women's Health Initiative. J Womens Health (Larchmt). 2013;22(11):915-929. (PubMed)
- Baron JA, Barry EL, Mott LA, et al. A trial of calcium and vitamin D for the prevention of colorectal adenomas. N Engl J Med. 2015;373(16):1519-1530. (PubMed)
- Holick MF. Calcium plus vitamin D and the risk of colorectal cancer. N Engl J Med. 2006;354(21):2287-2288; author reply 2287-2288. (PubMed)
- Barry EL, Peacock JL, Rees JR, et al. Vitamin D receptor genotype, vitamin D3 supplementation, and risk of colorectal adenomas: a randomized clinical trial. JAMA Oncol. 2017;3(5):628-635. (PubMed)
- Hiraki LT, Joshi AD, Ng K, et al. Joint effects of colorectal cancer susceptibility loci, circulating 25-hydroxyvitamin D and risk of colorectal cancer. PLoS One. 2014;9(3):e92212. (PubMed)
- Vidigal VM, Silva TD, de Oliveira J, Pimenta CAM, Felipe AV, Forones NM. Genetic polymorphisms of vitamin D receptor (VDR), CYP27B1 and CYP24A1 genes and the risk of colorectal cancer. Int J Biol Markers. 2017;32(2):e224-e230. (PubMed)
- Maalmi H, Ordonez-Mena JM, Schottker B, Brenner H. Serum 25-hydroxyvitamin D levels and survival in colorectal and breast cancer patients: Systematic review and meta-analysis of prospective cohort studies. 2014;50(8):1510-1521. (PubMed)
- Mohr SB, Garland CF, Gorham ED, Grant WB, Garland FC. Relationship between low ultraviolet B irradiance and higher breast cancer risk in 107 countries. Breast J. 2008;14(3):255-260. (PubMed)
- John EM, Schwartz GG, Dreon DM, Koo J. Vitamin D and breast cancer risk: the NHANES I Epidemiologic follow-up study, 1971-1975 to 1992. National Health and Nutrition Examination Survey. Cancer Epidemiol Biomarkers Prev. 1999;8(5):399-406. (PubMed)
- Kim Y, Je Y. Vitamin D intake, blood 25(OH)D levels, and breast cancer risk or mortality: a meta-analysis. Br J Cancer. 2014;110(11):2772-2784. (PubMed)
- Wang D, Velez de-la-Paz OI, Zhai JX, Liu DW. Serum 25-hydroxyvitamin D and breast cancer risk: a meta-analysis of prospective studies. Tumour Biol. 2013;34(6):3509-3517. (PubMed)
- Rose AA, Elser C, Ennis M, Goodwin PJ. Blood levels of vitamin D and early stage breast cancer prognosis: a systematic review and meta-analysis. Breast Cancer Res Treat. 2013;141(3):331-339. (PubMed)
- Sperati F, Vici P, Maugeri-Sacca M, et al. Vitamin D supplementation and breast cancer prevention: a systematic review and meta-analysis of randomized clinical trials. PLoS One. 2013;8(7):e69269. (PubMed)
- Hu K, Callen DF, Li J, Zheng H. Circulating vitamin D and overall survival in breast cancer patients: a dose-response meta-analysis of cohort studies. Integr Cancer Ther. 2017:1534735417712007. (PubMed)
- Mohr SB, Gorham ED, Kim J, Hofflich H, Garland CF. Meta-analysis of vitamin D sufficiency for improving survival of patients with breast cancer. Anticancer Res. 2014;34(3):1163-1166. (PubMed)
- Lu D, Jing L, Zhang S. Vitamin D receptor polymorphism and breast cancer risk: a meta-analysis. Medicine (Baltimore). 2016;95(18):e3535. (PubMed)
- Mun MJ, Kim TH, Hwang JY, Jang WC. Vitamin D receptor gene polymorphisms and the risk for female reproductive cancers: A meta-analysis. Maturitas. 2015;81(2):256-265. (PubMed)
- Gilbert R, Martin RM, Beynon R, et al. Associations of circulating and dietary vitamin D with prostate cancer risk: a systematic review and dose-response meta-analysis. Cancer Causes Control. 2011;22(3):319-340. (PubMed)
- van der Rhee H, Coebergh JW, de Vries E. Is prevention of cancer by sun exposure more than just the effect of vitamin D? A systematic review of epidemiological studies. Eur J Cancer. 2013;49(6):1422-1436. (PubMed)
- Tuohimaa P, Tenkanen L, Ahonen M, et al. Both high and low levels of blood vitamin D are associated with a higher prostate cancer risk: a longitudinal, nested case-control study in the Nordic countries. Int J Cancer. 2004;108(1):104-108. (PubMed)
- Xu Y, Shao X, Yao Y, et al. Positive association between circulating 25-hydroxyvitamin D levels and prostate cancer risk: new findings from an updated meta-analysis. J Cancer Res Clin Oncol. 2014;140(9):1465-1477. (PubMed)
- Grant WB, Karras SN, Bischoff-Ferrari HA, et al. Do studies reporting 'U'-shaped serum 25-hydroxyvitamin D-health outcome relationships reflect adverse effects? Dermatoendocrinol. 2016;8(1):e1187349. (PubMed)
- Wu X, Cheng J, Yang K. Vitamin D-related gene polymorphisms, plasma 25-hydroxy-vitamin D, cigarette smoke and non-small cell lung cancer (NSCLC) Risk. Int J Mol Sci. 2016;17(10). (PubMed)
- Zhang L, Wang S, Che X, Li X. Vitamin D and lung cancer risk: a comprehensive review and meta-analysis. Cell Physiol Biochem. 2015;36(1):299-305. (PubMed)
- Liao Y, Huang JL, Qiu MX, Ma ZW. Impact of serum vitamin D level on risk of bladder cancer: a systemic review and meta-analysis. Tumour Biol. 2015;36(3):1567-1572. (PubMed)
- Zhang H, Zhang H, Wen X, Zhang Y, Wei X, Liu T. Vitamin D deficiency and increased risk of bladder carcinoma: a meta-analysis. Cell Physiol Biochem. 2015;37(5):1686-1692. (PubMed)
- Lu D, Chen J, Jin J. Vitamin D status and risk of non-Hodgkin lymphoma: a meta-analysis. Cancer Causes Control. 2014;25(11):1553-1563. (PubMed)
- Prescott J, Bertrand KA, Poole EM, Rosner BA, Tworoger SS. Surrogates of long-term vitamin d exposure and ovarian cancer risk in two prospective cohort studies. Cancers (Basel). 2013;5(4):1577-1600. (PubMed)
- Khayatzadeh S, Feizi A, Saneei P, Esmaillzadeh A. Vitamin D intake, serum Vitamin D levels, and risk of gastric cancer: A systematic review and meta-analysis. J Res Med Sci. 2015;20(8):790-796. (PubMed)
- Gandini S, Raimondi S, Gnagnarella P, Dore JF, Maisonneuve P, Testori A. Vitamin D and skin cancer: a meta-analysis. Eur J Cancer. 2009;45(4):634-641. (PubMed)
- Deluca HF, Cantorna MT. Vitamin D: its role and uses in immunology. Faseb J. 2001;15(14):2579-2585. (PubMed)
- Agmon-Levin N, Mosca M, Petri M, Shoenfeld Y. Systemic lupus erythematosus one disease or many? Autoimmun Rev. 2012;11(8):593-595. (PubMed)
- Goodin DS. The epidemiology of multiple sclerosis: insights to disease pathogenesis. Handb Clin Neurol. 2014;122:231-266. (PubMed)
- Littorin B, Blom P, Scholin A, et al. Lower levels of plasma 25-hydroxyvitamin D among young adults at diagnosis of autoimmune type 1 diabetes compared with control subjects: results from the nationwide Diabetes Incidence Study in Sweden (DISS). Diabetologia. 2006;49(12):2847-2852. (PubMed)
- Pozzilli P, Manfrini S, Crino A, et al. Low levels of 25-hydroxyvitamin D3 and 1,25-dihydroxyvitamin D3 in patients with newly diagnosed type 1 diabetes. Horm Metab Res. 2005;37(11):680-683. (PubMed)
- Raab J, Giannopoulou EZ, Schneider S, et al. Prevalence of vitamin D deficiency in pre-type 1 diabetes and its association with disease progression. Diabetologia. 2014;57(5):902-908. (PubMed)
- Hypponen E, Laara E, Reunanen A, Jarvelin MR, Virtanen SM. Intake of vitamin D and risk of type 1 diabetes: a birth-cohort study. Lancet. 2001;358(9292):1500-1503. (PubMed)
- Sorensen IM, Joner G, Jenum PA, Eskild A, Torjesen PA, Stene LC. Maternal serum levels of 25-hydroxy-vitamin D during pregnancy and risk of type 1 diabetes in the offspring. Diabetes. 2012;61(1):175-178. (PubMed)
- Brekke HK, Ludvigsson J. Vitamin D supplementation and diabetes-related autoimmunity in the ABIS study. Pediatr Diabetes. 2007;8(1):11-14. (PubMed)
- Fronczak CM, Baron AE, Chase HP, et al. In utero dietary exposures and risk of islet autoimmunity in children. Diabetes Care. 2003;26(12):3237-3242. (PubMed)
- Marjamaki L, Niinisto S, Kenward MG, et al. Maternal intake of vitamin D during pregnancy and risk of advanced beta cell autoimmunity and type 1 diabetes in offspring. Diabetologia. 2010;53(8):1599-1607. (PubMed)
- Miettinen ME, Reinert L, Kinnunen L, et al. Serum 25-hydroxyvitamin D level during early pregnancy and type 1 diabetes risk in the offspring. Diabetologia. 2012;55(5):1291-1294. (PubMed)
- Smolders J, Thewissen M, Peelen E, et al. Vitamin D status is positively correlated with regulatory T cell function in patients with multiple sclerosis. PLoS One. 2009;4(8):e6635. (PubMed)
- Mokry LE, Ross S, Ahmad OS, et al. Vitamin D and risk of multiple sclerosis: a Mendelian randomization study. PLoS Med. 2015;12(8):e1001866. (PubMed)
- Staples J, Ponsonby AL, Lim L. Low maternal exposure to ultraviolet radiation in pregnancy, month of birth, and risk of multiple sclerosis in offspring: longitudinal analysis. BMJ. 2010;340:c1640. (PubMed)
- Bjornevik K, Riise T, Casetta I, et al. Sun exposure and multiple sclerosis risk in Norway and Italy: The EnvIMS study. Mult Scler. 2014; 20(8):1042-1049. (PubMed)
- McDowell TY, Amr S, Culpepper WJ, et al. Sun exposure, vitamin D and age at disease onset in relapsing multiple sclerosis. Neuroepidemiology. 2011;36(1):39-45. (PubMed)
- Munger KL, Levin LI, Hollis BW, Howard NS, Ascherio A. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis. JAMA. 2006;296(23):2832-2838. (PubMed)
- Munger KL, Zhang SM, O'Reilly E, et al. Vitamin D intake and incidence of multiple sclerosis. Neurology. 2004;62(1):60-65. (PubMed)
- Pierrot-Deseilligny C, Rivaud-Pechoux S, Clerson P, de Paz R, Souberbielle JC. Relationship between 25-OH-D serum level and relapse rate in multiple sclerosis patients before and after vitamin D supplementation. Ther Adv Neurol Disord. 2012;5(4):187-198. (PubMed)
- Ascherio A, Munger KL, White R, et al. Vitamin d as an early predictor of multiple sclerosis activity and progression. JAMA Neurol. 2014;71(3):306-314. (PubMed)
- Muris AH, Smolders J, Rolf L, et al. Vitamin D status does not affect disability progression of patients with multiple sclerosis over three year follow-up. PLoS One. 2016;11(6):e0156122. (PubMed)
- Kampman MT, Steffensen LH, Mellgren SI, Jorgensen L. Effect of vitamin D3 supplementation on relapses, disease progression, and measures of function in persons with multiple sclerosis: exploratory outcomes from a double-blind randomised controlled trial. Mult Scler. 2012;18(8):1144-1151. (PubMed)
- Soilu-Hanninen M, Aivo J, Lindstrom BM, et al. A randomised, double blind, placebo controlled trial with vitamin D3 as an add on treatment to interferon beta-1b in patients with multiple sclerosis. J Neurol Neurosurg Psychiatry. 2012;83(5):565-571. (PubMed)
- Mrad MF, El Ayoubi NK, Esmerian MO, Kazan JM, Khoury SJ. Effect of vitamin D replacement on immunological biomarkers in patients with multiple sclerosis. Clin Immunol. 2017;181:9-15. (PubMed)
- Muris AH, Smolders J, Rolf L, Thewissen M, Hupperts R, Damoiseaux J. Immune regulatory effects of high dose vitamin D3 supplementation in a randomized controlled trial in relapsing remitting multiple sclerosis patients receiving IFNbeta; the SOLARIUM study. J Neuroimmunol. 2016;300:47-56. (PubMed)
- O'Connell K, Sulaimani J, Basdeo SA, et al. Effects of vitamin D3 in clinically isolated syndrome and healthy control participants: A double-blind randomised controlled trial. Mult Scler J Exp Transl Clin. 2017;3(3):2055217317727296. (PubMed)
- Rosjo E, Steffensen LH, Jorgensen L, et al. Vitamin D supplementation and systemic inflammation in relapsing-remitting multiple sclerosis. J Neurol. 2015;262(12):2713-2721. (PubMed)
- Sotirchos ES, Bhargava P, Eckstein C, et al. Safety and immunologic effects of high- vs low-dose cholecalciferol in multiple sclerosis. Neurology. 2016;86(4):382-390. (PubMed)
- Bruce D, Whitcomb JP, August A, McDowell MA, Cantorna MT. Elevated non-specific immunity and normal Listeria clearance in young and old vitamin D receptor knockout mice. Int Immunol. 2009;21(2):113-122. (PubMed)
- Zwerina K, Baum W, Axmann R, et al. Vitamin D receptor regulates TNF-mediated arthritis. Ann Rheum Dis. 2011;70(6):1122-1129. (PubMed)
- Hitchon CA, Sun Y, Robinson DB, et al. Vitamin D receptor polymorphism rs2228570 (Fok1) is associated with rheumatoid arthritis in North American natives. J Rheumatol. 2012;39(9):1792-1797. (PubMed)
- Lee YH, Bae SC, Choi SJ, Ji JD, Song GG. Associations between vitamin D receptor polymorphisms and susceptibility to rheumatoid arthritis and systemic lupus erythematosus: a meta-analysis. Mol Biol Rep. 2011;38(6):3643-3651. (PubMed)
- Mosaad YM, Hammad EM, Fawzy Z, et al. Vitamin D receptor gene polymorphism as possible risk factor in rheumatoid arthritis and rheumatoid related osteoporosis. Hum Immunol. 2014;75(5):452-461. (PubMed)
- Zanetti M, Harris SS, Dawson-Hughes B. Ability of vitamin D to reduce inflammation in adults without acute illness. Nutr Rev. 2014;72(2):95-98. (PubMed)
- Merlino LA, Curtis J, Mikuls TR, Cerhan JR, Criswell LA, Saag KG. Vitamin D intake is inversely associated with rheumatoid arthritis: results from the Iowa Women's Health Study. Arthritis Rheum. 2004;50(1):72-77. (PubMed)
- Costenbader KH, Feskanich D, Holmes M, Karlson EW, Benito-Garcia E. Vitamin D intake and risks of systemic lupus erythematosus and rheumatoid arthritis in women. Ann Rheum Dis. 2008;67(4):530-535. (PubMed)
- Hiraki LT, Munger KL, Costenbader KH, Karlson EW. Dietary intake of vitamin D during adolescence and risk of adult-onset systemic lupus erythematosus and rheumatoid arthritis. Arthritis Care Res (Hoboken). 2012;64(12):1829-1836. (PubMed)
- Sen D, Ranganathan P. Vitamin D in rheumatoid arthritis: panacea or placebo? Discov Med. 2012;14(78):311-319. (PubMed)
- Lee YH, Bae SC. Vitamin D level in rheumatoid arthritis and its correlation with the disease activity: a meta-analysis. Clin Exp Rheumatol. 2016;34(5):827-833. (PubMed)
- Lin J, Liu J, Davies ML, Chen W. Serum vitamin D level and rheumatoid arthritis disease activity: review and meta-analysis. PLoS One. 2016;11(1):e0146351. (PubMed)
- Hansen KE, Bartels CM, Gangnon RE, Jones AN, Gogineni J. An evaluation of high-dose vitamin D for rheumatoid arthritis. J Clin Rheumatol. 2014;20(2):112-114. (PubMed)
- Buondonno I, Rovera G, Sassi F, et al. Vitamin D and immunomodulation in early rheumatoid arthritis: A randomized double-blind placebo-controlled study. PLoS One. 2017;12(6):e0178463. (PubMed)
- Dehghan A, Rahimpour S, Soleymani-Salehabadi H, Owlia MB. Role of vitamin D in flare ups of rheumatoid arthritis. Z Rheumatol. 2014;73(5):461-464. (PubMed)
- Yang J, Liu L, Zhang Q, Li M, Wang J. Effect of vitamin D on the recurrence rate of rheumatoid arthritis. Exp Ther Med. 2015;10(5):1812-1816. (PubMed)
- Gonzalez LA, Toloza SM, McGwin G, Jr., Alarcon GS. Ethnicity in systemic lupus erythematosus (SLE): its influence on susceptibility and outcomes. Lupus. 2013;22(12):1214-1224. (PubMed)
- Hsieh CC, Lin BF. Dietary factors regulate cytokines in murine models of systemic lupus erythematosus. Autoimmun Rev. 2011;11(1):22-27. (PubMed)
- Mao S, Huang S. Association between vitamin D receptor gene BsmI, FokI, ApaI and TaqI polymorphisms and the risk of systemic lupus erythematosus: a meta-analysis. Rheumatol Int. 2014;34(3):381-388. (PubMed)
- Monticielo OA, Teixeira Tde M, Chies JA, Brenol JC, Xavier RM. Vitamin D and polymorphisms of VDR gene in patients with systemic lupus erythematosus. Clin Rheumatol. 2012;31(10):1411-1421. (PubMed)
- Ruiz-Irastorza G, Egurbide MV, Olivares N, Martinez-Berriotxoa A, Aguirre C. Vitamin D deficiency in systemic lupus erythematosus: prevalence, predictors and clinical consequences. Rheumatology (Oxford). 2008;47(6):920-923. (PubMed)
- Toloza SM, Cole DE, Gladman DD, Ibanez D, Urowitz MB. Vitamin D insufficiency in a large female SLE cohort. Lupus. 2010;19(1):13-19. (PubMed)
- Amital H, Szekanecz Z, Szucs G, et al. Serum concentrations of 25-OH vitamin D in patients with systemic lupus erythematosus (SLE) are inversely related to disease activity: is it time to routinely supplement patients with SLE with vitamin D? Ann Rheum Dis. 2010;69(6):1155-1157. (PubMed)
- Terrier B, Derian N, Schoindre Y, et al. Restoration of regulatory and effector T cell balance and B cell homeostasis in systemic lupus erythematosus patients through vitamin D supplementation. Arthritis Res Ther. 2012;14(5):R221. (PubMed)
- Cutillas-Marco E, Marquina-Vila A, Grant W, Vilata-Corell J, Morales-Suarez-Varela M. Vitamin D and cutaneous lupus erythematosus: effect of vitamin D replacement on disease severity. Lupus. 2014;23(7):615-623. (PubMed)
- Abou-Raya A, Abou-Raya S, Helmii M. The effect of vitamin D supplementation on inflammatory and hemostatic markers and disease activity in patients with systemic lupus erythematosus: a randomized placebo-controlled trial. J Rheumatol. 2013;40(3):265-272. (PubMed)
- Lima GL, Paupitz J, Aikawa NE, Takayama L, Bonfa E, Pereira RM. Vitamin D supplementation in adolescents and young adults With juvenile systemic lupus erythematosus for improvement in disease activity and fatigue scores: a randomized, double-blind, placebo-controlled trial. Arthritis Care Res (Hoboken). 2016;68(1):91-98. (PubMed)
- Andreoli L, Dall'Ara F, Piantoni S, et al. A 24-month prospective study on the efficacy and safety of two different monthly regimens of vitamin D supplementation in pre-menopausal women with systemic lupus erythematosus. Lupus. 2015;24(4-5):499-506. (PubMed)
- Karimzadeh H, Shirzadi M, Karimifar M. The effect of Vitamin D supplementation in disease activity of systemic lupus erythematosus patients with Vitamin D deficiency: A randomized clinical trial. J Res Med Sci. 2017;22:4. (PubMed)
- Antico A, Tampoia M, Tozzoli R, Bizzaro N. Can supplementation with vitamin D reduce the risk or modify the course of autoimmune diseases? A systematic review of the literature. Autoimmun Rev. 2012;12(2):127-136. (PubMed)
- Wang TJ, Pencina MJ, Booth SL, et al. Vitamin D deficiency and risk of cardiovascular disease. Circulation. 2008;117(4):503-511. (PubMed)
- van Ballegooijen AJ, Kestenbaum B, Sachs MC, et al. Association of 25-Hydroxyvitamin D and parathyroid hormone with incident hypertension: MESA (Multi-Ethnic Study of Atherosclerosis). J Am Coll Cardiol. 2014;63(12):1214-1222. (PubMed)
- Kunutsor SK, Apekey TA, Steur M. Vitamin D and risk of future hypertension: meta-analysis of 283,537 participants. Eur J Epidemiol. 2013;28(3):205-221. (PubMed)
- Burgaz A, Orsini N, Larsson SC, Wolk A. Blood 25-hydroxyvitamin D concentration and hypertension: a meta-analysis. J Hypertens. 2011;29(4):636-645. (PubMed)
- Moody WE, Edwards NC, Madhani M, et al. Endothelial dysfunction and cardiovascular disease in early-stage chronic kidney disease: cause or association? Atherosclerosis. 2012;223(1):86-94. (PubMed)
- Chitalia N, Recio-Mayoral A, Kaski JC, Banerjee D. Vitamin D deficiency and endothelial dysfunction in non-dialysis chronic kidney disease patients. Atherosclerosis. 2012;220(1):265-268. (PubMed)
- Chitalia N, Ismail T, Tooth L, et al. Impact of vitamin d supplementation on arterial vasomotion, stiffness and endothelial biomarkers in chronic kidney disease patients. PLoS One. 2014;9(3):e91363. (PubMed)
- Mazidi M, Karimi E, Rezaie P, Vatanparast H. The impact of vitamin D supplement intake on vascular endothelial function; a systematic review and meta-analysis of randomized controlled trials. Food Nutr Res. 2017;61(1):1273574. (PubMed)
- Messa P, Curreri M, Regalia A, Alfieri CM. Vitamin D and the cardiovascular system: an overview of the recent literature. Am J Cardiovasc Drugs. 2014;14(1):1-14. (PubMed)
- Brondum-Jacobsen P, Benn M, Afzal S, Nordestgaard BG. No evidence that genetically reduced 25-hydroxyvitamin D is associated with increased risk of ischaemic heart disease or myocardial infarction: a Mendelian randomization study. Int J Epidemiol. 2015;44(2):651-661. (PubMed)
- Manousaki D, Mokry LE, Ross S, Goltzman D, Richards JB. Mendelian randomization studies do not support a role for vitamin D in coronary artery disease. Circ Cardiovasc Genet. 2016;9(4):349-356. (PubMed)
- Ford JA, MacLennan GS, Avenell A, Bolland M, Grey A, Witham M. Cardiovascular disease and vitamin D supplementation: trial analysis, systematic review, and meta-analysis. Am J Clin Nutr. 2014;100(3):746-755. (PubMed)
- Chin K, Appel LJ, Michos ED. Vitamin D, calcium, and cardiovascular disease: a"D"vantageous or "D"etrimental? An era of uncertainty. Curr Atheroscler Rep. 2017;19(1):5. (PubMed)
- Pradhan AD, Manson JE. Update on the Vitamin D and OmegA-3 trial (VITAL). J Steroid Biochem Mol Biol. 2016;155(Pt B):252-256. (PubMed)
- Neale RE, Armstrong BK, Baxter C, et al. The D-Health Trial: A randomized trial of vitamin D for prevention of mortality and cancer. Contemp Clin Trials. 2016;48:83-90. (PubMed)
- Scragg R, Stewart AW, Waayer D, et al. Effect of monthly high-dose vitamin D supplementation on cardiovascular disease in the vitamin D assessment study: a randomized clinical trial. JAMA Cardiol. 2017;2(6):608-616. (PubMed)
- Thomas GN, o Hartaigh B, Bosch JA, et al. Vitamin D levels predict all-cause and cardiovascular disease mortality in subjects with the metabolic syndrome: the Ludwigshafen Risk and Cardiovascular Health (LURIC) Study. Diabetes Care. 2012;35(5):1158-1164. (PubMed)
- Chiu KC, Chu A, Go VL, Saad MF. Hypovitaminosis D is associated with insulin resistance and beta cell dysfunction. Am J Clin Nutr. 2004;79(5):820-825. (PubMed)
- Shankar A, Sabanayagam C, Kalidindi S. Serum 25-hydroxyvitamin D levels and prediabetes among subjects free of diabetes. Diabetes Care. 2011;34(5):1114-1119. (PubMed)
- Deleskog A, Hilding A, Brismar K, Hamsten A, Efendic S, Ostenson CG. Low serum 25-hydroxyvitamin D level predicts progression to type 2 diabetes in individuals with prediabetes but not with normal glucose tolerance. Diabetologia. 2012;55(6):1668-1678. (PubMed)
- Khan H, Kunutsor S, Franco OH, Chowdhury R. Vitamin D, type 2 diabetes and other metabolic outcomes: a systematic review and meta-analysis of prospective studies. Proc Nutr Soc. 2013;72(1):89-97. (PubMed)
- Lucato P, Solmi M, Maggi S, et al. Low vitamin D levels increase the risk of type 2 diabetes in older adults: A systematic review and meta-analysis. Maturitas. 2017;100:8-15. (PubMed)
- George PS, Pearson ER, Witham MD. Effect of vitamin D supplementation on glycaemic control and insulin resistance: a systematic review and meta-analysis. Diabet Med. 2012;29(8):e142-150. (PubMed)
- Gulseth HL, Wium C, Angel K, Eriksen EF, Birkeland KI. Effects of vitamin D supplementation on insulin sensitivity and insulin secretion in subjects with type 2 diabetes and vitamin D deficiency: a randomized controlled trial. Diabetes Care. 2017;40(7):872-878. (PubMed)
- Lee CJ, Iyer G, Liu Y, et al. The effect of vitamin D supplementation on glucose metabolism in type 2 diabetes mellitus: A systematic review and meta-analysis of intervention studies. J Diabetes Complications. 2017;31(7):1115-1126. (PubMed)
- Talaei A, Mohamadi M, Adgi Z. The effect of vitamin D on insulin resistance in patients with type 2 diabetes. Diabetol Metab Syndr. 2013;5(1):8. (PubMed)
- Gezen-Ak D, Yilmazer S, Dursun E. Why vitamin d in Alzheimer's disease? The hypothesis. J Alzheimers Dis. 2014;40(2):257-269. (PubMed)
- Landel V, Annweiler C, Millet P, Morello M, Feron F. Vitamin D, cognition and Alzheimer's disease: the therapeutic benefit is in the D-tails. J Alzheimers Dis. 2016;53(2):419-444. (PubMed)
- Annweiler C, Schott AM, Rolland Y, Blain H, Herrmann FR, Beauchet O. Dietary intake of vitamin D and cognition in older women: a large population-based study. Neurology. 2010;75(20):1810-1816. (PubMed)
- Annweiler C, Rolland Y, Schott AM, et al. Higher vitamin D dietary intake is associated with lower risk of Alzheimer's disease: a 7-year follow-up. J Gerontol A Biol Sci Med Sci. 2012;67(11):1205-1211. (PubMed)
- Annweiler C, Fantino B, Schott AM, Krolak-Salmon P, Allali G, Beauchet O. Vitamin D insufficiency and mild cognitive impairment: cross-sectional association. Eur J Neurol. 2012;19(7):1023-1029. (PubMed)
- Hooshmand B, Lokk J, Solomon A, et al. Vitamin D in relation to cognitive impairment, cerebrospinal fluid biomarkers, and brain volumes. J Gerontol A Biol Sci Med Sci. 2014;69(9):1132-1138. (PubMed)
- Slinin Y, Paudel ML, Taylor BC, et al. 25-Hydroxyvitamin D levels and cognitive performance and decline in elderly men. Neurology. 2010;74(1):33-41. (PubMed)
- Slinin Y, Paudel M, Taylor BC, et al. Association between serum 25(OH) vitamin D and the risk of cognitive decline in older women. J Gerontol A Biol Sci Med Sci. 2012;67(10):1092-1098. (PubMed)
- Annweiler C, Milea D, Whitson HE, et al. Vitamin D insufficiency and cognitive impairment in Asians: a multi-ethnic population-based study and meta-analysis. J Intern Med. 2016;280(3):300-311. (PubMed)
- Annweiler C, Llewellyn DJ, Beauchet O. Low serum vitamin D concentrations in Alzheimer's disease: a systematic review and meta-analysis. J Alzheimers Dis. 2013;33(3):659-674. (PubMed)
- Balion C, Griffith LE, Strifler L, et al. Vitamin D, cognition, and dementia: a systematic review and meta-analysis. Neurology. 2012;79(13):1397-1405. (PubMed)
- Lopes da Silva S, Vellas B, Elemans S, et al. Plasma nutrient status of patients with Alzheimer's disease: Systematic review and meta-analysis. Alzheimers Dement. 2014;10(4):485-502. (PubMed)
- Shen L, Ji HF. Vitamin D deficiency is associated with increased risk of Alzheimer's disease and dementia: evidence from meta-analysis. Nutr J. 2015;14:76. (PubMed)
- Sommer I, Griebler U, Kien C, et al. Vitamin D deficiency as a risk factor for dementia: a systematic review and meta-analysis. BMC Geriatr. 2017;17(1):16. (PubMed)
- Olsson E, Byberg L, Karlstrom B, et al. Vitamin D is not associated with incident dementia or cognitive impairment: an 18-y follow-up study in community-living old men. Am J Clin Nutr. 2017;105(4):936-943. (PubMed)
- Mokry LE, Ross S, Morris JA, Manousaki D, Forgetta V, Richards JB. Genetically decreased vitamin D and risk of Alzheimer disease. Neurology. 2016;87(24):2567-2574. (PubMed)
- Annweiler C, Montero-Odasso M, Llewellyn DJ, Richard-Devantoy S, Duque G, Beauchet O. Meta-analysis of memory and executive dysfunctions in relation to vitamin D. J Alzheimers Dis. 2013;37(1):147-171. (PubMed)
- Annweiler C, Fantino B, Gautier J, Beaudenon M, Thiery S, Beauchet O. Cognitive effects of vitamin D supplementation in older outpatients visiting a memory clinic: a pre-post study. J Am Geriatr Soc. 2012;60(4):793-795. (PubMed)
- Stein MS, Scherer SC, Ladd KS, Harrison LC. A randomized controlled trial of high-dose vitamin D2 followed by intranasal insulin in Alzheimer's disease. J Alzheimers Dis. 2011;26(3):477-484. (PubMed)
- Annweiler C, Karras SN, Anagnostis P, Beauchet O. Vitamin D supplements: a novel therapeutic approach for Alzheimer patients. Front Pharmacol. 2014;5:6. (PubMed)
- Sato Y, Kikuyama M, Oizumi K. High prevalence of vitamin D deficiency and reduced bone mass in Parkinson's disease. Neurology. 1997;49(5):1273-1278. (PubMed)
- Evatt ML, Delong MR, Khazai N, Rosen A, Triche S, Tangpricha V. Prevalence of vitamin D insufficiency in patients with Parkinson disease and Alzheimer disease. Arch Neurol. 2008;65(10):1348-1352. (PubMed)
- Knekt P, Kilkkinen A, Rissanen H, Marniemi J, Saaksjarvi K, Heliovaara M. Serum vitamin D and the risk of Parkinson disease. Arch Neurol. 2010;67(7):808-811. (PubMed)
- Lv Z, Qi H, Wang L, et al. Vitamin D status and Parkinson's disease: a systematic review and meta-analysis. Neurol Sci. 2014;35(11):1723-1730. (PubMed)
- Shen L, Ji HF. Associations between vitamin D status, supplementation, outdoor work and risk of Parkinson's disease: a meta-analysis assessment. Nutrients. 2015;7(6):4817-4827. (PubMed)
- Zhao Y, Sun Y, Ji HF, Shen L. Vitamin D levels in Alzheimer's and Parkinson's diseases: a meta-analysis. Nutrition. 2013;29(6):828-832. (PubMed)
- Suzuki M, Yoshioka M, Hashimoto M, et al. Randomized, double-blind, placebo-controlled trial of vitamin D supplementation in Parkinson disease. Am J Clin Nutr. 2013;97(5):1004-1013. (PubMed)
- Dobson R, Yarnall A, Noyce AJ, Giovannoni G. Bone health in chronic neurological diseases: a focus on multiple sclerosis and parkinsonian syndromes. Pract Neurol. 2013;13(2):70-79. (PubMed)
- Torsney KM, Noyce AJ, Doherty KM, Bestwick JP, Dobson R, Lees AJ. Bone health in Parkinson's disease: a systematic review and meta-analysis. J Neurol Neurosurg Psychiatry. 2014;85(10):1159-66. (PubMed)
- van den Bos F, Speelman AD, Samson M, Munneke M, Bloem BR, Verhaar HJ. Parkinson's disease and osteoporosis. Age Ageing. 2013;42(2):156-162. (PubMed)
- Sato Y, Iwamoto J, Honda Y. Amelioration of osteoporosis and hypovitaminosis D by sunlight exposure in Parkinson's disease. Parkinsonism Relat Disord. 2011;17(1):22-26. (PubMed)
- Aghajafari F, Nagulesapillai T, Ronksley PE, Tough SC, O'Beirne M, Rabi DM. Association between maternal serum 25-hydroxyvitamin D level and pregnancy and neonatal outcomes: systematic review and meta-analysis of observational studies. BMJ. 2013;346:f1169. (PubMed)
- Perez-Lopez FR, Pasupuleti V, Mezones-Holguin E, et al. Effect of vitamin D supplementation during pregnancy on maternal and neonatal outcomes: a systematic review and meta-analysis of randomized controlled trials. Fertil Steril. 2015;103(5):1278-1288.e1274. (PubMed)
- Alzaim M, Wood RJ. Vitamin D and gestational diabetes mellitus. Nutr Rev. 2013;71(3):158-167. (PubMed)
- Lacroix M, Battista MC, Doyon M, et al. Lower vitamin D levels at first trimester are associated with higher risk of developing gestational diabetes mellitus. Acta Diabetol. 2014;51(4):609-616. (PubMed)
- Parlea L, Bromberg IL, Feig DS, Vieth R, Merman E, Lipscombe LL. Association between serum 25-hydroxyvitamin D in early pregnancy and risk of gestational diabetes mellitus. Diabet Med. 2012;29(7):e25-32. (PubMed)
- Lu M, Xu Y, Lv L, Zhang M. Association between vitamin D status and the risk of gestational diabetes mellitus: a meta-analysis. Arch Gynecol Obstet. 2016;293(5):959-966. (PubMed)
- Poel YH, Hummel P, Lips P, Stam F, van der Ploeg T, Simsek S. Vitamin D and gestational diabetes: a systematic review and meta-analysis. Eur J Intern Med. 2012;23(5):465-469. (PubMed)
- Wei SQ, Qi HP, Luo ZC, Fraser WD. Maternal vitamin D status and adverse pregnancy outcomes: a systematic review and meta-analysis. J Matern Fetal Neonatal Med. 2013;26(9):889-899. (PubMed)
- Zhang MX, Pan GT, Guo JF, Li BY, Qin LQ, Zhang ZL. Vitamin D deficiency increases the risk of gestational diabetes mellitus: a meta-analysis of observational studies. Nutrients. 2015;7(10):8366-8375. (PubMed)
- Triunfo S, Lanzone A, Lindqvist PG. Low maternal circulating levels of vitamin D as potential determinant in the development of gestational diabetes mellitus. J Endocrinol Invest. 2017;40(10):1049-1059. (PubMed)
- Asemi Z, Hashemi T, Karamali M, Samimi M, Esmaillzadeh A. Effects of vitamin D supplementation on glucose metabolism, lipid concentrations, inflammation, and oxidative stress in gestational diabetes: a double-blind randomized controlled clinical trial. Am J Clin Nutr. 2013;98(6):1425-1432. (PubMed)
- Jelsma JG, van Poppel MN, Galjaard S, et al. DALI: Vitamin D and lifestyle intervention for gestational diabetes mellitus (GDM) prevention: an European multicentre, randomised trial - study protocol. BMC Pregnancy Childbirth. 2013;13:142. (PubMed)
- Simmons D, Devlieger R, van Assche A, et al. Effect of Physical Activity and/or Healthy Eating on GDM Risk: The DALI Lifestyle Study. J Clin Endocrinol Metab. 2017;102(3):903-913. (PubMed)
- Galthen-Sorensen M, Andersen LB, Sperling L, Christesen HT. Maternal 25-hydroxyvitamin D level and fetal bone growth assessed by ultrasound: a systematic review. Ultrasound Obstet Gynecol. 2014;44(6):633-640. (PubMed)
- Shor DB, Barzel J, Tauber E, Amital H. The effects of maternal vitamin D on neonatal growth parameters. Eur J Pediatr. 2015;174(9):1169-1174. (PubMed)
- Cooper C, Harvey NC, Bishop NJ, et al. Maternal gestational vitamin D supplementation and offspring bone health (MAVIDOS): a multicentre, double-blind, randomised placebo-controlled trial. Lancet Diabetes Endocrinol. 2016;4(5):393-402. (PubMed)
- Handel MN, Frederiksen P, Osmond C, Cooper C, Abrahamsen B, Heitmann BL. Prenatal exposure to vitamin D from fortified margarine and risk of fractures in late childhood: period and cohort results from 222 000 subjects in the D-tect observational study. Br J Nutr. 2017;117(6):872-881. (PubMed)
- Rodda CP, Benson JE, Vincent AJ, Whitehead CL, Polykov A, Vollenhoven B. Maternal vitamin D supplementation during pregnancy prevents vitamin D deficiency in the newborn: an open-label randomized controlled trial. Clin Endocrinol (Oxf). 2015;83(3):363-368. (PubMed)
- Handel MN, Frederiksen P, Cohen A, Cooper C, Heitmann BL, Abrahamsen B. Neonatal vitamin D status from archived dried blood spots and future risk of fractures in childhood: results from the D-tect study, a population-based case-cohort study. Am J Clin Nutr. 2017;106(1):155-161. (PubMed)
- Bountouvi E, Douros K, Papadopoulou A. Can getting enough vitamin D during pregnancy reduce the risk of getting asthma in childhood? Front Pediatr. 2017;5:87. (PubMed)
- Goldring ST, Griffiths CJ, Martineau AR, et al. Prenatal vitamin D supplementation and child respiratory health: a randomised controlled trial. PLoS One. 2013;8(6):e66627. (PubMed)
- Chawes BL, Bonnelykke K, Stokholm J, et al. Effect of vitamin D3 supplementation during pregnancy on rsk of persistent wheeze in the offspring: a randomized clinical trial. JAMA. 2016;315(4):353-361. (PubMed)
- Litonjua AA, Carey VJ, Laranjo N, et al. Effect of prenatal supplementation with vitamin D on asthma or eecurrent wheezing in offspring by age 3 years: the VDAART randomized clinical trial. JAMA. 2016;315(4):362-370. (PubMed)
- Vahdaninia M, Mackenzie H, Helps S, Dean T. Prenatal intake of vitamins and allergic outcomes in the offspring: a systematic review and meta-analysis. J Allergy Clin Immunol Pract. 2017;5(3):771-778.e775. (PubMed)
- Weisse K, Winkler S, Hirche F, et al. Maternal and newborn vitamin D status and its impact on food allergy development in the German LINA cohort study. Allergy. 2013;68(2):220-228. (PubMed)
- Miettinen ME, Smart MC, Kinnunen L, et al. Maternal VDR variants rather than 25-hydroxyvitamin D concentration during early pregnancy are associated with type 1 diabetes in the offspring. Diabetologia. 2015;58(10):2278-2283. (PubMed)
- Sorensen IM, Joner G, Jenum PA, et al. Vitamin D-binding protein and 25-hydroxyvitamin D during pregnancy in mothers whose children later developed type 1 diabetes. Diabetes Metab Res Rev. 2016;32(8):883-890. (PubMed)
- Makela MJ, Puhakka T, Ruuskanen O, et al. Viruses and bacteria in the etiology of the common cold. J Clin Microbiol. 1998;36(2):539-542. (PubMed)
- Ginde AA, Mansbach JM, Camargo CA, Jr. Association between serum 25-hydroxyvitamin D level and upper respiratory tract infection in the Third National Health and Nutrition Examination Survey. Arch Intern Med. 2009;169(4):384-390. (PubMed)
- Murdoch DR, Slow S, Chambers ST, et al. Effect of vitamin D3 supplementation on upper respiratory tract infections in healthy adults: the VIDARIS randomized controlled trial. JAMA. 2012;308(13):1333-1339. (PubMed)
- Rees JR, Hendricks K, Barry EL, et al. Vitamin D3 supplementation and upper respiratory tract infections in a randomized, controlled trial. Clin Infect Dis. 2013;57(10):1384-1392. (PubMed)
- Tran B, Armstrong BK, Ebeling PR, et al. Effect of vitamin D supplementation on antibiotic use: a randomized controlled trial. Am J Clin Nutr. 2014;99(1):156-161. (PubMed)
- Grant CC, Kaur S, Waymouth E, et al. Reduced primary care respiratory infection visits following pregnancy and infancy vitamin D supplementation: a randomised controlled trial. Acta Paediatr. 2015;104(4):396-404. (PubMed)
- Martineau AR, Jolliffe DA, Hooper RL, et al. Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data. BMJ. 2017;356:i6583. (PubMed)
- Scragg R, Waayer D, Stewart AW, et al. The Vitamin D Assessment (ViDA) study: design of a randomized controlled trial of vitamin D supplementation for the prevention of cardiovascular disease, acute respiratory infection, falls and non-vertebral fractures. J Steroid Biochem Mol Biol. 2016;164:318-325. (PubMed)
- Manson JE, Bassuk SS, Buring JE, Group VR. Principal results of the VITamin D and OmegA-3 TriaL (VITAL) and updated meta-analyses of relevant vitamin D trials. J Steroid Biochem Mol Biol. 2020;198:105522. (PubMed)
- Gold DR, Litonjua AA, Carey VJ, et al. Lung VITAL: Rationale, design, and baseline characteristics of an ancillary study evaluating the effects of vitamin D and/or marine omega-3 fatty acid supplements on acute exacerbations of chronic respiratory disease, asthma control, pneumonia and lung function in adults. Contemp Clin Trials. 2016;47:185-195. (PubMed)
- Aglipay M, Birken CS, Parkin PC, et al. Effect of high-dose vs standard-dose wintertime vitamin D supplementation on viral upper respiratory tract infections in young healthy children. JAMA. 2017;318(3):245-254. (PubMed)
- Hueniken K, Aglipay M, Birken CS, et al. Effect of high-dose vitamin D supplementation on upper respiratory tract infection symptom severity in healthy children. Pediatr Infect Dis J. 2019;38(6):564-568. (PubMed)
- Loeb M, Dang AD, Thiem VD, et al. Effect of Vitamin D supplementation to reduce respiratory infections in children and adolescents in Vietnam: A randomized controlled trial. Influenza Other Respir Viruses. 2019;13(2):176-183. (PubMed)
- Kronbichler A, Kresse D, Yoon S, Lee KH, Effenberger M, Shin JI. Asymptomatic patients as a source of COVID-19 infections: A systematic review and meta-analysis. Int J Infect Dis. 2020;98:180-186. (PubMed)
- Wiersinga WJ, Rhodes A, Cheng AC, Peacock SJ, Prescott HC. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review. JAMA. 2020;324(8):782-793. (PubMed)
- Ferrari D, Locatelli M. No significant association between vitamin D and COVID-19. A retrospective study from a northern Italian hospital. Int J Vitam Nutr Res. 2020:1-4. (PubMed)
- Hastie CE, Mackay DF, Ho F, et al. Vitamin D concentrations and COVID-19 infection in UK Biobank. Diabetes Metab Syndr. 2020;14(4):561-565. (PubMed)
- Ma H, Zhou T, Heianza Y, Qi L. Habitual use of vitamin D supplements and risk of coronavirus disease 2019 (COVID-19) infection: a prospective study in UK Biobank. Am J Clin Nutr. 2021;113(5):1275-1281. (PubMed)
- Kaufman HW, Niles JK, Kroll MH, Bi C, Holick MF. SARS-CoV-2 positivity rates associated with circulating 25-hydroxyvitamin D levels. PLoS One. 2020;15(9):e0239252. (PubMed)
- Merzon E, Tworowski D, Gorohovski A, et al. Low plasma 25(OH) vitamin D level is associated with increased risk of COVID-19 infection: an Israeli population-based study. FEBS J. 2020;287(17):3693-3702. (PubMed)
- Meltzer DO, Best TJ, Zhang H, Vokes T, Arora V, Solway J. Association of vitamin D status and other clinical characteristics with COVID-19 test results. JAMA Netw Open. 2020;3(9):e2019722. (PubMed)
- D'Avolio A, Avataneo V, Manca A, et al. 25-Hydroxyvitamin D concentrations are lower in patients with positive PCR for SARS-CoV-2. Nutrients. 2020;12(5):1359. (PubMed)
- Ye K, Tang F, Liao X, et al. Does serum vitamin D level affect COVID-19 infection and its severity?-a case-control study. J Am Coll Nutr. 2020:1-8. (PubMed)
- Hernandez JL, Nan D, Fernandez-Ayala M, et al. Vitamin D status in hospitalized patients with SARS-CoV-2 infection. J Clin Endocrinol Metab. 2020;106(3):e1343-e1353. (PubMed)
- Im JH, Je YS, Baek J, Chung MH, Kwon HY, Lee JS. Nutritional status of patients with COVID-19. Int J Infect Dis. 2020;100:390-393. (PubMed)
- Abdollahi A, Kamali Sarvestani H, Rafat Z, et al. The association between the level of serum 25(OH) vitamin D, obesity, and underlying diseases with the risk of developing COVID-19 infection: A case-control study of hospitalized patients in Tehran, Iran. J Med Virol. 2020;93(4):2359-2364. (PubMed)
- Liu N, Sun J, Wang X, Zhang T, Zhao M, Li H. Low vitamin D status is associated with coronavirus disease 2019 outcomes: a systematic review and meta-analysis. Int J Infect Dis. 2021;104:58-64. (PubMed)
- Panagiotou G, Tee SA, Ihsan Y, et al. Low serum 25-hydroxyvitamin D (25[OH]D) levels in patients hospitalized with COVID-19 are associated with greater disease severity. Clin Endocrinol (Oxf). 2020;93(4):508-511. (PubMed)
- De Smet D, De Smet K, Herroelen P, Gryspeerdt S, Martens GA. Serum 25(OH)D level on hospital admission associated with COVID-19 stage and mortality. Am J Clin Pathol. 2020;155(3):381-388. (PubMed)
- Baktash V, Hosack T, Patel N, et al. Vitamin D status and outcomes for hospitalised older patients with COVID-19. Postgrad Med J. 2020. [Epub ahead of print] (PubMed)
- Radujkovic A, Hippchen T, Tiwari-Heckler S, Dreher S, Boxberger M, Merle U. Vitamin D deficiency and outcome of COVID-19 patients. Nutrients. 2020;12(9): 2757. (PubMed)
- Carpagnano GE, Di Lecce V, Quaranta VN, et al. Vitamin D deficiency as a predictor of poor prognosis in patients with acute respiratory failure due to COVID-19. J Endocrinol Invest. 2020;(4):765-771. (PubMed)
- Padhi S, Suvankar S, Panda VK, Pati A, Panda AK. Lower levels of vitamin D are associated with SARS-CoV-2 infection and mortality in the Indian population: An observational study. Int Immunopharmacol. 2020;88:107001. (PubMed)
- Hastie CE, Pell JP, Sattar N. Vitamin D and COVID-19 infection and mortality in UK Biobank. Eur J Nutr. 2020;60(1):545-548. (PubMed)
- Centers for Disease Control and Prevention. Underlying conditions and the higher risk for severe COVID-19. https://www.cdc.gov/covid/hcp/clinical-care/underlying-conditions.html. Accessed 8/11/25.
- Annweiler G, Corvaisier M, Gautier J, et al. Vitamin D supplementation associated to better survival in hospitalized frail elderly COVID-19 patients: the GERIA-COVID quasi-experimental study. Nutrients. 2020;12(11):3377. (PubMed)
- Annweiler C, Hanotte B, Grandin de l'Eprevier C, Sabatier JM, Lafaie L, Celarier T. Vitamin D and survival in COVID-19 patients: A quasi-experimental study. J Steroid Biochem Mol Biol. 2020;204:105771. (PubMed)
- Mesquita Kde C, Igreja AC, Costa IM. Atopic dermatitis and vitamin D: facts and controversies. An Bras Dermatol. 2013;88(6):945-953. (PubMed)
- Lee SA, Hong S, Kim HJ, Lee SH, Yum HY. Correlation between serum vitamin D level and the severity of atopic dermatitis associated with food sensitization. Allergy Asthma Immunol Res. 2013;5(4):207-210. (PubMed)
- Sudlow C, Gallacher J, Allen N, et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 2015;12(3):e1001779. (PubMed)
- Moffatt MF, Gut IG, Demenais F, et al. A large-scale, consortium-based genomewide association study of asthma. N Engl J Med. 2010;363(13):1211-1221. (PubMed)
- Paternoster L, Zhurov AI, Toma AM, et al. Genome-wide association study of three-dimensional facial morphology identifies a variant in PAX3 associated with nasion position. Am J Hum Genet. 2012;90(3):478-485. (PubMed)
- Manousaki D, Paternoster L, Standl M, et al. Vitamin D levels and susceptibility to asthma, elevated immunoglobulin E levels, and atopic dermatitis: A Mendelian randomization study. PLoS Med. 2017;14(5):e1002294. (PubMed)
- Javanbakht MH, Keshavarz SA, Djalali M, et al. Randomized controlled trial using vitamins E and D supplementation in atopic dermatitis. J Dermatolog Treat. 2011;22(3):144-150. (PubMed)
- Amestejani M, Salehi BS, Vasigh M, et al. Vitamin D supplementation in the treatment of atopic dermatitis: a clinical trial study. J Drugs Dermatol. 2012;11(3):327-330. (PubMed)
- Camargo CA, Jr., Ganmaa D, Sidbury R, Erdenedelger K, Radnaakhand N, Khandsuren B. Randomized trial of vitamin D supplementation for winter-related atopic dermatitis in children. J Allergy Clin Immunol. 2014;134(4):831-835.e831. (PubMed)
- Kim G, Bae JH. Vitamin D and atopic dermatitis: A systematic review and meta-analysis. Nutrition. 2016;32(9):913-920. (PubMed)
- Wat H, Dytoc M. Off-label uses of topical vitamin d in dermatology: a systematic review. J Cutan Med Surg. 2014;18(2):91-108. (PubMed)
- Xue LN, Xu KQ, Zhang W, Wang Q, Wu J, Wang XY. Associations between vitamin D receptor polymorphisms and susceptibility to ulcerative colitis and Crohn's disease: a meta-analysis. Inflamm Bowel Dis. 2013;19(1):54-60. (PubMed)
- Ananthakrishnan AN, Khalili H, Higuchi LM, et al. Higher predicted vitamin D status is associated with reduced risk of Crohn's disease. Gastroenterology. 2012;142(3):482-489. (PubMed)
- Sadeghian M, Saneei P, Siassi F, Esmaillzadeh A. Vitamin D status in relation to Crohn's disease: meta-analysis of observational studies. Nutrition. 2016;32(5):505-514. (PubMed)
- Jorgensen SP, Agnholt J, Glerup H, et al. Clinical trial: vitamin D3 treatment in Crohn's disease - a randomized double-blind placebo-controlled study. Aliment Pharmacol Ther. 2010;32(3):377-383. (PubMed)
- Yang L, Weaver V, Smith JP, Bingaman S, Hartman TJ, Cantorna MT. Therapeutic effect of vitamin d supplementation in a pilot study of Crohn's patients. Clin Transl Gastroenterol. 2013;4:e33. (PubMed)
- Raftery T, Martineau AR, Greiller CL, et al. Effects of vitamin D supplementation on intestinal permeability, cathelicidin and disease markers in Crohn's disease: Results from a randomised double-blind placebo-controlled study. United European Gastroenterol J. 2015;3(3):294-302. (PubMed)
- Anderson JL, May HT, Horne BD, et al. Relation of vitamin D deficiency to cardiovascular risk factors, disease status, and incident events in a general healthcare population. Am J Cardiol. 2010;106(7):963-968. (PubMed)
- Al Mheid I, Patel R, Murrow J, et al. Vitamin D status is associated with arterial stiffness and vascular dysfunction in healthy humans. J Am Coll Cardiol. 2011;58(2):186-192. (PubMed)
- Krause R, Buhring M, Hopfenmuller W, Holick MF, Sharma AM. Ultraviolet B and blood pressure. Lancet. 1998;352(9129):709-710. (PubMed)
- Kunutsor SK, Burgess S, Munroe PB, Khan H. Vitamin D and high blood pressure: causal association or epiphenomenon? Eur J Epidemiol. 2014;29(1):1-14. (PubMed)
- Pilz S, Gaksch M, Kienreich K, et al. Effects of vitamin D on blood pressure and cardiovascular risk factors: a randomized controlled trial. Hypertension. 2015;65(6):1195-1201. (PubMed)
- Arora P, Song Y, Dusek J, et al. Vitamin D therapy in individuals with prehypertension or hypertension: the DAYLIGHT trial. Circulation. 2015;131(3):254-262. (PubMed)
- Rostand SG. Ultraviolet light may contribute to geographic and racial blood pressure differences. Hypertension. 1997;30(2 Pt 1):150-156. (PubMed)
- Forman JP, Scott JB, Ng K, et al. Effect of vitamin D supplementation on blood pressure in blacks. Hypertension. 2013;61(4):779-785. (PubMed)
- Witham MD, Price RJ, Struthers AD, et al. Cholecalciferol treatment to reduce blood pressure in older patients with isolated systolic hypertension: the VitDISH randomized controlled trial. JAMA Intern Med. 2013;173(18):1672-1679. (PubMed)
- Oz F, Cizgici AY, Oflaz H, et al. Impact of vitamin D insufficiency on the epicardial coronary flow velocity and endothelial function. Coron Artery Dis. 2013;24(5):392-397. (PubMed)
- Liu LC, Voors AA, van Veldhuisen DJ, et al. Vitamin D status and outcomes in heart failure patients. Eur J Heart Fail. 2011;13(6):619-625. (PubMed)
- Shedeed SA. Vitamin D supplementation in infants with chronic congestive heart failure. Pediatr Cardiol. 2012;33(5):713-719. (PubMed)
- Boxer RS, Kenny AM, Schmotzer BJ, Vest M, Fiutem JJ, Pina IL. A randomized controlled trial of high dose vitamin D3 in patients with heart failure. JACC Heart Fail. 2013;1(1):84-90. (PubMed)
- Jiang WL, Gu HB, Zhang YF, Xia QQ, Qi J, Chen JC. Vitamin D supplementation in the treatment of chronic heart failure: a meta-analysis of randomized controlled trials. Clin Cardiol. 2016;39(1):56-61. (PubMed)
- Zittermann A, Ernst JB, Prokop S, et al. Effect of vitamin D on all-cause mortality in heart failure (EVITA): a 3-year randomized clinical trial with 4000 IU vitamin D daily. Eur Heart J. 2017;38(29):2279-2286. (PubMed)
- Norman AW, Henry HH. Vitamin D. In: Bowman BA, Russell RM, eds. Present Knowledge in Nutrition. 9th ed. Washington, D.C.: ILSI Press; 2006:198-210.
- Holick MF. Vitamin D: the underappreciated D-lightful hormone that is important for skeletal and cellular health. Curr Opin Endocrinol Diabetes. 2002;9:87-98.
- Terushkin V, Bender A, Psaty EL, Engelsen O, Wang SQ, Halpern AC. Estimated equivalency of vitamin D production from natural sun exposure versus oral vitamin D supplementation across seasons at two US latitudes. J Am Acad Dermatol. 2010;62(6):929 e921-929. (PubMed)
- Food and Nutrition Board, Institute of Medicine. Vitamin D. Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride. Washington, D.C.: National Academies Press; 1999:250-287. (National Academy Press)
- Ovesen L, Brot C, Jakobsen J. Food contents and biological activity of 25-hydroxyvitamin D: a vitamin D metabolite to be reckoned with? Ann Nutr Metab. 2003;47(3-4):107-113. (PubMed)
- Jakobsen J, Christensen T. Natural vitamin D in food: to what degree does 25-hydroxyvitaminn D contribute to the vitamin D activity in food? JMBR Plus. 2020: doi: 10.1002/jbm1004.10453.
- Tripkovic L, Lambert H, Hart K, et al. Comparison of vitamin D2 and vitamin D3 supplementation in raising serum 25-hydroxyvitamin D status: a systematic review and meta-analysis. Am J Clin Nutr. 2012;95(6):1357-1364. (PubMed)
- Logan VF, Gray AR, Peddie MC, Harper MJ, Houghton LA. Long-term vitamin D3 supplementation is more effective than vitamin D2 in maintaining serum 25-hydroxyvitamin D status over the winter months. Br J Nutr. 2013;109(6):1082-1088. (PubMed)
- Barger-Lux MJ, Heaney RP, Dowell S, Chen TC, Holick MF. Vitamin D and its major metabolites: serum levels after graded oral dosing in healthy men. Osteoporos Int. 1998;8(3):222-230. (PubMed)
- Cashman KD, Seamans KM, Lucey AJ, et al. Relative effectiveness of oral 25-hydroxyvitamin D3 and vitamin D3 in raising wintertime serum 25-hydroxyvitamin D in older adults. Am J Clin Nutr. 2012;95(6):1350-1356. (PubMed)
- Bischoff-Ferrari HA, Dawson-Hughes B, Stocklin E, et al. Oral supplementation with 25(OH)D3 versus vitamin D3: effects on 25(OH)D levels, lower extremity function, blood pressure, and markers of innate immunity. J Bone Miner Res. 2012;27(1):160-169. (PubMed)
- Jetter A, Egli A, Dawson-Hughes B, et al. Pharmacokinetics of oral vitamin D(3) and calcifediol. Bone. 2014;59:14-19. (PubMed)
- Navarro-Valverde C, Sosa-Henriquez M, Alhambra-Exposito MR, Quesada-Gomez JM. Vitamin D3 and calcidiol are not equipotent. J Steroid Biochem Mol Biol. 2016;164:205-208. (PubMed)
- Shieh A, Ma C, Chun RF, et al. Effects of cholecalciferol vs calcifediol on total and free 25-hydroxyvitamin D and parathyroid hormone. J Clin Endocrinol Metab. 2017;102(4):1133-1140. (PubMed)
- Vaes AMM, Tieland M, de Regt MF, Wittwer J, van Loon LJC, de Groot L. Dose-response effects of supplementation with calcifediol on serum 25-hydroxyvitamin D status and its metabolites: A randomized controlled trial in older adults. Clin Nutr. 2018;37(3):808-814. (PubMed)
- Graeff-Armas LA, Bendik I, Kunz I, Schoop R, Hull S, Beck M. Supplemental 25-hydroxycholecalciferol is more effective than cholecalciferol in raising serum 25-hydroxyvitamin D concentrations in older adults. J Nutr. 2020;150(1):73-81. (PubMed)
- Quesada-Gomez JM, Bouillon R. Is calcifediol better than cholecalciferol for vitamin D supplementation? Osteoporos Int. 2018;29(8):1697-1711. (PubMed)
- Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357(3):266-281. (PubMed)
- Vieth R. Vitamin D supplementation, 25-hydroxyvitamin D concentrations, and safety. Am J Clin Nutr. 1999;69(5):842-856. (PubMed)
- Heaney RP, Davies KM, Chen TC, Holick MF, Barger-Lux MJ. Human serum 25-hydroxycholecalciferol response to extended oral dosing with cholecalciferol. Am J Clin Nutr. 2003;77(1):204-210. (PubMed)
- Vieth R, Chan PC, MacFarlane GD. Efficacy and safety of vitamin D3 intake exceeding the lowest observed adverse effect level. Am J Clin Nutr. 2001;73(2):288-294. (PubMed)
- Knodel LC, Talbert RL. Adverse effects of hypolipidaemic drugs. Med Toxicol. 1987;2(1):10-32. (PubMed)
- McDuffie JR, Calis KA, Booth SL, Uwaifo GI, Yanovski JA. Effects of orlistat on fat-soluble vitamins in obese adolescents. Pharmacotherapy. 2002;22(7):814-822. (PubMed)
- Natural Medicines. Vitamin D. Professional handout/Drug interactions. Available at: https://naturalmedicines.therapeuticresearch.com. Accessed 6/11/17.
- Panday K, Gona A, Humphrey MB. Medication-induced osteoporosis: screening and treatment strategies. Ther Adv Musculoskelet Dis. 2014;6(5):185-202. (PubMed)
- Glass AR, Eil C. Ketoconazole-induced reduction in serum 1,25-dihydroxyvitamin D. J Clin Endocrinol Metab. 1986;63(3):766-769. (PubMed)
Disclaimer
The Linus Pauling Institute's Micronutrient Information Center provides scientific information on the health aspects of dietary factors and supplements, food, and beverages for the general public. The information is made available with the understanding that the author and publisher are not providing medical, psychological, or nutritional counseling services on this site. The information should not be used in place of a consultation with a competent health care or nutrition professional.
The information on dietary factors and supplements, food, and beverages contained on this website does not cover all possible uses, actions, precautions, side effects, and interactions. It is not intended as nutritional or medical advice for individual problems. Liability for individual actions or omissions based upon the contents of this site is expressly disclaimed.
You may not copy, modify, distribute, display, transmit, perform, publish or sell any of the copyrightable material on this website. You may hyperlink to this website but must include the following statement:
"This link leads to a website provided by the Linus Pauling Institute at Oregon State University. [Your name] is not affiliated or endorsed by the Linus Pauling Institute or Oregon State University."